Abstract
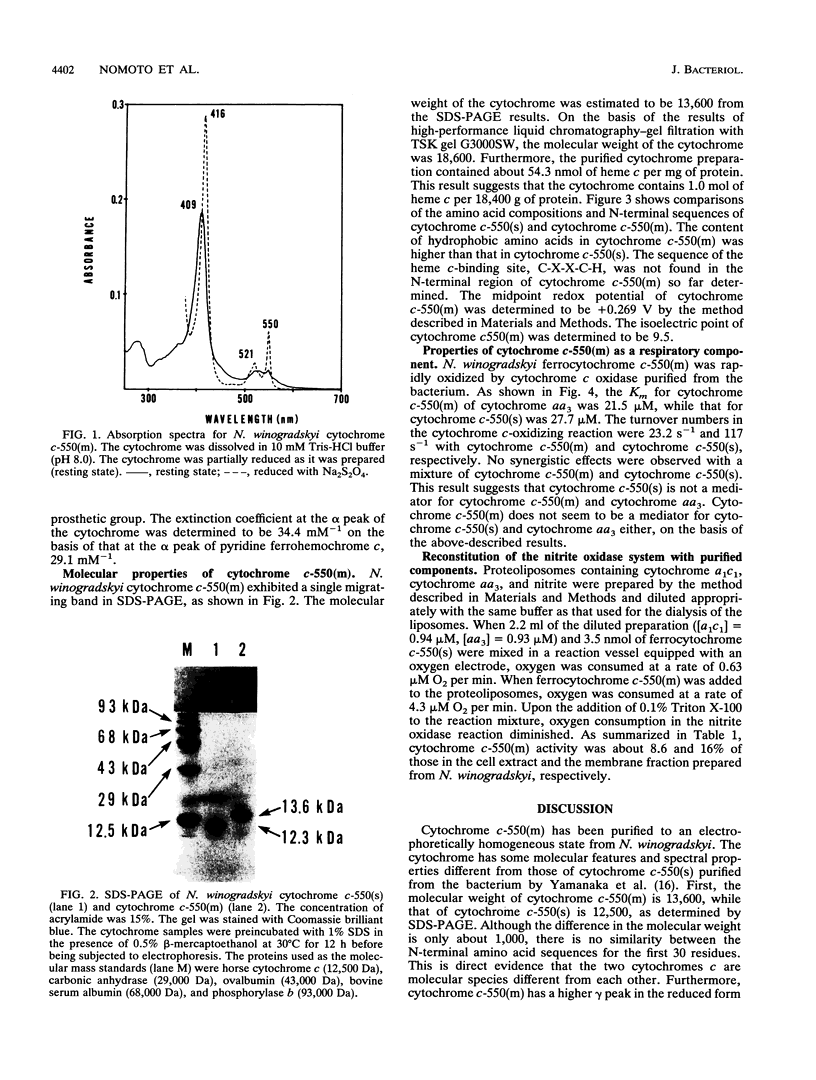
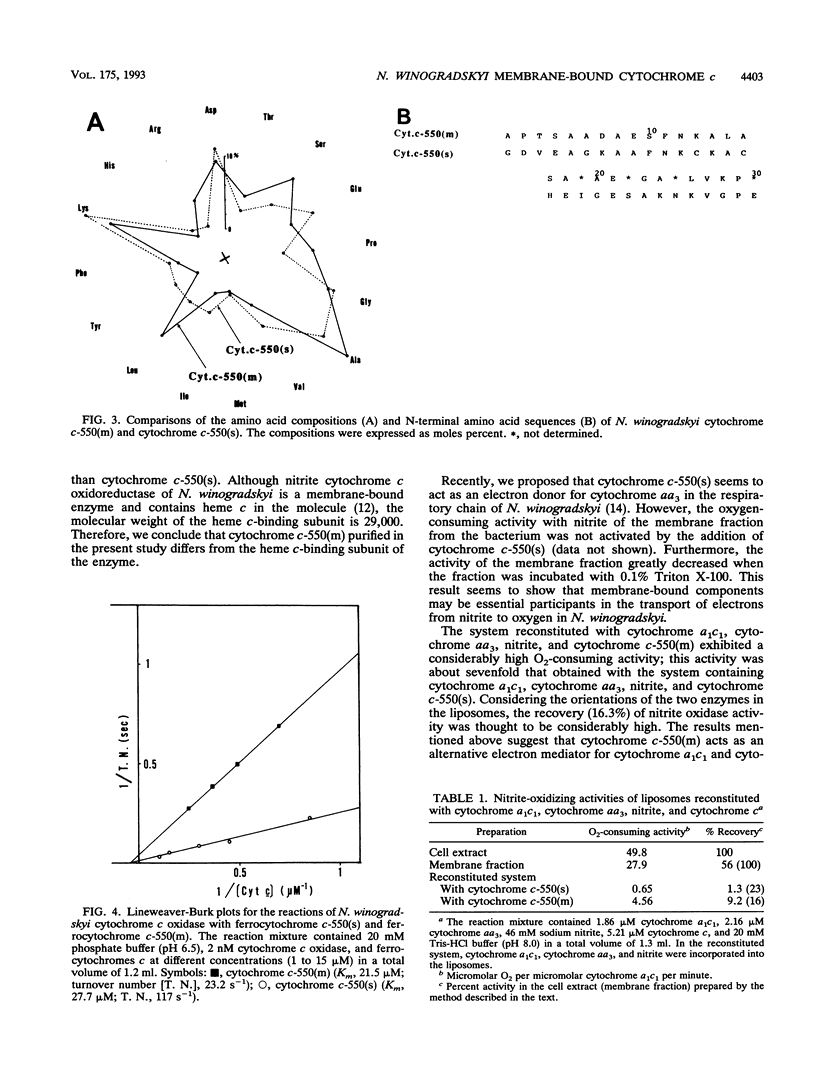
We purified membrane-bound cytochrome c-550 [cytochrome c-550(m)] to an electrophoretically homogeneous state from Nitrobacter winogradskyi. The cytochrome showed peaks at 409 and 525 nm in the oxidized form and peaks at 416, 521, and 550 nm in the reduced form. The molecular weight of the cytochrome was estimated to be 18,400 on the basis of protein and heme c contents and 18,600 by gel filtration. The N-terminal amino acid sequence of cytochrome c-550(m) was determined to be A-P-T-S-A-A-D-A-E-S-F-N-K-A-L-A-S-A-?-A-E-?-G-A-?-L-V-K-P. We previously purified soluble cytochrome c-550 cytochrome c-550(s)] from N. winogradskyi and determined its complete amino acid sequence (Y. Tanaka, Y. Fukumori, and T. Y. Yamanaka, Biochim. Biophys. Acta 707:14-20, 1982). Although the sequence of cytochrome c-550(m) was completely different from that of cytochrome c-550(s), ferrocytochrome c-550(m) was rapidly oxidized by the cytochrome c oxidase of the bacterium. Furthermore, the liposomes into which nitrite cytochrome c oxidoreductase, cytochrome c oxidase, and nitrite were incorporated showed nitrite oxidase activity in the presence of cytochrome c-550(m). These results suggest that cytochrome c-550(m) may be an alternative electron mediator between nitrite cytochrome c oxidoreductase and cytochrome c oxidase.
Full text
PDF


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- DAVENPORT H. E., HILL R. The preparation and some properties of cytochrome f. Proc R Soc Lond B Biol Sci. 1952 Apr 24;139(896):327–345. doi: 10.1098/rspb.1952.0016. [DOI] [PubMed] [Google Scholar]
- Fukuoka M., Fukumori Y., Yamanaka T. Nitrobacter winogradskyi cytochrome a1c1 is an iron-sulfur molybdoenzyme having hemes a and c. J Biochem. 1987 Sep;102(3):525–530. doi: 10.1093/oxfordjournals.jbchem.a122084. [DOI] [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- O'Farrell P. H. High resolution two-dimensional electrophoresis of proteins. J Biol Chem. 1975 May 25;250(10):4007–4021. [PMC free article] [PubMed] [Google Scholar]
- Sone N., Yoshida M., Hirata H., Kagawa Y. Reconstitution of vesicles capable of energy transformation from phospholipids and adenosine triphosphatase of a thermophilic bacterium. J Biochem. 1977 Feb;81(2):519–528. doi: 10.1093/oxfordjournals.jbchem.a131485. [DOI] [PubMed] [Google Scholar]
- Tanaka Y., Fukumori Y., Yamanaka T. The complete amino acid sequence of Nitrobacter agilis cytochrome c-550. Biochim Biophys Acta. 1982 Sep 22;707(1):14–20. doi: 10.1016/0167-4838(82)90390-9. [DOI] [PubMed] [Google Scholar]
- Yamanaka T., Fukumori Y. The nitrite oxidizing system of Nitrobacter winogradskyi. FEMS Microbiol Rev. 1988 Dec;4(4):259–270. doi: 10.1016/0378-1097(88)90246-7. [DOI] [PubMed] [Google Scholar]
- Yamanaka T., Kamita Y., Fukumori Y. Molecular and enzymatic properties of "cytochrome aa3"-type terminal oxidase derived from Nitrobacter agilis. J Biochem. 1981 Jan;89(1):265–273. doi: 10.1093/oxfordjournals.jbchem.a133190. [DOI] [PubMed] [Google Scholar]