Abstract
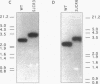
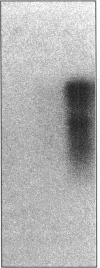
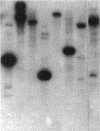
A new glutamine synthetase gene, glnN, which encodes a polypeptide of 724 amino acid residues (M(r), 79,416), has been identified in the unicellular cyanobacterium Synechocystis sp. strain PCC 6803; this is the second gene that encodes a glutamine synthetase (GS) in this cyanobacterium. The functionality of this gene was evidenced by its ability to complement an Escherichia coli glnA mutant and to support Synechocystis growth in a strain whose glnA gene was inactivated by insertional mutagenesis. In this mutant (strain SJCR3), as well as in the wild-type strain, the second GS activity was subject to regulation by the nitrogen source, being strongly enhanced in nitrogen-free medium. Transcriptional fusion of a chloramphenicol acetyltransferase (cat) gene with the 5'-upstream region of glnN suggested that synthesis of the second Synechocystis GS is regulated at the transcriptional level. Furthermore, the level of glnN mRNA, a transcript of about 2,300 bases, was found to be strongly increased in nitrogen-free medium. The glnN product is similar to the GS subunits of Bacteroides fragilis and Butyrivibrio fibrisolvens, two obligate anaerobic bacteria whose GSs are markedly different from other prokaryotic and eukaryotic GSs. However, significant similarity is evident in the five regions which are homologous in all of the GSs so far described. The new GS gene was also found in other cyanobacteria but not in N2-fixing filamentous species.
Full text
PDF





Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Almassy R. J., Janson C. A., Hamlin R., Xuong N. H., Eisenberg D. Novel subunit-subunit interactions in the structure of glutamine synthetase. 1986 Sep 25-Oct 1Nature. 323(6086):304–309. doi: 10.1038/323304a0. [DOI] [PubMed] [Google Scholar]
- Behrmann I., Hillemann D., Pühler A., Strauch E., Wohlleben W. Overexpression of a Streptomyces viridochromogenes gene (glnII) encoding a glutamine synthetase similar to those of eucaryotes confers resistance against the antibiotic phosphinothricyl-alanyl-alanine. J Bacteriol. 1990 Sep;172(9):5326–5334. doi: 10.1128/jb.172.9.5326-5334.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bradford M. M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem. 1976 May 7;72:248–254. doi: 10.1016/0003-2697(76)90527-3. [DOI] [PubMed] [Google Scholar]
- Cai Y. P., Wolk C. P. Use of a conditionally lethal gene in Anabaena sp. strain PCC 7120 to select for double recombinants and to entrap insertion sequences. J Bacteriol. 1990 Jun;172(6):3138–3145. doi: 10.1128/jb.172.6.3138-3145.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chiurazzi M., Meza R., Lara M., Lahm A., Defez R., Iaccarino M., Espín G. The Rhizobium leguminosarum biovar phaseoli glnT gene, encoding glutamine synthetase III. Gene. 1992 Sep 21;119(1):1–8. doi: 10.1016/0378-1119(92)90060-3. [DOI] [PubMed] [Google Scholar]
- Devereux J., Haeberli P., Smithies O. A comprehensive set of sequence analysis programs for the VAX. Nucleic Acids Res. 1984 Jan 11;12(1 Pt 1):387–395. doi: 10.1093/nar/12.1part1.387. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Elhai J., Wolk C. P. A versatile class of positive-selection vectors based on the nonviability of palindrome-containing plasmids that allows cloning into long polylinkers. Gene. 1988 Aug 15;68(1):119–138. doi: 10.1016/0378-1119(88)90605-1. [DOI] [PubMed] [Google Scholar]
- Elmorjani K., Liotenberg S., Houmard J., de Marsac N. T. Molecular characterization of the gene encoding glutamine synthetase in the cyanobacterium Calothrix sp. PCC 7601. Biochem Biophys Res Commun. 1992 Dec 30;189(3):1296–1302. doi: 10.1016/0006-291x(92)90214-6. [DOI] [PubMed] [Google Scholar]
- Espin G., Moreno S., Wild M., Meza R., Iaccarino M. A previously unrecognized glutamine synthetase expressed in Klebsiella pneumoniae from the glnT locus of Rhizobium leguminosarum. Mol Gen Genet. 1990 Sep;223(3):513–516. doi: 10.1007/BF00264461. [DOI] [PubMed] [Google Scholar]
- Ferino F., Chauvat F. A promoter-probe vector-host system for the cyanobacterium, Synechocystis PCC6803. Gene. 1989 Dec 14;84(2):257–266. doi: 10.1016/0378-1119(89)90499-x. [DOI] [PubMed] [Google Scholar]
- Fisher R., Tuli R., Haselkorn R. A cloned cyanobacterial gene for glutamine synthetase functions in Escherichia coli, but the enzyme is not adenylylated. Proc Natl Acad Sci U S A. 1981 Jun;78(6):3393–3397. doi: 10.1073/pnas.78.6.3393. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goodman H. J., Woods D. R. Cloning and nucleotide sequence of the Butyrivibrio fibrisolvens gene encoding a type III glutamine synthetase. J Gen Microbiol. 1993 Jul;139(7):1487–1493. doi: 10.1099/00221287-139-7-1487. [DOI] [PubMed] [Google Scholar]
- Hill R. T., Parker J. R., Goodman H. J., Jones D. T., Woods D. R. Molecular analysis of a novel glutamine synthetase of the anaerobe Bacteroides fragilis. J Gen Microbiol. 1989 Dec;135(12):3271–3279. doi: 10.1099/00221287-135-12-3271. [DOI] [PubMed] [Google Scholar]
- Markwell M. A., Haas S. M., Bieber L. L., Tolbert N. E. A modification of the Lowry procedure to simplify protein determination in membrane and lipoprotein samples. Anal Biochem. 1978 Jun 15;87(1):206–210. doi: 10.1016/0003-2697(78)90586-9. [DOI] [PubMed] [Google Scholar]
- Marqués S., Florencio F. J., Candau P. Ammonia assimilating enzymes from cyanobacteria: in situ and in vitro assay using high-performance liquid chromatography. Anal Biochem. 1989 Jul;180(1):152–157. doi: 10.1016/0003-2697(89)90104-8. [DOI] [PubMed] [Google Scholar]
- Meeks J. C., Wolk C. P., Lockau W., Schilling N., Shaffer P. W., Chien W. S. Pathways of assimilation of [13N]N2 and 13NH4+ by cyanobacteria with and without heterocysts. J Bacteriol. 1978 Apr;134(1):125–130. doi: 10.1128/jb.134.1.125-130.1978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mohamed A., Jansson C. Influence of light on accumulation of photosynthesis-specific transcripts in the cyanobacterium Synechocystis 6803. Plant Mol Biol. 1989 Dec;13(6):693–700. doi: 10.1007/BF00016024. [DOI] [PubMed] [Google Scholar]
- Mérida A., Candau P., Florencio F. J. In vitro reactivation of in vivo ammonium-inactivated glutamine synthetase from Synechocystis sp. PCC 6803. Biochem Biophys Res Commun. 1991 Dec 16;181(2):780–786. doi: 10.1016/0006-291x(91)91258-e. [DOI] [PubMed] [Google Scholar]
- Mérida A., Candau P., Florencio F. J. Regulation of glutamine synthetase activity in the unicellular cyanobacterium Synechocystis sp. strain PCC 6803 by the nitrogen source: effect of ammonium. J Bacteriol. 1991 Jul;173(13):4095–4100. doi: 10.1128/jb.173.13.4095-4100.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mérida A., Flores E., Florencio F. J. Regulation of Anabaena sp. strain PCC 7120 glutamine synthetase activity in a Synechocystis sp. strain PCC 6803 derivative strain bearing the Anabaena glnA gene and a mutated host glnA gene. J Bacteriol. 1992 Jan;174(2):650–654. doi: 10.1128/jb.174.2.650-654.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mérida A., Leurentop L., Candau P., Florencio F. J. Purification and properties of glutamine synthetases from the cyanobacteria Synechocystis sp. strain PCC 6803 and Calothrix sp. strain PCC 7601. J Bacteriol. 1990 Aug;172(8):4732–4735. doi: 10.1128/jb.172.8.4732-4735.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Orr J., Haselkorn R. Regulation of glutamine synthetase activity and synthesis in free-living and symbiotic Anabaena spp. J Bacteriol. 1982 Nov;152(2):626–635. doi: 10.1128/jb.152.2.626-635.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Orr J., Keefer L. M., Keim P., Nguyen T. D., Wellems T., Heinrikson R. L., Haselkorn R. Purification, physical characterization, and NH2-terminal sequence of glutamine synthetase from the cyanobacterium Anabaena 7120. J Biol Chem. 1981 Dec 25;256(24):13091–13098. [PubMed] [Google Scholar]
- Pesole G., Bozzetti M. P., Lanave C., Preparata G., Saccone C. Glutamine synthetase gene evolution: a good molecular clock. Proc Natl Acad Sci U S A. 1991 Jan 15;88(2):522–526. doi: 10.1073/pnas.88.2.522. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rawlings D. E., Jones W. A., O'Neill E. G., Woods D. R. Nucleotide sequence of the glutamine synthetase gene and its controlling region from the acidophilic autotroph Thiobacillus ferrooxidans. Gene. 1987;53(2-3):211–217. doi: 10.1016/0378-1119(87)90009-6. [DOI] [PubMed] [Google Scholar]
- Sanger F., Nicklen S., Coulson A. R. DNA sequencing with chain-terminating inhibitors. Proc Natl Acad Sci U S A. 1977 Dec;74(12):5463–5467. doi: 10.1073/pnas.74.12.5463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shaw W. V. Chloramphenicol acetyltransferase from chloramphenicol-resistant bacteria. Methods Enzymol. 1975;43:737–755. doi: 10.1016/0076-6879(75)43141-x. [DOI] [PubMed] [Google Scholar]
- Smith M. W., Feng D. F., Doolittle R. F. Evolution by acquisition: the case for horizontal gene transfers. Trends Biochem Sci. 1992 Dec;17(12):489–493. doi: 10.1016/0968-0004(92)90335-7. [DOI] [PubMed] [Google Scholar]
- Wagner S. J., Thomas S. P., Kaufman R. I., Nixon B. T., Stevens S. E., Jr The glnA gene of the cyanobacterium Agmenellum quadruplicatum PR-6 is nonessential for ammonium assimilation. J Bacteriol. 1993 Feb;175(3):604–612. doi: 10.1128/jb.175.3.604-612.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yamashita M. M., Almassy R. J., Janson C. A., Cascio D., Eisenberg D. Refined atomic model of glutamine synthetase at 3.5 A resolution. J Biol Chem. 1989 Oct 25;264(30):17681–17690. doi: 10.2210/pdb2gls/pdb. [DOI] [PubMed] [Google Scholar]
- de Bruijn F. J., Rossbach S., Schneider M., Ratet P., Messmer S., Szeto W. W., Ausubel F. M., Schell J. Rhizobium meliloti 1021 has three differentially regulated loci involved in glutamine biosynthesis, none of which is essential for symbiotic nitrogen fixation. J Bacteriol. 1989 Mar;171(3):1673–1682. doi: 10.1128/jb.171.3.1673-1682.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]