Abstract
The steroid hormone 20-hydroxyecdysone (referred to here as ecdysone) directs Drosophila metamorphosis by activating a series of genetic regulatory hierarchies. ETS domain transcription factors encoded by the ecdysone-inducible E74 early gene, E74A and E74B, act at the top of these hierarchies to coordinate the induction of target genes. We have ectopically expressed these E74 isoforms to understand their regulatory functions during the onset of metamorphosis. We show that E74 can regulate its own transcription, most likely through binding sites within its gene. Ectopic expression of E74B can partially repress the E78B and DHR3 orphan receptor genes, suggesting a role for E74 in the appropriate timing of early-late gene expression. Furthermore, E74A is both necessary and sufficient for E78B induction, implicating E74A as a key regulator of E78B expression. We also show, consistent with our studies of E74 loss-of-function mutations, that E74B is a potent repressor of late gene transcription and E74A is sufficient to prematurely induce the L71–1 late gene. However, ectopic expression of both Broad-Complex and E74A activators in an E74B mutant background is not sufficient to prematurely induce all late genes, indicating that other factors contribute to this regulatory circuit. These observations demonstrate that the steroid-triggered switch in E74 transcription factor isoforms plays a central role in the proper timing of secondary-response gene expression.
Keywords: steroid hormones, gene regulation, Drosophila development
Extensive studies have shown that steroid hormones interact with specific nuclear receptors to reprogram gene expression within target cells (1, 2). Our understanding of the events that occur downstream from the receptor, however, remains obscure. Few hormone-induced target genes have been identified, and their biological functions are often unclear. We are studying the hormonal regulation of insect metamorphosis as a model system for defining the molecular mechanisms of steroid hormone action. In the fruit fly, Drosophila melanogaster, pulses of the steroid hormone ecdysone direct the major postembryonic developmental transitions, including molting and metamorphosis (3). A high titer ecdysone pulse at the end of the third, and final, larval instar triggers puparium formation and the onset of prepupal development, and is followed by another ecdysone pulse 10 hr later that triggers head eversion and the prepupal-pupal transition.
Studies of the puffing patterns of the larval salivary gland polytene chromosomes have provided a unique opportunity to visualize the effects of a steroid hormone on gene activity (4, 5). About six early puff genes are rapidly and directly induced by ecdysone. Some of these genes encode regulatory proteins that both repress their own expression and induce more than 100 secondary-response genes that reside within the late puffs. The late puff products, in turn, are thought to direct the appropriate function of the salivary gland during prepupal development.
Molecular studies have confirmed and extended this hierarchical model of ecdysone action (see refs. 6, 7 for recent reviews). Four early ecdysone-inducible puff genes have been described. One of these, encoded by the 63F early puff, encodes a calcium-binding protein related to calmodulin, providing the potential for crosstalk between steroid and calcium signaling pathways (8). The other three early puff genes—the Broad-Complex (BR-C), E74, and E75—encode families of site-specific DNA-binding proteins that function as transcription factors (9–11). The BR-C encodes multiple protein isoforms, each containing one of four possible pairs of zinc fingers (designated Z1-Z4) (9, 12). E74 encodes two protein isoforms, E74A and E74B, that share a common C-terminal ETS DNA-binding domain fused to unique N-terminal sequences (11). The BR-C and E74 are essential for critical developmental responses to ecdysone and function together to regulate overlapping sets of secondary-response target genes (13–16). Studies with staged animals and cultured larval organs have indicated that the timing and levels of early gene transcription are a direct consequence of changes in ecdysone titer (17, 18). Some early mRNAs, including the BR-C and E74B, are induced early in the third larval instar by low titer pulses of ecdysone, whereas the high titer late larval ecdysone pulse represses E74B transcription and induces E74A. These responses lead to waves of ecdysone-induced gene expression during the onset of metamorphosis (19, 20).
The late puffs fall into two categories, early-late and late-late, distinguished by their timing and ecdysone regulation (21). The DHR3 and E78B early-late puff genes encode orphan members of the nuclear receptor superfamily, and thus may function as transcriptional regulators much like the early genes (22, 23). Also like the early genes, both DHR3 and E78B are induced directly by ecdysone. Unlike the early genes, however, maximal DHR3 and E78B transcription is dependent on ecdysone-induced protein synthesis (23, 24). Mutations in E78 have no detectable effect on development, although they do show subtle alterations in the puffing response to ecdysone (25). In contrast, DHR3 mutations lead to lethality during embryonic development (G. Carney and M. Bender, personal communication). Ectopic expression experiments have revealed that DHR3 can both repress early gene transcription at puparium formation and induce the βFTZ-F1 mid-prepupal competence factor (26, 27).
Two late-late puff genes have been described at the molecular level. One gene has been isolated from the 4F late puff and five pairs of L71 genes have been isolated from the 71E late puff (28, 29). In contrast to the early and early-late genes, these late-late genes are expressed in one tissue at one stage during development; they are coordinately induced in the salivary glands at puparium formation and repressed 12–14 hr later. The L71 genes encode small, apparently secreted, polypeptides that resemble defensins and venom toxins. This final secretion from the salivary glands into the space between the imaginal hypoderm and prepupal cuticle may protect the animal from bacterial infection during metamorphosis (29). The BR-C and E74 are essential for appropriate late gene expression, consistent with their induction as a secondary-response to ecdysone (14, 30, 31). This has been most extensively demonstrated with L71–6, which is induced directly by both BR-C and E74A proteins (32, 33).
In this study, we continue our functional analysis of E74 by examining the effects of ectopic E74A and E74B expression on target gene transcription. We show that the E74 isoforms can positively regulate one another, suggesting that the cluster of E74 binding sites in the middle of the E74 gene is of functional significance. Ectopic expression of E74B can partially repress DHR3 and E78B and efficiently repress the 4F and L71 late genes. Furthermore, E74A is both necessary and sufficient for E78B induction, supporting a key role for E74A in E78B expression. In contrast, ectopic E74A is sufficient to induce only low levels of transcription of some late genes, even in an E74B mutant background and in the presence of the BR-C Z1 activator. We conclude that the ecdysone-triggered isoform switch from an E74B repressor to an E74A inducer is necessary, but not sufficient, for the proper timing of late gene induction at puparium formation. These studies demonstrate that changes in hormone titer are transduced through the induction and repression of different transcription factors to coordinate secondary-response gene expression during development.
MATERIALS AND METHODS
Construction of Transgenic Fly Stocks.
The 3.2-kb SpeI-StuI restriction fragment containing the E74A coding region was inserted between the EcoRI and StuI sites of the pCaSpeR-hs vector (34). This P element was introduced into the germ line of w; ry506 P[ry+Δ2–3](99B) embryos by microinjection (35) and eight independent transformant lines were recovered. One line, carrying a single homozygous viable P element insertion on the second chromosome, designated P[hs-E74A], generated the highest levels of E74A mRNA in response to heat shock and was used for the studies reported here.
A 4.3-kb HindIII (filled in)–XbaI restriction fragment containing the E74B coding region and 1.0 kb of the 3′ untranslated region was inserted between the HpaI and XbaI sites of pCaSpeR-hs (34). This P element was coinjected along with the helper plasmid π25.7wc into the germ line of y Df(1)w67c23 embryos (36), and two independent transformant lines were recovered. One line carrying a single homozygous viable P element insertion on the second chromosome, designated P[hs-E74B], generated high levels of E74B mRNA in response to heat shock and was used for the studies reported here.
Ectopic Expression.
Third instar larvae were maintained on food containing 0.05% bromophenol blue and staged based on the clearance of dye from their guts (37). Heat treatment had no apparent effect on the timing of gut clearance. For heat shock, staged animals were placed in 1.5-ml microcentrifuge tubes, transferred to a 35°C water bath for 30 min, and allowed to recover for 2 hr at 25°C, after which RNA was extracted from the whole animals. To test the effects of ectopic E74A and/or Z1 expression in E74B mutants, Tb+ larvae from the following crosses were selected for heat treatment and RNA isolation: (1) for E74B mutants with no ectopic expression: w; Df(3l)st-81k19/TM6C Sb Tb virgin females crossed with w; E74DL-1/TM6C Sb Tb males, (2) for ectopic E74A: w; Df(3l)st-81k19/TM6C Sb Tb virgin females crossed with w; P[hs-E74A]; E74DL-1/TM6C Sb Tb males, (3) for ectopic Z1: w P[hs(BRC-Q1-Z1)]; Df(3)st-81k19/TM6C Sb Tb virgin females crossed with w; E74DL-1/TM6C Sb Tb males, (4) for ectopic E74A and Z1: w P[hs(BRC-Q1-Z1)]; Df(3L) st-81k19/TM6C Sb Tb virgin females crossed with w; P[hs-E74A]; E74DL-1/TM6C Sb Tb males. The P[hs(BRC-Q1-Z1)] line was kindly provided by K. Crossgrove and G. Guild (University of Pennsylvania) (33). The E74 mutations have been described previously (15).
Northern Blot Hybridization.
Total RNA was isolated as described (37), fractionated by formaldehyde agarose gel electrophoresis, and transferred to nylon membranes (NEN/DuPont). Hybridization and washing were performed using probes specific for each ecdysone-regulated gene, as described (19, 31).
RESULTS AND DISCUSSION
Heat Shock Directs High Levels of Ectopic E74A and E74B Expression.
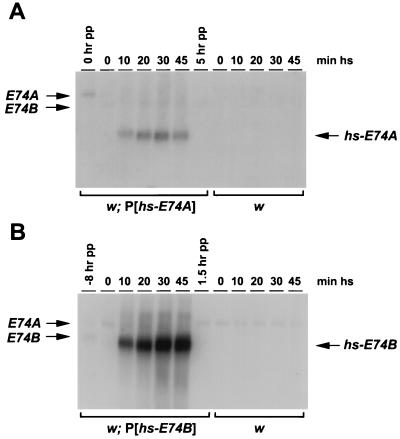
Full-length E74A or E74B coding regions were inserted into the pCaSpeR-hs P-element vector and introduced into the Drosophila genome by germ-line transformation (36). One transformant line that efficiently expressed either E74A or E74B under the control of the hsp70 promoter was selected for further characterization. In P[hs-E74A] prepupae, a 3.2-kb E74A transcript accumulates in response to a 10-min heat shock at 35°C, and maximal levels of mRNA are induced after a 30-min heat treatment (Fig. 1A). Similarly, a 4.4-kb E74B transcript accumulates in P[hs-E74B] prepupae after a 10-min heat shock, and maximal mRNA levels are obtained by 30 min (Fig. 1B). The amount of hs-E74A mRNA is severalfold higher than that normally seen in newly formed prepupae, whereas the amount of hs-E74B mRNA is significantly higher than endogenous E74B mRNA in late larvae. No hs-E74A or hs-E74B transcripts are detectable in the absence of heat treatment or in w control animals (Fig. 1).
Figure 1.
Time course of ectopic E74 induction. (A) Both w and w; P[hs-E74A] animals were synchronized at puparium formation and aged 4 hr. These prepupae were then heat-shocked at 35°C for the number of minutes indicated. RNA was extracted from whole animals, and the endogenous and heat-induced E74A and E74B mRNAs were detected by Northern blot hybridization as described (31). RNA was also extracted from non-heat-induced w; P[hs-E74A] 0-hr prepupae (0 hr pp) as a positive control for normal levels of endogenous E74A mRNA, and from non-heat-induced w; P[hs-E74A] 5-hr prepupae (5 hr pp) as a negative control for E74A expression. (B) Both w and w; P[hs-E74B] animals were synchronized at puparium formation and heat-shocked at 35°C for the number of minutes indicated. RNA was extracted from whole animals, and the endogenous and heat-induced E74A and E74B mRNAs detected by Northern blot hybridization. RNA was also extracted from non-heat-induced w; P[hs-E74B] −8-hr larvae (−8 hr pp) as a positive control for normal levels of endogenous E74B mRNA, and from non-heat-induced w; P[hs-E74B] 1.5-hr prepupae (1.5 hr pp) as a negative control for E74B expression.
E74 Autoregulation.
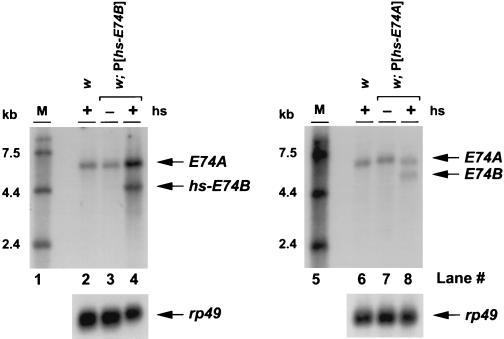
Analysis of E74 loss-of-function mutations revealed that E74B is required for proper levels of E74A transcription in late prepupae, suggesting that E74B induces E74A at this stage in development (31). Furthermore, the presence of three adjacent E74 binding sites in the middle of the E74 gene suggest that this regulation may be direct (38, 39). To determine whether ectopic E74A or E74B expression had any effect on E74 transcription, control, P[hs-E74A], and P[hs-E74B] late third instar larvae were heat-shocked for 30 min and allowed to recover for 2 hr. RNA was extracted from newly formed prepupae selected from this population of heat-treated animals, and E74 transcription was analyzed by Northern blot hybridization (Fig. 2). As expected, E74B mRNA is not detectable in control newly formed prepupae, and E74A is expressed (17) (Fig. 2, lanes 2, 6). The levels of E74A transcription, however, are clearly increased in the presence of ectopic E74B expression (Fig. 2, lane 4). This result is consistent with our E74B mutant analysis, and suggests that E74B directly induces E74A transcription. E74B must, however, exert this effect together with one or more stage-specific regulators because E74A transcription is selectively reduced in E74B mutant late prepupae, and remains unaffected in mutant late third instar larvae (31).
Figure 2.
E74 regulates its own transcription. Both w (lane 2) and w; P[hs-E74B] (lane 4) late third instar larvae or w (lane 6) and w; P[hs-E74A] (lane 8) late third instar larvae were heat-shocked at 35°C for 30 min and allowed to recover at 25°C for 2 hr. Newly formed prepupae were selected from the population of animals, and RNA was extracted and analyzed by Northern blot hybridization to detect E74 transcription. RNA was also extracted from non-heat-induced w; P[hs-E74B] newly formed prepupae (lane 3) or w; P[hs-E74A] newly formed prepupae as a control (lane 7). Heat-induced E74B transcripts (hs-E74B) are still detectable 2 hr after heat shock, but heat-induced E74A transcripts are not detectable at this time (lanes 4 and 8). Hybridization to detect rp49 mRNA was used to confirm equivalent loading and transfer in each lane. The numbers to the left of the blots denote RNA marker sizes (M) in kilobases (kb) (lanes 1 and 5).
Unexpectedly, the complementary experiment revealed that ectopic expression of E74A is sufficient to induce E74B transcription in newly formed prepupae (Fig. 2, lane 8). This result is difficult to rationalize, as E74A is not detectable when E74B transcription is induced during development (17), and E74A mutations have no effect on E74B expression (31).
E74 Regulates DHR3 and E78B Early-Late Gene Expression.
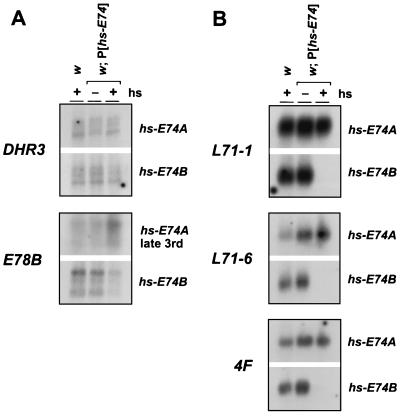
DHR3 and E78B respond in different ways to a loss of E74 activity. The peak of DHR3 expression in early prepupae is shifted earlier in E74B mutants, whereas E78B is submaximally induced in E74A mutant prepupae (31). Consistent with these loss-of-function phenotypes, DHR3 transcription is slightly repressed in the presence of ectopic E74B expression and unaffected by E74A (Fig. 3A). The inefficiency of DHR3 repression suggests that this is an indirect effect of E74B. Interestingly, ectopic E74B expression also leads to repression of E78B, suggesting that there may exist coordinate pathways for early-late gene regulation by E74. E78B transcription is unaffected by ectopic E74A expression in newly formed prepupae (data not shown), but a distinct induction can be detected in the presence of ectopic E74A in late third instar larvae (Fig. 3A). This observation is consistent with the E74A loss-of-function phenotype and indicates that E74A is both necessary and sufficient for proper E78B induction. E74A thus functions as at least one of the ecdysone-induced proteins required for maximal E78B induction at puparium formation (23).
Figure 3.
Ectopic E74B represses late gene transcription. The Northern blots of w, w; P[hs-E74A], and w; P[hs-E74B] RNA described in Fig. 2 were hybridized with a radiolabeled probe to detect (A) DHR3 mRNA or (B) L71–1, L71–6, and 4F late mRNAs. A different time point was used to detect E78B mRNA (A), where w and w; P[hs-E74A] late third instar larvae were subjected to heat shock and recovery approximately 8 hr before puparium formation. This time corresponds to the initial induction of E78B, which occurs earlier than that of DHR3. The slightly reduced levels of L71–6 and 4F mRNA in control prepupae reflect standard experimental variation for this stage in development.
E74B Is a Potent Repressor of Late Gene Transcription.
The duration of L71 transcription expands by several hours in E74B mutants, suggesting that E74B may normally repress their expression (31). In support of this hypothesis, ectopic E74B completely prevents L71–1 and L71–6 induction in newly formed prepupae (Fig. 3B). The similar effect on 4F transcription suggests that E74B may function as a general repressor of late gene transcription. This is consistent with our earlier observation that a subset of late puffs that normally peak in size in 1- to 2-hr prepupae are significantly enlarged in newly formed E74B mutant prepupae (15).
The observation that E74B is necessary and sufficient to repress late gene transcription confirms a role for E74B in the late larval salivary gland ecdysone regulatory hierarchy. This result also implies a dual function for E74B in a single tissue during third instar larval development: as an activator of salivary gland glue gene transcription (31), and as a repressor of late gene activity. An understanding of the mechanism whereby E74B is able to mediate these opposite functions awaits biochemical analysis. However, as proposed for E74A, part of the mechanism is likely to involve interactions between E74B and other factors such as those encoded by the BR-C (16).
E74A and the BR-C Are Not Sufficient for Proper Late Gene Induction, Even in the Absence of the E74B Repressor.
Unlike E74B, which represses late gene transcription, E74A appears to function as a specific late gene activator. The L71 late genes are transcribed at reduced levels and with a 2-hr delay in E74A mutants (31). To test whether E74A is sufficient and necessary to obtain proper late gene transcription, we examined L71–1, L71–6, and 4F mRNAs in transgenic animals ectopically expressing E74A at different developmental stages. We find that ectopic E74A expression has no apparent effect on the levels of late gene transcription in newly formed prepupae (Fig. 3B). However, ectopic expression of E74A in mid-third instar larvae is sufficient to prematurely induce low levels of L71–1 mRNA (Fig. 4), consistent with the loss-of-function E74A phenotype and demonstrating that E74A is sufficient to induce at least a subset of late genes.
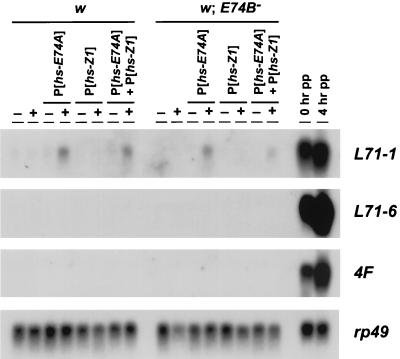
Figure 4.
BR-C Z1 and E74A are not sufficient to prematurely induce proper late gene transcription. Transgenic fly stocks were constructed that could ectopically express either E74A (P[hs-E74A]) or the BR-C Z1 protein (P[hs-Z1]), or both factors together, in wild-type (w) and E74B mutant (w; E74B−) backgrounds. Mid-third instar larvae from these lines were subjected to a 30-min heat shock at 37°C and allowed to recover at room temperature for 2 hr (+), or left at room temperature as a control (−). Both E74A and Z1 mRNA are efficiently induced by heat shock at this stage in development (data not shown). RNA was then extracted and analyzed by Northern blot hybridization to detect L71–1, L71–6, and 4F transcription. RNA isolated from staged 0 and 4-hr w prepupae was used as a positive control. Hybridization to detect rp49 mRNA was used to confirm equivalent loading and transfer in each lane.
Our inability to induce proper levels of salivary gland late gene transcription with ectopic E74A expression could be due to either the presence of the E74B repressor or the absence of another essential inducer of late genes. One candidate for this essential inducer is encoded by the BR-C Z1 isoform, corresponding to the rbp+ function (40). The rbp+ function is essential for late gene expression and exerts its effects through direct interaction of the Z1 protein with late promoters (14, 33, 41). To determine if E74A functions together with the BR-C Z1 isoform to induce late genes, we constructed transgenic lines that could ectopically express either E74A, Z1, or both proteins in either wild-type or E74B mutant larvae. We predicted that if E74A and Z1 are sufficient for late gene induction, we should see premature late gene expression only in an E74B mutant background and only in the presence of ectopic E74A and Z1. The results clearly show, however, that this is not the case (Fig. 4). No L71–6 or 4F transcription can be detected in mid-third instar larvae as a result of ectopic E74A and/or Z1 expression. Furthermore, the absence of E74B has no effect on the ability of E74A and/or Z1 to induce late promoter activity (Fig. 4).
These results demonstrate that, although E74 and the BR-C are necessary for proper L71 induction, they are not sufficient for this response and thus other regulators must contribute to this pathway. One candidate is the crooked legs (crol) gene (P.P.D. and C.S.T., unpublished work). crol mutants have no effect on E74A transcription in late third instar larvae, but show a delay in L71 induction in prepupae that is similar to that seen in E74A mutants. It is also possible that another repressor acts on the late genes in third instar larvae. One candidate for such a repressor is the ecdysone receptor itself, which exerts negative control on the late puffs, preventing their premature induction by ecdysone (21). Further studies of late gene regulation should provide a more complete understanding of this regulatory circuit.
The Ecdysone-Triggered Switch from E74B Repressor to E74A Inducer Is Required for the Timing of Late Gene Induction.
We have shown that E74A and E74B transcription is precisely coordinated by dynamic changes in ecdysone concentration (17). E74B is induced by a low ecdysone concentration and repressed by higher hormone concentrations. Furthermore, the ecdysone concentration required for 50% maximal E74B repression is similar to that required for 50% maximal E74A induction. Thus, each rise in ecdysone titer directs an obligate switch in E74 isoforms, with a period of E74B expression preceding a period of E74A activity. The timing and amounts of E74B expression are, therefore, a function of the rate at which the ecdysone titer increases. Cultured larval organs treated with a high concentration of ecdysone will result in minimal E74B expression, for about 1- to 2-hr duration, whereas E74B is expressed for about a day when the ecdysone titer rises slowly, as in third instar larvae (17, 19).
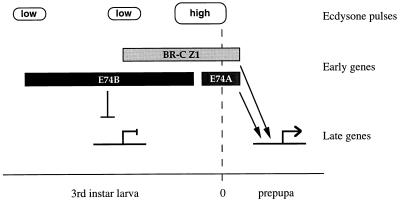
The observation that E74B is a potent repressor of late gene transcription provides a function for this elegant switch in E74 isoforms (Fig. 5). We propose that the products of rbp and E74B, which are induced in early third instar larvae by low titer pulses of ecdysone, act opposingly on the late promoters. We suggest that the presence of E74B protein counteracts Z1 and other activators that might be present, directly preventing late gene induction until the end of larval development when E74B is repressed and E74A is induced. The presence of both Z1 and E74A in newly formed prepupae then contribute to the coordinate induction of late gene transcription, together with other positive regulators such as those encoded by the crol locus. The ETS DNA-binding domain shared by E74A and E74B allows these factors to oppositely regulate the same target genes with distinct temporal specificity, permitting the tight coupling of rises in ecdysone titer to the induction of secondary-response genes. It is interesting to note that a similar switch between negative and positive ETS domain transcription factors has been described during Drosophila eye and ventral ectoderm development, with the opposing effects of yan and pointed (42, 43).
Figure 5.
A model for the regulation of L71 late gene transcription by E74 and the BR-C. The timing of BR-C Z1, E74B, and E74A expression in late larval salivary glands is represented by boxes. Low titer ecdysone pulses in early and mid-third instar larvae induce the E74B protein and BR-C Z1 isoform, respectively (18–20). Z1 acts as a general activator of L71 gene transcription, but is prevented from inducing L71 promoter activity during larval development by the presence of E74B. Just before puparium formation, the high titer late larval ecdysone pulse represses E74B and induces E74A. The presence of both Z1 and E74A proteins in newly formed prepupae contributes to the coordination of late gene induction. Our studies also show that one or more other repressors and/or inducers must contribute to this regulatory circuit.
The general features of this model provide a mechanism for obtaining the exquisite temporal specificity of the many salivary gland late puffs by a relatively small number of interacting regulatory molecules. The early induction of an activator such as the BR-C Z1 isoform may prime the genome for global change, by providing a general regulator required for local control. The expression of combinations of repressors and activators, derived from promoters with different ecdysone sensitivities, would then serve to counteract or enhance the activity of Z1 at specific late puff loci, leading to the observed waves of coordinate late puff formation. Further studies of late gene regulation should provide a more detailed understanding of these regulatory circuits and establish a foundation for understanding how dynamic changes in hormone titer can lead to diverse biological responses through the coordinate regulation of large batteries of stage-specific secondary-response genes.
Acknowledgments
We thank members of the Thummel laboratory, past and present, for comments and criticisms that have contributed to this study, and G. Guild for providing the P[hs(BRC-Q1-Z1)] transformant line and for helpful comments during the course of this work. This study was supported by the National Institutes of Health Genetics Training Grant 5T32GM-07464 (J.C.F.) and the Howard Hughes Medical Institute (P.P.D. and C.S.T.).
References
- 1.Beato M, Herrlich P, Schütz G. Cell. 1995;83:851–857. doi: 10.1016/0092-8674(95)90201-5. [DOI] [PubMed] [Google Scholar]
- 2.Tsai M-J, O’Malley B W. Annu Rev Biochem. 1994;63:451–486. doi: 10.1146/annurev.bi.63.070194.002315. [DOI] [PubMed] [Google Scholar]
- 3.Riddiford L M. In: Hormones and Drosophila Development. Bate M, Martinez-Arias A, editors. Vol. 2. Plainview, NY: Cold Spring Harbor Lab. Press; 1993. pp. 899–939. [Google Scholar]
- 4.Becker H J. Chromosoma. 1959;10:654–678. doi: 10.1007/BF00396591. [DOI] [PubMed] [Google Scholar]
- 5.Ashburner M, Chihara C, Meltzer P, Richards G. Cold Spring Harbor Symp Quant Biol. 1974;38:655–662. doi: 10.1101/sqb.1974.038.01.070. [DOI] [PubMed] [Google Scholar]
- 6.Russell S, Ashburner M. In: Ecdysone-Regulated Chromosome Puffing in Drosophila melanogaster. Gilbert L I, Tata J R, Atkinson B G, editors. New York: Academic; 1996. pp. 109–144. [Google Scholar]
- 7.Thummel C S. Trends Genet. 1996;12:306–310. doi: 10.1016/0168-9525(96)10032-9. [DOI] [PubMed] [Google Scholar]
- 8.Andres A J, Thummel C S. Development. 1995;121:2667–2679. doi: 10.1242/dev.121.8.2667. [DOI] [PubMed] [Google Scholar]
- 9.DiBello P R, Withers D A, Bayer C A, Fristrom J W, Guild G M. Genetics. 1991;129:385–397. doi: 10.1093/genetics/129.2.385. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Segraves W A, Hogness D S. Genes Dev. 1990;4:204–219. doi: 10.1101/gad.4.2.204. [DOI] [PubMed] [Google Scholar]
- 11.Burtis K C, Thummel C S, Jones C W, Karim F D, Hogness D S. Cell. 1990;61:85–99. doi: 10.1016/0092-8674(90)90217-3. [DOI] [PubMed] [Google Scholar]
- 12.Bayer C A, Holley B, Fristrom J W. Dev Biol. 1996;177:1–14. doi: 10.1006/dbio.1996.0140. [DOI] [PubMed] [Google Scholar]
- 13.Kiss I, Beaton A H, Tardiff J, Fristrom D, Fristrom J W. Genetics. 1988;118:247–259. doi: 10.1093/genetics/118.2.247. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Guay P S, Guild G M. Genetics. 1991;129:169–175. doi: 10.1093/genetics/129.1.169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Fletcher J C, Burtis K C, Hogness D S, Thummel C S. Development. 1995;121:1455–1465. doi: 10.1242/dev.121.5.1455. [DOI] [PubMed] [Google Scholar]
- 16.Fletcher J C, Thummel C S. Genetics. 1995;141:1025–1035. doi: 10.1093/genetics/141.3.1025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Karim F D, Thummel C S. Genes Dev. 1991;5:1067–1079. doi: 10.1101/gad.5.6.1067. [DOI] [PubMed] [Google Scholar]
- 18.Karim F D, Thummel C S. EMBO J. 1992;11:4083–4093. doi: 10.1002/j.1460-2075.1992.tb05501.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Andres A J, Fletcher J C, Karim F K, Thummel C S. Dev Biol. 1993;160:388–404. doi: 10.1006/dbio.1993.1315. [DOI] [PubMed] [Google Scholar]
- 20.Huet F, Puiz C, Richards G. Development. 1993;118:613–627. doi: 10.1242/dev.118.2.613. [DOI] [PubMed] [Google Scholar]
- 21.Ashburner M, Richards G. Dev Biol. 1976;54:241–255. doi: 10.1016/0012-1606(76)90302-x. [DOI] [PubMed] [Google Scholar]
- 22.Koelle M R, Segraves W A, Hogness D S. Proc Natl Acad Sci USA. 1992;89:6167–6171. doi: 10.1073/pnas.89.13.6167. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Stone B L, Thummel C S. Cell. 1993;75:307–320. doi: 10.1016/0092-8674(93)80072-m. [DOI] [PubMed] [Google Scholar]
- 24.Horner M A, Chen T, Thummel C S. Dev Biol. 1995;168:490–502. doi: 10.1006/dbio.1995.1097. [DOI] [PubMed] [Google Scholar]
- 25.Russell S R H, Heimbeck G, Goddard C M, Carpenter A T C, Ashburner M. Genetics. 1996;144:159–170. doi: 10.1093/genetics/144.1.159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Lam, G. T., Jiang, C. & Thummel, C. S. (1997) Development, in press. [DOI] [PubMed]
- 27.White, K. P., Hurban, P., Watanabe, T. & Hogness, D. S. (1997) Science, in press. [DOI] [PubMed]
- 28.Wolfner M. Ph.D. thesis. Stanford, CA: Stanford University; 1980. [Google Scholar]
- 29.Wright L G, Chen T, Thummel C S, Guild G M. J Mol Biol. 1996;255:387–400. doi: 10.1006/jmbi.1996.0032. [DOI] [PubMed] [Google Scholar]
- 30.Karim F D, Guild G M, Thummel C S. Development. 1993;118:977–988. doi: 10.1242/dev.118.3.977. [DOI] [PubMed] [Google Scholar]
- 31.Fletcher J C, Thummel C S. Development. 1995;121:1411–1421. doi: 10.1242/dev.121.5.1411. [DOI] [PubMed] [Google Scholar]
- 32.Urness L D, Thummel C S. EMBO J. 1995;14:6239–6246. doi: 10.1002/j.1460-2075.1995.tb00314.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Crossgrove K, Bayer C A, Fristrom J W, Guild G M. Dev Biol. 1996;180:745–758. doi: 10.1006/dbio.1996.0343. [DOI] [PubMed] [Google Scholar]
- 34.Thummel C S, Pirrota V. Dros Info Serv. 1992;71:150. [Google Scholar]
- 35.Robertson H M, Preston C R, Phillis R W, Johnson-Schlitz D M, Benz W K, Engels W R. Genetics. 1988;118:461–470. doi: 10.1093/genetics/118.3.461. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Rubin G M, Spradling A C. Science. 1982;218:348–353. doi: 10.1126/science.6289436. [DOI] [PubMed] [Google Scholar]
- 37.Andres A J, Thummel C S. In: Methods for Quantitative Analysis of Transcription in Larvae and Prepupae. Goldstein L S B, Fyrberg E A, editors. New York: Academic; 1994. pp. 565–573. [DOI] [PubMed] [Google Scholar]
- 38.Urness L D, Thummel C S. Cell. 1990;63:47–61. doi: 10.1016/0092-8674(90)90287-o. [DOI] [PubMed] [Google Scholar]
- 39.Karim F D. Ph.D. thesis. Salt Lake City: University of Utah; 1992. [Google Scholar]
- 40.Emery I F, Bedian V, Guild G M. Development. 1994;120:3275–3287. doi: 10.1242/dev.120.11.3275. [DOI] [PubMed] [Google Scholar]
- 41.Belyaeva E S, Vlassova I E, Biyasheva Z M, Kakpakov V T, Richards G, Zhimulev I F. Chromosoma. 1981;84:207–219. doi: 10.1007/BF00399132. [DOI] [PubMed] [Google Scholar]
- 42.O’Neill E M, Rebay I, Tjian R, Rubin G M. Cell. 1994;78:137–147. doi: 10.1016/0092-8674(94)90580-0. [DOI] [PubMed] [Google Scholar]
- 43.Gabay L, Scholz H, Golembo M, Klaes A, Shilo B, Klämbt C. Development. 1996;122:3355–3362. doi: 10.1242/dev.122.11.3355. [DOI] [PubMed] [Google Scholar]