Abstract
The molecular defects responsible for tumorigenesis in adult de novo acute myeloid leukemia (AML) with a normal karyotype or an additional copy of one chromosome (i.e., trisomy) remain largely unknown. We recently discovered that approximately 90% of adult patients with de novo AML and trisomy 11 (+11) as a sole abnormality and 11% of adult patients with de novo AML and normal cytogenetics carry a molecular rearrangement of the ALL1 (MLL, HRX, or HTRX) gene. The rearranged ALL1 gene has been shown to result from the direct tandem duplication of a portion of ALL1 itself. To better understand the underlying mechanisms of leukemogenesis, we asked whether in cytogenetically normal cases one or both chromosomes carry the mutated allele and whether in trisomic cases the mutation is present in one, two, or three chromosomes. Herein we show that in cytogenetically normal cases of AML and in cases with +11 as a sole cytogenetic abnormality, only one chromosome contains the mutated ALL1 allele. Thus a single mutated ALL1 allele with the partial tandem duplication is sufficient for ALL1-associated leukemogenesis, irrespective of the number of normal genes present. The frequently occurring specific association of +11 and ALL1 gene mutation in the leukemic clone remains unexplained.
Reciprocal chromosomal translocations or inversions have led to the identification of the majority of genetic defects known to contribute to malignant transformation in acute myeloid leukemia (AML) (1, 2). However, normal diploid chromosomes or clonal evidence for trisomy (+) of chromosome 21 (+21), 8 (+8), or 11 (+11) as a sole abnormality on cytogenetic analysis of leukemic blasts account for approximately 45% of AML cases (3). In the majority of these cases, the specific genetic mutations responsible for AML have not been identified. Recently, we discovered the first consistent molecular defect in cancer associated with trisomy while studying patients diagnosed with AML and +11 as a sole cytogenetic abnormality (4, 5). The gene, located at 11q23, called ALL1 (6, 7), MLL (8), HRX (9), or HTRX (10), was found to be rearranged in 10 of 11 (91%) AML patients with +11 as a sole cytogenetic abnormality, without cytogenetic abnormality at 11q23. In each case where additional material was available, the ALL1 gene was found to have undergone a partial tandem duplication of an internal portion of itself (4, 5, 11). More recently, we have discovered that approximately 11% of adult patients with de novo AML and normal cytogenetics have rearranged ALL1 as the result of a partial tandem duplication of the gene (12). More than 90% of the partial duplications span either exons 2 through 6 or exons 2 through 8, and each has been identified in adult de novo AML cases with +11 or normal cytogenetics. In all cases, the unique fusion resulting from partially duplicated sequence of the ALL1 gene has been found to be in frame, predicting a partially duplicated protein product. The mechanism by which the partial tandem duplication contributes to leukemogenesis is currently unknown, as is the mechanism of all rearrangements involving ALL1. Moreover, the mechanism underlying the frequent specific association of +11 with ALL1 gene mutation is unexplained.
In the current study, we analyze AML blasts from patients with the partial tandem duplication of ALL1 and either +11 as a sole cytogenetic abnormality or normal cytogenetics. We demonstrate that in each instance, only one chromosome 11 contains the partially duplicated ALL1 allele.
MATERIALS AND METHODS
Patients Specimens.
Bone marrow or blood blasts were obtained from five patients who were diagnosed with AML and +11 as a sole cytogenetic abnormality (three patients) or AML and normal cytogenetics (two patients) (Table 1). The diagnosis of AML was based on standard French–American–British morphological and cytochemical criteria (13, 14). All five patients were previously shown to rearrange ALL1 on Southern blot analysis with the B859 probe and were found to have the partial tandem duplication of ALL1 by reverse transcription-coupled PCR and direct DNA sequencing (11, 12).
Table 1.
Allelic dosage of the partial tandem duplication of ALL1 in AML blasts
| UPN | Karyotype* | ALL1 partial duplication* | Copies of ALL1 exon 18† | Copies of ALL1 exon 5† |
|---|---|---|---|---|
| C‡ | 46,XY | N/A | 2 | 2 |
| 23 | 47,XX,+11 | Exon 2–exon 6 | 2.92 ± 0.026 | 3.86 ± 0.071 |
| 56 | 47,XY,+11 | Exon 4a–exon 6 | 2.99 ± 0.035 | 3.83 ± 0.075 |
| 108 | 47,XY,+11 | Exon 2–exon 8 | 2.98 ± 0.024 | 3.89 ± 0.051 |
| 144 | 46,XX | Exon 2–exon 8 | 2.01 ± 0.023 | 3.05 ± 0.030 |
| 211 | 46,XX | Exon 2–exon 6 | 2.01 ± 0.003 | 3.13 ± 0.006 |
As quantitated by phosphorimage analysis, relative to one-half of the signal intensity measured from the same amount of DNA obtained from normal diploid cells (C) hybridized to the same probe.
C, control of diploid cells from a normal donor; N/A, not applicable.
Cytogenetic Analyses.
The criteria used to define a cytogenetic clone and descriptions of karyotypes followed the recommendations of the International System for Human Cytogenetic Nomenclature (15).
DNA Preparation and Probes for Southern Blot Analysis.
Preparation of DNA and Southern blot analysis were carried out as described (11). The B859 probe was used in Southern blot analysis. B859 is an 859-bp BamHI fragment of ALL1 cDNA that spans the breakpoint cluster region defined by exons 5–11 of the ALL1 gene (7).
Determination of Number of Chromosomes Carrying the Partial Tandem Duplication of ALL1.
Cryopreserved bone marrow or blood specimens containing ≥90% leukemic blasts were utilized. In instances where specimens contained <90% leukemic cells (two cases), blasts were positively selected using a CD34+ biotin-avidin Ceprate column (Cellpro, Bothel, WA), according to the manufacturer’s instructions; 95% blasts were recovered as determined by Wright–Giemsa staining. Genomic DNA was quantitated three times by UV spectrophotometry and blotted (5 μg per well) onto a positively charged nylon membrane with a Bio-Dot SF Microfiltration Apparatus (Bio-Rad) for a slot blot hybridization procedure. A single-copy 2.2-kb genomic probe (2.2XX) from the SCL gene located on chromosome 1p was used to quantitate the amount of DNA loaded (16). A 528-bp cDNA probe from exon 18 of ALL1, derived from normal genomic DNA by PCR, was used as a single-copy probe for chromosome 11, as exon 18 lies outside of the duplication of ALL1. A 240-bp cDNA probe from exon 5 of ALL1 was derived from normal genomic DNA by PCR and was used to determine the number of chromosomes containing the partial tandem duplication of ALL1, as exon 5 lies within the duplicated region (11). The probes were labeled with [32P]dCTP and hybridized to the slot blot by standard techniques (17). DNA was quantitated by measuring the signal intensity of the blot with a Molecular Dynamics PhosphorImager and the imagequant computer program (Molecular Dynamics). The recorded signal intensity values of 2.2XX, exon 5, and exon 18 probes were first corrected for any differences in DNA loading, as measured by the 2.2XX probe signal intensity from normal DNA and patient DNA. The relative signal intensity from patient DNA (Yr) was calculated as follows: Yr = Y/(X/2), where Y is the exon probe signal intensity recorded from patient DNA and X is the exon probe signal intensity recorded from normal diploid DNA. The relative signal intensity from normal DNA (Xr) was calculated as follows: Xr = X/(X/2). Xr is therefore always equal to two. Thus, the Yr and Xr are graphed relative to one-half of the measured probe signal intensity from normal diploid DNA. Each blot was probed three times, and the results from each experiment represent the signal intensity (mean ± SEM).
RESULTS AND DISCUSSION
Specimen Characteristics.
Specimens from five patients were analyzed (Table 1). All patients had AML classified by the French–American–British system as FAB M1 or M2. AML blasts from unique patient numbers (UPNs) 23, 56, and 108 had +11 as the sole clonal cytogenetic abnormality; UPNs 144 and 211 had normal cytogenetics. All patients demonstrated ALL1 gene rearrangements by Southern blotting, and the partial tandem duplication of ALL1 by reverse transcription-coupled PCR and direct DNA sequencing. Each partial tandem duplication included exon 5 (11, 12).
Determination of Number of Chromosomes Carrying the Mutated ALL1 Allele.
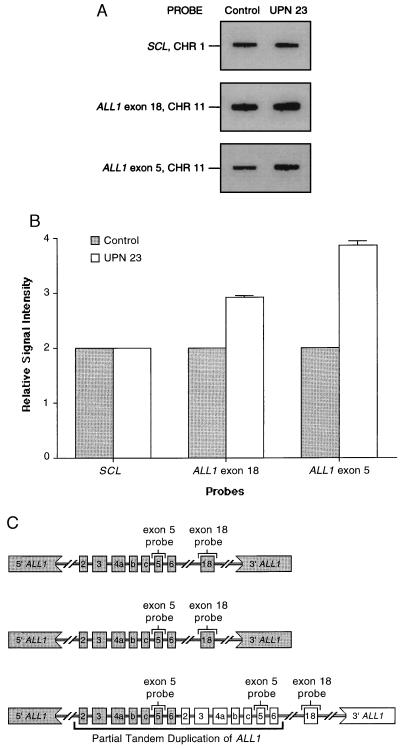
When DNA obtained from diploid cells of a normal individual is hybridized, the signal intensity with the ALL1 exon 18 probe and the ALL1 exon 5 probe is indicative of one copy of each ALL1 exon on each chromosome 11. Therefore, an arbitrary value of two is assigned to each signal. In AML blasts containing +11 as a sole cytogenetic abnormality, the signal intensity with the ALL1 exon 18 probe should be three, relative to the signal intensity detected in DNA from normal cells. This is because the extra chromosome 11 should contain a single copy of ALL1 exon 18, downstream of the partially duplicated region of ALL1 (5, 11). However, the relative signal intensity of the ALL1 exon 5 probe could vary from four to six, depending on the number of chromosomes 11 carrying the mutated ALL1 allele. Each chromosome 11 without the partial duplication of ALL1 should have a relative signal intensity of one with the ALL1 exon 5 probe, while each chromosome 11 with the partial duplication of ALL1 should have a relative signal intensity of two with the ALL1 exon 5 probe. AML blasts obtained from three different cases displaying +11 as a sole cytogenetic abnormality and the partial duplication of ALL1 each had a relative signal intensity of three with the ALL1 exon 18 probe and a relative signal intensity of four with the ALL1 exon 5 probe. This is consistent with the AML blasts from each case having only one chromosome 11 with the partially duplicated ALL1 allele (Table 1 and Fig. 1).
Figure 1.
Dosage of the wild-type and partially duplicated ALL1 alleles in AML blasts of a patient (UPN 23) with +11 as a sole cytogenetic abnormality. (A) The pattern obtained with hybridization using SCL, ALL1 exon 18, and ALL1 exon 5 probes. The signal intensity detected with the SCL probe from chromosome 1 was the same for the normal control and UPN 23 (see Table 1), indicating that equivalent amounts of DNA were loaded. However, the signal intensity for the ALL1 exon 18 and the ALL1 exon 5 are visibly different when comparing the equal amounts of DNA from the normal control and UPN 23. (B) Graphical depiction of the quantitative phosphorimage analysis of signal intensity from SCL, ALL1 exon 18, and ALL1 exon 5 blots shown in A. Each measurement is relative to one-half the signal intensity in the normal donor using the same probe. This shows that UPN 23 with +11 had three copies of ALL1 exon 18 that is outside the duplication, but four copies of ALL1 exon 5 that is within the duplication. (C) A schematic diagram of ALL1 allelic dosage in AML with +11 and the partial duplication, incorporating data obtained above. This shows that the three copies of ALL1 exon 18 and four copies of ALL1 exon 5 can be accounted for with only one of three chromosomes containing the partial tandem duplication of ALL1. The two remaining chromosomes 11 each contain a wild-type ALL1 allele.
We quantitated the dose of wild-type and mutated ALL1 alleles in AML blasts obtained from two patients with AML, normal cytogenetics, and the partial tandem duplication of the gene. The signal intensity measured with patient DNA hybridized to the ALL1 exon 18 probe was equal to the signal intensity measured with an equivalent amount of normal donor DNA hybridized to the ALL1 exon 18 probe (Table 1). This is consistent with the euploid number of chromosomes in both populations of cells and with the fact that the ALL1 exon 18 is downstream of the partially duplicated region of ALL1. However, in both cases of AML, the signal intensity measured with patient DNA hybridized to the ALL1 exon 5 probe was approximately 50% greater than the signal intensity measured with an equivalent amount of normal donor DNA hybridized to the ALL1 exon 5 probe. This is consistent with three copies of ALL1 exon 5 in the DNA obtained from AML blasts with normal cytogenetics, relative to two copies of ALL1 exon 5 in normal cells (Table 1). Thus, in each of these two cases, only one chromosome 11 has a mutated ALL1 allele containing two copies of ALL1 exon 5 in the partial duplication of ALL1.
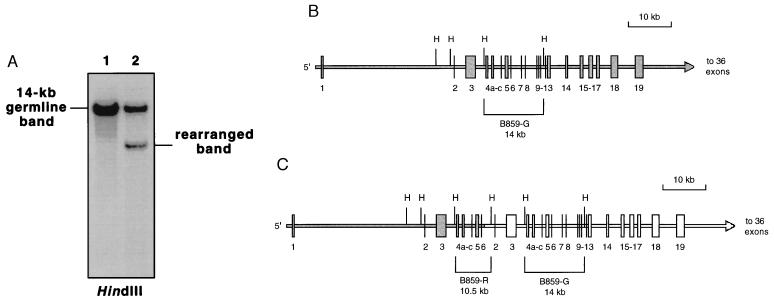
The partially duplicated ALL1 allele contains both rearranged and germ-line DNA fragments on Southern blot analysis with the B859 probe (Fig. 2C). Hence, the presence of a germ-line band on analysis of leukemic blasts with the duplication does not in itself provide conclusive evidence for the existence of a wild-type ALL1 allele. We therefore demonstrated the existence of a wild-type ALL1 allele by assessing allelic dosage on Southern blot analysis with DNA obtained from a case of AML with the duplication spanning exons 2–6, normal cytogenetics, and 99% blasts. By using a HindIII restriction digest and the B859 probe, a germ-line band of 14 kb spanning exons 5–11 was noted with nearly double the intensity of the smaller rearranged band predicted by the partial duplication. No other bands were seen (Fig. 2A). A schematic of these data is shown in Fig. 2 B and C. These results demonstrate that in AML with normal cytogenetics and the duplication, one chromosome 11 has a wild-type ALL1 allele. An identical Southern blot analysis was performed with >95% blasts from a case of AML with +11 as a sole cytogenetic abnormality and the duplication, demonstrating two wild-type ALL1 alleles (data not shown).
Figure 2.
Analysis of ALL1 germ-line and rearranged bands in an AML patient with a normal karyotype (UPN 211) and the partial tandem duplication of ALL1 spanning exons 2–6. (A) Southern blot analysis of HindIII digests of normal control (lane 1) and patient (lane 2) genomic DNA using the B859 probe. Patient genomic DNA was obtained from a specimen containing 99% blasts. Phosphorimage analysis showed the signal intensity of the patient’s germ-line band to be approximately twice that of the rearranged bands (data not shown). (B) Schematic of the genomic structure of the wild-type ALL1 allele showing location of HindIII (H) restriction sites within the gene. Numbered vertical lines and boxes indicate exons. The brackets indicate the 14-kb germ-line (G) ALL1 band detected with the B859 probe after HindIII digestion. (C) Schematic of the genomic structure of the partial tandem duplication of ALL1 involving a portion of the gene spanning exons 2–6, within the white region. The size of the rearranged (R) band that results after a restriction digest of genomic DNA with HindIII and hybridization with the B859 probe is indicated. Note that the chromosome 11 with the partially duplicated ALL1 allele also has a germ-line band after HindIII digestion and hybridization with the B859 probe.
In the current study, we establish that the partial tandem duplication of ALL1 is found on a single chromosome in five cases of AML, regardless of whether the karyotype of AML blasts is normal or displays +11 as a sole cytogenetic abnormality. Thus, this work demonstrates that a single mutated ALL1 allele with the partial tandem duplication is sufficient for ALL1-associated leukemogenesis. The lack of an additional mutated ALL1 allele in cases of AML with +11 as a sole cytogenetic abnormality also leads us to speculate that the extra copy of chromosome 11 containing the wild-type allele provides a selective growth or survival advantage over normal cells and over leukemic cells with only one wild-type and mutated ALL1 allele. Indeed, despite a strong association between a variety of human malignant phenotypes and certain trisomies, there is little understanding of how trisomy may lead to neoplasia. Gene amplification in somatic cells does occur frequently in certain neoplasias (18, 19) and presumably plays a role in some cancers consistently associated with trisomy. It is conceivable that increased gene dosage of other loci on chromosome 11 could also contribute to, or be responsible for, a selective growth or survival advantage afforded by the +11. However, the documented absence of an extra copy of chromosome 11 in leukemic blasts that have normal cytogenetics and the ALL1 duplication (12) suggests that the additional chromosome is not critical for ALL1-associated leukemogenesis.
The mechanism by which the trisomy occurs in these cases also remains obscure. Cavenee et al. (20) postulated a chromosomal mechanism of mitotic nondisjunction with loss of the wild-type chromosome and reduplication of the mutant chromosome to account for a retinoblastoma containing three copies of a chromosome 13, each with evidence suggesting a mutant allele at the Rb-1 locus. However, in striking contrast, the cases of AML with +11 that we studied appear to have two chromosomes 11 containing the wild-type allele at the ALL1 locus and only one chromosome 11 with a mutant allele at the ALL1 locus. This difference emphasizes the different modes of action of the Rb-1 gene (a tumor suppressor gene whose mutations are recessive and lead to loss of function) and the ALL1 gene (a likely transcription factor gene in which the tandem duplication may act dominantly through a gain of function). We postulate a mechanism of mitotic nondisjunction as the somatic genetic event to account for the +11 observed in some of our cases. We have evidence that Alu-mediated recombination is a molecular mechanism that can be responsible for ALL1 partial tandem duplication observed in these somatic cells (ref. 21 and M.P.S. and M.A.C., unpublished observations). Whether the recombination occurs between two different chromosome 11 alleles or between sister chromatids of a single allele during mitosis is unknown. We cannot exclude the possibility that the molecular rearrangement and the emergence of trisomy occurred as different components of a single event. It is clear that in five cases of AML with the partial duplication and either normal cytogenetics or +11, the unaffected chromosomes 11 in the leukemic blasts have a wild-type ALL1 allele.
In all 30 AML cases with the partial tandem duplication of ALL1 and either normal cytogenetics or +11 studied thus far, RNA PCR and DNA sequence analysis have consistently revealed the unique fusion of exons to be in frame, demonstrating that the partially duplicated gene is transcribed into mRNA capable of encoding a partially duplicated protein (5, 11, 12, 21). Structural and functional evidence to date indicates that the wild-type ALL1 is likely to encode a transcription factor that has a role in morphogenesis and hematopoiesis (22, 23). The manner by which the partially duplicated ALL1 protein may elicit leukemic growth remains uncertain. Dominant negative effects have been described in many such proteins and would be consistent with the dosage analysis of the mutated ALL1 allele described in this report. Characterizaton of the wild-type and mutated ALL1 proteins is underway and should provide additional insights into its role in during normal hematopoiesis and leukemogenesis.
Acknowledgments
We thank Dr. Peter Aplan for his critical review of the manuscript and Ms. Dung Nguyen for her editorial assistance. This work was supported by Grants CA-37027, CA-65670, and CA-57974; a Translational Research Grant from the Leukemia Society of America; the Lady Tata Memorial Trust Fund; and the Coleman Leukemia Research Fund.
ABBREVIATIONS
- AML
acute myeloid leukemia
- +
trisomy
- UPN
unique patient number
References
- 1.Croce C M. Cell. 1987;49:155–156. doi: 10.1016/0092-8674(87)90552-6. [DOI] [PubMed] [Google Scholar]
- 2.Caligiuri M A, Strout M P, Gilliland D G. Sem Oncol. 1997;24:32–44. [PubMed] [Google Scholar]
- 3.Heim S, Mitelman F. Cancer Cytogenetics. New York: Wiley; 1995. [Google Scholar]
- 4.Caligiuri M A, Schichman S A, Strout M P, Mrozek K, Baer M R, Frankel S R, Barcos M, Herzig G P, Croce C M, Bloomfield C D. Cancer Res. 1994;54:370–373. [PubMed] [Google Scholar]
- 5.Schichman S A, Caligiuri M A, Gu Y, Strout M P, Canaani E, Bloomfield C D, Croce C M. Proc Natl Acad Sci USA. 1994;91:6236–6239. doi: 10.1073/pnas.91.13.6236. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Cimino G, Moir D T, Canaani O, Williams K, Crist W M, Katzav S, Cannizzaro L, Lange B, Nowell P C, Croce C M, Canaani E. Cancer Res. 1991;51:6712–6714. [PubMed] [Google Scholar]
- 7.Gu Y, Nakamura T, Alder H, Prasad R, Canaani O, Cimino G, Croce C M, Canaani E. Cell. 1992;71:701–708. doi: 10.1016/0092-8674(92)90603-a. [DOI] [PubMed] [Google Scholar]
- 8.Ziemin-van der Poel S, McCabe N R, Gill H J, Espinosa R, III, Patel Y, Harden A, Rubinelli P, Smith S D, LeBeau M M, Rowley J D, Diaz M O. Proc Natl Acad Sci USA. 1991;88:10735–10739. doi: 10.1073/pnas.88.23.10735. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Tkachuk D C, Kohler S, Cleary M L. Cell. 1992;71:691–700. doi: 10.1016/0092-8674(92)90602-9. [DOI] [PubMed] [Google Scholar]
- 10.Djabali M, Selleri L, Parry P, Bower M, Young B D, Evans G A. Nat Genet. 1992;2:113–118. doi: 10.1038/ng1092-113. [DOI] [PubMed] [Google Scholar]
- 11.Caligiuri M A, Strout M P, Schichman S A, Mrozek K, Arthur D C, Herzig G P, Baer M R, Schiffer C A, Heinonen K, Knuutila S, Nousiainen T, Ruutu T, Block A W, Schulman P, Pedersen-Bjergaard J, Croce C M, Bloomfield C D. Cancer Res. 1996;56:1418–1425. [PubMed] [Google Scholar]
- 12.Caligiuri M A, Strout M P, Arthur D C, Baer M R, Shah D, Block A W, Mrozek K, Yu F, Oberkircher A, Lawrence D, Knuutila S, Ruutu T, Herzig G P, Schiffer C A, Bloomfield C D. Proc Am Soc Clin Oncol. 1996;15:1060. (abstr.). [Google Scholar]
- 13.Bennett J M, Catovsky D, Daniel M T, Flandrin G, Galton D A, Gralnick H R, Sultan C. Ann Intern Med. 1985;103:620–625. doi: 10.7326/0003-4819-103-4-620. [DOI] [PubMed] [Google Scholar]
- 14.Bennett J M, Catovsky D, Daniel M T, Flandrin G, Galton D A, Gralnick H R, Sultan C. Br J Haematol. 1982;51:189–199. [PubMed] [Google Scholar]
- 15.Mitelman F. Guidelines for Cancer Cytogenetics: Supplement to an International System for Human Cytogenetic Nomenclature. Basel: Karger; 1991. [Google Scholar]
- 16.Aplan P D, Lombardi D P, Reaman G H, Sather H N, Hammond G D, Kirsch I R. Blood. 1992;79:1327–1333. [PubMed] [Google Scholar]
- 17.Sambrook J, Fritsch E, Maniatis T. Molecular Cloning: A Laboratory Manual. 2nd Ed. Plainview, NY: Cold Spring Harbor Lab. Press; 1989. [Google Scholar]
- 18.Bishop J M. Cell. 1991;64:235–248. doi: 10.1016/0092-8674(91)90636-d. [DOI] [PubMed] [Google Scholar]
- 19.Oliner J D, Kinzler K W, Meltzer P S, George D L, Vogelstein B. Nature (London) 1992;358:80–83. doi: 10.1038/358080a0. [DOI] [PubMed] [Google Scholar]
- 20.Cavenee W K, Dryja T P, Phillips R A, Benedict W F, Godbout R, Gallie B L, Murphree A L, Strong L C, White R L. Nature (London) 1983;305:779–784. doi: 10.1038/305779a0. [DOI] [PubMed] [Google Scholar]
- 21.Schichman S A, Caligiuri M A, Strout M P, Carter S L, Gu Y, Canaani E, Bloomfield C D, Croce C M. Cancer Res. 1994;54:4277–4280. [PubMed] [Google Scholar]
- 22.Bernard O A, Berger R. Genes Chromosomes Cancer. 1995;13:75–85. doi: 10.1002/gcc.2870130202. [DOI] [PubMed] [Google Scholar]
- 23.Yu B D, Hess J L, Horning S E, Brown G A, Korsmeyer S J. Nature (London) 1995;378:505–508. doi: 10.1038/378505a0. [DOI] [PubMed] [Google Scholar]