Abstract
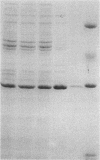
A clone of Yersinia pseudotuberculosis DNA carrying the ascA gene was constructed, and the corresponding protein was successfully overexpressed in Escherichia coli. A protocol consisting of DEAE-cellulose and Sephadex G-100 column chromatography was developed and led to a nearly homogeneous purification of the ascA product. Initial characterization showed that the ascA-encoded protein is actually the alpha-D-glucose-1-phosphate cytidylyltransferase which catalyzes the first step of the biosynthesis of CDP-ascarylose (CDP-3,6-dideoxy-L-arabino-hexose), converting alpha-D-glucose-1-phosphate to CDP-D-glucose. In contrast to early studies suggesting that this enzyme was a monomeric protein of 111 kDa, the purified cytidylyltransferase from Y. pseudotuberculosis was found to consist of four identical subunits, each with a molecular mass of 29 kDa. This assignment is supported by the fact that the ascA gene, as a part of the ascarylose biosynthetic cluster, exhibits high sequence homology with other nucleotidylyltransferases, and its product shows high cytidylyltransferase activity. Subsequent amino acid comparison with other known nucleotidylyltransferases has allowed a definition of the important active-site residues within this essential catalyst. These comparisons have also afforded the inclusion of the cytidylyltransferase into the mechanistic convergence displayed by this fundamental class of enzyme.
Full text
PDF








Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Andrews P. Estimation of the molecular weights of proteins by Sephadex gel-filtration. Biochem J. 1964 May;91(2):222–233. doi: 10.1042/bj0910222. [DOI] [PMC free article] [PubMed] [Google Scholar]
- BERNSTEIN R. L., ROBBINS P. W. CONTROL ASPECTS OF URIDINE 5'-DIPHOSPHATE GLUCOSE AND THYMIDINE 5'-DIPHOSPHATE GLUCOSE SYNTHESIS BY MICROBIAL ENZYMES. J Biol Chem. 1965 Jan;240:391–397. [PubMed] [Google Scholar]
- Baecker P. A., Furlong C. E., Preiss J. Biosynthesis of bacterial glycogen. Primary structure of Escherichia coli ADP-glucose synthetase as deduced from the nucleotide sequence of the glg C gene. J Biol Chem. 1983 Apr 25;258(8):5084–5088. [PubMed] [Google Scholar]
- Birnboim H. C., Doly J. A rapid alkaline extraction procedure for screening recombinant plasmid DNA. Nucleic Acids Res. 1979 Nov 24;7(6):1513–1523. doi: 10.1093/nar/7.6.1513. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brede G., Fjaervik E., Valla S. Nucleotide sequence and expression analysis of the Acetobacter xylinum uridine diphosphoglucose pyrophosphorylase gene. J Bacteriol. 1991 Nov;173(21):7042–7045. doi: 10.1128/jb.173.21.7042-7045.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brutlag D. L., Dautricourt J. P., Maulik S., Relph J. Improved sensitivity of biological sequence database searches. Comput Appl Biosci. 1990 Jul;6(3):237–245. doi: 10.1093/bioinformatics/6.3.237. [DOI] [PubMed] [Google Scholar]
- Darzins A., Frantz B., Vanags R. I., Chakrabarty A. M. Nucleotide sequence analysis of the phosphomannose isomerase gene (pmi) of Pseudomonas aeruginosa and comparison with the corresponding Escherichia coli gene manA. Gene. 1986;42(3):293–302. doi: 10.1016/0378-1119(86)90233-7. [DOI] [PubMed] [Google Scholar]
- Devereux J., Haeberli P., Smithies O. A comprehensive set of sequence analysis programs for the VAX. Nucleic Acids Res. 1984 Jan 11;12(1 Pt 1):387–395. doi: 10.1093/nar/12.1part1.387. [DOI] [PMC free article] [PubMed] [Google Scholar]
- GINSBURG V., O'BRIEN J., HALL C. W. Enzymic synthesis of cytidine 5'-diphosphate D-glucose. Biochem Biophys Res Commun. 1962 Feb 20;7:1–4. doi: 10.1016/0006-291x(62)90132-8. [DOI] [PubMed] [Google Scholar]
- HEATH E. C., ELBEIN A. D. The enzymatic synthesis of guanosine diphosphate colitose by a mutant strain of Escherichia coli. Proc Natl Acad Sci U S A. 1962 Jul 15;48:1209–1216. doi: 10.1073/pnas.48.7.1209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hanessian S. Deoxy sugars. Adv Carbohydr Chem Biochem. 1966;21:143–207. doi: 10.1016/s0096-5332(08)60317-3. [DOI] [PubMed] [Google Scholar]
- Haugen T. H., Preiss J. Biosynthesis of bacterial glycogen. The nature of the binding of substrates and effectors to ADP-glucose synthase. J Biol Chem. 1979 Jan 10;254(1):127–136. [PubMed] [Google Scholar]
- Henikoff S. Unidirectional digestion with exonuclease III creates targeted breakpoints for DNA sequencing. Gene. 1984 Jun;28(3):351–359. doi: 10.1016/0378-1119(84)90153-7. [DOI] [PubMed] [Google Scholar]
- Hill M. A., Kaufmann K., Otero J., Preiss J. Biosynthesis of bacterial glycogen. Mutagenesis of a catalytic site residue of ADP-glucose pyrophosphorylase from Escherichia coli. J Biol Chem. 1991 Jul 5;266(19):12455–12460. [PubMed] [Google Scholar]
- Ideguchi T., Hu C., Kim B. H., Nishise H., Yamashita J., Kakuno T. An open reading frame in the Rhodospirillum rubrum plasmid, pKY1, similar to algA, encoding the bifunctional enzyme phosphomannose isomerase-guanosine diphospho-D-mannose pyrophosphorylase (PMI-GMP). Biochim Biophys Acta. 1993 Mar 20;1172(3):329–331. doi: 10.1016/0167-4781(93)90224-2. [DOI] [PubMed] [Google Scholar]
- Jiang X. M., Neal B., Santiago F., Lee S. J., Romana L. K., Reeves P. R. Structure and sequence of the rfb (O antigen) gene cluster of Salmonella serovar typhimurium (strain LT2). Mol Microbiol. 1991 Mar;5(3):695–713. doi: 10.1111/j.1365-2958.1991.tb00741.x. [DOI] [PubMed] [Google Scholar]
- Kappel W. K., Preiss J. Biosynthesis of bacterial glycogen: purification and characterization of ADPglucose pyrophosphorylase with modified regulatory properties from Escherichia coli B mutant CL1136-504. Arch Biochem Biophys. 1981 Jun;209(1):15–28. doi: 10.1016/0003-9861(81)90252-6. [DOI] [PubMed] [Google Scholar]
- Katsube T., Kazuta Y., Tanizawa K., Fukui T. Expression in Escherichia coli of UDP-glucose pyrophosphorylase cDNA from potato tuber and functional assessment of the five lysyl residues located at the substrate-binding site. Biochemistry. 1991 Sep 3;30(35):8546–8551. doi: 10.1021/bi00099a008. [DOI] [PubMed] [Google Scholar]
- Kazuta Y., Omura Y., Tagaya M., Nakano K., Fukui T. Identification of lysyl residues located at the substrate-binding site in UDP-glucose pyrophosphorylase from potato tuber: affinity labeling with uridine di- and triphosphopyridoxals. Biochemistry. 1991 Sep 3;30(35):8541–8545. doi: 10.1021/bi00099a007. [DOI] [PubMed] [Google Scholar]
- Kimata K., Suzuki S. Studies on cytidine diphosphate glucose pyrophosphorylase and related enzymes of Azotobacter vinelandii. J Biol Chem. 1966 Mar 10;241(5):1099–1113. [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Leung P. S., Preiss J. Biosynthesis of bacterial glycogen: primary structure of Salmonella typhimurium ADPglucose synthetase as deduced from the nucleotide sequence of the glgC gene. J Bacteriol. 1987 Sep;169(9):4355–4360. doi: 10.1128/jb.169.9.4355-4360.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lindquist L., Kaiser R., Reeves P. R., Lindberg A. A. Purification, characterization and HPLC assay of Salmonella glucose-1-phosphate thymidylyl-transferase from the cloned rfbA gene. Eur J Biochem. 1993 Feb 1;211(3):763–770. doi: 10.1111/j.1432-1033.1993.tb17607.x. [DOI] [PubMed] [Google Scholar]
- Lo S. F., Miller V. P., Lei Y., Thorson J. S., Liu H. W., Schottel J. L. CDP-6-deoxy-delta 3,4-glucoseen reductase from Yersinia pseudotuberculosis: enzyme purification and characterization of the cloned gene. J Bacteriol. 1994 Jan;176(2):460–468. doi: 10.1128/jb.176.2.460-468.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lüderitz O., Staub A. M., Westphal O. Immunochemistry of O and R antigens of Salmonella and related Enterobacteriaceae. Bacteriol Rev. 1966 Mar;30(1):192–255. doi: 10.1128/br.30.1.192-255.1966. [DOI] [PMC free article] [PubMed] [Google Scholar]
- MAYER R. M., GINSBURG V. PURIFICATION AND PROPERTIES OF CYTIDINE DIPHOSPHATE D-GLUCOSE PYROPHOSPHORYLASE FROM SALMONELLA PARATYPHI A. J Biol Chem. 1965 May;240:1900–1904. [PubMed] [Google Scholar]
- MELO A., GLASER L. THE NUCLEOTIDE SPECIFICITY AND FEEDBACK CONTROL OF THYMIDINE DIPHOSPHATE D-GLUCOSE PYROPHOSPHORYLASE. J Biol Chem. 1965 Jan;240:398–405. [PubMed] [Google Scholar]
- Matsuhashi S., Matsuhashi M., Brown J. G., Strominger J. L. Enzymatic synthesis of cytidine diphosphate 3,6-dideoxyhexoses. 3. Cytidine diphosphate D-glucose oxidoreductase. J Biol Chem. 1966 Sep 25;241(18):4283–4287. [PubMed] [Google Scholar]
- Matsuhashi S., Matsuhashi M., Strominger J. L. Enzymatic synthesis of cytidine diphosphate 3,6-dideoxyhexoses. I. Over-all reactions. J Biol Chem. 1966 Sep 25;241(18):4267–4274. [PubMed] [Google Scholar]
- Nikaido H., Nikaido K. Enzymatic synthesis of cytidine diphosphate 3,6-dideoxyhexoses in Salmonella. J Biol Chem. 1966 Mar 25;241(6):1376–1385. [PubMed] [Google Scholar]
- Parsons T. F., Preiss J. Biosynthesis of bacterial glycogen. Incorporation of pyridoxal phosphate into the allosteric activator site and an ADP-glucose-protected pyridoxal phosphate binding site of Escherichia coli B ADP-glucose synthase. J Biol Chem. 1978 Sep 10;253(17):6197–6202. [PubMed] [Google Scholar]
- Pissowotzki K., Mansouri K., Piepersberg W. Genetics of streptomycin production in Streptomyces griseus: molecular structure and putative function of genes strELMB2N. Mol Gen Genet. 1991 Dec;231(1):113–123. doi: 10.1007/BF00293829. [DOI] [PubMed] [Google Scholar]
- Preiss J., Romeo T. Physiology, biochemistry and genetics of bacterial glycogen synthesis. Adv Microb Physiol. 1989;30:183–238. doi: 10.1016/s0065-2911(08)60113-7. [DOI] [PubMed] [Google Scholar]
- Raetz C. R. Biochemistry of endotoxins. Annu Rev Biochem. 1990;59:129–170. doi: 10.1146/annurev.bi.59.070190.001021. [DOI] [PubMed] [Google Scholar]
- Rick P. D., Wolski S., Barr K., Ward S., Ramsay-Sharer L. Accumulation of a lipid-linked intermediate involved in enterobacterial common antigen synthesis in Salmonella typhimurium mutants lacking dTDP-glucose pyrophosphorylase. J Bacteriol. 1988 Sep;170(9):4008–4014. doi: 10.1128/jb.170.9.4008-4014.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rubenstein P. A., Strominger J. L. Enzymatic synthesis of cytidine diphosphate 3,6-dideoxyhexosis. IX. Purification and properties of the cytidine diphosphate-D-glucose pyrophosphorylase from Pasteurella pseudotuberculosis. type V. J Biol Chem. 1974 Jun 25;249(12):3789–3796. [PubMed] [Google Scholar]
- Sanger F., Nicklen S., Coulson A. R. DNA sequencing with chain-terminating inhibitors. Proc Natl Acad Sci U S A. 1977 Dec;74(12):5463–5467. doi: 10.1073/pnas.74.12.5463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saraste M., Sibbald P. R., Wittinghofer A. The P-loop--a common motif in ATP- and GTP-binding proteins. Trends Biochem Sci. 1990 Nov;15(11):430–434. doi: 10.1016/0968-0004(90)90281-f. [DOI] [PubMed] [Google Scholar]
- Shinabarger D., Berry A., May T. B., Rothmel R., Fialho A., Chakrabarty A. M. Purification and characterization of phosphomannose isomerase-guanosine diphospho-D-mannose pyrophosphorylase. A bifunctional enzyme in the alginate biosynthetic pathway of Pseudomonas aeruginosa. J Biol Chem. 1991 Feb 5;266(4):2080–2088. [PubMed] [Google Scholar]
- Smith-White B. J., Preiss J. Comparison of proteins of ADP-glucose pyrophosphorylase from diverse sources. J Mol Evol. 1992 May;34(5):449–464. doi: 10.1007/BF00162999. [DOI] [PubMed] [Google Scholar]
- Stevenson G., Lee S. J., Romana L. K., Reeves P. R. The cps gene cluster of Salmonella strain LT2 includes a second mannose pathway: sequence of two genes and relationship to genes in the rfb gene cluster. Mol Gen Genet. 1991 Jun;227(2):173–180. doi: 10.1007/BF00259668. [DOI] [PubMed] [Google Scholar]
- Sá-Correia I., Darzins A., Wang S. K., Berry A., Chakrabarty A. M. Alginate biosynthetic enzymes in mucoid and nonmucoid Pseudomonas aeruginosa: overproduction of phosphomannose isomerase, phosphomannomutase, and GDP-mannose pyrophosphorylase by overexpression of the phosphomannose isomerase (pmi) gene. J Bacteriol. 1987 Jul;169(7):3224–3231. doi: 10.1128/jb.169.7.3224-3231.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ueguchi C., Ito K. Multicopy suppression: an approach to understanding intracellular functioning of the protein export system. J Bacteriol. 1992 Mar;174(5):1454–1461. doi: 10.1128/jb.174.5.1454-1461.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vesterberg O. Staining of protein zones after isoelectric focusing in polyacrylamide gels. Biochim Biophys Acta. 1971 Aug 27;243(2):345–348. doi: 10.1016/0005-2795(71)90094-8. [DOI] [PubMed] [Google Scholar]
- Walker J. E., Saraste M., Runswick M. J., Gay N. J. Distantly related sequences in the alpha- and beta-subunits of ATP synthase, myosin, kinases and other ATP-requiring enzymes and a common nucleotide binding fold. EMBO J. 1982;1(8):945–951. doi: 10.1002/j.1460-2075.1982.tb01276.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weigel T. M., Liu L. D., Liu H. W. Mechanistic studies of the biosynthesis of 3,6-dideoxyhexoses in Yersinia pseudotuberculosis: purification and characterization of CDP-4-keto-6-deoxy-D-glucose-3-dehydrase. Biochemistry. 1992 Feb 25;31(7):2129–2139. doi: 10.1021/bi00122a034. [DOI] [PubMed] [Google Scholar]
- Yu Y., Russell R. N., Thorson J. S., Liu L. D., Liu H. W. Mechanistic studies of the biosynthesis of 3,6-dideoxyhexoses in Yersinia pseudotuberculosis. Purification and stereochemical analysis of CDP-D-glucose oxidoreductase. J Biol Chem. 1992 Mar 25;267(9):5868–5875. [PubMed] [Google Scholar]
- du Jardin P., Berhin A. Isolation and sequence analysis of a cDNA clone encoding a subunit of the ADP-glucose pyrophosphorylase of potato tuber amyloplasts. Plant Mol Biol. 1991 Feb;16(2):349–351. doi: 10.1007/BF00020568. [DOI] [PubMed] [Google Scholar]