Abstract
Selective androgen receptor modulators (SARMs) are a novel class of androgen receptor (AR) ligands that might change the future of androgen therapy dramatically. With improved pharmacokinetic characteristics and tissue-selective pharmacological activities, SARMs are expected to greatly extend the clinical applications of androgens to osteoporosis, muscle wasting, male contraception and diseases of the prostate. Mechanistic studies with currently available SARMs will help to define the contributions of differential tissue distribution, tissue-specific expression of 5α-reductase, ligand-specific regulation of gene expression and AR interactions with tissue-specific coactivators to their observed tissue selectivity, and lead to even greater expansion of selective anabolic therapies.
Androgen receptor ligands
Androgens are important in male physiology because of their essential roles in male sexual differentiation, male puberty changes, maintenance of muscle and bone mass, prostate growth, and spermatogenesis in adults. The androgen receptor (AR) is a member of the steroid and nuclear receptor superfamily, and an important drug target. Various AR ligands have been discovered and developed for the treatment of, for example, male hypogonadism, muscle wasting, anemia, benign prostate hyperplasia (BPH) and prostate cancer. Pharmacologically, AR ligands are classified as agonists (androgens) and antagonists (anti-androgens), based on their ability to either activate or inhibit the transcription of AR target genes. Structurally, there are steroidal and non-steroidal ligands. The endogenous androgens, testosterone (T) and dihydrotestosterone (DHT), are both steroidal AR agonists.
Steroids
In the early years (1950s) of development of AR ligands, research efforts were devoted solely to structural modification of endogenous steroids and led to the discovery of various synthetic steroids, including agonists and antagonists [1-3]. T and DHT are not available orally, so most of the early structural modifications were made to either improve the oral bioavailability of the steroidal ligands or to separate the androgenic and anabolic activities of androgens. T esters and 17α-alkylated steroids are often used to improve either the duration of action or the bioavailability of steroidal androgens. T esters work as delayed-release forms of T, but produce inconsistent pharmacokinetic profiles that severely hamper their clinical use. 17α-alkylated androgens possess much improved oral bioavailability and half-life, but their chronic use is often associated with severe hepatotoxicity. Further development of steroidal ligands has been limited by the rigid structural features of the steroidal plane and its perceived, but recently disproved, importance for AR binding [4-6].
The clearest therapeutic indication for androgens is hypogonadism in men. Various T preparations and synthetic steroidal androgens (e.g. methyltestosterone and fluoxymesterone) have been used to treat both primary (testicular) and secondary (lack of gonadotropins) hypogonadism. Although treatment with T is effective generally, the efficacy of treatment and serum T levels need to be monitored carefully because of the inconsistent pharmacokinetic profile of most preparations. Orally available synthetic steroidal androgens (e.g. nandrolone and oxandrolone) are also used, but the application of these ligands is limited by their hepatotoxicity and cross-reactivity with other steroid receptors. Overall, direct supplementation with T appears to be relatively safe, with concerns related to the increasing incidence of BPH and prostate cancer in older men representing the greatest barrier to expanded clinical use of androgens. In an attempt to separate the androgenic and anabolic activities of androgens, 19-norandrogens (e.g. nandrolone) have been designed. Separation of the androgenic and anabolic activities of androgens in different tissues (e.g. androgenic tissues such as the prostate and skin versus anabolic tissues like muscle and bone) might overcome many of these limitations and concerns. However, most 19-norandrogens retain the unfavorable pharmacokinetic and toxicological properties of their steroidal relatives. Therefore, the clinical applications of androgens are limited by poor tissue selectivity, pharmacokinetics and toxicity.
Non-steroidal ligands allow more flexibility in the choices of various pharmacophores and further structural modification, and were proposed and developed to overcome the limitations of steroidal ligands by achieving higher specificity for AR, and improved oral bioavailability, in vivo pharmacokinetics and tissue selectivity. The earliest of these compounds, which were discovered in the 1970s, act as antagonists and are still used in the management of prostate cancer (Table 1). Although these nonsteroidal anti-androgens possess two of the desirable properties, they do not have meaningful tissue selectivity. Interest in developing non-steroidal androgens has been kindled by the successful development and marketing of selective estrogen receptor modulators (SERMs) in the 1980s and 1990s. The major purpose in developing non-steroidal, selective androgen receptor modulators (SARMs) is to achieve tissue-selective modulation of AR action, so minimizing undesirable side-effects that are normally associated with steroidal androgens and capitalizing on more potent anabolic effects to exploit previously untenable therapeutic applications of AR ligands.
TABLE 1.
Non-steroidal anti-androgens
SARMs
In general, SARMs are defined as tissue-selective AR ligands [7]. An ideal SARM should also have (i) high specificity for the AR, (ii) improved oral bioavailability and a pharmacokinetic profile that allows once-a-day administration and, most importantly, (iii) desirable, tissue-selective pharmacological activities. The major discriminating criterion is tissue selectivity of the ligand in vivo, so both agonists and antagonists, and steroidal and non-steroidal ligands might be classified as SARMs. The major goal in the development of SARMs is to avoid the undesirable side-effects of treatment by improving the tissue selectivity of the ligands. This would greatly extend the clinical applications of these ligands beyond primary and secondary hypogonadism. Other potential applications for SARMs include hormone-replacement therapy, osteoporosis, muscle wasting, male contraception, BPH and prostate cancer [8].
The dual effects of SARMs in muscle and bone might be particularly advantageous for the treatment of osteoporosis, because an increase in muscle mass and strength might help rebuild bone mass and also reduce the risk of fracture because of a reduced chance of falls. Although the protective effects of endogenous T on bone are mediated partially by an indirect action through the estrogen receptor (ER) following the conversion of T to estradiol by aromatase (Figure 1), direct activation of the AR also has strong anabolic and anti-resorptive effects on bone [9]. With the exception of parathyroid hormone, which requires injection and is limited to the treatment of severe osteoporosis because its side-effects and risk of osteosarcoma, all the treatments for osteoporosis that are available currently are anti-resorptive agents. An orally bioavailable SARM with strong anabolic effects on bone would, thus, be a unique alternative for the treatment of osteoporosis, including aging-related (i.e. loss of endogenous estrogen and/or androgen) osteoporosis and glucocorticoid-induced (chronic use of corticosteroids) osteoporosis. Such SARMs with little or no androgenic activities would be safer choices considering the increased risk of prostatic diseases in elderly men and concerns related to the use of T. Similarly, the anabolic actions of SARMs in skeletal muscle can also be used to treat muscle-wasting conditions such as frailty and those caused by severe burn injury, cancer, end-stage renal disease and AIDS. The increase in net protein balance afforded by an anabolic SARM would provide an innovative, safer approach to help rebuild muscle mass and strength.
FIGURE 1.

Three modes of action of T. T might act directly through ARs, be converted to DHT by 5α-reductase before binding to ARs, and be aromatized to estrogen and act through the ER.
One of the biggest potential advantages of anabolic SARMs is that these ligands might be safe to use in women. With improved tissue selectivity, SARMs that maintain the anabolic actions of androgens without causing the undesirable virilizing effects associated with traditional androgen therapies would greatly expand the therapeutic options for androgen use in women, opening the door to potential applications in hormone-replacement therapy (HRT) in postmenopausal women, sexual function, osteoporosis, and disease- and aging-related muscle wasting conditions in women.
In addition to their androgenic and anabolic activities, androgens also regulate the function of the hypothalamus–pituitary–testis axis, including the feedback regulation of the release of gonadotropin releasing hormone (GnRH) from the hypothalamus and luteinizing hormone (LH) from the pituitary, and the downstream production of T (Leydig cell) and spermatogenesis (Sertoli cell). Supraphysiological concentrations of T have suppressive effects on GnRH release and spermatogenesis, and can be used for male contraception. However, large variability has been observed in the spermatogenic response to T therapy, and supraphysiological concentrations of T often cause undesirable side-effects such as acne, changes in serum lipoproteins and potential exacerbation of BPH. Combination therapy with progestins, which suppress gonadotropin release, and physiological concentrations of exogenous T is a more effective, safer strategy for male contraception [10], but the application of this method is hampered by the unpractical dosing regimens of the steroidal androgens. Nonsteroidal AR agonists with improved oral availability might provide the opportunity to develop a ‘male pill’. Pharmacologically, efficient suppression of GnRH and gonadotropins at the hypothalamic and pituitary levels requires a strong AR agonist that has good CNS distribution and/or selectivity. In addition, strong agonist activity in peripheral tissues is necessary to maintain normal androgen functions in the body.
The brain is also an important androgen-target tissue because of the role of T in mood, libido and cognition. It is recognized that androgen administration helps to improve libido and mood in both men and women. Therefore, the positive effects of SARMs on libido would be crucial when used as a male contraceptive, and beneficial when used for HRT. Furthermore, sufficient distribution of SARMs in the CNS is important for maintaining libido during treatment.
As discussed previously, hepatotoxicity and changes in the serum lipid profile are common adverse effects of traditional androgen therapy. Hepatotoxicity caused by synthetic steroidal androgens is related more to the chemical structure and metabolism of these ligands [11], whereas changes in lipid profile are related more to AR action [12,13]. The most consistent change caused by androgen therapy is a decrease in serum high-density lipoprotein cholesterol (HDL-C) levels, which is sometimes considered as a risk factor for cardiovascular disease [14]. However, physiological doses of T cause only minor decreases (10–20%) in serum HDL-C levels, rather than the significant decreases induced by supraphysiological doses of T. Further, it is crucial to note that the minor decreases in HDL-C might reflect accelerated reverse cholesterol transport and, ultimately, provide cardiovascular benefit [14]. Although SARMs that have no effects on HDL-C profile might be considered optimal, minor decreases in HDL-C should be well tolerated. One possible exception to this might be the single-agent use of SARMs in male contraception, where supraphysiological doses are likely to be required for efficient suppression of GnRH and gonadotropins. As such, the absence of adverse effects on lipid profile will be necessary considering the safety of long-term administration necessary for contraception.
The risk of prostatic disease in men, mainly BPH and prostate cancer, increases with age. Urinary obstruction is the main symptom of BPH, and it appears to be caused by both physical obstruction (static or mechanic component) and/or contractions of smooth muscles under α-adrenergic receptor-mediated sympathetic stimulation (dynamic component) [15]. Stimulation by both androgens and estrogen is believed to be responsible for the enlargement of the tissue. As such, suppression of AR and ER actions with an aromatase inhibitor (Figure 1) [16-18] has been used as therapeutically in BPH. Androgen suppression primarily causes the regression of the epithelial elements of the prostate, whereas aromatase inhibitors are believed to suppress the size of the stromal component and the stromal–epithelial interactions in the prostate. The most commonly used drugs for androgen suppression include anti-androgens (e.g. flutamide) and 5α-reductase inhibitors (e.g. finasteride). Anti-androgen treatment causes severe side-effects because the complete, nonselective inhibition of AR action; 5α-reductase inhibitors are considered more ‘tissue selective’ because of tissue-specific expression of 5α-reductase. However, inhibition of 5α-reductase increases estrogen production in adipose tissue, breast (gynecomastia) and prostate (proliferation of the stromal components). SARMs with either partial agonist or anti-androgen actions in the prostate might provide a novel approach for androgen suppression in BPH treatment, without causing side-effects in the anabolic tissue and hypothalamus–pituitary–testis axis. SARMs with partial-agonist activity in the prostate and full-agonist activity in muscle and other peripheral tissues would decrease prostate size (because of their ability to work as antagonists in the presence of endogenous DHT) while avoiding the side-effects caused by nonselective inhibition by anti-androgens. Tissue selective anti-androgens are a novel approach for the treatment of BPH and prostate cancer, and can be considered as a new class of SARMs. These ligands should behave as antagonists in the prostate with either no activity or agonist activity in other target tissues. However, discovering this new class of ligands might be challenging because the molecular mechanism of AR action is not well understood.
AR action
AR is expressed mainly in androgen target tissues such as the prostate, skeletal muscle, liver, skin and CNS, with the highest expression observed in the prostate, adrenal gland and epididymis, determined by real-time PCR [19]. Unlike ER, which has two isoforms, ERα and ERβ, as distinct gene products [20], only one AR gene has been identified in humans. Both T and its active metabolite, DHT, are endogenous androgens that activate the AR.
Although T is the major form of circulating androgen, most is converted to DHT by 5α-reductase (type 1 and type 2) in the prostate, skin and liver [21]; a small amount of T (0.2%) is also converted to estradiol by aromatase (Figure 1). Both DHT and estradiol are active ‘metabolites’, so T is thought to have three modes of action. It might directly act through the AR in target tissues in which 5α-reductase is either not present or present at very low levels, it might be converted to DHT (5–10% in blood, >90% in prostate) [22] by 5α-reductase before binding to AR, and it might be aromatized to estrogen that acts through the ER [1]. Because DHT is a potent androgen that binds to the AR with a higher affinity than T [23], the conversion of T to DHT is considered a natural ‘amplification’ process in ‘DHT-dependent’ tissues because 5α-reductase is not expressed in all androgen-target tissues. Type 2 5α-reductase is present mainly in the prostate whereas type 1 5α-reductase is found mainly in liver and skin [24]. Both isozymes are expressed at a much lower level in other peripheral tissues, including skeletal muscle, and the CNS. By contrast, estrogen has a major role in regulating metabolic processes [25,26], mood and cognition [27], cardiovascular disease [28,29], sexual function including libido [30], and bone turnover in men [31,32].
At the cellular level, unbound AR is located mainly in the cytoplasm where it is associated with a complex of heat shock proteins (HSPs), mainly through the interactions with the ligand binding domain (LBD) [33]. On agonist binding, AR goes through a series of conformational changes, the HSPs dissociate from the AR, and the transformed AR undergoes dimerization, post-translational modification and translocation to the nucleus. The translocated receptor then binds to various androgen response elements (AREs), and recruits other transcription regulators (including co-activators and co-repressors) [34] and transcriptional machinery [35], which ensures the transactivation of AR-regulated gene expression. This process is the classical genomic function of AR.
In addition to the genomic pathway, a non-genomic AR pathway has been reported in oocytes [36], skeletal muscle cells [37], osteoblasts [38,39] and prostate cancer cells [40,41]. Compared to the genomic pathway, the non-genomic actions of steroid receptors are characterized by the rapidity of action (seconds to ∼1 hour) and interaction with plasma membrane-associated signaling pathways [42]. Nevertheless, the structural basis for non-genomic activity is the direct interaction between AR and cytosolic proteins from different signaling pathways [43]. However, the physiological role of the non-genomic effects of the AR are yet to be defined.
Regardless of the pathway, AR activity involves ligand-modulated conformational change of the receptor, which further affects the interactions between AR and other proteins, either co-regulators and transcription factors (genomic pathway) or signaling cascade proteins (non-genomic pathway). Although the interactions between AR and other proteins are crucial for AR functions, not all protein–protein interactions are characterized. The interactions between AR and co-regulators are best understood, and are mediated by different functional domains of the receptor [1], mainly through the N-terminal domain (NTD) and LBD, and, occasionally, the DNA-binding domain [34]. Structural biology studies [44]* indicate that ligand binding might regulate LBD-mediated interactions directly by affecting the conformation and/or surface topology of the LBD. In addition, AR might undergo N/C (N-terminal and C-terminal) interaction on agonist binding, so ligand binding might also regulate the conformation of the NTD and its interaction with co-regulators indirectly.
Most of the interactions between the LBD and the co-regulators identified so far have been mapped to a specific surface region, called activation function 2 (AF2), of the LBD. Both co-activator signature motifs, including LxxLL and FxxLF, and NTD motifs (F23xxLF27 and W433xxLF437) interact specifically with the AF2 and regulate AR transcriptional activation. Several regions of the NTD [45], including AF1, have also been shown to be important for the recruitment of co-regulators, but the interface is not yet defined because of the lack of detailed structural information on the NTD. AR agonist-binding appears to preferentially recruit co-activators, whereas antagonist binding seems to preferentially recruit corepressors [46]. It has been shown that co-repressor binding motifs (NCoR box) also interact specifically with the AF2 region in either the unbound AR or antagonist-bound AR [46]. It is clear that ligand binding regulates the conformation of the LBD and subsequent protein–protein interactions, which makes it possible to regulate AR action by introducing ligand-specific conformational changes in the receptor.
Strategies to achieve tissue selectivity
As mentioned above, different clinical applications prefer different tissue-selectivity profiles for the AR ligand. As multiple mechanisms of action might contribute to the tissue selectivity of AR ligands, various strategies might be used to achieve tissue-selective pharmacological activity in vivo.
One early trend of thought was that high-affinity AR ligands distribute preferentially to the tissues with high concentrations of AR, like the prostate, which would limit the activities of these ligands in other tissues and reduce the side-effects of therapy. Animal studies [47] show higher uptake of radio-labeled androgens in the prostate, and the tissue-uptake efficiency and selectivity of different ligands seem to be related to their binding affinity for the AR and their resistance to metabolism. This raises the possibility of developing anti-androgens with high-binding affinity as tissue selective anti-androgens. However, recent studies [48] dispel this notion and indicate that preferential tissue distribution has either little or no role in tissue selectivity, especially for non-steroidal ligands.
Of the mechanisms that have been proposed to achieve tissue selectivity of AR ligands, the most definitive evidence exists for the role of 5α-reductase. The tissue-specific expression 5α-reductase makes it a unique contributor to tissue selectivity. Specific inhibition of the type 2 enzyme by finasteride blocks the conversion of T to DHT in the prostate, and T exhibits partial agonist activity in the regrowth of this tissue in castrated rats (Figure 2a) [22]. Therefore, for SARMs such as T, which have relatively low intrinsic activity and/or potency (Figure 2b) and do not interact with 5α-reductase [49], tissue selectivity (Figure 2c) might be caused simply by their partial agonist activity in the prostate. Most SARMs (Table 2) with verified tissue selectivity appear to be partial agonists in the prostate, which indicates that their tissue selectivity is related, at least partially, to the tissue-specific expression of 5α-reductase.
FIGURE 2.
Tissue-specific expression of 5α-reductase contributes to the tissue selectivity of SARMs. (a) Partial agonist activity of T in the prostate when co-administered with finasteride, a type 2 5α-reductase inhibitor. Redrawn with permission from Ref. [22]. (b) Partial agonist activity of a SARM (aryl propionamide analog S-4) in the prostate when administered alone. Redrawn with permission from Ref. [54]. (c) Full agonist activity of the aryl propionamide analog S-4 in the levator ani muscle. Redrawn with permission from Ref. [54]. Data in (a) was obtained using a prostate regrowth model in castrated rats [22], whereas data in (b,c) were obtained using a prostate maintenance model in castrated rats [54]. Because S-4 has similar effects in maintenance [54] and re-growth [60] models, the more detailed dose–response data from the maintenance model are used in (b) and (c).
TABLE 2.
Non-steroidal AR agonists
| Chemotype | General chemical structure | Lead compound | |
|---|---|---|---|
| Quinolinone analogs |  |
LGD2226 |  |
| Aryl propionamide analogs | S4 | ||
| S1 | |||
| C6 | |||
| Hydantoin analogs | 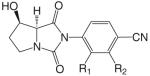 |
BMS564929 |  |
| Tetrahydro-quinoline analogs | 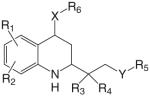 |
S40503 | 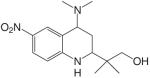 |
If tissue selectivity can be achieved by lack of interaction with 5α-reductase, it might also be achieved by tissue-specific metabolism of the ligands by 5α-reductase. Several approaches might make use of the potential tissue-specific conversion to develop SARMs, including (i) inactive parent compounds that are activated by type 2 5α-reductase in the prostate to form anti-androgens, (ii) AR agonists that are inactivated by type 2 5α-reductase in the prostate, and (iii) AR agonists that are converted to anti-androgens only by type 2 5α-reductase in the prostate. The feasibility of these approaches might be limited by the chemical structures of 5α-reductase substrates. However, the common interaction of T as substrate and ligand of 5α-reductase and AR, respectively, indicate that this might be exploited as another unique therapeutic application of SARMs.
As the mechanism of action of AR becomes better characterized, and more is learned about the tissue selectivity of SERMs, more research efforts have been devoted to the development of SARMs that target AR action at the molecular level. It has been proposed that either ligand-specific or tissue-specific regulation of gene expression might also serve as novel mechanism for tissue selectivity [1]. More specifically, ligand binding might initiate specific conformational changes in the LBD, to further modulate co-regulator recruitment and cause differential regulation of target gene expression. By contrast, co-regulator recruitment might also be affected by tissue-specific expression of the co-regulators, so ligand-specific interactions between AR and co-regulators might take place in some tissues but not the others. In this case, detailed characterization of the protein–protein interaction profile in the presence of different AR ligands is crucial to identifying SARMs. Furthermore, the tissue selectivity of these ligands needs to be verified in vivo. Although there is intense research interest in this area, it has not been proved that ligand-specific regulation of gene expression contributes directly to the in vivo tissue selectivity of SARMs.
The non-genomic pathway of AR action has been identified, but is not well understood. It is not clear how non-genomic effects contribute to AR function overall, and there is little structural information about how ligand binding affects the interaction between AR and proteins in signaling cascades. Nevertheless, by learning from the latest developments in SERM research [50], it is reasonable to speculate that tissue selectivity and unique therapeutic uses of AR ligands might be achieved by either separation of the genomic and non-genomic functions of the receptor using specific ligands [42,51], or separation of different, non-genomic pathways of AR. However, structural features that are essential for achieving this separation are yet to be identified.
Current development of SARMs
The discovery and development of SARMs is at an early stage. Non-steroidal AR agonists were reported first by Dalton et al. (aryl propionamide) [52] and Edwards et al. (quinoline) [53] in 1998. Several different structural classes of non-steroidal AR agonists have since been discovered and developed (Table 2), most of which adopt the pharmacophores from the non-steroidal anti-androgens (Table 1) that were developed in the 1970s and 1980s. The concept of a tissue-selective AR modulator was documented first in 1999, and the aryl propionamide SARMs were the first to demonstrate tissue selectivity in vivo in 2003 [54], followed later that year by a tetrahydroquinoline (THQ) SARM [55], and quinoline SARMs in 2006 [56]. Most of current research efforts have focused on the development of non-steroidal AR agonists as tissue-selective agents, but there is also interest in developing tissue-selective AR antagonist, and steroidal SARMs.
The chemistry of various classes of AR ligands has been reviewed extensively recently [1,57-59], and the in vivo pharmacological activities of these ligands have also been compared in detail according to the information released [58]. Most non-steroidal AR agonists are available orally, bind to AR with high affinity and demonstrate different degrees of tissue-selective pharmacological activities. Many compounds are still under preclinical development, with a few entering either phase I or phase II clinical trials, but no SARM has entered the market to date [58]. Only compounds with verified in vivo tissue selectivity (Table 2) are discussed in this review.
The leading compounds identified from the four major structural classes (Table 2), including the quinolinone analog LGD2226 [56], aryl propionamide analogs S1, S4 [49,54,60] and C6 [61], the bicyclic hydantoin analog BMS564929†, and the tetrahydroquinoline analog S-40503 [55], share similar pharmacological profiles, which include strong anabolic activity in skeletal muscle and bone, and partial agonist activity in the prostate. Because each of these lead compounds is orally available [58] and tissue selective in vivo, their potential applications in treatment have been assessed using various animal models, including muscle wasting (LGD2226, S4 and BMS56929) [56,60]†, osteoporosis (LGD2226, S4 and S-40503) [55,56,60], male contraception (C6) [61] and BPH (S1) [49]. Although the mechanisms of action of these ligands have not been investigated fully, available data [22,49] indicate that the tissue-specific expression of 5α-reductase contributes, at least partially, to the tissue selectivity of these ligands. These ligands might be valuable tools to understand the mechanisms of tissue selectivity, and further explore and identify novel strategies for the discovery of SARMs.
Compared with the development of tissue-selective agonist, there has been less progress in the development of tissue-selective anti-androgens. Earlier approaches focused on the development of peripheral-selective anti-androgens to reduce the elevation of serum T caused by blockage of the hypothalamus–pituitary–testis axis. Although animal studies [62] indicate that bicalutamide is a peripheral-selective agent, this selectivity does not apply to humans [63] because of species differences in the tissue distribution of bicalutamide.
Non-steroidal AR agonists with partial agonist activity might also be considered as tissue-selective anti-androgens because they behave as antagonist in the prostate in the presence of endogenous DHT. Animal studies indicate that these SARMs might be used to treat BPH [49], but their application in the treatment of prostate cancer has not been reported. The possibility of developing SARMs with pure anti-androgenic activity in the prostate and with either no activity or agonist activity in other target tissue for the treatment of prostate cancer, has not been reported.
Although most research has focused on the development of small molecules that modulate AR conformation and function, some research groups [64] are exploring the potential of peptide antagonists that can specifically disrupt the interactions between AR and co-regulators, and block AR action in the prostate. Further identification of receptor–protein interactions that are crucial and specific to AR-regulated gene expression in the prostate is required for the further development of these peptide antagonists. Also, the in vivo delivery and stability of these peptide ligands might be problematic but small, non-peptide molecules that mimic peptide binding might be a more realistic approach [65].
Targeting the interactions between AR and co-regulators might also be used as a strategy to design tissue-selective AR agonists, as discussed in the previous section. Ligand-specific conformational change of the LBD could alter receptor co-regulator interaction profile as assessed by peptide probes [44]*. However, for currently identified SARMs (ligands with verified in vivo tissue selectivity), it is not clear if ligand binding initiates ligand-specific interactions between the AR and co-regulators, or whether the potential ligand-specific interactions contribute to the observed tissue selectivity of these ligands. More detailed mechanism studies are required to acheive the rational design of novel SARMs based on the protein-interaction profile characterized in vitro.
Although structural modification of the steroidal ligands has been limited by the rigid structure of the steroidal plane, continuous research efforts [44] have been devoted to the development of steroidal SARMs. Preliminary in vitro studies [44] with some of the lead compounds have demonstrated ligand-specific changes in AR–coregulator interactions. However, the tissue selectivity and pharmacokinetic profile of these ligands in vivo have not been reported. As such, it is unclear whether these ligands have pharmacological activities in vivo, and whether the in vitro observations are related to these.
Future directions and challenges
Research into SARMs has made dramatic progress over the past decade, and several lead compounds with various chemical structures and in vivo tissue selectivity have been identified and developed, with some proceeding to clinical trials. Most of the discovery and development efforts continue to be devoted to nonsteroidal AR agonists as anabolic SARMs. Meanwhile, there is growing interest in the search for non-steroidal AR antagonists and peptide antagonist as tissue-selective anti-androgens. The mechanism of tissue selectivity of SARMs remains unclear, although tissue-specific expression of 5α-reductase appears to have a role for many of the SARMs that are identified currently. It is not clear if other mechanisms related to SARM action contribute to the observed tissue selectivity in vivo. Future mechanism-based drug design will rely on further investigations of the mechanisms of action of known SARMs, including co-regulator recruitment during the genomic actions of ARs, signaling pathways involved in the non-genomic effects of ARs, and potential tissue-specific distribution.
Clinically, SARMs with improved pharmacokinetic characteristics and tissue selectivity might expand the therapeutic applications of AR ligands to include HRT, muscle wasting, osteoporosis, male contraception, BPH and prostate cancer. Anabolic SARMs are safe for use in women, so combination therapy with SARMs and SERMs might introduce revolutionary changes to HRT in men and women, and be used to treat osteoporosis, prostatic diseases and a wide array of conditions associated with muscle wasting and frailty.
Footnotes
Disclosure Statement
James T. Dalton is an employee of GTx, Inc., Memphis, TN, USA.
Gao, W. and Dalton, J.T. (2005) Selective androgen receptor modulator (SARM) binding initiates unique surface topology of androgen receptor AF2 region that alters N/C interaction and co-activator recruitment. 2006 Keystone Symposia, Tissue-Selective Nuclear Receptors, Abstract 207
Hamann, L.G. (2004) Discovery and preclinical profile of a highly potent and muscle selective androgen receptor modulator (SARM). 227th National Meeting of the American Chemical Society Medicinal Chemistry Division
References
- 1.Gao W, et al. Chemistry and structural biology of androgen receptor. Chem. Rev. 2005;105:3352–3370. doi: 10.1021/cr020456u. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Fang H, et al. Study of 202 natural, synthetic, and environmental chemicals for binding to the androgen receptor. Chem. Res. Toxicol. 2003;16:1338–1358. doi: 10.1021/tx030011g. [DOI] [PubMed] [Google Scholar]
- 3.Burger A, Abraham DJ. Burger's Medicinal Chemistry and Drug Discovery. Wiley; 2003. [Google Scholar]
- 4.Bohl CE, et al. Structural basis for antagonism and resistance of bicalutamide in prostate cancer. Proc. Natl. Acad. Sci. U. S. A. 2005;102:6201–6206. doi: 10.1073/pnas.0500381102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Matias PM, et al. Structural evidence for ligand specificity in the binding domain of the human androgen receptor. Implications for pathogenic gene mutations. J. Biol. Chem. 2000;275:26164–26171. doi: 10.1074/jbc.M004571200. [DOI] [PubMed] [Google Scholar]
- 6.Colombo D, et al. A comparative molecular modeling study of dydrogesterone with other progestational agents through theoretical calculations and nuclear magnetic resonance spectroscopy. J. Steroid Biochem. Mol. Biol. 2006;98:56–62. doi: 10.1016/j.jsbmb.2005.07.009. [DOI] [PubMed] [Google Scholar]
- 7.Negro-Vilar A. Selective androgen receptor modulators (SARMs): a novel approach to androgen therapy for the new millennium. J. Clin. Endocrinol. Metab. 1999;84:3459–3462. doi: 10.1210/jcem.84.10.6122. [DOI] [PubMed] [Google Scholar]
- 8.Segal S, et al. Therapeutic potential of the SARMs: revisiting the androgen receptor for drug discovery. Expert Opin. Investig. Drugs. 2006;15:377–387. doi: 10.1517/13543784.15.4.377. [DOI] [PubMed] [Google Scholar]
- 9.Kawano H, et al. Suppressive function of androgen receptor in bone resorption. Proc. Natl. Acad. Sci. U. S. A. 2003;100:9416–9421. doi: 10.1073/pnas.1533500100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Chen J, et al. Discovery and therapeutic promise of selective androgen receptor modulators. Mol. Interv. 2005;5:173–188. doi: 10.1124/mi.5.3.7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Foye WO, et al. Foye's Principles of Medicinal Chemistry. Lippincott Williams & Wilkins; 2002. [Google Scholar]
- 12.Wu FC, von Eckardstein A. Androgens and coronary artery disease. Endocr. Rev. 2003;24:183–217. doi: 10.1210/er.2001-0025. [DOI] [PubMed] [Google Scholar]
- 13.Mooradian AD, et al. Biological actions of androgens. Endocr. Rev. 1987;8:1–28. doi: 10.1210/edrv-8-1-1. [DOI] [PubMed] [Google Scholar]
- 14.Eckardstein A, Wu FC. Testosterone and atherosclerosis. Growth Horm IGF Res. 2003;13(Suppl A):S72–84. doi: 10.1016/s1096-6374(03)00059-5. [DOI] [PubMed] [Google Scholar]
- 15.Chacon A, Monga M. Medical management of benign prostatic hyperplasia. Geriatr. Nephrol. Urol. 1999;9:39–48. doi: 10.1023/a:1008308819463. [DOI] [PubMed] [Google Scholar]
- 16.Radlmaier A, et al. Estrogen reduction by aromatase inhibition for benign prostatic hyperplasia: results of a double-blind, placebo-controlled, randomized clinical trial using two doses of the aromatase-inhibitor atamestane. Atamestane Study Group. Prostate. 1996;29:199–208. doi: 10.1002/(SICI)1097-0045(199610)29:4<199::AID-PROS1>3.0.CO;2-7. [DOI] [PubMed] [Google Scholar]
- 17.Gingell JC, et al. Placebo controlled double-blind study to test the efficacy of the aromatase inhibitor atamestane in patients with benign prostatic hyperplasia not requiring operation. The Schering 90.062 Study Group. J. Urol. 1995;154:399–401. doi: 10.1097/00005392-199508000-00020. [DOI] [PubMed] [Google Scholar]
- 18.Habenicht UF, et al. Management of benign prostatic hyperplasia with particular emphasis on aromatase inhibitors. J. Steroid Biochem. Mol. Biol. 1993;44:557–563. doi: 10.1016/0960-0760(93)90259-y. [DOI] [PubMed] [Google Scholar]
- 19.Keller ET, et al. The androgen receptor: a mediator of diverse responses. Front. Biosci. 1996;1:d59–d71. doi: 10.2741/a116. [DOI] [PubMed] [Google Scholar]
- 20.Mosselman S, et al. ER beta: identification and characterization of a novel human estrogen receptor. FEBS Lett. 1996;392:49–53. doi: 10.1016/0014-5793(96)00782-x. [DOI] [PubMed] [Google Scholar]
- 21.Thigpen AE, et al. Molecular genetics of steroid 5 alpha-reductase 2 deficiency. J. Clin. Invest. 1992;90:799–809. doi: 10.1172/JCI115954. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Wright AS, et al. Androgen-induced regrowth in the castrated rat ventral prostate: role of 5alpha-reductase. Endocrinology. 1999;140:4509–4515. doi: 10.1210/endo.140.10.7039. [DOI] [PubMed] [Google Scholar]
- 23.Oettel M. Testosterone metabolism, dose-response relationships and receptor polymorphisms: selected pharmacological/toxicological considerations on benefits versus risks of testosterone therapy in men. Aging Male. 2003;6:230–256. doi: 10.1080/13685530312331309772. [DOI] [PubMed] [Google Scholar]
- 24.Russell DW, et al. The molecular genetics of steroid 5 alpha-reductases. Recent Prog. Horm. Res. 1994;49:275–284. doi: 10.1016/b978-0-12-571149-4.50018-0. [DOI] [PubMed] [Google Scholar]
- 25.Oettel M. Is there a role for estrogens in the maintenance of men's health? Aging Male. 2002;5:248–257. [PubMed] [Google Scholar]
- 26.de Ronde W, et al. The importance of oestrogens in males. Clin. Endocrinol. (Oxf.) 2003;58:529–542. doi: 10.1046/j.1365-2265.2003.01669.x. [DOI] [PubMed] [Google Scholar]
- 27.Barrett-Connor E, et al. Endogenous sex hormones and cognitive function in older men. J. Clin. Endocrinol. Metab. 1999;84:3681–3685. doi: 10.1210/jcem.84.10.6086. [DOI] [PubMed] [Google Scholar]
- 28.Van Pottelbergh I, et al. Differential contribution of testosterone and estradiol in the determination of cholesterol and lipoprotein profile in healthy middle-aged men. Atherosclerosis. 2003;166:95–102. doi: 10.1016/s0021-9150(02)00308-8. [DOI] [PubMed] [Google Scholar]
- 29.Mukherjee TK, et al. Testosterone attenuates expression of vascular cell adhesion molecule-1 by conversion to estradiol by aromatase in endothelial cells: implications in atherosclerosis. Proc. Natl. Acad. Sci. U. S. A. 2002;99:4055–4060. doi: 10.1073/pnas.052703199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Toda K, et al. Oestrogen at the neonatal stage is critical for the reproductive ability of male mice as revealed by supplementation with 17beta-oestradiol to aromatase gene (Cyp19) knockout mice. J. Endocrinol. 2001;168:455–463. doi: 10.1677/joe.0.1680455. [DOI] [PubMed] [Google Scholar]
- 31.Falahati-Nini A, et al. Relative contributions of testosterone and estrogen in regulating bone resorption and formation in normal elderly men. J. Clin. Invest. 2000;106:1553–1560. doi: 10.1172/JCI10942. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Orwoll ES. Men, bone and estrogen: unresolved issues. Osteoporos. Int. 2003;14:93–98. doi: 10.1007/s00198-002-1332-9. [DOI] [PubMed] [Google Scholar]
- 33.Pratt WB, Toft DO. Steroid receptor interactions with heat shock protein and immunophilin chaperones. Endocr. Rev. 1997;18:306–360. doi: 10.1210/edrv.18.3.0303. [DOI] [PubMed] [Google Scholar]
- 34.Heinlein CA, Chang C. Androgen receptor (AR) coregulators: an overview. Endocr. Rev. 2002;23:175–200. doi: 10.1210/edrv.23.2.0460. [DOI] [PubMed] [Google Scholar]
- 35.Shang Y, et al. Formation of the androgen receptor transcription complex. Mol. Cell. 2002;9:601–610. doi: 10.1016/s1097-2765(02)00471-9. [DOI] [PubMed] [Google Scholar]
- 36.Lutz LB, et al. Selective modulation of genomic and nongenomic androgen responses by androgen receptor ligands. Mol. Endocrinol. 2003;17:1106–1116. doi: 10.1210/me.2003-0032. [DOI] [PubMed] [Google Scholar]
- 37.Estrada M, et al. Testosterone stimulates intracellular calcium release and mitogen-activated protein kinases via a G protein-coupled receptor in skeletal muscle cells. Endocrinology. 2003;144:3586–3597. doi: 10.1210/en.2002-0164. [DOI] [PubMed] [Google Scholar]
- 38.Kousteni S, et al. Nongenotropic, sex-nonspecific signaling through the estrogen or androgen receptors: dissociation from transcriptional activity. Cell. 2001;104:719–730. [PubMed] [Google Scholar]
- 39.Zagar Y, et al. Signaling cross-talk from Gbeta4 subunit to Elk-1 in the rapid action of androgens. J. Biol. Chem. 2004;279:2403–2413. doi: 10.1074/jbc.M309132200. [DOI] [PubMed] [Google Scholar]
- 40.Kampa M, et al. The human prostate cancer cell line LNCaP bears functional membrane testosterone receptors that increase PSA secretion and modify actin cytoskeleton. FASEB J. 2002;16:1429–1431. doi: 10.1096/fj.02-0131fje. [DOI] [PubMed] [Google Scholar]
- 41.Unni E, et al. Changes in androgen receptor nongenotropic signaling correlate with transition of LNCaP cells to androgen independence. Cancer Res. 2004;64:7156–7168. doi: 10.1158/0008-5472.CAN-04-1121. [DOI] [PubMed] [Google Scholar]
- 42.Norman AW, et al. Steroid-hormone rapid actions, membrane receptors and a conformational ensemble model. Nat. Rev. Drug Discov. 2004;3:27–41. doi: 10.1038/nrd1283. [DOI] [PubMed] [Google Scholar]
- 43.Migliaccio A, et al. Steroid-induced androgen receptor-oestradiol receptor beta-Src complex triggers prostate cancer cell proliferation. EMBO J. 2000;19:5406–5417. doi: 10.1093/emboj/19.20.5406. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Sathya G, et al. Pharmacological uncoupling of androgen receptor-mediated prostate cancer cell proliferation and prostate-specific antigen secretion. Cancer Res. 2003;63:8029–8036. [PubMed] [Google Scholar]
- 45.Callewaert L, et al. Interplay between two hormone-independent activation domains in the androgen receptor. Cancer Res. 2006;66:543–553. doi: 10.1158/0008-5472.CAN-05-2389. [DOI] [PubMed] [Google Scholar]
- 46.Hodgson MC, et al. The androgen receptor recruits nuclear receptor corepressor (N-CoR) in the presence of mifepristone via its N and C termini revealing a novel molecular mechanism for androgen receptor antagonists. J. Biol. Chem. 2005;280:6511–6519. doi: 10.1074/jbc.M408972200. [DOI] [PubMed] [Google Scholar]
- 47.Carlson KE, Katzenellenbogen JA. A comparative study of the selectivity and efficiency of target tissue uptake of five tritium-labeled androgens in the rat. J. Steroid Biochem. 1990;36:549–561. doi: 10.1016/0022-4731(90)90172-o. [DOI] [PubMed] [Google Scholar]
- 48.Parent EE, et al. Synthesis and biological evaluation of a nonsteroidal bromine-76-labeled androgen receptor ligand 3-[76Br]bromohydroxyflutamide. Nucl. Med. Biol. 2006;33:705–713. doi: 10.1016/j.nucmedbio.2006.05.009. [DOI] [PubMed] [Google Scholar]
- 49.Gao W, et al. Comparison of the pharmacological effects of a novel selective androgen receptor modulator, the 5alpha-reductase inhibitor finasteride, and the antiandrogen hydroxyflutamide in intact rats: new approach for benign prostate hyperplasia. Endocrinology. 2004;145:5420–5428. doi: 10.1210/en.2004-0627. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Bjornstrom L, Sjoberg M. Estrogen receptor-dependent activation of AP-1 via non-genomic signalling. Nucl. Recept. 2004;2:3. doi: 10.1186/1478-1336-2-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Kousteni S, et al. Reversal of bone loss in mice by nongenotropic signaling of sex steroids. Science. 2002;298:843–846. doi: 10.1126/science.1074935. [DOI] [PubMed] [Google Scholar]
- 52.Dalton JT, et al. Discovery of nonsteroidal androgens. Biochem. Biophys. Res. Commun. 1998;244:1–4. doi: 10.1006/bbrc.1998.8209. [DOI] [PubMed] [Google Scholar]
- 53.Edwards JP, et al. New nonsteroidal androgen receptor modulators based on 4-(trifluoromethyl)-2(1H)-pyrrolidino[3,2-g] quinolinone. Bioorg. Med. Chem. Lett. 1998;8:745–750. doi: 10.1016/s0960-894x(98)00107-3. [DOI] [PubMed] [Google Scholar]
- 54.Yin D, et al. Pharmacodynamics of selective androgen receptor modulators. J. Pharmacol. Exp. Ther. 2003;304:1334–1340. doi: 10.1124/jpet.102.040840. [DOI] [PubMed] [Google Scholar]
- 55.Hanada K, et al. Bone anabolic effects of S-40503, a novel nonsteroidal selective androgen receptor modulator (SARM), in rat models of osteoporosis. Biol. Pharm. Bull. 2003;26:1563–1569. doi: 10.1248/bpb.26.1563. [DOI] [PubMed] [Google Scholar]
- 56.Miner JN, et al. An orally-active selective androgen receptor modulator is efficacious on bone, muscle and sex function with reduced impact on prostate. Endocrinology. 2006 doi: 10.1210/en.2006-0793. 10.1210/en.2006-0793 ( http://endo.endojournals.org) [DOI] [PubMed] [Google Scholar]
- 57.Cadilla R, Turnbull P. Selective androgen receptor modulators in drug discovery: medicinal chemistry and therapeutic potential. Curr. Top. Med. Chem. 2006;6:245–270. doi: 10.2174/156802606776173456. [DOI] [PubMed] [Google Scholar]
- 58.Gao W, et al. Pharmacokinetics and pharmacodynamics of nonsteroidal androgen receptor ligands. Pharm. Res. 2006;23:1641–1658. doi: 10.1007/s11095-006-9024-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Buijsman RC, et al. Non-steroidal steroid receptor modulators. Curr. Med. Chem. 2005;12:1017–1075. doi: 10.2174/0929867053764671. [DOI] [PubMed] [Google Scholar]
- 60.Gao W, et al. Selective androgen receptor modulator (SARM) treatment improves muscle strength and body composition, and prevents bone loss in orchidectomized rats. Endocrinology. 2005;146:4887–4897. doi: 10.1210/en.2005-0572. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Chen J, et al. A selective androgen receptor modulator for hormonal male contraception. J. Pharmacol. Exp. Ther. 2005;312:546–553. doi: 10.1124/jpet.104.075424. [DOI] [PubMed] [Google Scholar]
- 62.Furr BJ, Tucker H. The preclinical development of bicalutamide: pharmacodynamics and mechanism of action. Urology. 1996;47(Suppl 1A):13–25. doi: 10.1016/s0090-4295(96)80003-3. discussion 29–32. [DOI] [PubMed] [Google Scholar]
- 63.Cockshott ID. Bicalutamide: clinical pharmacokinetics and metabolism. Clin. Pharmacokinet. 2004;43:855–878. doi: 10.2165/00003088-200443130-00003. [DOI] [PubMed] [Google Scholar]
- 64.Chang CY, et al. Development of peptide antagonists for the androgen receptor using combinatorial peptide phage display. Mol. Endocrinol. 2005;19:2478–2490. doi: 10.1210/me.2005-0072. [DOI] [PubMed] [Google Scholar]
- 65.Arnold LA, et al. Discovery of small molecule inhibitors of the interaction of the thyroid hormone receptor with transcriptional coregulators. J. Biol. Chem. 2005;280:43048–43055. doi: 10.1074/jbc.M506693200. [DOI] [PubMed] [Google Scholar]






