Abstract
High risk human papillomaviruses (HPVs) known to be closely associated with cervical cancer, such as HPV16 and HPV18, have the potential to immortalize human epithelial cells in culture. Four lines of HPV-transfected keratinocytes were analyzed by comparative genomic hybridization at different time points after transfection. A number of chromosomal imbalances was found to be highly characteristic for the cultures progressing toward immortality. Whereas several of these were new and previously not found as recurrent aberrations in cervical tumors, some were identical to chromosomal changes observed during cervical carcinogenesis. The data put new emphasis on the studied cell system as a relevant model for HPV-induced pathogenesis.
Specific types of human papillomavirus (HPV) are known to play a key role in the etiology of genital tumors. In particular, HPV16 and HPV18 are regarded as key factors in the origin of cervical cancer (1). In the majority of dysplastic lesions of the cervix, one of these two virus types is found. Invasive cervical cancer is preceded by premalignant lesions [cervical intraepithelial neoplasia (CIN)] ranging from mild and moderate dysplasia (CIN-I and -II) to severe dysplasia (CIN-III), also termed “carcinoma in situ.” Progression from a premalignant lesion to invasive cancer usually requires several years, if not decades. There is a large body of evidence for a multistep nature of cervical carcinogenesis. In addition to the viral genes E6 and E7, which are considered as factors necessary but not sufficient for oncogenesis, genetic mutations in specific genes within the host cell genome also are thought to contribute to malignant progression (2).
Oncogenic genital HPV types such as HPV16, HPV18, and HPV33 are capable of immortalizing human primary epithelial cells in culture (3, 4). This property is not shared by HPV6 or HPV11, which generally induce benign lesions only (5, 6). The immortalization represents the first step in the multistage process of HPV-induced malignant transformation. Although immortalization defines a phenotypic state of cells in culture, there is evidence suggesting that this state corresponds to low grade HPV-positive precancerous lesions of the cervix uteri (7). Cell culture model systems for HPV-associated tumorigenesis are therefore useful with respect to the analysis of cervical carcinogenesis. The viral proteins E6 and E7 contribute to immortalization by interfering with key regulatory proteins of the host cell, such as p53 and pRB, respectively (8, 9). In particular, the expression of the E6 protein, based on its interaction with p53, appears to be responsible for chromosomal instability in infected cells (10–12).
HPV-immortalized cells can acquire a malignant phenotype, e.g., after transfection with the ras oncogene, by a combination of γ-irradiation and long term passage in culture or even by long term passage alone. This has been used to establish a cell culture system for HPV-induced tumorigenicity, which may serve to delineate the sequence of events involved in cell transformation (13–18). For some of these cell lines, genetic analyses were conducted by complementation assays using somatic cell fusion experiments. It was shown that the immortal and the tumorigenic phenotype are both recessive and that they are independent from each other (17, 19). Studying transformed cell lines of different origin, Pereira–Smith and colleagues reported evidence for at least four different complementation groups for immortalization (20, 21). HPV-immortalized cells as well as cells derived from cervical cancers could be assigned to one such complementation group, suggesting that there is a common mechanism associated with HPV-induced immortalization (17, 19).
To identify genetic imbalances that are related to immortalization and possibly also to an early stage in the development of precancerous lesions, we analyzed HPV-transfected keratinocytes by the technique of comparative genomic hybridization (CGH) (22). For CGH, whole genomic tumor DNA is cohybridized with a differently labeled normal genomic DNA to normal metaphase chromosomes (22–24). Subsequent comparison of the hybridization signal intensities of the two DNA probes along the chromosomes allows the assessment of chromosomal imbalances; over- and underrepresented chromosomal regions in the tumor DNA are indicated by increased or decreased fluorescence ratio values. In this study, HPV16- or HPV18-immortalized keratinocyte cell lines from different origin were analyzed by CGH at different intervals after transfection.
MATERIALS AND METHODS
Cell Culture Conditions.
HPV-immortalized cells were established by transfection of human primary foreskin keratinocytes (from different donors) with the cloned complete HPV16 (25) or HPV18 (26) genome and subsequent selection for unlimited growth in culture (3). Primary or secondary cultures at near confluence were transfected with HPV16 or HPV18 DNA either by DNA–calcium phosphate precipitation (27) or the use of Lipofectamid (Life Technologies, Gaithersburg, MD). For lipofection, 50 μl of the polycationic lipid was mixed with 10 μg of DNA for transfection of a 100-mm culture dish. Cells were subcultured at a ratio of 1:2 whenever they had reached confluency. At each passage, 50% of the cells was either frozen in liquid nitrogen or was used for DNA extraction. DNA was extracted at four or five time points to assess the status of the viral DNA during the process of immortalization and to identify genetic changes that may be related to this phenotype.
Southern Blot Analysis.
Total DNA was isolated from cells by proteinase K digestion and phenol extraction. DNA was digested with restriction enzymes, electrophoretically separated on 0.7% agarose gels, and blotted onto nylon membranes (GeneScreenPlus, NEN). Hybridization was performed at 42°C in 50% formamide, 5× SSC, 50 mM sodium phosphate (pH 6.5), 5 × Denhardt’s solution, 0.1 mg/ml tRNA, and 1% SDS, with gel-purified, 32P-labeled HPV16 or HPV18 DNA (>5 × 108 cpm/μg) as probe. The filters were exposed to Kodak X-Omat AR films at −70°C with an intensifying screen.
Comparative Genomic Hybridization.
Genomic DNA extracted from each cell line at different time points was used as test DNA for comparative genomic hybridization. Preparation of metaphase chromosomes, labeling of DNA, and hybridization were performed as described (28). Metaphase chromosomes were prepared from female peripheral blood leukocytes, and control DNA was isolated from peripheral blood lymphocytes of a healthy male individual. Test and control DNA were labeled by nick-translation with biotin 16–dUTP and digoxigenin 11–dUTP, respectively. One microgram of test DNA, 1 μg of control DNA, and 80 μg of human Cot-1 DNA were coprecipitated in ethanol and resuspended in a 12-μl hybridization cocktail. After denaturation, probe DNA and Cot-1 DNA were allowed to preanneal for 25 min at 37°C to suppress signals from repeated sequences. Hybridization to denatured chromosomes was carried out for 3 days at 37°C. After posthybridization washes in 50% formamide/2× SSC at 42°C and in 0.1× SSC at 60°C, biotinylated test DNA was detected with avidin–fluorescein isothiocyanate and digoxigenin-labeled control DNA with mouse anti-digoxigenin antibody conjugated to tetramethylrhodamine B isothiocyanate. Chromosomes were counterstained with 4′, 6-diamidino-2-phenylindole, and specimens were mounted in antifade solution (Vectashield, Vector Laboratories).
Analysis of CGH.
For image acquisition, an epifluorescence microscope (Zeiss Axioplan) equipped with the Cytovision System (Applied Imaging, Sunderland, Tyne & Wear, UK) was used. Control of the camera, image acquisition, and CGH image analysis were performed by the system software (cytovision, Version 2.47). Our laboratory actively contributed to the establishment of the cytovision analysis procedure and extensively tested its performance using multiple tumor specimens. The sensitivity of the cytovision software in detecting chromosomal imbalances was proven to be equivalent to that of a previously developed, dedicated CGH software (29). Chromosomes were identified on inverted 4′, 6-diamidino-2-phenylindole images by the automated karyotyping feature integrated in the software. The ratio of fluorescein isothiocyanate/tetramethylrhodamine B isothiocyanate fluorescence intensities was calculated along each individual chromosome. The average ratio profile was calculated from 10 to 13 metaphase spreads and plotted next to chromosome ideograms. Chromosomal imbalances were detected on the basis of the ratio profile deviating from the balanced value (fluorescein isothiocyanate/tetramethylrhodamine B isothiocyanate = 1). The diagnostic thresholds for over- and underrepresentations were 1.25 and 0.75, respectively. These thresholds were established in previous CGH studies for the software application developed by Du Manoir and coworkers (29, 30) as well as for the cytovision system applied in this study. These thresholds corresponded to the values expected for trisomy or monosomy present in 50% of diploid cells. Chromosome regions were scored as imbalanced regions when the ratio profile either reached or exceeded the diagnostic thresholds. High copy number amplification was scored when a strong distinct hybridization signal could be easily detected by visual inspection and/or when the ratio of green to red fluorescence exceeded the value of 2.0.
The use of metaphase cells from a female donor did not allow us to obtain a CGH profile for the Y chromosome. However, because of its high content of heterochromatin (often resulting in gross ratio variations despite only small variations in the fluorescence intensity), this chromosome is often excluded from CGH analysis. The same applies to the centromeric regions. Other chromosomal regions have been recognized as being difficult in CGH analyses (29, 31). Of these, chromosome region 1p32-pter and chromosome 19 were excluded from the analysis for reasons specified elsewhere (28, 32). CGH data regarding these two regions were not scored in the results.
RESULTS
Establishing HPV16- and HPV18-Immortalized Keratinocyte Cell Lines.
Human primary foreskin keratinocytes were transfected with cloned HPV DNA and cultured with continual passages at low split ratios (1:2). Whereas mock-transfected cells senesced within eight passages, all HPV-transfected cultures showed a prolonged lifespan. Four of five cultures developed into cell lines, i.e., became immortalized. The cell lines are termed “HPK” (human papillomavirus-immortalized keratinocytes) I to V. Establishment of HPK IA and II has been reported (3, 33). HPK IV did not acquire an immortal phenotype and was therefore excluded from cytogenetic analysis. The other four lines could be cultured beyond 150 passages and are therefore considered immortal. HPK IA, II, and III were immortalized by HPV16 and cell line HPK V by HPV18.
Status of HPV–DNA in HPK Cells.
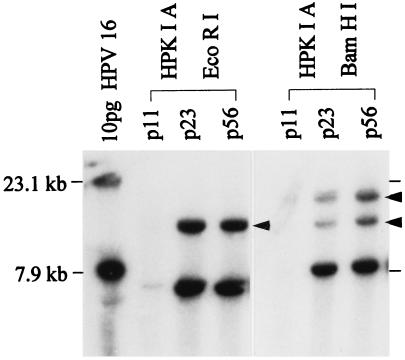
Transfected viral DNA could be detected by Southern blot analysis within the first two passages posttransfection. This was followed by several passages in which no viral DNA could be detected. Faint bands specific for viral DNA were first apparent in HPK IA and II-V cells at passage 11, 4, 7, 5, and 12, respectively (for an example, see Fig. 1). Beside the characteristic HPV16 restriction fragments obtained after digestion with a single- (BamHI, 7.9 kb) or double-cutting (EcoRI, 7.3 kb) enzyme, bands of higher molecular mass (>7.9 kb; see arrowheads in Fig. 1), indicative of integrated viral DNA, are clearly evident for HPKIA cells of passage 23. The signal intensity of this band pattern remained unaltered during further passaging (see p56), which is consistent with the fact that this cell line emerged by clonal expansion of a common progenitor cell (3, 18).
Figure 1.
Detection of HPV16 DNA in a keratinocyte cell culture (HPK IA) by Southern blot analysis at different time points after transfection. Ten picograms of HPV16 DNA excised from vector by BamHI digestion (7.9 kb) is included as a sensitivity marker for hybridization and represents approximately one viral copy per cell. Total genomic cell line DNA was digested with EcoRI or BamHI. The 7.3-kb EcoRI band and the 7.9-kb BamHI band are characteristic for cleavage of the full length HPV16 genome whereas high molecular mass EcoRI and BamHI bands (arrowheads) are indicative of viral integration.
CGH Analysis of HPK Cell Lines at Different Time Points After HPV Transfection.
The CGH results obtained from HPK IA-III and V at different time points, i.e., passage p0 to p72, after HPV16 or HPV18 transfection are presented in Table 1.
Table 1.
Chromosomal imbalances in four HPV-transfected keratinocyte cell lines at different time points after virus transfection
| HPKIA | p11 | none | ||||||||||||||||
| p17 | +5p | +5q | +8p | +8q | −10p | +11 | +16 | +20 | +21q | +22q | ||||||||
| p23 | −4 | +5q | −6q | +8q | +9 | −10p | +11 | +14q | +16 | +20 | ||||||||
| p56 | +3q | (−4)* | +5 | −6 | +8q | +9 | −10p | +11 | +14q | +15q | +16 | +20 | ||||||
| HPKII | p1 | none | ||||||||||||||||
| p6 | −10p | +12q | +14q | +16q | +20q | |||||||||||||
| p17 | −4q | +5 | +6p | −10p | +11 | +14q | −15 | +16 | +20 | +22q | −X | |||||||
| p48 | −1q | −4 | +5p | −6q | +7p | −7q | −10p | +11 | +12q | +14q | 15 h | +16 | +20 | +22q | −Xp | −Xq | ||
| HPKIII | p2 | none | ||||||||||||||||
| p14 | +3q | +8 | −X | |||||||||||||||
| p42 | +3q | −6q | −7q | +8 | +9q | −10 | +11 | +12q | +14q | +16 | +17 | +20 | −X | |||||
| p55 | +3q | +8 | +9q | +11q | +12q | +14q | +16 | +17 | +20q | +22q | −X | |||||||
| p72 | +3q | −4 | +5q | −6q | +7p | +8 | +9 | +11 | +12q | +14q | +16 | +17 | +20 | +22q | (−X)* | |||
| HPKV | p0 | none | ||||||||||||||||
| p12 | none | |||||||||||||||||
| p22 | −3p | −10p | +10q | +17q | ||||||||||||||
| p35 | −3p | +3q | −4 | −10p | +10q | +11q |
Even when only a portion of a chromosome arm was found imbalanced, the entire arm is listed. Details of the extension of an imbalanced region are presented in Fig. 2. p, passage upon virus transfection; h, high copy number amplification on chromosome band 15q23-q24; +, gained chromosome material [rev ish enh according to ISCN 1995 (62)]; −, lost chromosome material [rev ish dim according to ISCN 1995 (62)].
The CGH profile of these chromosomes shows a strong deviation from the balanced state and reaches the diagnostic threshold for deletion in only two (chromosome 4) and three (chromosome X) regions. However, the profile of the whole chromosome does not entirely exceeds this threshold, but, because this was the case in the previous time point analyzed, we report it here in parentheses.
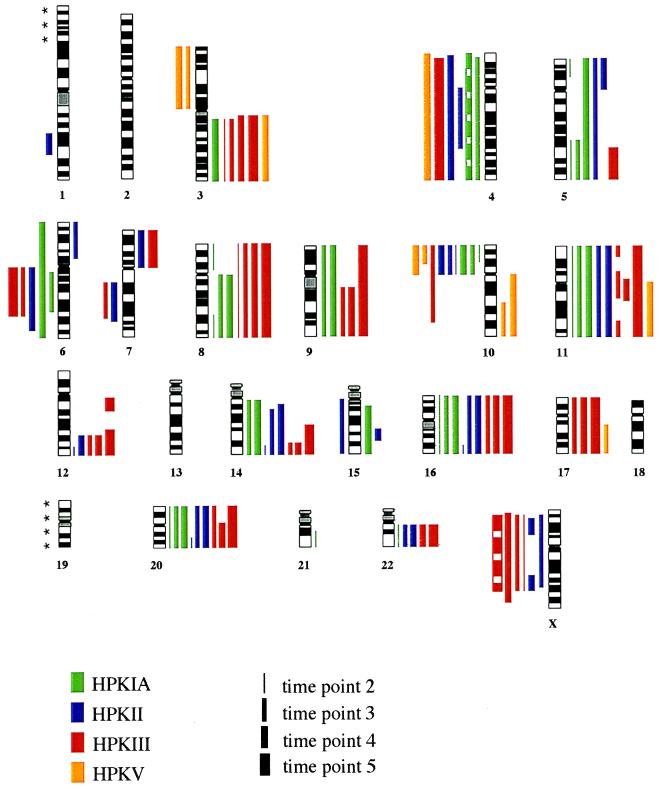
Of interest, the pattern of chromosomal imbalances found by CGH was very similar in the four cell lines investigated. Chromosomal gains or losses were not found in any of the four lines at the first time point after HPV transfection. p6 in HPKII was the earliest passage in which chromosomal changes were detected. The chromosomal imbalances detected in the analyzed cell lines at the later time points are summarized in Fig. 2 and Table 1. As documented in Fig. 2, the chromosomes or chromosomal regions most often involved in loss of material were chromosomes 4 and 10p (in all four cell lines) and chromosome 6q (in three cell lines) whereas gain of chromosomal material involved chromosome 11q (in four cell lines) and chromosomes 3q, 5q, 14q, 16, 20, and 22q (in three cell lines each). The CGH profiles of chromosomes 3, 4, 10, and 11 of each cell line at the different time points are shown in Fig. 3. It should be noted that the profile of chromosome 3q in HPK II deviates from the value indicating a balanced state but does not reach the diagnostic threshold. Therefore, this case was not scored imbalanced (see Fig. 2 and Table 1) although the analyzed culture of HPK II likely contains a subclone carrying the respective gain of chromosome 3q material.
Figure 2.
Summary of the chromosomal imbalances detected in four HPV-immortalized keratinocyte cell lines at different time points. Vertical lines on the left of chromosome ideograms indicate loss of genetic material; vertical lines on the right indicate gain of genetic material. Each line is presented by a color as indicated at the bottom. Chromosomal imbalances were not found at the first time point analyzed; imbalances found at the second, third, fourth, and fifth time points are drawn with lines of increasing thickness as indicated at the bottom. Interrupted lines indicate ratio values close to, but not reaching, the diagnostic threshold, possibly because of the presence of a subclone carrying this chromosomal imbalance (see text). ∗, The distal part of chromosome 1p and the chromosome 19 are labeled with asterisks because imbalances found in these two regions were not scored for reasons mentioned in Materials and Methods.
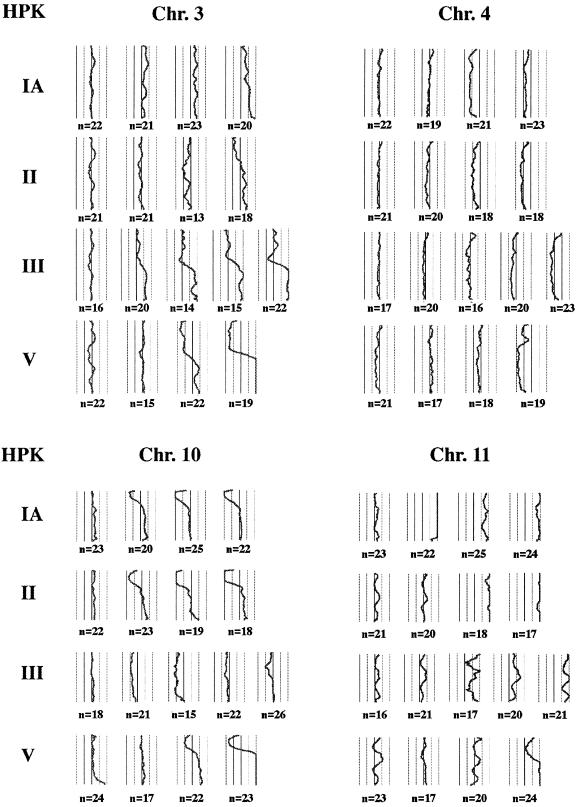
Figure 3.
CGH average ratio profiles of chromosomes 3, 4, 10, and 11 at the different time points analyzed for each of the four HPK cell lines. The ratios of fluorescein isothiocyanate/tetramethylrhodamine B isothiocyanate fluorescence are plotted along the chromosome ideograms. The central line indicates the intensity ratio of a balanced copy number (value of 1), and the thresholds for over- (value of 1.25) and underrepresentation (value of 0.75) are indicated by the adjacent right and left lines, respectively (the far right and left lines represent the values of 1.5 and 0.5, respectively). This figure is comprised of black and white prints of CGH profiles obtained with the cytovision CGH software package.
Chromosomal imbalances increase in number from the second to the last time point analyzed (Fig. 2). Furthermore, with the increase of passage, the CGH profiles indicate that gain or loss of chromosomal material deviates in most cases more strongly from the balanced state (Fig. 3). This could be representative for a continuous progression of a subclone of cells containing the genomic imbalance or for a successive increase (or decrease) of the copy number of a given chromosome region per cell.
In one instance, a chromosomal region was found lost at one time point but gained at a later point in the same cell line: In cell line HPK II, loss of material of chromosome 15 was observed at time point 3 (p17) whereas at p48 a high copy number amplification was found on the same chromosome within band 15q23-q24. However, this could be the result of an amplification originating from the remaining chromosome 15 homolog.
DISCUSSION
To the present day, reports about tumor-specific chromosomal aberrations in cervical carcinoma are limited (34), and no specific virus-associated landmark aberrations have been identified. The two largest cytogenetic studies of primary cervical carcinomas (35, 36) analyzed series of 43 and 150 cases. The most prevalent chromosomal imbalances were additional copies of chromosomes 3, 5, 19, and 20 as well as loss of chromosomes 1, 2, 4, 11, 14, 15, and 22. Furthermore, numerous structural aberrations were observed by chromosome banding analysis, with chromosomes 1, 11, and 17 being most frequently involved (35–39). In the present study of HPV-transfected keratinocytes, a number of characteristic genetic changes were identified that previously were not recognized in HPV-positive cervical cancer by using cytogenetic techniques. In particular, the imbalances found in all cell lines, loss of the short arm of chromosome 10 and gain of chromosome 11q, have not been described before as a recurrent genomic change in this tumor type. These aberrations appear early after HPV transfection. Chromosomes 4 and 10p are strong candidate regions for harboring putative suppressor gene(s), and 11q is a strong candidate region for proto-oncogene(s), playing a key role in immortalization of keratinocytes upon HPV transfection. Other studies have suggested a putative tumor suppressor gene on 11q functionally inactive in cervical cancer (40–42), so our model system seems to depict a different gene regarding chromosome 11.
Whereas some of the second most common genetic imbalances (three of four cell lines) have been reported—i.e., gain of chromosome arms 3q, 5q, and 20q, loss of chromosome arm 6q (35–37, 43)—gain of material from chromosome arms 14q and 22q, as well as from chromosome 16, was not yet identified. It should be noted that, although gain of material of chromosome 3q could be scored for only three cell lines (see Results), the fourth line HPK II probably also contains a subclone of cells with this genomic imbalance.
In a recent study, Heselmeyer et al. (44) described a correlation between genotype and phenotype in premalignant lesions and invasive carcinoma of the cervix by analyzing DNA that was isolated after microdissection of tissue sections. Chromosome 3q was found overrepresented in 9 of 10 invasive carcinoma samples, and detection of 3q gain by CGH depicted the transition from severe dysplasia to invasive carcinoma because this imbalance was found only in one (of 13) severe dysplasia. Of interest, we also found a high frequency of gain of chromosome 3q. The tumors analyzed by Heselmeyer et al. (44) were also HPV-positive, so gain of 3q could be considered an aberration associated with HPV infection. Loss of material of the short arm of chromosome 3, a genomic change indicated by a number of loss of heterozygosity studies (45–50), was much less frequently found in our study and in the study of Heselmeyer et al. (44). A deletion of 3p could be the result of isochromosome formation with retainment of i(3q) and subsequent loss of i(3p), a mechanism suggested by the coincidence of 3p loss and 3q gain found in HPK V as well as in three cases of the above mentioned study (44). However, gain of 3q is much more frequent, so this aberration seems to be the important one of chromosome 3, and loss of 3p might occur more as a consequence of one of the mechanisms by which 3q gain is generated. In another study of HPV-transfected keratinocytes, a few allelic losses were identified. However, none of them was characteristic because they occurred in only one–fourth (10p, 11q, and 18q) or two–fourths (3p) of the cell lines (51). However, it seems noteworthy that the one loss at 10p coincides with one of the regions that was found lost in all cell lines investigated in the present study by CGH. The comparison of both studies clearly demonstrates the potential of CGH for a genome-wide screening for chromosomal imbalances because the analysis of allelic losses is less comprehensive and is not suited for the identification of chromosomal gains.
Experiments using microcell-mediated chromosome transfer revealed that different chromosomes can confer senescence when introduced into different immortal cell lines (52–61). Ning and colleagues (56) reported that microcell-mediated transfer of a single copy of chromosome 4 in HeLa cells as well as in other cell lines belonging to the same complementation group B as defined by Pereira–Smith et al. (21) (namely a bladder carcinoma and a glioblastoma cell line) resulted in loss of proliferation and reversal of the immortal phenotype. This observation provides evidence that a gene modified in immortal cell lines assigned to complementation group B is present on human chromosome 4 and that the normal counterpart of this gene is required for expression of the senescent phenotype. In our in vitro model system, chromosome 4 was found recurrently lost during the process of immortalization (Table 1), indicating that loss of this chromosome is a process preceding malignancy. When genes controlling cellular senescence are inactivated by mutation or deletion, they presumably activate the process of immortalization (63). Therefore, our data nicely fit the model of senescence genes on chromosome 4 (56) involved in the immortalization of premalignant cells. Loss of chromosome 4 also was described in 30% of cervical carcinomas (44).
Transfection of normal chromosome 2 has been reported to induce cellular senescence in the human cervical carcinoma cell line SiHa (61); however, no deletion of this chromosome was observed in the cell lines analyzed in this study. In contrast, the underrepresented chromosome region of 6q, occurring in three cell lines, also has been described as a candidate for suppression of the immortal phenotype for complementation group A (60). However, cervical carcinoma cell lines for complementation group A have not been identified.
Reznikoff et al. (10) immortalized another cell type, human uroepithelial cells, by the HPV16 E6 and/or E7 genes with known oncogenic potential. Chromosome analysis of later cell culture passages revealed only a few changes in the two lines expressing the viral E7 oncoprotein, which is known to bind and inactivate pRB. In contrast, in both cell lines expressing oncoprotein E6, which binds to the cellular p53 protein and thus interferes with its function, more chromosomal changes were found. This is consistent with the concept of p53 as one of the key regulators of genomic integrity mediating cell cycle arrest after DNA damage, with p53 inactivation resulting in karyotypic instability (10, 64–66). There is no similarity between the changes found in both E6 positive lines, so the data suggest a general genotypic instability in the uroepithelial cells. This is clearly distinct from our results obtained by transfecting human keratinocytes with whole HPV genomes. There is no evidence for a loss of chromosome regions 13q or 17p that would indicate inactivation of the cellular pRB and p53 protein genes, but such an inactivation could occur via the interference of the viral E7 and E6 proteins, respectively, because the cell lines reported in the present study express both viral oncoproteins (refs. 3 and 33 and unpublished data). However, the chromosomal imbalances identified in our study were highly characteristic and cannot be attributed to a general karyotypic instability. If the immortalization of the cell lines is related to the inhibition of p53 by E6, a mechanism would have to be postulated by which specific chromosomal changes occur, which are not corrected by the inhibition of the p53-mediated cell cycle arrest. It is tempting to attribute the mechanism, which leads to characteristic changes, to the role of other HPV genes, in particular because Reznikoff et al. (10) only transfected HPV E6 and E7 genes in their study. However, it should be noted that several of the chromosomal imbalances found in the E6- or E7-immortalized uroepithelial cell lines (10) were identified as recurrent aberrations in our study of keratinocytes upon HPV infection.
In conclusion, we demonstrate a high specificity of chromosomal imbalances associated with transfection of high risk HPVs. Several of these imbalances fit to previous data describing chromosomal regions involved in immortalization or associated with transition to malignancy in HPV-infected cervical tumors. The striking similarities of our data obtained by the CGH analysis of HPV-transfected cultured cells and the analysis of in vivo tumor samples strongly support the adequacy of the established cell lines as an in vitro model to study immortalization and tumorigenesis in HPV-associated tumors. Therefore, analysis of these cell lines at various time points will allow further elucidation of the molecular pathogenetic mechanisms of high risk HPVs. In this study, several new chromosomal aberrations were identified as a result of viral transfection, so analysis of the respective chromosomal regions in other HPV-associated tumors seems warranted.
Acknowledgments
We thank Applied Imaging (Sunderland, Tyne & Wear, UK) for a fruitful collaboration and for the supply of the cytovision system for CGH analysis.
ABBREVIATIONS
- HPV
human papillomavirus
- CGH
comparative genomic hybridization
- HPK
human papillomavirus-immortalized keratinocytes
References
- 1.zur Hausen H. Curr Top Microbiol Immunol. 1994;186:131–156. doi: 10.1007/978-3-642-78487-3_8. [DOI] [PubMed] [Google Scholar]
- 2.zur Hausen H. Cancer Res. 1989;49:4677–4681. [PubMed] [Google Scholar]
- 3.Dürst M, Dzarlieva-Petrusevska R T, Boukamp P, Fusenig N E, Gissmann L. Oncogene. 1987;1:251–256. [PubMed] [Google Scholar]
- 4.Pirisi L, Yasumoto S, Feller M, Doniger J, DiPaolo J A. J Virol. 1987;6:1061–1066. doi: 10.1128/jvi.61.4.1061-1066.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Schlegel R, Phelps W C, Zhang Y-L, Barbosa M. EMBO J. 1988;7:3181–3187. doi: 10.1002/j.1460-2075.1988.tb03185.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Woodworth C D, Doniger J, DiPaolo J A. J Virol. 1989;63:159–164. doi: 10.1128/jvi.63.1.159-164.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.McDougall J K. Curr Topics Microbiol Immunol. 1994;186:101–119. doi: 10.1007/978-3-642-78487-3_6. [DOI] [PubMed] [Google Scholar]
- 8.Hoppe-Seyler F, Butz K. J Mol Med. 1995;73:529–538. doi: 10.1007/BF00195138. [DOI] [PubMed] [Google Scholar]
- 9.zur Hausen H. Biochim Biophys Acta. 1996;1288:F55–F78. doi: 10.1016/0304-419x(96)00020-0. [DOI] [PubMed] [Google Scholar]
- 10.Reznikoff C A, Belair C, Savelieva E, Zhai Y, Pfeifer K, Yeager T, Thompson K J, DeVries S, Bindley C, Newton M A, Sekhon G, Waldman F. Genes Dev. 1994;8:2227–2240. doi: 10.1101/gad.8.18.2227. [DOI] [PubMed] [Google Scholar]
- 11.White A E, Livanos E M, Tlsty T D. Genes Dev. 1994;8:666–677. doi: 10.1101/gad.8.6.666. [DOI] [PubMed] [Google Scholar]
- 12.Havre P A, Yuan J, Hedrick L, Cho K R, Glazer P M. Cancer Res. 1995;55:4420–4424. [PubMed] [Google Scholar]
- 13.Kaur P, McDougall J K. J Virol. 1988;62:1917–1924. doi: 10.1128/jvi.62.6.1917-1924.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Dürst M, Gallahan D, Jay G, Rhim J S. Virology. 1989;173:767–771. doi: 10.1016/0042-6822(89)90595-3. [DOI] [PubMed] [Google Scholar]
- 15.Smith P P, Bryant E M, Kaur P, McDougall J K. Int J Cancer. 1989;44:1124–1131. doi: 10.1002/ijc.2910440631. [DOI] [PubMed] [Google Scholar]
- 16.Pecoraro G, Lee M, Morgan D, Defendi V. Am J Pathol. 1991;138:1–8. [PMC free article] [PubMed] [Google Scholar]
- 17.Chen T-M, Pecoraro G, Defendi V. Cancer Res. 1993;53:1167–1171. [PubMed] [Google Scholar]
- 18.Dürst M, Seagon S, Wanschura S, zur Hausen H, Bullerdiek J. Cancer Genet Cytogenet. 1995;85:105–112. doi: 10.1016/0165-4608(95)00155-7. [DOI] [PubMed] [Google Scholar]
- 19.Seagon S, Dürst M. Cancer Res. 1994;54:5593–5598. [PubMed] [Google Scholar]
- 20.Pereira-Smith O M, Smith J R. Science. 1983;221:964–966. doi: 10.1126/science.6879195. [DOI] [PubMed] [Google Scholar]
- 21.Pereira-Smith O M, Smith J R. Proc Natl Acad Sci USA. 1988;85:6042–6046. doi: 10.1073/pnas.85.16.6042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Kallioniemi A, Kallioniemi O-P, Sudar D, Rutovitz D, Gray J W, Waldman F, Pinkel D. Science. 1992;258:818–821. doi: 10.1126/science.1359641. [DOI] [PubMed] [Google Scholar]
- 23.Du Manoir S, Speicher M R, Joos S, Schröck E, Popp S, Döhner H, Kovacs G, Robert-Nicoud M, Lichter P, Cremer T. Hum Genet. 1993;90:590–610. doi: 10.1007/BF00202476. [DOI] [PubMed] [Google Scholar]
- 24.Joos S, Scherthan H, Speicher M R, Schlegel J, Cremer T, Lichter P. Hum Genet. 1993;90:584–589. doi: 10.1007/BF00202475. [DOI] [PubMed] [Google Scholar]
- 25.Dürst M, Gissmann L, Ikenberg H, zur Hausen H. Proc Natl Acad Sci USA. 1983;80:3812–3815. doi: 10.1073/pnas.80.12.3812. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Boshart M, Gissmann L, Ikenberg H, Kleinheinz A, Scheurlen W, zur Hausen H. EMBO J. 1984;3:1151–1157. doi: 10.1002/j.1460-2075.1984.tb01944.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Graham F L, van der Eb A J. Virology. 1973;52:456–467. doi: 10.1016/0042-6822(73)90341-3. [DOI] [PubMed] [Google Scholar]
- 28.Lichter P, Bentz M, du Manoir S, Joos S. In: Human Chromosomes. Verma R S, Babu A, editors. New York: McGraw Hill; 1995. pp. 191–210. [Google Scholar]
- 29.Du Manoir S, Schröck E, Bentz M, Speicher M R, Joos S, Ried T, Lichter P, Cremer T. Cytometry. 1995;19:27–41. doi: 10.1002/cyto.990190105. [DOI] [PubMed] [Google Scholar]
- 30.Bentz M, Döhner H, Huck K, Schütz B, Ganser A, Joos S, du Manoir S, Lichter P. Genes Chromosom Cancer. 1995;12:193–200. doi: 10.1002/gcc.2870120306. [DOI] [PubMed] [Google Scholar]
- 31.Kallioniemi O-P, Kallioniemi A, Piper J, Isola J, Waldman F M, Gray J W, Pinkel D. Genes Chromosom Cancer. 1994;10:231–243. doi: 10.1002/gcc.2870100403. [DOI] [PubMed] [Google Scholar]
- 32.Solinas-Toldo S, Wallrapp C, Müller-Pillasch F, Bentz M, Gress T, Lichter P. Cancer Res. 1996;56:3803–3807. [PubMed] [Google Scholar]
- 33.Dürst M, Bosch F X, Glitz D, Schneider A, zur Hausen H. J Virol. 1991;65:796–804. doi: 10.1128/jvi.65.2.796-804.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Heim S, Mitelman F. Cancer Cytogenetics. New York: Wiley–Liss; 1995. pp. 389–407. [Google Scholar]
- 35.Atkin N B, Baker M C, Fox M F. Cancer Genet Cytogenet. 1990;44:229–241. doi: 10.1016/0165-4608(90)90052-c. [DOI] [PubMed] [Google Scholar]
- 36.Sreekantaiah C, De Braekeleer M, Haas O. Cancer Genet Cytogenet. 1991;53:75–81. doi: 10.1016/0165-4608(91)90116-c. [DOI] [PubMed] [Google Scholar]
- 37.Atkin N B, Baker M C. Cancer Genet Cytogenet. 1982;7:209–222. doi: 10.1016/0165-4608(82)90068-1. [DOI] [PubMed] [Google Scholar]
- 38.Atkin N B, Baker M C. Cancer Genet Cytogenet. 1984;13:189–207. doi: 10.1016/0165-4608(84)90043-8. [DOI] [PubMed] [Google Scholar]
- 39.Atkin N B, Baker M C. Cancer Genet Cytogenet. 1989;37:229–233. doi: 10.1016/0165-4608(89)90053-8. [DOI] [PubMed] [Google Scholar]
- 40.Srivatsan E S, Misra B C, Venugopalan M, Wilczynski S P. Am J Hum Genet. 1991;49:868–877. [PMC free article] [PubMed] [Google Scholar]
- 41.Hampton G M, Penny L A, Baergen R N, Larson A, Brewer C, Liao S, Busby-Earle R M C, Williams A W R, Steel C M, Bird C C, Stanbridge E J, Evans G A. Proc Natl Acad Sci USA. 1994;91:6953–6957. doi: 10.1073/pnas.91.15.6953. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Bethwaite P B, Koreth J, Herrington C S, McGee J D. Br J Cancer. 1995;71:814–818. doi: 10.1038/bjc.1995.157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Mitra A B, Rao P H, Pratap M. Cancer Genet Cytogenet. 1994;76:56–58. doi: 10.1016/0165-4608(94)90072-8. [DOI] [PubMed] [Google Scholar]
- 44.Heselmeyer K, Schröck E, Du Manoir S, Blegent H, Shah K, Steinbeck R, Auer G, Ried T. Proc Natl Acad Sci USA. 1996;93:479–484. doi: 10.1073/pnas.93.1.479. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Yokota J, Tsukada Y, Nakajima T, Gotoh M, Shimosato Y, Mori N, Tsunokawa Y, Sugimura T, Terada M. Cancer Res. 1989;49:3598–3601. [PubMed] [Google Scholar]
- 46.Chung G T Y, Huang D P, Lo K W, Chan M K M, Wong F W S. Anticancer Res. 1992;12:1485–1490. [PubMed] [Google Scholar]
- 47.Jones M H, Nakamura Y. Oncogene. 1992;7:1631–1634. [PubMed] [Google Scholar]
- 48.Kohno T, Takayama H, Hamaguchi M, Takano H, Yamaguchi N, Tsuda H, Hirohashi S, Vissing H, Shimizu M, Oshimura M, Yokota J. Oncogene. 1993;8:1825–1832. [PubMed] [Google Scholar]
- 49.Karlsen F, Rabbitts P H, Sundresan V, Hagmar B. Int J Cancer. 1994;58:787–792. doi: 10.1002/ijc.2910580606. [DOI] [PubMed] [Google Scholar]
- 50.Mitra A B, Murty V V V S, Li R G, Pratap M, Luthra U K, Chaganti R S K. Cancer Res. 1994;54:4481–4487. [PubMed] [Google Scholar]
- 51.Steenbergen R D M, Walboomers J M M, Meijer C J L M, van der Raaij-Helmer E M H, Parker J N, Chow L T, Broker T R, Snijders P J F. Oncogene. 1996;13:1249–1257. [PubMed] [Google Scholar]
- 52.Koi M, Morita H, Yamada H, Satoh H, Barrett J C, Oshimura M. Mol Carcinog. 1989;2:12–21. doi: 10.1002/mc.2940020103. [DOI] [PubMed] [Google Scholar]
- 53.Sugawara O, Oshimura M, Koi M, Annab L A, Barrett J C. Science. 1990;247:707–710. doi: 10.1126/science.2300822. [DOI] [PubMed] [Google Scholar]
- 54.Yamada H, Wake N, Fujimoto S, Barrett J C, Oshimura M. Oncogene. 1990;5:1141–1147. [PubMed] [Google Scholar]
- 55.Klein C B, Conway K, Wang X W, Bhamra R K, Lin X, Cohen M D, Annab L, Barrett J C, Costa M. Science. 1991;251:796–799. doi: 10.1126/science.1990442. [DOI] [PubMed] [Google Scholar]
- 56.Ning Y, Weber J L, Killary A M, Ledbetter D H, Smith J R, Pereira-Smith O M. Proc Natl Acad Sci USA. 1991;88:5635–5639. doi: 10.1073/pnas.88.13.5635. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Koi M, Johnson L A, Kalikin L M, Little P F R, Nakamura Y, Feinberg A P. Science. 1993;260:361–364. doi: 10.1126/science.8469989. [DOI] [PubMed] [Google Scholar]
- 58.Ogata T, Ayusawa D, Namba M, Takahashi E, Oshimura M, Oishi M. Mol Cell Biol. 1993;13:6036–6043. doi: 10.1128/mcb.13.10.6036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Hensler P J, Annab L A, Barrett J C, Pereira-Smith O M. Mol Cell Biol. 1994;14:2291–2297. doi: 10.1128/mcb.14.4.2291. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Sandhu A K, Hubbard K, Kaur G P, Jha K K, Ozer H L, Athwal R S. Proc Natl Acad Sci USA. 1994;91:5498–5502. doi: 10.1073/pnas.91.12.5498. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Uejima H, Mitsuya K, Kugoh H, Horikawa I, Oshimura M. Genes Chromosomes Cancer. 1995;14:120–127. doi: 10.1002/gcc.2870140206. [DOI] [PubMed] [Google Scholar]
- 62.Mitelman F, editor. ISCN 1995: An International System for Human Cytogenetic Nomenclature. Basel: Karger; 1995. [Google Scholar]
- 63.Sasaki M, Honda T, Yamada H, Wake N, Barrett J C, Oshimura M. Cancer Res. 1994;54:6090–6093. [PubMed] [Google Scholar]
- 64.Kastan M B, Onyekwere O, Sidransky D, Vogelstein B, Craig R W. Cancer Res. 1991;51:6304–6311. [PubMed] [Google Scholar]
- 65.Lane D P. Nature (London) 1992;358:15–16. doi: 10.1038/358015a0. [DOI] [PubMed] [Google Scholar]
- 66.Fritsche M, Haessler C, Brandner G. Oncogene. 1993;8:307–318. [PubMed] [Google Scholar]