Abstract
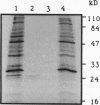
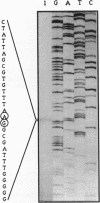
beta-Ketoadipate:succinyl-coenzyme A transferase (beta-ketoadipate:succinyl-CoA transferase) (EC 2.8.3.6) carries out the penultimate step in the conversion of benzoate and 4-hydroxybenzoate to tricarboxylic acid cycle intermediates in bacteria utilizing the beta-ketoadipate pathway. This report describes the characterization of a DNA fragment from Pseudomonas putida that encodes this enzyme. The fragment complemented mutants defective in the synthesis of the CoA transferase, and two proteins of sizes appropriate to encode the two nonidentical subunits of the enzyme were produced in Escherichia coli when the fragment was placed under the control of a phage T7 promoter. DNA sequence analysis revealed two open reading frames, designated pcaI and pcaJ, that were separated by 8 bp, suggesting that they may comprise an operon. A comparison of the deduced amino acid sequence of the P. putida CoA transferase genes with the sequences of two other bacterial CoA transferases and that of succinyl-CoA:3-ketoacid CoA transferase from pig heart suggests that the homodimeric structure of the mammalian enzyme may have resulted from a gene fusion of the bacterial alpha and beta subunit genes during evolution. Conserved functional groups important to the catalytic activity of CoA transferases were also identified.
Full text
PDF







Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Barker H. A., Jeng I. M., Neff N., Robertson J. M., Tam F. K., Hosaka S. Butyryl-CoA:acetoacetate CoA-transferase from a lysine-fermenting Clostridium. J Biol Chem. 1978 Feb 25;253(4):1219–1225. [PubMed] [Google Scholar]
- Blair J. B. Skeletal muscle coenzyme A transferase. Purification and properties. J Biol Chem. 1969 Feb 10;244(3):951–954. [PubMed] [Google Scholar]
- Bradford M. M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem. 1976 May 7;72:248–254. doi: 10.1016/0003-2697(76)90527-3. [DOI] [PubMed] [Google Scholar]
- Buckel W., Dorn U., Semmler R. Glutaconate CoA-transferase from Acidaminococcus fermentans. Eur J Biochem. 1981 Aug;118(2):315–321. doi: 10.1111/j.1432-1033.1981.tb06404.x. [DOI] [PubMed] [Google Scholar]
- Crawford I. P., Eberly L. DNA sequence of the tryptophan synthase genes of Pseudomonas putida. Biochimie. 1989 Apr;71(4):521–531. doi: 10.1016/0300-9084(89)90183-1. [DOI] [PubMed] [Google Scholar]
- Cánovas J. L., Stanier R. Y. Regulation of the enzymes of the beta-ketoadipate pathway in Moraxella calcoacetica. 1. General aspects. Eur J Biochem. 1967 May;1(3):289–300. doi: 10.1007/978-3-662-25813-2_40. [DOI] [PubMed] [Google Scholar]
- Devereux J., Haeberli P., Smithies O. A comprehensive set of sequence analysis programs for the VAX. Nucleic Acids Res. 1984 Jan 11;12(1 Pt 1):387–395. doi: 10.1093/nar/12.1part1.387. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Doten R. C., Ngai K. L., Mitchell D. J., Ornston L. N. Cloning and genetic organization of the pca gene cluster from Acinetobacter calcoaceticus. J Bacteriol. 1987 Jul;169(7):3168–3174. doi: 10.1128/jb.169.7.3168-3174.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Durham D. R., Stirling L. A., Ornston L. N., Perry J. J. Intergeneric evolutionary homology revealed by the study of protocatechuate 3,4-dioxygenase from Azotobacter vinelandii. Biochemistry. 1980 Jan 8;19(1):149–155. doi: 10.1021/bi00542a023. [DOI] [PubMed] [Google Scholar]
- Entsch B., Nan Y., Weaich K., Scott K. F. Sequence and organization of pobA, the gene coding for p-hydroxybenzoate hydroxylase, an inducible enzyme from Pseudomonas aeruginosa. Gene. 1988 Nov 30;71(2):279–291. doi: 10.1016/0378-1119(88)90044-3. [DOI] [PubMed] [Google Scholar]
- Gerischer U., Dürre P. Cloning, sequencing, and molecular analysis of the acetoacetate decarboxylase gene region from Clostridium acetobutylicum. J Bacteriol. 1990 Dec;172(12):6907–6918. doi: 10.1128/jb.172.12.6907-6918.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hadero A., Crawford I. P. Nucleotide sequence of the genes for tryptophan synthase in Pseudomonas aeruginosa. Mol Biol Evol. 1986 May;3(3):191–204. doi: 10.1093/oxfordjournals.molbev.a040388. [DOI] [PubMed] [Google Scholar]
- Hanahan D. Studies on transformation of Escherichia coli with plasmids. J Mol Biol. 1983 Jun 5;166(4):557–580. doi: 10.1016/s0022-2836(83)80284-8. [DOI] [PubMed] [Google Scholar]
- Hartnett C., Neidle E. L., Ngai K. L., Ornston L. N. DNA sequences of genes encoding Acinetobacter calcoaceticus protocatechuate 3,4-dioxygenase: evidence indicating shuffling of genes and of DNA sequences within genes during their evolutionary divergence. J Bacteriol. 1990 Feb;172(2):956–966. doi: 10.1128/jb.172.2.956-966.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harwood C. S., Rivelli M., Ornston L. N. Aromatic acids are chemoattractants for Pseudomonas putida. J Bacteriol. 1984 Nov;160(2):622–628. doi: 10.1128/jb.160.2.622-628.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hattori M., Sakaki Y. Dideoxy sequencing method using denatured plasmid templates. Anal Biochem. 1986 Feb 1;152(2):232–238. doi: 10.1016/0003-2697(86)90403-3. [DOI] [PubMed] [Google Scholar]
- Hedges R. W., Jacob A. E., Crawford I. P. Wide ranging plasmid bearing the Pseudomonas aeruginosa tryptophan synthase genes. Nature. 1977 May 19;267(5608):283–284. doi: 10.1038/267283a0. [DOI] [PubMed] [Google Scholar]
- Hosokawa K. Regulation of synthesis of early enzymes of p-hydroxybenzoate pathway in Pseudomonas putida. J Biol Chem. 1970 Oct 25;245(20):5304–5308. [PubMed] [Google Scholar]
- Howard J. B., Zieske L., Clarkson J., Rathe L. Mechanism-based fragmentation of coenzyme A transferase. Comparison of alpha 2-macroglobulin and coenzyme A transferase thiol ester reactions. J Biol Chem. 1986 Jan 5;261(1):60–65. [PubMed] [Google Scholar]
- Hughes E. J., Shapiro M. K., Houghton J. E., Ornston L. N. Cloning and expression of pca genes from Pseudomonas putida in Escherichia coli. J Gen Microbiol. 1988 Nov;134(11):2877–2887. doi: 10.1099/00221287-134-11-2877. [DOI] [PubMed] [Google Scholar]
- Keen N. T., Tamaki S., Kobayashi D., Trollinger D. Improved broad-host-range plasmids for DNA cloning in gram-negative bacteria. Gene. 1988 Oct 15;70(1):191–197. doi: 10.1016/0378-1119(88)90117-5. [DOI] [PubMed] [Google Scholar]
- Lee S. Y., Rasheed S. A simple procedure for maximum yield of high-quality plasmid DNA. Biotechniques. 1990 Dec;9(6):676–679. [PubMed] [Google Scholar]
- Lin T. W., Bridger W. A. Sequence of a cDNA clone encoding pig heart mitochondrial CoA transferase. J Biol Chem. 1992 Jan 15;267(2):975–978. [PubMed] [Google Scholar]
- Moore S. A., Jencks W. P. Formation of active site thiol esters of CoA transferase and the dependence of catalysis on specific binding interactions. J Biol Chem. 1982 Sep 25;257(18):10893–10907. [PubMed] [Google Scholar]
- Ornston L. N., Parke D. Properties of an inducible uptake system for beta-ketoadipate in Pseudomonas putida. J Bacteriol. 1976 Feb;125(2):475–488. doi: 10.1128/jb.125.2.475-488.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ornston L. N., Parke D. The evolution of induction mechanisms in bacteria: insights derived from the study of the beta-ketoadipate pathway. Curr Top Cell Regul. 1977;12:209–262. doi: 10.1016/b978-0-12-152812-6.50011-1. [DOI] [PubMed] [Google Scholar]
- Ornston L. N., Stanier R. Y. The conversion of catechol and protocatechuate to beta-ketoadipate by Pseudomonas putida. J Biol Chem. 1966 Aug 25;241(16):3776–3786. [PubMed] [Google Scholar]
- Ornston L. N. The conversion of catechol and protocatechuate to beta-ketoadipate by Pseudomonas putida. IV. Regulation. J Biol Chem. 1966 Aug 25;241(16):3800–3810. [PubMed] [Google Scholar]
- Parke D., Ornston L. N. Constitutive synthesis of enzymes of the protocatechuate pathway and of the beta-ketoadipate uptake system in mutant strains of Pseudomonas putida. J Bacteriol. 1976 Apr;126(1):272–281. doi: 10.1128/jb.126.1.272-281.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Parke D., Rynne F., Glenn A. Regulation of phenolic catabolism in Rhizobium leguminosarum biovar trifolii. J Bacteriol. 1991 Sep;173(17):5546–5550. doi: 10.1128/jb.173.17.5546-5550.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pearson W. R., Lipman D. J. Improved tools for biological sequence comparison. Proc Natl Acad Sci U S A. 1988 Apr;85(8):2444–2448. doi: 10.1073/pnas.85.8.2444. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Raibaud O., Schwartz M. Positive control of transcription initiation in bacteria. Annu Rev Genet. 1984;18:173–206. doi: 10.1146/annurev.ge.18.120184.001133. [DOI] [PubMed] [Google Scholar]
- Russell J. J., Patel M. S. Purification and properties of succinyl-CoA:3-oxo-acid CoA-transferase from rat brain. J Neurochem. 1982 May;38(5):1446–1452. doi: 10.1111/j.1471-4159.1982.tb07924.x. [DOI] [PubMed] [Google Scholar]
- Scherf U., Buckel W. Purification and properties of 4-hydroxybutyrate coenzyme A transferase from Clostridium aminobutyricum. Appl Environ Microbiol. 1991 Sep;57(9):2699–2702. doi: 10.1128/aem.57.9.2699-2702.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sharp J. A., Edwards M. R. Purification and properties of succinyl-coenzyme A-3-oxo acid coenzyme A-transferase from sheep kidney. Biochem J. 1978 Sep 1;173(3):759–765. doi: 10.1042/bj1730759. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shine J., Dalgarno L. The 3'-terminal sequence of Escherichia coli 16S ribosomal RNA: complementarity to nonsense triplets and ribosome binding sites. Proc Natl Acad Sci U S A. 1974 Apr;71(4):1342–1346. doi: 10.1073/pnas.71.4.1342. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sramek S. J., Frerman F. E. Escherichia coli coenzyme A-transferase: kinetics, catalytic pathway and structure. Arch Biochem Biophys. 1975 Nov;171(1):27–35. doi: 10.1016/0003-9861(75)90003-x. [DOI] [PubMed] [Google Scholar]
- Sramek S. J., Frerman F. E. Purification and properties of Escherichia coli coenzyme A-transferase. Arch Biochem Biophys. 1975 Nov;171(1):14–26. doi: 10.1016/0003-9861(75)90002-8. [DOI] [PubMed] [Google Scholar]
- Stanier R. Y., Ornston L. N. The beta-ketoadipate pathway. Adv Microb Physiol. 1973;9(0):89–151. [PubMed] [Google Scholar]
- Stewart G. S., Lubinsky-Mink S., Jackson C. G., Cassel A., Kuhn J. pHG165: a pBR322 copy number derivative of pUC8 for cloning and expression. Plasmid. 1986 May;15(3):172–181. doi: 10.1016/0147-619x(86)90035-1. [DOI] [PubMed] [Google Scholar]
- Tabor S., Richardson C. C. A bacteriophage T7 RNA polymerase/promoter system for controlled exclusive expression of specific genes. Proc Natl Acad Sci U S A. 1985 Feb;82(4):1074–1078. doi: 10.1073/pnas.82.4.1074. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tung K. K., Wood W. A. Purification, new assay, and properties of coenzyme A transferase from Peptostreptococcus elsdenii. J Bacteriol. 1975 Dec;124(3):1462–1474. doi: 10.1128/jb.124.3.1462-1474.1975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- West S. E., Iglewski B. H. Codon usage in Pseudomonas aeruginosa. Nucleic Acids Res. 1988 Oct 11;16(19):9323–9335. doi: 10.1093/nar/16.19.9323. [DOI] [PMC free article] [PubMed] [Google Scholar]
- White H., Jencks W. P. Properties of succinyl-CoA:3-ketoacid coenzyme A transferase. J Biol Chem. 1976 Mar 25;251(6):1708–1711. [PubMed] [Google Scholar]
- White H., Solomon F., Jencks W. P. Utilization of the inactivation rate of coenzyme A transferase by thiol reagents to determine properties of the enzyme-CoA intermediate. J Biol Chem. 1976 Mar 25;251(6):1700–1707. [PubMed] [Google Scholar]
- Wierenga R. K., Terpstra P., Hol W. G. Prediction of the occurrence of the ADP-binding beta alpha beta-fold in proteins, using an amino acid sequence fingerprint. J Mol Biol. 1986 Jan 5;187(1):101–107. doi: 10.1016/0022-2836(86)90409-2. [DOI] [PubMed] [Google Scholar]
- Wiesenborn D. P., Rudolph F. B., Papoutsakis E. T. Coenzyme A transferase from Clostridium acetobutylicum ATCC 824 and its role in the uptake of acids. Appl Environ Microbiol. 1989 Feb;55(2):323–329. doi: 10.1128/aem.55.2.323-329.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yeh W. K., Fletcher P., Ornston L. N. Evolutionary divergence of co-selected beta-ketoadipate enol-lactone hydrolases in Acinetobacter calcoaceticus. J Biol Chem. 1980 Jul 10;255(13):6342–6346. [PubMed] [Google Scholar]
- Yeh W. K., Ornston L. N. Evolutionarily homologous alpha 2 beta 2 oligomeric structures in beta-ketoadipate succinyl-CoA transferases from Acinetobacter calcoaceticus and Pseudomonas putida. J Biol Chem. 1981 Feb 25;256(4):1565–1569. [PubMed] [Google Scholar]
- Yeh W. K., Ornston L. N. p-Chloromercuribenzoate specifically modifies thiols associated with the active sites of beta-ketoadipate enol-lactone hydrolase and succinyl CoA: beta-ketoadipate CoA transferase. Arch Microbiol. 1984 Jun;138(2):102–105. doi: 10.1007/BF00413008. [DOI] [PubMed] [Google Scholar]