Abstract
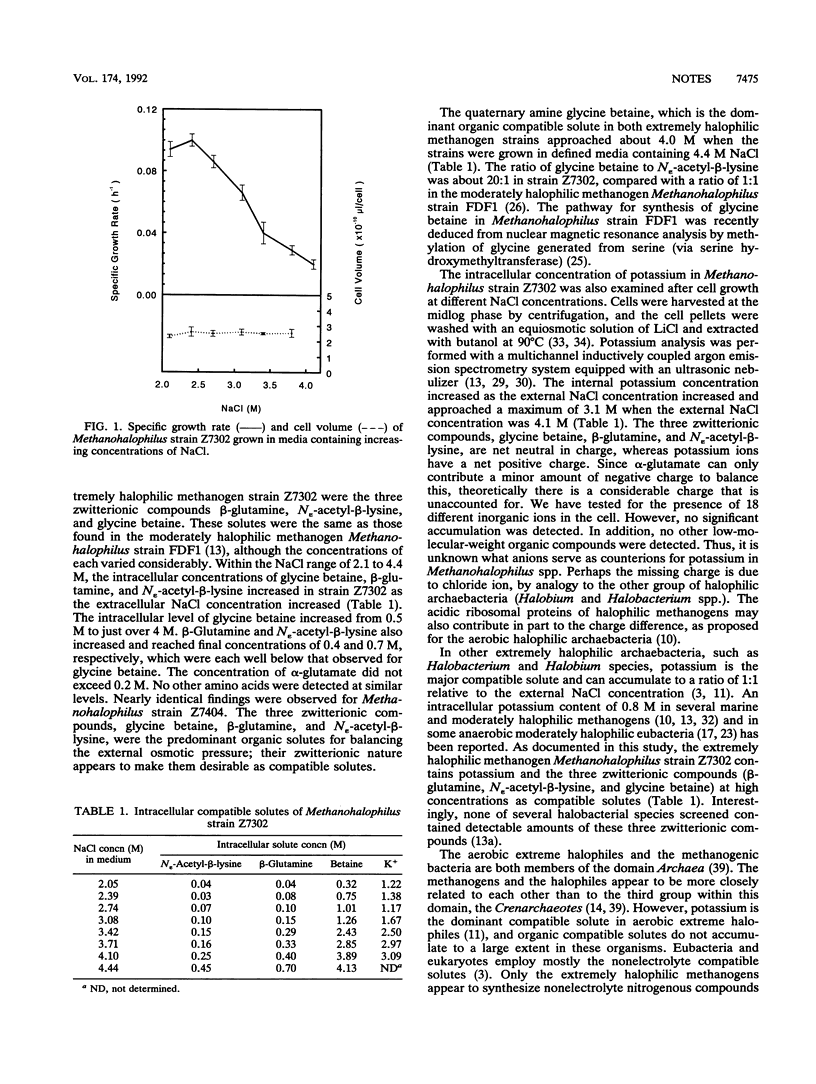
Methanohalophilus strain Z7302 was previously isolated from a hypersaline environment and grows over a range of NaCl concentrations from 1.7 to 4.4 M. We examined the relationships between cell growth rate, cell volume, and intracellular solute concentrations with increasing salinity. This extremely halophilic methanogen synthesized three zwitterionic compounds, beta-glutamine, N epsilon-acetyl-beta-lysine, and glycine betaine, and also accumulated potassium ion as compatible solutes to balance the external and internal osmotic pressures. Potassium and glycine betaine were the predominant compatible solutes when Methanohalophilus strain Z7302 was grown at high external NaCl concentrations and approached intracellular levels of 3 and 4 M, respectively.
Full text
PDF


Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Ben-Amotz A., Avron M. Accumulation of metabolites by halotolerant algae and its industrial potential. Annu Rev Microbiol. 1983;37:95–119. doi: 10.1146/annurev.mi.37.100183.000523. [DOI] [PubMed] [Google Scholar]
- Booth I. R., Higgins C. F. Enteric bacteria and osmotic stress: intracellular potassium glutamate as a secondary signal of osmotic stress? FEMS Microbiol Rev. 1990 Jun;6(2-3):239–246. doi: 10.1111/j.1574-6968.1990.tb04097.x. [DOI] [PubMed] [Google Scholar]
- Cover W. H., Martinez R. J., Rittenberg S. C. Permeability of the boundary layers of Bdellovibrio bacteriovorus 109J and its bdelloplasts to small hydrophilic molecules. J Bacteriol. 1984 Feb;157(2):385–390. doi: 10.1128/jb.157.2.385-390.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Csonka L. N., Hanson A. D. Prokaryotic osmoregulation: genetics and physiology. Annu Rev Microbiol. 1991;45:569–606. doi: 10.1146/annurev.mi.45.100191.003033. [DOI] [PubMed] [Google Scholar]
- Csonka L. N. Physiological and genetic responses of bacteria to osmotic stress. Microbiol Rev. 1989 Mar;53(1):121–147. doi: 10.1128/mr.53.1.121-147.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ferguson T. J., Mah R. A. Isolation and characterization of an h(2)-oxidizing thermophilic methanogen. Appl Environ Microbiol. 1983 Jan;45(1):265–274. doi: 10.1128/aem.45.1.265-274.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kiene R. P., Oremland R. S., Catena A., Miller L. G., Capone D. G. Metabolism of reduced methylated sulfur compounds in anaerobic sediments and by a pure culture of an estuarine methanogen. Appl Environ Microbiol. 1986 Nov;52(5):1037–1045. doi: 10.1128/aem.52.5.1037-1045.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lai M. C., Sowers K. R., Robertson D. E., Roberts M. F., Gunsalus R. P. Distribution of compatible solutes in the halophilic methanogenic archaebacteria. J Bacteriol. 1991 Sep;173(17):5352–5358. doi: 10.1128/jb.173.17.5352-5358.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Masui M., Wada S. Intracellular concentrations of Na+, K+, and cl minus of a moderately halophilic bacterium. Can J Microbiol. 1973 Oct;19(10):1181–1186. doi: 10.1139/m73-191. [DOI] [PubMed] [Google Scholar]
- Mathrani I. M., Boone D. R. Isolation and characterization of a moderately halophilic methanogen from a solar saltern. Appl Environ Microbiol. 1985 Jul;50(1):140–143. doi: 10.1128/aem.50.1.140-143.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meikle A. J., Reed R. H., Gadd G. M. Osmotic adjustment and the accumulation of organic solutes in whole cells and protoplasts of Saccharomyces cerevisiae. J Gen Microbiol. 1988 Nov;134(11):3049–3060. doi: 10.1099/00221287-134-11-3049. [DOI] [PubMed] [Google Scholar]
- Mizuno T., Mizushima S. Signal transduction and gene regulation through the phosphorylation of two regulatory components: the molecular basis for the osmotic regulation of the porin genes. Mol Microbiol. 1990 Jul;4(7):1077–1082. doi: 10.1111/j.1365-2958.1990.tb00681.x. [DOI] [PubMed] [Google Scholar]
- Paterek J. R., Smith P. H. Isolation and characterization of a halophilic methanogen from great salt lake. Appl Environ Microbiol. 1985 Oct;50(4):877–881. doi: 10.1128/aem.50.4.877-881.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roberts M. F., Lai M. C., Gunsalus R. P. Biosynthetic pathways of the osmolytes N epsilon-acetyl-beta-lysine, beta-glutamine, and betaine in Methanohalophilus strain FDF1 suggested by nuclear magnetic resonance analyses. J Bacteriol. 1992 Oct;174(20):6688–6693. doi: 10.1128/jb.174.20.6688-6693.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Robertson D. E., Lai M. C., Gunsalus R. P., Roberts M. F. Composition, Variation, and Dynamics of Major Osmotic Solutes in Methanohalophilus Strain FDF1. Appl Environ Microbiol. 1992 Aug;58(8):2438–2443. doi: 10.1128/aem.58.8.2438-2443.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Robertson D. E., Lesage S., Roberts M. F. Beta-aminoglutaric acid is a major soluble component of Methanococcus thermolithotrophicus. Biochim Biophys Acta. 1989 Sep 15;992(3):320–326. doi: 10.1016/0304-4165(89)90091-3. [DOI] [PubMed] [Google Scholar]
- Robertson D. E., Roberts M. F., Belay N., Stetter K. O., Boone D. R. Occurrence of beta-glutamate, a novel osmolyte, in marine methanogenic bacteria. Appl Environ Microbiol. 1990 May;56(5):1504–1508. doi: 10.1128/aem.56.5.1504-1508.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sowers K. R., Robertson D. E., Noll D., Gunsalus R. P., Roberts M. F. N epsilon-acetyl-beta-lysine: an osmolyte synthesized by methanogenic archaebacteria. Proc Natl Acad Sci U S A. 1990 Dec;87(23):9083–9087. doi: 10.1073/pnas.87.23.9083. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sprott G. D., Jarrell K. F. K+, Na+, and Mg2+ content and permeability of Methanospirillum hungatei and Methanobacterium thermoautotrophicum. Can J Microbiol. 1981 Apr;27(4):444–451. doi: 10.1139/m81-067. [DOI] [PubMed] [Google Scholar]
- Sprott G. D., Shaw K. M., Jarrell K. F. Ammonia/potassium exchange in methanogenic bacteria. J Biol Chem. 1984 Oct 25;259(20):12602–12608. [PubMed] [Google Scholar]
- Vreeland R. H. Mechanisms of halotolerance in microorganisms. Crit Rev Microbiol. 1987;14(4):311–356. doi: 10.3109/10408418709104443. [DOI] [PubMed] [Google Scholar]
- Welsh D. T., Reed R. H., Herbert R. A. The role of trehalose in the osmoadaptation of Escherichia coli NCIB 9484: interaction of trehalose, K+ and glutamate during osmoadaptation in continuous culture. J Gen Microbiol. 1991 Apr;137(4):745–750. doi: 10.1099/00221287-137-4-745. [DOI] [PubMed] [Google Scholar]
- Whatmore A. M., Reed R. H. Determination of turgor pressure in Bacillus subtilis: a possible role for K+ in turgor regulation. J Gen Microbiol. 1990 Dec;136(12):2521–2526. doi: 10.1099/00221287-136-12-2521. [DOI] [PubMed] [Google Scholar]
- Woese C. R., Kandler O., Wheelis M. L. Towards a natural system of organisms: proposal for the domains Archaea, Bacteria, and Eucarya. Proc Natl Acad Sci U S A. 1990 Jun;87(12):4576–4579. doi: 10.1073/pnas.87.12.4576. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yancey P. H., Clark M. E., Hand S. C., Bowlus R. D., Somero G. N. Living with water stress: evolution of osmolyte systems. Science. 1982 Sep 24;217(4566):1214–1222. doi: 10.1126/science.7112124. [DOI] [PubMed] [Google Scholar]



