Abstract
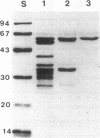
The xynC gene of Fibrobacter succinogenes S85 codes for a 66.4-kDa xylanase which consists of three distinct domains separated by two flexible regions rich in serine residues. Domains A and B of XynC code for catalytic domains with 56.5% identity and 9.6% similarity with each other, and both domains share homology with xylanases of Ruminococcus flavefaciens, Neocallimastix patriciarum, Clostridium acetobutylicum, Bacillus pumilus, Bacillus subtilis, and Bacillus circulans. More than 88% of the xylanase activity of Escherichia coli cells carrying the original 13-kb recombinant plasmid was released from intact cells by cold water washes. The major products of hydrolysis of xylan by both domains were xylose and xylobiose, indicating that the xynC gene product exhibits catalytic properties similar to those of the XynA xylanases from R. flavefaciens and N. patriciarum. So far, these features are not shared broadly with bacteria from other environments and may indicate specific selection for this domain structure in the highly competitive environment of the rumen.
Full text
PDF






Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bolivar F., Backman K. Plasmids of Escherichia coli as cloning vectors. Methods Enzymol. 1979;68:245–267. doi: 10.1016/0076-6879(79)68018-7. [DOI] [PubMed] [Google Scholar]
- Bradford M. M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem. 1976 May 7;72:248–254. doi: 10.1016/0003-2697(76)90527-3. [DOI] [PubMed] [Google Scholar]
- Bueno A., Vazquez de Aldana C. R., Correa J., Villa T. G., del Rey F. Synthesis and secretion of a Bacillus circulans WL-12 1,3-1,4-beta-D-glucanase in Escherichia coli. J Bacteriol. 1990 Apr;172(4):2160–2167. doi: 10.1128/jb.172.4.2160-2167.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cavicchioli R., East P. D., Watson K. endAFS, a novel family E endoglucanase gene from Fibrobacter succinogenes AR1. J Bacteriol. 1991 May;173(10):3265–3268. doi: 10.1128/jb.173.10.3265-3268.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Flint H. J., Martin J., McPherson C. A., Daniel A. S., Zhang J. X. A bifunctional enzyme, with separate xylanase and beta(1,3-1,4)-glucanase domains, encoded by the xynD gene of Ruminococcus flavefaciens. J Bacteriol. 1993 May;175(10):2943–2951. doi: 10.1128/jb.175.10.2943-2951.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gilbert H. J., Hazlewood G. P., Laurie J. I., Orpin C. G., Xue G. P. Homologous catalytic domains in a rumen fungal xylanase: evidence for gene duplication and prokaryotic origin. Mol Microbiol. 1992 Aug;6(15):2065–2072. doi: 10.1111/j.1365-2958.1992.tb01379.x. [DOI] [PubMed] [Google Scholar]
- Gilkes N. R., Henrissat B., Kilburn D. G., Miller R. C., Jr, Warren R. A. Domains in microbial beta-1, 4-glycanases: sequence conservation, function, and enzyme families. Microbiol Rev. 1991 Jun;55(2):303–315. doi: 10.1128/mr.55.2.303-315.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hanahan D. Studies on transformation of Escherichia coli with plasmids. J Mol Biol. 1983 Jun 5;166(4):557–580. doi: 10.1016/s0022-2836(83)80284-8. [DOI] [PubMed] [Google Scholar]
- Hopp T. P., Woods K. R. Prediction of protein antigenic determinants from amino acid sequences. Proc Natl Acad Sci U S A. 1981 Jun;78(6):3824–3828. doi: 10.1073/pnas.78.6.3824. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee J. M., Hu Y., Zhu H., Cheng K. J., Krell P. J., Forsberg C. W. Cloning of a xylanase gene from the ruminal fungus Neocallimastix patriciarum 27 and its expression in Escherichia coli. Can J Microbiol. 1993 Jan;39(1):134–139. doi: 10.1139/m93-020. [DOI] [PubMed] [Google Scholar]
- Matte A., Forsberg C. W. Purification, characterization, and mode of action of endoxylanases 1 and 2 from Fibrobacter succinogenes S85. Appl Environ Microbiol. 1992 Jan;58(1):157–168. doi: 10.1128/aem.58.1.157-168.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McDermid K. P., Forsberg C. W., MacKenzie C. R. Purification and properties of an acetylxylan esterase from Fibrobacter succinogenes S85. Appl Environ Microbiol. 1990 Dec;56(12):3805–3810. doi: 10.1128/aem.56.12.3805-3810.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McGavin M. J., Forsberg C. W., Crosby B., Bell A. W., Dignard D., Thomas D. Y. Structure of the cel-3 gene from Fibrobacter succinogenes S85 and characteristics of the encoded gene product, endoglucanase 3. J Bacteriol. 1989 Oct;171(10):5587–5595. doi: 10.1128/jb.171.10.5587-5595.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Neu H. C., Heppel L. A. The release of enzymes from Escherichia coli by osmotic shock and during the formation of spheroplasts. J Biol Chem. 1965 Sep;240(9):3685–3692. [PubMed] [Google Scholar]
- Sipat A., Taylor K. A., Lo R. Y., Forsberg C. W., Krell P. J. Molecular cloning of a xylanase gene from Bacteroides succinogenes and its expression in Escherichia coli. Appl Environ Microbiol. 1987 Mar;53(3):477–481. doi: 10.1128/aem.53.3.477-481.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smith D. C., Forsberg C. W. alpha-Glucuronidase and Other Hemicellulase Activities of Fibrobacter succinogenes S85 Grown on Crystalline Cellulose or Ball-Milled Barley Straw. Appl Environ Microbiol. 1991 Dec;57(12):3552–3557. doi: 10.1128/aem.57.12.3552-3557.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Teather R. M., Erfle J. D. DNA sequence of a Fibrobacter succinogenes mixed-linkage beta-glucanase (1,3-1,4-beta-D-glucan 4-glucanohydrolase) gene. J Bacteriol. 1990 Jul;172(7):3837–3841. doi: 10.1128/jb.172.7.3837-3841.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Teather R. M., Wood P. J. Use of Congo red-polysaccharide interactions in enumeration and characterization of cellulolytic bacteria from the bovine rumen. Appl Environ Microbiol. 1982 Apr;43(4):777–780. doi: 10.1128/aem.43.4.777-780.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xue G. P., Gobius K. S., Orpin C. G. A novel polysaccharide hydrolase cDNA (celD) from Neocallimastix patriciarum encoding three multi-functional catalytic domains with high endoglucanase, cellobiohydrolase and xylanase activities. J Gen Microbiol. 1992 Nov;138(11):2397–2403. doi: 10.1099/00221287-138-11-2397. [DOI] [PubMed] [Google Scholar]
- Yang R. C., MacKenzie C. R., Narang S. A. Nucleotide sequence of a Bacillus circulans xylanase gene. Nucleic Acids Res. 1988 Jul 25;16(14B):7187–7187. doi: 10.1093/nar/16.14.7187. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zappe H., Jones W. A., Woods D. R. Nucleotide sequence of a Clostridium acetobutylicum P262 xylanase gene (xynB). Nucleic Acids Res. 1990 Apr 25;18(8):2179–2179. doi: 10.1093/nar/18.8.2179. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang J. X., Flint H. J. A bifunctional xylanase encoded by the xynA gene of the rumen cellulolytic bacterium Ruminococcus flavefaciens 17 comprises two dissimilar domains linked by an asparagine/glutamine-rich sequence. Mol Microbiol. 1992 Apr;6(8):1013–1023. doi: 10.1111/j.1365-2958.1992.tb02167.x. [DOI] [PubMed] [Google Scholar]