Abstract
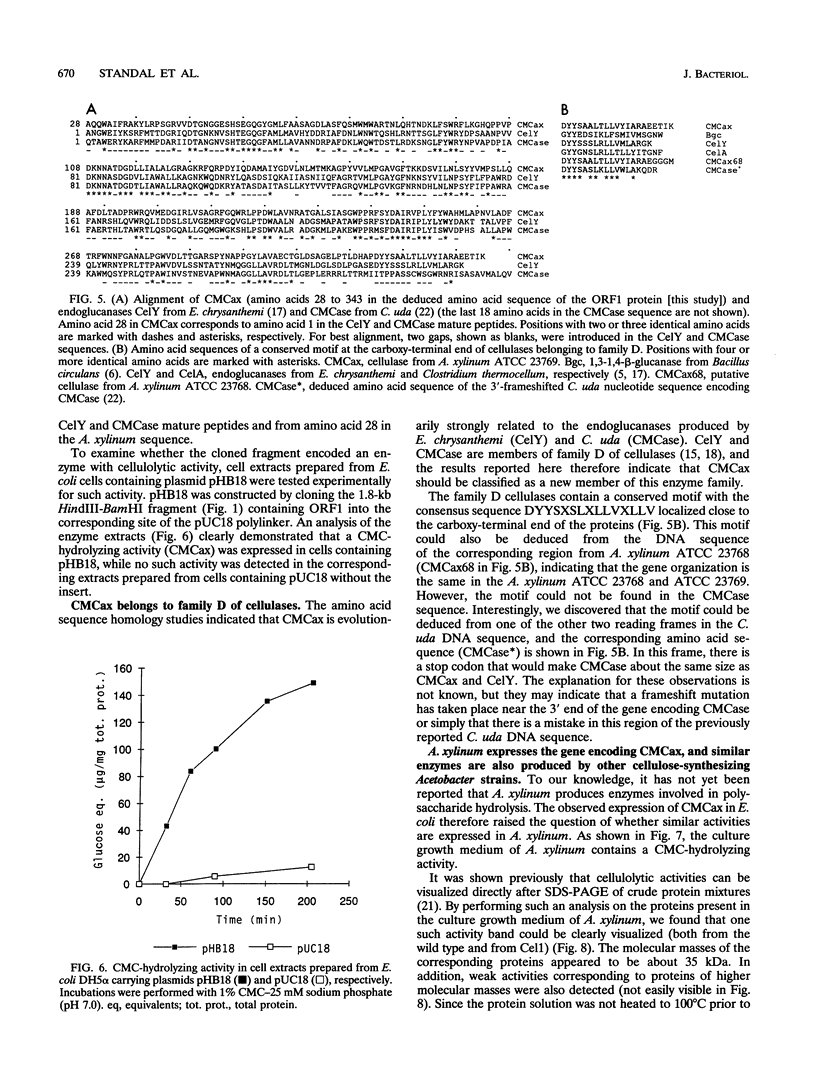
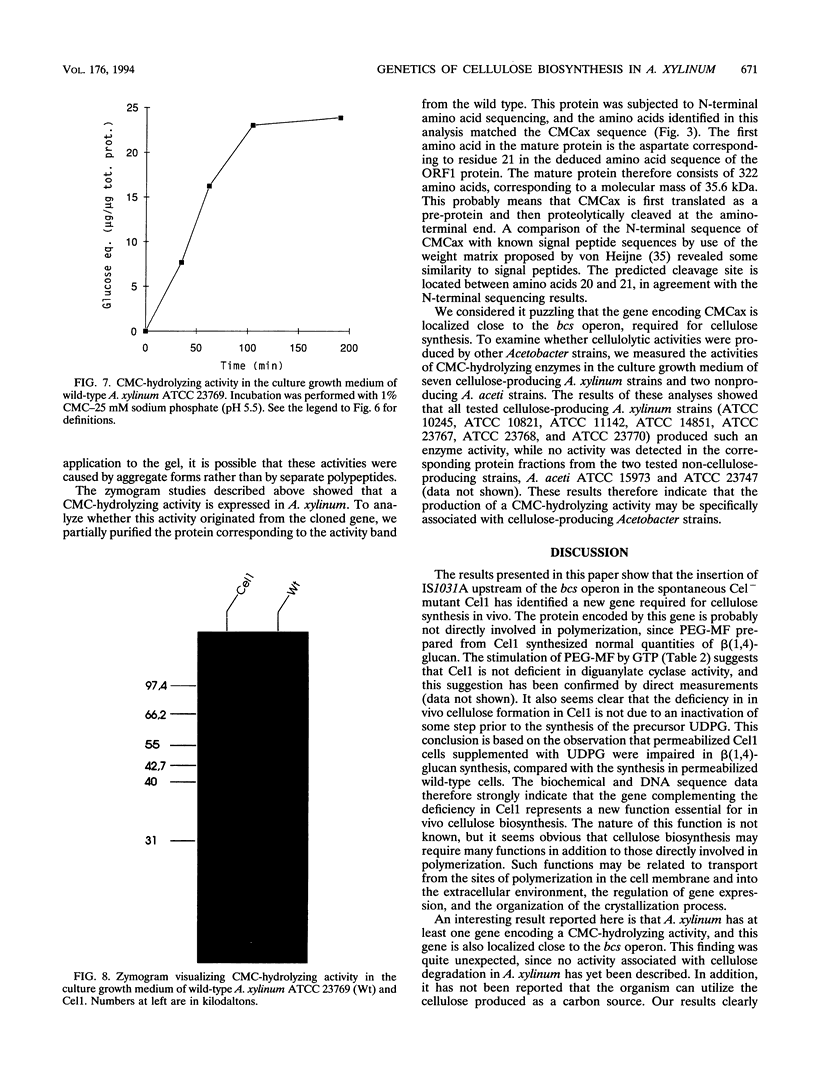
Recently, it was shown that a cellulose-negative mutant (Cel1) of Acetobacter xylinum ATCC 23769 carried an insertion of an indigenous transposable element (IS1031A) about 500 bp upstream of the bcs operon, required for cellulose synthesis. Here we show that Cel1 can be complemented by wild-type DNA covering the insertion point. Nucleotide sequencing of this region revealed the presence of two open reading frames, ORF1 and ORF2. ORF2, which is disrupted by the IS1031A insertion in Cel1, potentially encodes the complementing function. ORF1 encodes a protein (CMCax) with significant homology to previously described endoglucanases. A cloned DNA fragment containing ORF1 expressed a carboxymethyl cellulose-hydrolyzing activity in Escherichia coli. In A. xylinum, CMCax is secreted into the culture growth medium. The CMCax mature protein consists of 322 amino acids and has a molecular mass of 35.6 kDa.
Full text
PDF





Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Aloni Y., Delmer D. P., Benziman M. Achievement of high rates of in vitro synthesis of 1,4-beta-D-glucan: activation by cooperative interaction of the Acetobacter xylinum enzyme system with GTP, polyethylene glycol, and a protein factor. Proc Natl Acad Sci U S A. 1982 Nov;79(21):6448–6452. doi: 10.1073/pnas.79.21.6448. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Altschul S. F., Gish W., Miller W., Myers E. W., Lipman D. J. Basic local alignment search tool. J Mol Biol. 1990 Oct 5;215(3):403–410. doi: 10.1016/S0022-2836(05)80360-2. [DOI] [PubMed] [Google Scholar]
- Becker A., Kleickmann A., Arnold W., Pühler A. Analysis of the Rhizobium meliloti exoH/exoK/exoL fragment: ExoK shows homology to excreted endo-beta-1,3-1,4-glucanases and ExoH resembles membrane proteins. Mol Gen Genet. 1993 Apr;238(1-2):145–154. doi: 10.1007/BF00279541. [DOI] [PubMed] [Google Scholar]
- Bueno A., Vazquez de Aldana C. R., Correa J., del Rey F. Nucleotide sequence of a 1,3-1,4-beta-glucanase-encoding gene in Bacillus circulans WL-12. Nucleic Acids Res. 1990 Jul 25;18(14):4248–4248. doi: 10.1093/nar/18.14.4248. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bureau T. E., Brown R. M. In vitro synthesis of cellulose II from a cytoplasmic membrane fraction of Acetobacter xylinum. Proc Natl Acad Sci U S A. 1987 Oct;84(20):6985–6989. doi: 10.1073/pnas.84.20.6985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Béguin P., Cornet P., Aubert J. P. Sequence of a cellulase gene of the thermophilic bacterium Clostridium thermocellum. J Bacteriol. 1985 Apr;162(1):102–105. doi: 10.1128/jb.162.1.102-105.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Coucheron D. H. A family of IS1031 elements in the genome of Acetobacter xylinum: nucleotide sequences and strain distribution. Mol Microbiol. 1993 Jul;9(1):211–218. doi: 10.1111/j.1365-2958.1993.tb01682.x. [DOI] [PubMed] [Google Scholar]
- Coucheron D. H. An Acetobacter xylinum insertion sequence element associated with inactivation of cellulose production. J Bacteriol. 1991 Sep;173(18):5723–5731. doi: 10.1128/jb.173.18.5723-5731.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Devereux J., Haeberli P., Smithies O. A comprehensive set of sequence analysis programs for the VAX. Nucleic Acids Res. 1984 Jan 11;12(1 Pt 1):387–395. doi: 10.1093/nar/12.1part1.387. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Durland R. H., Toukdarian A., Fang F., Helinski D. R. Mutations in the trfA replication gene of the broad-host-range plasmid RK2 result in elevated plasmid copy numbers. J Bacteriol. 1990 Jul;172(7):3859–3867. doi: 10.1128/jb.172.7.3859-3867.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fellay R., Krisch H. M., Prentki P., Frey J. Omegon-Km: a transposable element designed for in vivo insertional mutagenesis and cloning of genes in gram-negative bacteria. Gene. 1989;76(2):215–226. doi: 10.1016/0378-1119(89)90162-5. [DOI] [PubMed] [Google Scholar]
- Fukaya M., Tayama K., Tamaki T., Tagami H., Okumura H., Kawamura Y., Beppu T. Cloning of the Membrane-Bound Aldehyde Dehydrogenase Gene of Acetobacter polyoxogenes and Improvement of Acetic Acid Production by Use of the Cloned Gene. Appl Environ Microbiol. 1989 Jan;55(1):171–176. doi: 10.1128/aem.55.1.171-176.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gilkes N. R., Henrissat B., Kilburn D. G., Miller R. C., Jr, Warren R. A. Domains in microbial beta-1, 4-glycanases: sequence conservation, function, and enzyme families. Microbiol Rev. 1991 Jun;55(2):303–315. doi: 10.1128/mr.55.2.303-315.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guiseppi A., Aymeric J. L., Cami B., Barras F., Creuzet N. Sequence analysis of the cellulase-encoding celY gene of Erwinia chrysanthemi: a possible case of interspecies gene transfer. Gene. 1991 Sep 30;106(1):109–114. doi: 10.1016/0378-1119(91)90573-t. [DOI] [PubMed] [Google Scholar]
- HESTRIN S., SCHRAMM M. Synthesis of cellulose by Acetobacter xylinum. II. Preparation of freeze-dried cells capable of polymerizing glucose to cellulose. Biochem J. 1954 Oct;58(2):345–352. doi: 10.1042/bj0580345. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Henrissat B., Claeyssens M., Tomme P., Lemesle L., Mornon J. P. Cellulase families revealed by hydrophobic cluster analysis. Gene. 1989 Sep 1;81(1):83–95. doi: 10.1016/0378-1119(89)90339-9. [DOI] [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Morag E., Bayer E. A., Lamed R. Relationship of cellulosomal and noncellulosomal xylanases of Clostridium thermocellum to cellulose-degrading enzymes. J Bacteriol. 1990 Oct;172(10):6098–6105. doi: 10.1128/jb.172.10.6098-6105.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Norrander J., Kempe T., Messing J. Construction of improved M13 vectors using oligodeoxynucleotide-directed mutagenesis. Gene. 1983 Dec;26(1):101–106. doi: 10.1016/0378-1119(83)90040-9. [DOI] [PubMed] [Google Scholar]
- Ross P., Mayer R., Benziman M. Cellulose biosynthesis and function in bacteria. Microbiol Rev. 1991 Mar;55(1):35–58. doi: 10.1128/mr.55.1.35-58.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sanger F., Nicklen S., Coulson A. R. DNA sequencing with chain-terminating inhibitors. Proc Natl Acad Sci U S A. 1977 Dec;74(12):5463–5467. doi: 10.1073/pnas.74.12.5463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saxena I. M., Lin F. C., Brown R. M., Jr Cloning and sequencing of the cellulose synthase catalytic subunit gene of Acetobacter xylinum. Plant Mol Biol. 1990 Nov;15(5):673–683. doi: 10.1007/BF00016118. [DOI] [PubMed] [Google Scholar]
- Saxena I. M., Lin F. C., Brown R. M., Jr Identification of a new gene in an operon for cellulose biosynthesis in Acetobacter xylinum. Plant Mol Biol. 1991 Jun;16(6):947–954. doi: 10.1007/BF00016067. [DOI] [PubMed] [Google Scholar]
- Valla S., Coucheron D. H., Fjaervik E., Kjosbakken J., Weinhouse H., Ross P., Amikam D., Benziman M. Cloning of a gene involved in cellulose biosynthesis in Acetobacter xylinum: complementation of cellulose-negative mutants by the UDPG pyrophosphorylase structural gene. Mol Gen Genet. 1989 May;217(1):26–30. doi: 10.1007/BF00330938. [DOI] [PubMed] [Google Scholar]
- Valla S., Coucheron D. H., Kjosbakken J. Conjugative transfer of the naturally occurring plasmids of Acetobacter xylinum by IncP-plasmid-mediated mobilization. J Bacteriol. 1986 Jan;165(1):336–339. doi: 10.1128/jb.165.1.336-339.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wong H. C., Fear A. L., Calhoon R. D., Eichinger G. H., Mayer R., Amikam D., Benziman M., Gelfand D. H., Meade J. H., Emerick A. W. Genetic organization of the cellulose synthase operon in Acetobacter xylinum. Proc Natl Acad Sci U S A. 1990 Oct;87(20):8130–8134. doi: 10.1073/pnas.87.20.8130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- von Heijne G. A new method for predicting signal sequence cleavage sites. Nucleic Acids Res. 1986 Jun 11;14(11):4683–4690. doi: 10.1093/nar/14.11.4683. [DOI] [PMC free article] [PubMed] [Google Scholar]





