Abstract
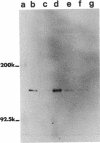
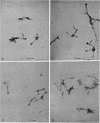
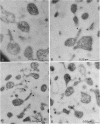
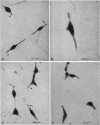
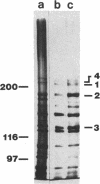
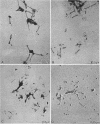
The subcellular location of the phase-variable cytadherence-accessory protein HMW3 in Mycoplasma pneumoniae has been examined by biochemical and immunoelectron microscopic techniques. Analysis by Western blot (immunoblot) with HMW3-specific antiserum established the presence of this protein within the M. pneumoniae Triton X-100-insoluble fraction or triton shell. Immunogold labeling of Triton-extracted mycoplasmas with affinity-purified antibodies localized HMW3 to the terminal knob on the rodlike extensions of the triton shell, a location that would correspond to the adherence organelle in whole mycoplasmas. Treatment of triton shells with KI resulted in the selective removal of the adherence-accessory proteins HMW1 to HMW4. Analysis of these triton shells by transmission electron microscopy revealed dramatic ultrastructural changes in the filamentous network and core structure. Immunogold labeling of KI-extracted shells reflected the removal of HMW3 from the disrupted tip structure. An examination of ultrathin sections of wild-type cells by transmission electron microscopy following labeling with HMW3-specific antibodies provided further evidence for the nonrandom distribution of HMW3 and its localization to the terminal portion of filamentous cell extensions. Most colloidal gold molecules were associated with the cell interior, but limited peripheral labeling of the terminal region was also observed. Postfixation antibody labeling of whole cells suggested limited exposure of HMW3 on the mycoplasma surface at the tip structure. However, prefixation antibody labeling failed to indicate surface exposure, raising some uncertainty regarding the relationship of HMW3 with the mycoplasma membrane.
Full text
PDF









Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Baseman J. B., Cole R. M., Krause D. C., Leith D. K. Molecular basis for cytadsorption of Mycoplasma pneumoniae. J Bacteriol. 1982 Sep;151(3):1514–1522. doi: 10.1128/jb.151.3.1514-1522.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baseman J. B., Morrison-Plummer J., Drouillard D., Puleo-Scheppke B., Tryon V. V., Holt S. C. Identification of a 32-kilodalton protein of Mycoplasma pneumoniae associated with hemadsorption. Isr J Med Sci. 1987 May;23(5):474–479. [PubMed] [Google Scholar]
- Biberfeld G., Biberfeld P. Ultrastructural features of Mycoplasma pneumoniae. J Bacteriol. 1970 Jun;102(3):855–861. doi: 10.1128/jb.102.3.855-861.1970. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bredt W., Feldner J., Kahane I. Adherence of mycoplasmas to cells and inert surfaces: phenomena, experimental models and possible mechanisms. Isr J Med Sci. 1981 Jul;17(7):586–588. [PubMed] [Google Scholar]
- Collier A. M., Clyde W. A., Jr Appearance of Mycoplasma pneumoniae in lungs of experimentally infected hamsters and sputum from patients with natural disease. Am Rev Respir Dis. 1974 Dec;110(6):765–773. doi: 10.1164/arrd.1974.110.6P1.765. [DOI] [PubMed] [Google Scholar]
- Collier A. M., Clyde W. A. Relationships Between Mycoplasma pneumoniae and Human Respiratory Epithelium. Infect Immun. 1971 May;3(5):694–701. doi: 10.1128/iai.3.5.694-701.1971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Feldner J., Göbel U., Bredt W. Mycoplasma pneumoniae adhesin localized to tip structure by monoclonal antibody. Nature. 1982 Aug 19;298(5876):765–767. doi: 10.1038/298765a0. [DOI] [PubMed] [Google Scholar]
- Göbel U., Speth V., Bredt W. Filamentous structures in adherent Mycoplasma pneumoniae cells treated with nonionic detergents. J Cell Biol. 1981 Nov;91(2 Pt 1):537–543. doi: 10.1083/jcb.91.2.537. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hayflick L. Tissue cultures and mycoplasmas. Tex Rep Biol Med. 1965 Jun;23(Suppl):285+–285+. [PubMed] [Google Scholar]
- Hu P. C., Cole R. M., Huang Y. S., Graham J. A., Gardner D. E., Collier A. M., Clyde W. A., Jr Mycoplasma pneumoniae infection: role of a surface protein in the attachment organelle. Science. 1982 Apr 16;216(4543):313–315. doi: 10.1126/science.6801766. [DOI] [PubMed] [Google Scholar]
- Hu P. C., Collier A. M., Baseman J. B. Surface parasitism by Mycoplasma pneumoniae of respiratory epithelium. J Exp Med. 1977 May 1;145(5):1328–1343. doi: 10.1084/jem.145.5.1328. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kahane I., Tucker S., Leith D. K., Morrison-Plummer J., Baseman J. B. Detection of the major adhesin P1 in triton shells of virulent Mycoplasma pneumoniae. Infect Immun. 1985 Dec;50(3):944–946. doi: 10.1128/iai.50.3.944-946.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Krause D. C., Baseman J. B. Inhibition of mycoplasma pneumoniae hemadsorption and adherence to respiratory epithelium by antibodies to a membrane protein. Infect Immun. 1983 Mar;39(3):1180–1186. doi: 10.1128/iai.39.3.1180-1186.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Krause D. C., Leith D. K., Baseman J. B. Reacquisition of specific proteins confers virulence in Mycoplasma pneumoniae. Infect Immun. 1983 Feb;39(2):830–836. doi: 10.1128/iai.39.2.830-836.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Krause D. C., Winkler H. H., Wood D. O. Cosmid cloning of Rickettsia prowazekii antigens in Escherichia coli K-12. Infect Immun. 1985 Jan;47(1):157–165. doi: 10.1128/iai.47.1.157-165.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Lipman R. P., Clyde W. A., Jr, Denny F. W. Characteristics of virulent, attenuated, and avirulent Mycoplasma pneumoniae strains. J Bacteriol. 1969 Nov;100(2):1037–1043. doi: 10.1128/jb.100.2.1037-1043.1969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Markwell M. A., Haas S. M., Bieber L. L., Tolbert N. E. A modification of the Lowry procedure to simplify protein determination in membrane and lipoprotein samples. Anal Biochem. 1978 Jun 15;87(1):206–210. doi: 10.1016/0003-2697(78)90586-9. [DOI] [PubMed] [Google Scholar]
- Meng K. E., Pfister R. M. Intracellular structures of Mycoplasma pneumoniae revealed after membrane removal. J Bacteriol. 1980 Oct;144(1):390–399. doi: 10.1128/jb.144.1.390-399.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Niggli V., Burger M. M. Interaction of the cytoskeleton with the plasma membrane. J Membr Biol. 1987;100(2):97–121. doi: 10.1007/BF02209144. [DOI] [PubMed] [Google Scholar]
- Oakley B. R., Kirsch D. R., Morris N. R. A simplified ultrasensitive silver stain for detecting proteins in polyacrylamide gels. Anal Biochem. 1980 Jul 1;105(2):361–363. doi: 10.1016/0003-2697(80)90470-4. [DOI] [PubMed] [Google Scholar]
- Ogle K. F., Lee K. K., Krause D. C. Cloning and analysis of the gene encoding the cytadherence phase-variable protein HMW3 from Mycoplasma pneumoniae. Gene. 1991 Jan 2;97(1):69–75. doi: 10.1016/0378-1119(91)90011-y. [DOI] [PubMed] [Google Scholar]
- Ogle K. F., Lee K. K., Krause D. C. Nucleotide sequence analysis reveals novel features of the phase-variable cytadherence accessory protein HMW3 of Mycoplasma pneumoniae. Infect Immun. 1992 Apr;60(4):1633–1641. doi: 10.1128/iai.60.4.1633-1641.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Powell D. A., Hu P. C., Wilson M., Collier A. M., Baseman J. B. Attachment of Mycoplasma pneumoniae to respiratory epithelium. Infect Immun. 1976 Mar;13(3):959–966. doi: 10.1128/iai.13.3.959-966.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stevens M. K., Krause D. C. Disulfide-linked protein associated with Mycoplasma pneumoniae cytadherence phase variation. Infect Immun. 1990 Oct;58(10):3430–3433. doi: 10.1128/iai.58.10.3430-3433.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stevens M. K., Krause D. C. Localization of the Mycoplasma pneumoniae cytadherence-accessory proteins HMW1 and HMW4 in the cytoskeletonlike Triton shell. J Bacteriol. 1991 Feb;173(3):1041–1050. doi: 10.1128/jb.173.3.1041-1050.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tjian R., Stinchcomb D., Losick R. Antibody directed against Bacillus subtilis rho factor purified by sodium dodecyl sulfate slab gel electrophoresis. Effect on transcription by RNA polymerase in crude extracts of vegetative and sporulating cells. J Biol Chem. 1975 Nov 25;250(22):8824–8828. [PubMed] [Google Scholar]
- Towbin H., Staehelin T., Gordon J. Electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose sheets: procedure and some applications. Proc Natl Acad Sci U S A. 1979 Sep;76(9):4350–4354. doi: 10.1073/pnas.76.9.4350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wall F., Pfister R. M., Somerson N. L. Freeze-fracture confirmation of the presence of a core in the specialized tip structure of Mycoplasma pneumoniae. J Bacteriol. 1983 May;154(2):924–929. doi: 10.1128/jb.154.2.924-929.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wilson M. H., Collier A. M. Ultrastructural study of Mycoplasma pneumoniae in organ culture. J Bacteriol. 1976 Jan;125(1):332–339. doi: 10.1128/jb.125.1.332-339.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]