Abstract
Sterols such as cholesterol are important components of cellular membranes. They are not uniformly distributed among organelles and maintaining the proper distribution of sterols is critical for many cellular functions. Both vesicular and non-vesicular pathways move sterols between membranes and into and out of cells. There is growing evidence that a number of non-vesicular transport pathways operate in cells and, in the past few years, a number of proteins have been proposed to facilitate this transfer. Some are soluble sterol transfer proteins that may move sterol between membranes. Others are integral membranes proteins that mediate sterol efflux, uptake from cells, and perhaps intracellular sterol transfer as well. In most cases, the mechanisms and regulation of these proteins remains poorly understood. This review summarizes our current knowledge of these proteins and how they could contribute to intracellular sterol trafficking and distribution.
1. Introduction
Cholesterol is required for the viability of mammalian cells and has many important functions [1-3]. It contributes to maintaining the permeability barrier of the plasma membrane (PM) and is also critical for intracellular trafficking. The ability of cholesterol to form cholesterol sphingolipid-enriched microdomains in membranes is likely also important for the proper distribution of lipids and proteins within cells [2, 4-6]. In addition, cholesterol metabolites, such as steroid hormones, oxysterols, and bile acids have many important physiological functions.
Cells tightly regulate cholesterol levels by a number of mechanisms, but primarily via a feedback pathway that controls the synthesis and uptake of cholesterol [7]. This pathway uses the sterol-gated transport of the membrane-bound transcription factor sterol regulatory element binding protein (SREBP) from the endoplasmic reticulum (ER) to the Golgi complex. When sterol levels are low, SREBP is moved to the Golgi complex where it is cleaved to a soluble form that enters the nucleus and increases expression of genes required for cholesterol synthesis and uptake. Cholesterol synthesis occurs mostly in the ER [8]. Exogenous cholesterol is obtained primarily from plasma lipoproteins such as low- and high-density lipoproteins (LDL and HDL). Cholesteryl esters in lipoproteins are hydrolyzed in late endodomes and lysosomes and the free cholesterol is then trafficked from these compartments to the rest of the cell. Cholesterol can also be effluxed to external acceptors in some cell types. In addition, excess cholesterol can be esterified with fatty acids and stored in cytosolic lipid droplets (LDs).
Cholesterol and other lipids are not uniformly distributed in cells [9-11]. Maintaining proper cholesterol distribution is essential for many cellular functions including membrane trafficking and cell signaling. The highest concentrations of cholesterol are found in the PM, where it has been estimated to be about 35-45% of the lipid in the PM [9, 12, 13]. Approximately 65 to 80% of the free cholesterol in cells is in the PM. In contrast, the ER and mitochondria contain only very low amounts of cholesterol [14-16]. The cholesterol content of the membranes of the Golgi complex is probably intermediate between that of the ER and PM and it has been proposed that there is a cholesterol gradient across the membranes of the secretory pathway [17-19]. A substantial amount of free cholesterol is also found in early endosomes and endocytic recycling compartments (ERC) [20-22].
Many non-mammalian eukaryotic cells contain sterols other than cholesterol. These sterols, such as sitosterol in some plants and ergosterol in fungi, differ slightly from cholesterol but have similar physical properties and intracellular distribution [23-27]. Sterols likely have comparable functions in most eukaryotic cells.
How cells maintain large differences in the concentration of sterols in various organelles is not well understood. Cholesterol is moved among cellular compartments by a combination of vesicular and non-vesicular pathways [9, 11, 28, 29]. Since there is continuous membrane vesicular trafficking between the various compartments of the secretory and endocytic pathways, some of which have very different cholesterol concentrations, cholesterol is probably sorted during transport vesicle formation. For example, to maintain the high concentration of cholesterol in the PM, cholesterol could be sorted into vesicles headed toward to the PM and out of vesicles moving cargo away from the PM. There is some evidence that such sorting occurs, cholesterol and sphingomyelin are partially excluded from COPI-coated vesicles, which form in the Golgi complex [30]. How cholesterol sorting during vesicle formation occurs is not well understood. The sorting of cholesterol and other lipids could be driven by their affinity for microdomains and the effects of membrane curvature [31, 32]. Cholesterol can also be moved between a number of cellular compartments or effluxed from cells by non-vesicular transport pathways. This review summarizes our current knowledge of these pathways. First, I outline the known non-vesicular cholesterol transport pathways in cells. Second I discuss the role of soluble transfer proteins (LPTs) in non-vesicular cholesterol transport. Third, the roles of membranes proteins in facilitating sterol uptake and efflux and possibly transport between cellular membranes will be addressed. Finally, I discuss the directionality and regulation of non-vesicular transport and how it might contribute to maintaining intracellular sterol distribution.
2. Non-vesicular sterol transport pathways
There is growing evidence that sterols (and other lipids) can move between many intracellular compartments by non-vesicular mechanisms. However, demonstrating this in cells is not trivial for a number of reasons. First, tracking the distribution and movement of sterols in vivo is challenging. A number of methods are used and have been thoroughly reviewed previously [33]. Second, sterols are moved between some cellular compartments by a combination of vesicular and non-vesicular pathways. Finally, unlike most lipids that have two long fatty-acyl chains, cholesterol can spontaneously move between bilayers unassisted by any proteins; and this process has been demonstrated both with pure liposomes and membranes derived from cells [34-36]. Though this movement is relatively slow compared to rates at which sterols are trafficked between membranes, it complicates the analysis of sterol transport and measurements of the sterol concentration in cell membranes. Thus, while non-vesicular transport pathways may move sterols between many cellular compartments, I focus here on the cases where there is compelling evidence for such transport. These pathways and some of the proteins that may be involved are shown in Figure 1.
Figure 1.
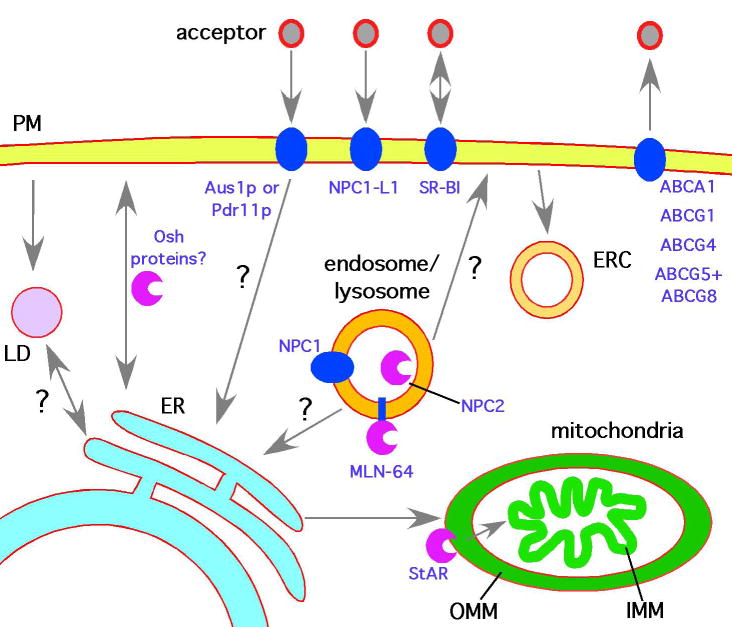
Non-vesicular sterol transport pathways in cells. Pathways that are only speculative are marked with a “?”. Non-vesicular sterol transport pathways probably move sterols between the ER and many organelles, perhaps at membrane contact sites. Organelles shown: endoplasmic reticulum (ER), plasma membrane (PM), inner mitochondrial membrane (IMM), outer mitochondrial membrane (OMM), endocytic recycling compartment (ERC), lipid droplet (LD), and external acceptor, which could be a lipoproteins or detergent micelle depending on cell type. Some of the proteins mentioned in the text are shown in blue. It should be noted that all of these proteins are not expressed in the same cells.
2.1 Transfer of cholesterol to the outer and inner mitochondrial membranes
There is no known vesicular trafficking between mitochondria and other cellular compartments. Since mitochondria cannot synthesize most lipids including sterols, they must get them by non-vesicular transfer pathways. This has been demonstrated for phospholipids [37] and is likely true of sterols as well, though little is know about this transport. Cholesterol is also moved from the outer to the inner mitochondrial membranes by a non-vesicular mechanism in some cells. This transport is required for the biogenesis of steroids, which are derived from cholesterol. The first step in steroid synthesis requires the cleavage of the cholesterol side chain to form pregnenolone by the cytochrome P450 side chain cleavage enzyme, located in the inner membrane of mitochondria [38]. The non-vesicular transfer of cholesterol from the outer to inner mitochondrial membrane is the rate limiting step in steroid biogenesis [39] and requires the steroidogenic acute regulator protein (StAR), which is discussed below.
2.2 PM to ERC sterol transport
In order to study sterol uptake and trafficking in live cells, a number of studies have used the naturally fluorescent sterol Dehydroergosterol (DHE). This sterol is a good analogue of cholesterol because it has similar physical properties and behavior in biological membranes [22, 40-45]. Maxfield and collegues found that exogenous DHE was rapidly trafficked from the PM to the ERC with a half-time of only 2.5 minutes and indeed that a substantial portion of the free DHE and cholesterol in cells is in this compartment [22]. This transport was only slightly slowed by depleting cells of ATP, while the vesicular movement of transferrin from the PM to the ERC was blocked by the same conditions. Using density gradient centrifugation, they were able to show that transport of exogenous [3H]cholesterol to the ERC is probably also energy-independent. Thus, the rapid transfer of sterols from the PM to the ERC is likely non-vesicular.
2.3 Sterol movement to and from lipid droplets (LDs)
LDs serve as lipid storage depots and consist of a core of steryl ester and triglyceride surrounded by a monolayer of phospholipids and various proteins. There is no known vesicular transport to or from these organelles. In macrophage foam cells, exogenous DHE was transferred not to the ERC but to lipid droplets (LDs) instead [46]. As with PM to ERC DHE transfer, the movement of DHE from the PM to LDs in foam cells was very rapid, with a half-time of 1.5 minutes, and did not require energy. This transport is therefore likely non-vesicular.
The movement of sterols between LDs and the ER is also non-vesicular and may require proteins to transfer lipids between these organelles in some cases. Steryl esters are synthesized in the ER, but reside primarily in LDs. How they move to LDs is not well understood. LDs probably derive from the ER and it has been suggested that the phospholipid mononlayer of LDs remains continuous with the outer leaflet of the ER [47, 48]. If this is the case, then steryl esters could simply diffuse between the ER and LDs via these connections. However, it remains possible that some LDs are not connected to the ER and the movement of steryl esters to these LDs requires proteins to facilitate this transfer. The movement of sterols from LDs to the ER (and other membranes) could also require such proteins. When sterols are mobilized from LDs, enzymes on these organelles hydrolyze steryl esters and the free sterols must then be transferred to the ER and other membranes.
Ergosterol biosynthesis in the yeast S cerevisiae may also require non-vesicular sterol transfer between the ER and LDs. In this organism, ergosterol is synthesized by a series of enzymes known as Erg proteins. While most are located on the ER, a few are found localized either exclusively or partially on LDs [24, 49-52]. Thus, ergosterol biosynthesis in S. cerevisiae may require the non-vesicular transfer of sterol intermediates between the ER and LDs. This process may be facilitated by transport proteins.
2.4 Sterol transfer between the ER and PM
After cholesterol is synthesized in the ER, it is rapidly transferred to the PM with a half-time of 10 to 20 minutes in mammalian cells [53, 54]. Treating cells with brefeldin A, which causes Golgi complex disassembly and blocks the movement of proteins from the ER to the PM, only slightly slows the delivery of newly synthesized cholesterol to the PM [54-56]. Similar results were obtained with the yeast S. cerevisiae; newly synthesized ergosterol was moved between the ER and PM with a half-time of about 10 minutes. This rate was not significantly reduced in a number of SEC mutants with conditional defects in proteins required for vesicular trafficking between the ER and PM, including Sec18p, the yeast homologue of N-ethylmaleimide-sensitive factor (NSF) in mammalian cells [57]. Sec18p is needed for most vesicular transport in cells [58, 59]. Another group found that ER to PM sterol transport in yeast was partially blocked (20-50%) in some SEC mutants, but that ergosterol could still move from the ER to the PM by a SEC-independent pathway [60]. Taken together, these findings suggest that in both yeast and mammalian cells, a non-vesicular pathway can likely move a substantial portion newly synthesized sterols from the ER to the PM. It is not known what fraction of newly synthesized sterol moves to the PM by this pathway, though it can accommodate most if not all of the flux.
There is also good evidence that non-vesicular pathways can move sterols from the PM back to the ER. These studies make use of the fact that the enzymes that esterify free sterols are primarily localized in the ER; sterol esterification is used as an indication of sterol transfer to the ER. In the yeast S. cerevisiae, exogenous sterols enter the PM, are moved the ER, and esterified. This transfer is not blocked in a number of SEC mutants, indicating that it is likely non-vesicular [61]. PM to ER cholesterol transfer in mammalian cells may also be non-vesicular. Treating mammalian cells with sphingomyelinease causes cholesterol to redistribute to internal compartments, including the ER [62, 63]. This transport was not inhibited by ATP-depletion or other treatments that affect known vesicular transport pathways, including addition of N-ethylmaleimide and inhibitors of phosphatidylinositol 3-kinase [64]. Though the authors suggest a novel vesicular pathway moves PM cholesterol to the ER, their results are also consistent with non-vesicular transfer.
It is important to note that, in yeast, at least one protein may be trafficked from the ER to the PM by a pathway that does not need Sec18p [65], indicating that a novel vesicular transport pathway may operate between the ER and PM. Thus it remains possible that not all SEC-independent sterol transfer between the ER and PM is nonvesicular.
2.5 Sterol efflux from cells
Many mammalian cells move cholesterol and other sterols from the PM to external acceptors. This is an important mechanism for regulating cellular cholesterol levels. Sterol desorption or efflux from the PM is mediated by a number of proteins, primarily ATP binding cassette (ABC) transporters, which will be discussed below.
3. Mechanisms of non-vesicular sterol transport in cells
Some of the known and potential mechanisms of sterol transfer between membranes are shown in Figure 2. As mentioned above, sterols can move between membrane unassisted by any protein (Figure 2A) [34-36]. For liposomes, the rate-limiting step in the process is likely the rate at which cholesterol desorbs or escapes from membranes [34]. Though it has been known for some time this rate is affected by the lipid composition and shape of membranes, more recent studies point to the importance of interactions between sterols and phospholipids in determining rates of cholesterol desorption from membranes. These theoretical and experimental studies suggest that sterols in membranes exist in two pools, one with low chemical activity and a second with high chemical activity. Sterol with high chemical activity has a higher escape potential (or fugacity) than sterol with low chemical activity. There are two related models of what determines sterol fugacity in membranes. In one, the small headgroup of sterols is not sufficient to shield the hydrophobic portion of these molecules from water [66]. Consequently, sterols tend to take cover under the “umbrella” of the larger headgroups of phospholipids and sterol-sterol interactions are disfavored. Shielded sterols have low chemical activity, but once the sterol concentration of a membrane exceeds the capacity of phospholipids to provide cover, the excess sterol has a high chemical activity. In a second model, McConnell and coworkers suggest that sterols tend to associate with phospholipids, particularly those with saturated acyl chains, and form “condensed complexes.” Sterols that partition into these complexes have lower chemical activity than those outside. Both models predict that the chemical activity or fugacity of sterols increases substantially when sterol concentration in membranes exceeds the ability of phospholipids to partner with sterols. Beautiful work from Lange, Steck, and coworkers has confirmed this prediction in cells [67-69]. Just as the chemical activity of sterols in membranes determines its propensity to escape membranes, the ability of cellular membranes to act as sterol acceptors is at least partially also determined by their lipid composition [70].
Figure 2.
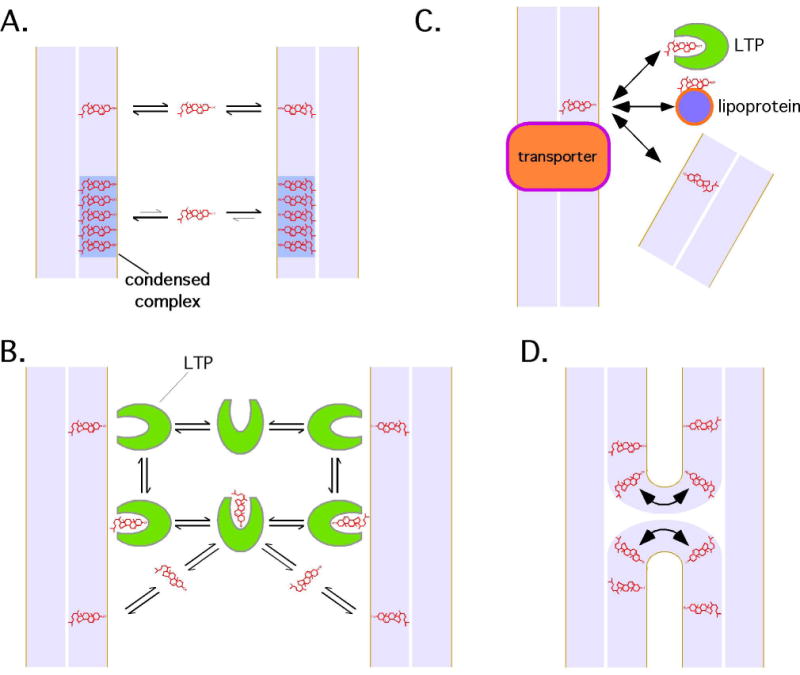
Possible mechanisms of non-vesicular sterol transfer. Sterols are shown in red, the membranes bilayer is light blue. A. Spontaneous movement of sterols through the aqueous phase. Sterol in “condensed complexes,” perhaps sterol-sphingolipid enriched microdomains (see text), probably desorbs from membranes more slowly than sterols outside such domains. B. Transfer by soluble lipid transfer proteins (LTPs). These proteins could bind membranes directly when they extract or deliver sterols. Alternatively, they may bind sterols in the aqueous phase. C. Sterol transfer by membrane proteins. These proteins could facilitate transfer between membranes and external acceptors like lipoproteins during sterol uptake or efflux. They could also facilitate sterol movement to LTPs or directly to another membrane. D. Sterols and other lipids could be exchanged between membranes that do not subsequently fuse by transient hemifusion. There is no evidence that this mechanism is used in cells.
Although sterols can spontaneously move between membranes through the aqueous phase, this process is slow compared to rates of non-vesicular lipid transfer in vivo and is likely facilitated in some cases by soluble lipid transfer proteins (LTPs) (Figure 2B). These proteins tend to bind lipid with a 1:1 stochimetry in a hydrophobic tunnel or pit, sometimes covered by a hinged lid domain. LTPs may directly bind membranes, extract sterols, and deliver them to acceptor membranes or could bind and/or release sterols in the aqueous phase. Possible sterol LTPs are discussed below.
Membrane proteins can also facilitate the movement of sterols into and out of membranes (Figure 2C). They may transfer sterols between membranes and LTPs or other soluble acceptors, other membranes, or the aqueous phase. Membrane proteins can directly transport sterols or indirectly affect sterol transfer by modulating the chemical activity of sterols in a membrane. For example, they could change the distribution of lipids between the leaflets of a membrane or the lateral organization of lipids within membranes.
Finally, it is possible that proteins could facilitate lipid transfer between membranes by causing transient hemifusion between two membranes (Figure 2D). In hemifused membranes, lipids in one of the two leaflets of each bilayer can mix. Hemifused membranes that allow lipid exchange have been shown to be intermediates during membrane fusion reactions [71]. However, there is no evidence that transient hemifusion is used to move lipid between membranes that do not subsequently fuse.
3.1 Membrane contact sites
Many organelles have domains of close apposition with each other. This is particularly true of the ER, regions of which can be seen closely apposed to virtually all other cellular organelles. Since the ER extends throughout the cell, some of these contacts may be coincidental but probably not all. These regions, known as membrane contact sites (MCSs), may facilitate the non-vesicular transfer of sterols and other lipids [72, 73]. Indeed, there is substantial evidence consistent with a role for MCSs in non-vesicular lipid transfer between the ER and a number of organelles [37, 50, 51, 74-79]. The small distances LTPs would have to diffuse between membranes at MCSs might facilitate the transfer of sterols and other lipids. More importantly, close apposition of two membranes might enhance lipid transfer between them by LTPs since it would increase the probability a LTP that has extracted a lipid from one membrane would collide with and deliver the lipid to the second membrane. In some cases, LTPs may bind simultaneously to both donor and acceptor membranes. For example, CERT, which transfers ceramides from the ER to the Golgi complex, has domains that allow it to interact with both organelles: a plextrin homology (PH) domain, which binds phosphoinositides on the Golgi complex, and a FFAT motif that binds the ER resident protein VAP [80]. Many oxysterol-binding protein (OSBP)-related proteins (ORPs), also have these domains [81].
Little is known about the proteins responsible for maintaining MCSs. The one exception is the nucleus vacuole junction (NVJ) in the yeast S. cerevisiae, which is the site of piecemeal microautophagy of nuclear material [82]. This structure is held together by Vac8p on the vacuolar membrane and Nvj1p on the outer nuclear membrane [83]. Though some of the proteins recruited to the NVJ have been identified, including the ORP Osh1p, the role of the NVJ in microautophagy or other cellular processes remains obscure. It is not yet known if the NVJ is important for the transfer of sterols or other lipids between these organelles.
4. Sterol transfer proteins
As outlined above, there is strong evidence for non-vesicular sterol transfer among some cellular compartments. However, in most cases the proteins required have not been identified. A number of soluble sterol-binding proteins have been proposed to transfer sterols among cellular compartments. These fall into five families. Though some members of these protein families can extract sterols from membranes and transfer sterols between membranes in vitro, it is less clear that they do in cells and what their other functions are. In this way, they are similar to phospholipid-transfer proteins (PL-TPs), which have been studied for many years [84-86]. These proteins transfer phospholipids between membranes in vitro but many probably do not in transfer lipids between membranes in cells. Instead, they may deliver lipids directly to metabolic enzymes or function as lipid sensors that regulate other enzymes in response to binding lipids.
4.1 Steroidogenic acute regulatory protein (StAR) and StAR-related lipid transfer (START) proteins
StAR is required for the efficient transfer of cholesterol from the outer mitochondrial membrane (OMM) to the inner membrane (IMM), where it is converted to pregnenolone, the first step in steroid biogenesis [87]. The protein is synthesized with an N-terminal mitochondrial targeting sequence and the mature form of the proteins binds cholesterol with a 1:1 stoichiometry [88]. In addition, StAR functions as an LTP in vitro, rapidly moving cholesterol from liposomes to isolated mitochondria and microsomes [89].
The mechanism of StAR action in cells is controversial. Since StAR is synthesized with a cleavable targeting sequence and the cytosolic form of the protein is short lived, it is simplest to imagine that StAR shuttles cholesterol between the OMM and IMM. However, this seems not to be correct. Surprisingly, StAR lacking its targeting sequence (N-62 StAR) is still functional in cells (i.e., it stimulates pregnenolone production), suggesting it acts on the cytosolic face of the OMM [90, 91]. This is consistent with further studies in which StAR was fused to proteins localized to each of the mitochondrial compartments. StAR was only active when it was fused to the C-termius of Tom20p, which faces the cytosolic surface of the OMM [92]. Moreover, slowing the entry of the wild-type protein into mitochondria increased its activity, consistent with the idea that only the short-lived cytosolic form of the protein is active. Taken together, these findings suggest that StAR acts on the cytosolic face of the OMM and directly stimulates cholesterol transfer from the OMM to the IMM. How it does this remains unclear [93, 94].
One possibility is that StAR mobilizes cholesterol in the OMM so that it can be transferred to the IMM by another protein. This could be the function of the peripheral benzodiazepine receptor (PBR), an integral membrane protein in the OMM [95]. Consistent with this idea, PBR is highly expressed in steroidogenic cells and PBR ligands stimulate steroid biosynthesis. More importantly, disruption of the PBR gene dramatically reduces steroid production in Leydig tumor cells [96]. In addition, PBR has a cytosolically exposed cholesterol recognition amino acid consensus (CRAC) domain which is required for cholesterol transfer to the IMM [95]. Whether StAR delivers cholesterol to PBR or PBR affects cholesterol transport indirectly, perhaps by affecting the chemical activity of OMM cholesterol, remains to be determined.
Humans have at least 14 START-containing proteins in addition to StAR [97, 98]. Two of these, MLN64 and STARD5, are know to bind cholesterol [88, 99]. Others bind different lipids, including phospholipids and ceramides, or have unknown ligands [97]. Little is know about STARD5 function, but it could be an LTP [99, 100].
MLN64 might also function as an LTP. Unlike the other START proteins in humans, it is an integral membane protein. The amino terminal half of the protein has four transmembranes domains that target the protein to the limiting membrane of late endosomes [101]. The C-terminal half consists of a cytosolic START domain that binds cholesterol with a 1:1 stoichiometry [88]. This region of the protein has been crystallized and revealed to contain a hydrophobic tunnel that can bind a single cholesterol molecule, consistent with a role in cholesterol transfer [88]. In vitro, the START domain facilitates the transfer of sterols to liposomes and mitochondria [102]. Indeed, it has been proposed that MLN64 mediates sterol transfer from the endosomal membrane, to either an unknown acceptor or another membrane [101, 102]. The N-terminal region of the protein, which contains the transmembrane segments, has been termed the MENTAL domain and shown to bind a photoactivatable sterol analogue [103]. This domain homodimerizes and heterodimerizes with another proteins called MENTHO, which has substantial homology to the MENTAL domain of MLN64 but lacks START domain. It has been proposed that MLN64 binds cholesterol with its MENTAL domain that is then extracted by the START domain and transferred to a cytosolic acceptor or another membrane [103].
4.2 NPC2
Niemann Pick Type C (NPC) is a rare lipid storage disease characterized by the accumulation of cholesterol and other lipids in a late endosomal/lysosomal compartment [104-107]. The disease is caused by mutations in either of two genes, NPC1 and NPC2 [108]. The accumulated evidence suggests that both proteins are part of a pathway required for the trafficking of LDL-derived cholesterol out of lysosomes, but the exact functions of the proteins in cells remains enigmatic [109]. NPC1 is a membrane protein found on the limiting membrane of endosomes [110, 111], while NPC2 is a small, soluble glycoprotein inside endosomal/lysosomal compartments [112]. NPC2 binds cholesterol with a molecular stoichiometery of 1 [113, 114]. The structure of apo-NPC2 has been solved and, surprisingly, revealed that it has a hydrophobic tunnel that seems too small to accommodate a cholesterol molecule [115]. Therefore, it likely undergoes a conformational change during cholesterol binding that increases the volume of the binding tunnel. In an elegant study, it was recently shown that NPC2 can rapidly deliver cholesterol to liposomes by a collisional mechanism, consistent with a role in non-vesicular cholesterol transfer in cells [116].
4.3 ORPs
Oxysterol-binding protein (OSBP) was the founding member of a large family of lipid-binding proteins. It was initially identified because it bound oxysterols, oxygenated cholesterol derivatives that are potent regulators of cholesterol metabolism in mammals [117]. A large number of OSBP-related proteins (ORPs) have been identified in eukaryotes from yeast to humans, which have at least twelve [81]. All contain a lipid-binding OSBP-related domain (ORD) and most contain additional domains. They are thought to play roles in numerous cellular processes including regulation of lipid distribution and metabolism, cell signaling, and vesicular transport.
Recently, ORPs have also been suggested to function as soluble sterol transporters [118, 119]. This evidence comes from work in the yeast S. cerevisiae, which has seven ORPs called Osh proteins (Osh stands for OSBP-homolog). As mentioned above, the transfer of sterols between the ER and PM in this yeast is unaffected in numerous mutants with conditional defects in proteins required for vesicular trafficking between these compartments. Yeast requires any one of the seven Osh proteins for viability and it had previously been shown that intracellular sterol distribution is altered in a strain missing six of these and having a conditional defect in the seventh (oshΔ osh4-1) [120, 121]. The transport of exogenous sterols from the PM to the ER also slows significantly in the oshΔ osh4-1 strain [119], suggesting that Osh proteins could be soluble sterol transfer proteins [119]. The transfer of newly synthesized sterol from the ER to the PM also slows significantly in the oshΔ osh4-1 strain [122]. Consistent with a role for Osh proteins in non-vesicular sterol transfer, Osh4p (also known as Kes1p) binds cholesterol with nanomolar affinity [118] and transfers cholesterol and ergosterol between liposomes in vitro [119]. The structure of Osh4p is also consistent with a role in lipid transfer. It reveals that Osh4p binds sterols and oxysterols in a hydrophobic tunnel covered by a flexible “lid” region. Near the entrance of the tunnel are a number of highly conserved charged resides that are likely required interacting with the charged surface of membranes. Mutation of many of these residues significantly reduced the ability of the protein to transfer sterols in vitro and function in cells [118, 119].
It seems likely that a number of other Osh proteins also bind sterols and transport them in cells. Cells missing any one of the seven Osh proteins have only little to no defect in PM to ER sterol transfer, suggesting that a number of these proteins may function as sterol transfer proteins [119]. Osh5p, the Osh protein most similar to Osh4p, can bind and transfer sterols as well as Osh4p in vitro (unpublished observation).
It is interesting to note that, in vitro, sterol transfer by Osh4p is specifically enhanced by a phosphoinositide (PIP), specifically PI(4,5)P2 [119]. This is also true of Osh5p (unpublished observation). This lipid is highly enriched in some membranes, particularly the PM [123], and PI(4,5)P2-simulation of sterol transfer by Osh4p and could regulate sterol transfer to and from this organelle. Indeed, since most of the Osh proteins probably bind to PIPs [124, 125], PIP-binding may regulate the function of a number of the Osh proteins. Consistent with this, depletion of PI(4)P and PI(4,5)P2 in cells substantially slows sterol transport from the PM to the ER [119].
It remains possible that primary function of Osh proteins in vivo is not nonvesicular sterol transfer [11, 122]. To date, there is no evidence that any of the mammalian ORPs function as sterol transfer proteins. On the other hand, there is substantial evidence that OSBP functions as a lipid sensor [126, 127] and that ORP1L is part of a complex that activates endosomal dynein motors [128]. In addition, there is still some sterol transfer between the PM and ER in both directions even in cells lacking Osh proteins, albeit 5 to 10 times more slowly than in wild-type cells [119, 122]. This residual transport probably occurs too rapidly to be passive diffusion of sterols unassisted by any carrier proteins, suggesting that proteins other than the Osh proteins may also transfer sterols between the ER and PM. S. cerevisiae lacks homologs of START proteins, sterol carrier protein 2 (SCP2), or caveolins (other candidate sterol transfer proteins discussed in the following sections), but could contain novel transport proteins. (It does contain an NPC2 homolog, but this resides in the vacuole [129].) Thus, it is difficult to rule out that Osh proteins, like many PL-TPs, do not function at lipid transfer proteins in cells and only indirectly affect lipid transport between the ER and PM. Whether and how any of the Osh proteins might function as lipid sensors remains to be determined.
4.4 SCP-2
SCP-2 is a small 13.3kDa protein that is also known as non-specific lipid transfer protein (nsL-TP) because of its ability to transfer sterols and a variety of other lipids between membranes in vitro [130, 131]. This protein is formed by the cleavage of either of two larger proteins termed SCP-x and pro-SCP-2, both of which are encoded by the same gene by two distinct promoters. The SCP-2 domain is at the C-terminus of both proteins. SCP-x also contains an N-terminal 3-oxoacyl-CoA thiolase domain, while pro-SCP2 has a small N-terminal 2kDa domain. However, in many tissues much of the SCP-x remains intact. Both proteins are made with a C-terminal peroxisomal targeting signals and are likely involved in peroxisomal β-oxidiation of fatty acids, perhaps by facilitating substrate presentation to the enzymes involved. Indeed, mice lacking the gene encoding SCP-x/pro-SCP-2 have defects in branched fatty acid catabolism [132].
In addition to its role fatty acid oxidiation in peroxisomes, it has been suggested that SCP-2 may transfer sterols and other lipids between some cellular compartments. Despite having a peroxisomal targeting signal, a fraction of SCP-2 is found in the cytosol [133, 134]. Since there are numerous studies demonstrating that SCP-2 can transfer cholesterol between liposomes and/or biological membranes (thoroughly reviewed in [131]), it remains possible that cytosolic SCP-2 might facilitate non-vesicular sterol transfer in cells as well. A number of papers suggest that cellular cholesterol distribution and trafficking may be altered when SCP-2 expression levels are either increased or decreased [135-141], consistent with a role in intracellular sterol transport, but this awaits definitive proof.
4.5 Caveolins
Caveolins are abundant membrane proteins that associate with cholesterol and sphingolipids. They can oligomerize, leading to the formation of caveolae, cholesterol and sphingolipid-rich invaginations found in some cell types. Numerous functions have been ascribed to caveolins and they likely play important roles in maintaining cholesterol homeostasis and distribution in cells (for recent reviews see [142, 143]). Smart and co-workers suggest that that caveolins transfer cholesterol from the ER to the PM as part of a soluble complex that also contains cholesterol, heat shock protein 56, cyclophilin A, and cyclophilin 40 [144-146]. Treating cells with cyclosporin A or rapamycin disrupts this complex and dramatically slows the transfer of newly synthesized cholesterol from the ER to caveolae in the PM. However, subsequent work by another group found ER to PM cholesterol transport did not slow in cyclosporin A treated cells [56]. The role of caveolin in non-vesicular cholesterol transport is uncertain. It remains to be determined if ER to PM cholesterol transfer slows in cells lacking caveolin or whether the cavolin complex described by Smart and co-workers transfers cholesterol in vitro.
5. Proteins that mediate sterol efflux and uptake
A number of other integral membrane proteins have been shown to play import roles in facilitating non-vesicular sterol transport, primarily between the plasma membrane and exogenous acceptors. Some also play important roles in intracellular sterol trafficking. The mechanisms of these transporters and how they promote sterol transfer remains poorly understood.
5.1 ABC-transporters and sterol efflux
At least four ABC-transporters mediate sterol efflux to external acceptors in mammalian cells: ABCA1, ABCG1, ABCG4, and ABCG5/ABCG8. ABCA1 facilitates the transfer of cholesterol and phospholipids to lipid-free apolipoproteins including apolipoprotein I (apoI) [147]. Loss of ABCA1 function causes Tangier disease, which is characterized by HDL deficiency, sterol accumulation in some tissues, and atherosclorosis. The mechanism of cholesterol efflux by ABCA1 remains controversial. Several reports indicated that cholesterol and phospholipid dissociation occur sequentially [148-150], leading to the model that ABCA1 stimulates phosopholipid export to apoI which can subsequently bind desorbed cholesterol. Alternatively, ABCA1 could mediate simultaneous export of cholesterol and phospholipids to apoI [151, 152]. In either case, it remains unclear whether ABCA1 directly binds and moves lipids to apoI or indirectly affects the chemical activity of cholesterol and perhaps phospholipids, promoting their export to the apoI. A fraction of ABCAI is located in internal endocytic compartments and, interestingly, there is some evidence that this internal protein may facilitate cholesterol efflux from late endosomes; this process slowed in cells expressing an ACBA1 mutant with impaired internalization from the PM [153]. Indeed, it remains possible that ABCA1 is actually active internally, promoting cholesterol movement to the PM where it can be effluxed from cells.
ABCG1 and ABCG4 mediate cholesterol export to HDL. A number of studies have shown that reducing expression of either transporter decreases cholesterol efflux to HDL, while overexpression stimulates cholesterol export [154-158]. How these transporters mediated cholesterol efflux is not known. Unlike ABCA1, ABCG1 and ABCG4 can only mediate the efflux of cholesterol to phospholipid containing acceptors and not lipid free apoI [155, 157, 158].
ABCG5 and ABCG8 form a heterodimer and are expressed on the apical surface of enterocytes, the absorptive cells of the intestine, and in hepatocytes [159]. Defects in these genes cause sitosterolemia, a rare disorder characterized by the accumulation of sitosterol and other plant sterols and hypercholesterolemia. ABCG5/ABCG8 limits the uptake of non-cholesterol sterols by effluxing them from enterocytes [160, 161]. In hepatocytes, they are required for efficient cholesterol secretion into bile [162, 163]. Though the mechanism of transport is not yet understood, in a remarkable study they have been reconstituted in vitro [164]. This work showed that ABCG5/ABCG8 transports cholesterol but not a cholesterol enantiomer. Thus, the transporter likely makes direct contact with the cholesterol rather than indirectly causing sterol desorption from membranes. This is a significant step toward understanding how ABC transporters mediate sterol efflux.
5.2 Sterol uptake
A number of proteins that promote sterol uptake have been identified. The scavenger receptor class B type I (SR-BI) mediates the bi-directional selective transfer of a variety of lipids between cells and lipoproteins including cholesterol and cholesteryl esters [165]. The mechanism of the lipid transfer by SR-BI has been studied for many years but is still not fully understood. For a thorough review, see [165]. SR-BI binds a wide variety of lipoportein acceptors and small unilamellar vesicles [166]. Unlike ABC-transporters, SR-BI does not bind ATP or use ATP hydrolysis to drive transfer. The protein, which is mostly in the PM, is predicted to have a large, heavily glycosylated extracellular domain [167, 168]. This domain may form a hydrophobic channel that facilitates lipid movement between bound lipoproteins and the PM [169, 170]. SR-BI directly mediates cholesterol transfer directly since the purified protein reconstituted into liposomes is functional and will transfer cholesterol from HDL [171]. It likely accelerates the diffusion of cholesterol and other lipids between lipoproteins and the PM and a number of mechanisms for how it does this have been proposed [172]. There is some indication that it increases the chemical activity of cholesterol in the PM [173, 174].
NPC1-like 1 proteins (NPC1L1) mediates the uptake of dietary sterols in the intestine (for a review, see [175]). This protein, like ABCG5/ABCG8, is expressed on the apical surface of enterocytes and was found to be the target of ezetimibe, a drug that inhibits cholesterol absorption [176, 177]. NPC1L1 is an integral membrane protein that contains a sterol-sensing domain (SSD) in the membrane. This domain is also found in NPC1 [178] and some other membrane proteins implicated in cellular cholesterol homeostasis and trafficking [179]. Though the SSD domain has been shown to be required for cholesterol-binding by some proteins, including NPC1 [180, 181], it probably does not play a role in moving cholesterol into or out of membranes. Rather, it may be important for regulating the trafficking of SSD-containing proteins in response to cellular cholesterol levels [179]. Indeed, there is some evidence that some NPC1L1 traffics from the ERC to the cell surface when cellular cholesterol is depleted [182]. It is not known if this requires a functional SSD domain.
The mechanism of NPC1L1-mediated cholesterol uptake is not understood. There is some evidence that NPC1 facilitates the uptake of small apolar molecules including acriflavine and oleic acid but not cholesterol when it is expressed in E. coli [183]. How this relates to cholesterol uptake by NPC1L1 (or cholesterol exit from lysosomes by NPC1) is not clear. Alternatively, NPC1L1 may regulate proteins that take up cholesterol directly.
In the yeast S. cerevisiae, either of two ABC-transporters, Aus1p and Pdr11p, are required for the efficient uptake of exogenous sterols [184]. Surprisingly, they are also needed for efficient PM to ER sterol transfer; after exogenous sterols enter the PM, they are more rapidly transferred to the ER in cells containing Aus1p and Pdr11p than those lacking them [61]. Thus, Aus1p and Pdr11p may facilitate sterol movement out of the PM to internal compartments and only indirectly facilitate the uptake of exogenous sterol by mediating sterol export from the PM. Whether they directly move sterols or indirectly facilitate sterol movement by altering the chemical activity of PM sterols is not known. Alternatively, they could recruit sterol transfer proteins to the PM and thus promote sterol removal from the PM.
6. Non-vesicular sterol transport and intracellular distribution
The concentration of sterols in cellular compartments varies widely. What role might non-vesicular sterol transfer play in establishing and maintaining these differences? One possibility is that it does not significantly contribute [11, 57]. In this model, differences in the sterol concentration of various organelles are driven primarily by their ability to form “condensed complexes.” Sterols in these complexes have low chemical activity and would not be readily extracted by proteins that facilitate non-vesicular sterol transfer. In contrast, sterols not in such complexes would have a high chemical activity and would be rapidly equilibrated between membranes by non-vesicular transfer pathways. Membranes with a high capacity to form condensed complexes, like the PM, would therefore have a higher sterol concentration than organelles with a low capacity, like the ER or mitochondria. In this model, non-vesicular sterol transport pathways would not contribute to maintaining differences in the sterol concentration of various organelles, but rather ensure that cellular membranes have similar amounts of cholesterol with high chemical activity. Consistent with this view, it has long been known that depleting the PM of sphingomyelin (an abundant cholesterol binding partner) by treating cells with sphingomyelinase causes a rapid transfer of cholesterol to the internal compartments [62, 63].
Alternatively, it remains possible that proteins that facilitate non-vesicular sterol transfer between membranes are regulated so that they affect intracellular sterol distribution. One way they are likely regulated is by being localized to particular membranes or MCSs in the cell. Such targeting could promoter sterol transfer to or extraction from specific membranes. For example, CERT, which moves ceramide from the ER to the Golgi, has domains that target it to both membranes. Some sterol transfer proteins, such as ORPs, might be similarly regulated. It is also possible that some sterol transfer proteins might be simulated to extract or deliver sterol to a particular membrane by a partner protein or lipid localized there. For sample, Osh4p stimulation by PI(4,5)P2 probably serves to regulate sterol transfer to or from membranes containing this lipid. This stimulation may affect intracellular sterol distribution. As more is learned about the mechanisms of sterol transfer proteins, we will begin to understand how they are regulated and affect sterol distribution.
7. Conclusions
There is growing evidence that sterols and other lipids move between cellular compartments by non-vesicular mechanisms. How these transport pathways contribute to the maintenance of intracellular sterol distribution and homeostasis remains poorly understood. In the past few years, a number of proteins that facilitate this movement have been identified. It seems likely that many more remain to be discovered and new techniques for identifying these proteins are needed. We are also just beginning to understand the mechanisms of lipid transfer proteins. Though some likely move sterol directly, others may affect rates of sterol transfer by altering the physical properties of membranes by, for example, altering the distribution and organization of lipids in a bilayer or bending membranes. Understanding these mechanisms and how sterol transfer proteins are regulated remains a major challenge for the future.
Acknowledgments
I thank J. Hanover and members of my lab for reading this manuscript. This work was supported by the intramural research program of the National Institute of Diabetes and Digestive and Kidney Diseases.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- 1.Yeagle PL. Cholesterol and the cell membrane. Biochim Biophys Acta. 1985;822:267–287. doi: 10.1016/0304-4157(85)90011-5. [DOI] [PubMed] [Google Scholar]
- 2.Simons K, Ikonen E. How cells handle cholesterol. Science. 2000;290:1721–1726. doi: 10.1126/science.290.5497.1721. [DOI] [PubMed] [Google Scholar]
- 3.Ohvo-Rekila H, Ramstedt B, Leppimaki P, Slotte JP. Cholesterol interactions with phospholipids in membranes. Prog Lipid Res. 2002;41:66–97. doi: 10.1016/s0163-7827(01)00020-0. [DOI] [PubMed] [Google Scholar]
- 4.Sprong H, van der Sluijs P, van Meer G. How proteins move lipids and lipids move proteins. Nat Rev Mol Cell Biol. 2001;2:504–513. doi: 10.1038/35080071. [DOI] [PubMed] [Google Scholar]
- 5.Brown DA, London E. Structure and function of sphingolipid- and cholesterol-rich membrane rafts. J Biol Chem. 2000;275:17221–17224. doi: 10.1074/jbc.R000005200. [DOI] [PubMed] [Google Scholar]
- 6.Mukherjee S, Maxfield FR. Role of membrane organization and membrane domains in endocytic lipid trafficking. Traffic. 2000;1:203–211. doi: 10.1034/j.1600-0854.2000.010302.x. [DOI] [PubMed] [Google Scholar]
- 7.Goldstein JL, DeBose-Boyd RA, Brown MS. Protein sensors for membrane sterols. Cell. 2006;124:35–46. doi: 10.1016/j.cell.2005.12.022. [DOI] [PubMed] [Google Scholar]
- 8.Reinhart MP, Billheimer JT, Faust JR, Gaylor JL. Subcellular localization of the enzymes of cholesterol biosynthesis and metabolism in rat liver. J Biol Chem. 1987;262:9649–9655. [PubMed] [Google Scholar]
- 9.Liscum L, Munn NJ. Intracellular cholesterol transport. Biochim Biophys Acta. 1999;1438:19–37. doi: 10.1016/s1388-1981(99)00043-8. [DOI] [PubMed] [Google Scholar]
- 10.Maxfield FR, Wustner D. Intracellular cholesterol transport. J Clin Invest. 2002;110:891–898. doi: 10.1172/JCI16500. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Maxfield FR, Menon AK. Intracellular sterol transport and distribution. Curr Opin Cell Biol. 2006;18:379–385. doi: 10.1016/j.ceb.2006.06.012. [DOI] [PubMed] [Google Scholar]
- 12.Zambrano F, Fleischer S, Fleischer B. Lipid composition of the Golgi apparatus of rat kidney and liver in comparison with other subcellular organelles. Biochim Biophys Acta. 1975;380:357–369. doi: 10.1016/0005-2760(75)90104-6. [DOI] [PubMed] [Google Scholar]
- 13.Yeagle PL. The Membrane of Cells. Academic Press, Inc.; New York: 1993. [Google Scholar]
- 14.Lange Y, Steck TL. Quantitation of the pool of cholesterol associated with acyl-CoA:cholesterol acyltransferase in human fibroblasts. J Biol Chem. 1997;272:13103–13108. doi: 10.1074/jbc.272.20.13103. [DOI] [PubMed] [Google Scholar]
- 15.Li Y, Ge M, Ciani L, Kuriakose G, Westover EJ, Dura M, Covey DF, Freed JH, Maxfield FR, Lytton J, Tabas I. Enrichment of endoplasmic reticulum with cholesterol inhibits sarcoplasmic-endoplasmic reticulum calcium ATPase-2b activity in parallel with increased order of membrane lipids: implications for depletion of endoplasmic reticulum calcium stores and apoptosis in cholesterol-loaded macrophages. J Biol Chem. 2004;279:37030–37039. doi: 10.1074/jbc.M405195200. [DOI] [PubMed] [Google Scholar]
- 16.Lange Y. Disposition of intracellular cholesterol in human fibroblasts. J Lipid Res. 1991;32:329–339. [PubMed] [Google Scholar]
- 17.Orci L, Montesano R, Meda P, Malaisse-Lagae F, Brown D, Perrelet A, Vassalli P. Heterogeneous distribution of filipin--cholesterol complexes across the cisternae of the Golgi apparatus. Proc Natl Acad Sci U S A. 1981;78:293–297. doi: 10.1073/pnas.78.1.293. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Coxey RA, Pentchev PG, Campbell G, Blanchette-Mackie EJ. Differential accumulation of cholesterol in Golgi compartments of normal and Niemann-Pick type C fibroblasts incubated with LDL: a cytochemical freeze-fracture study. J Lipid Res. 1993;34:1165–1176. [PubMed] [Google Scholar]
- 19.Bretscher MS, Munro S. Cholesterol and the Golgi apparatus. Science. 1993;261:1280–1281. doi: 10.1126/science.8362242. [DOI] [PubMed] [Google Scholar]
- 20.Evans WH, Hardison WG. Phospholipid, cholesterol, polypeptide and glycoprotein composition of hepatic endosome subfractions. Biochem J. 1985;232:33–36. doi: 10.1042/bj2320033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Hornick CA, Hui DY, DeLamatre JG. A role for retrosomes in intracellular cholesterol transport from endosomes to the plasma membrane. Am J Physiol. 1997;273:C1075–1081. doi: 10.1152/ajpcell.1997.273.3.C1075. [DOI] [PubMed] [Google Scholar]
- 22.Hao M, Lin SX, Karylowski OJ, Wustner D, McGraw TE, Maxfield FR. Vesicular and non-vesicular sterol transport in living cells. The endocytic recycling compartment is a major sterol storage organelle. J Biol Chem. 2002;277:609–617. doi: 10.1074/jbc.M108861200. [DOI] [PubMed] [Google Scholar]
- 23.Nes WR. Role of sterols in membranes. Lipids. 1974;9:596–612. doi: 10.1007/BF02532509. [DOI] [PubMed] [Google Scholar]
- 24.Zinser E, Paltauf F, Daum G. Sterol composition of yeast organelle membranes and subcellular distribution of enzymes involved in sterol metabolism. J Bacteriol. 1993;175:2853–2858. doi: 10.1128/jb.175.10.2853-2858.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Zinser E, Daum G. Isolation and biochemical characterization of organelles from the yeast, Saccharomyces cerevisiae. Yeast. 1995;11:493–536. doi: 10.1002/yea.320110602. [DOI] [PubMed] [Google Scholar]
- 26.Bhat RA, Panstruga R. Lipid rafts in plants. Planta. 2005;223:5–19. doi: 10.1007/s00425-005-0096-9. [DOI] [PubMed] [Google Scholar]
- 27.Bagnat M, Keranen S, Shevchenko A, Shevchenko A, Simons K. Lipid rafts function in biosynthetic delivery of proteins to the cell surface in yeast. Proc Natl Acad Sci U S A. 2000;97:3254–3259. doi: 10.1073/pnas.060034697. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Prinz W. Cholesterol trafficking in the secretory and endocytic systems. Semin Cell Dev Biol. 2002;13:197–203. doi: 10.1016/s1084-9521(02)00048-4. [DOI] [PubMed] [Google Scholar]
- 29.Soccio RE, Breslow JL. Intracellular Cholesterol Transport. Arterioscler Thromb Vasc Biol. 2004;4:1150–1160. doi: 10.1161/01.ATV.0000131264.66417.d5. [DOI] [PubMed] [Google Scholar]
- 30.Brugger B, Sandhoff R, Wegehingel S, Gorgas K, Malsam J, Helms JB, Lehmann WD, Nickel W, Wieland FT. Evidence for segregation of sphingomyelin and cholesterol during formation of COPI-coated vesicles. J Cell Biol. 2000;151:507–518. doi: 10.1083/jcb.151.3.507. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Roux A, Cuvelier D, Nassoy P, Prost J, Bassereau P, Goud B. Role of curvature and phase transition in lipid sorting and fission of membrane tubules. Embo J. 2005;24:1537–1545. doi: 10.1038/sj.emboj.7600631. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Baumgart T, Hess ST, Webb WW. Imaging coexisting fluid domains in biomembrane models coupling curvature and line tension. Nature. 2003;425:821–824. doi: 10.1038/nature02013. [DOI] [PubMed] [Google Scholar]
- 33.Wustner D, Herrmann A, Hao M, Maxfield FR. Rapid nonvesicular transport of sterol between the plasma membrane domains of polarized hepatic cells. J Biol Chem. 2002;277:30325–30336. doi: 10.1074/jbc.M202626200. [DOI] [PubMed] [Google Scholar]
- 34.McLean LR, Phillips MC. Mechanism of cholesterol and phosphatidylcholine exchange or transfer between unilamellar vesicles. Biochemistry. 1981;20:2893–2900. doi: 10.1021/bi00513a028. [DOI] [PubMed] [Google Scholar]
- 35.Phillips MC, Johnson WJ, Rothblat GH. Mechanisms and consequences of cellular cholesterol exchange and transfer. Biochim Biophys Acta. 1987;906:223–276. doi: 10.1016/0304-4157(87)90013-x. [DOI] [PubMed] [Google Scholar]
- 36.Frolov A, Woodford JK, Murphy EJ, Billheimer JT, Schroeder F. Spontaneous and protein-mediated sterol transfer between intracellular membranes. J Biol Chem. 1996;271:16075–16083. doi: 10.1074/jbc.271.27.16075. [DOI] [PubMed] [Google Scholar]
- 37.Voelker DR. Bridging gaps in phospholipid transport. Trends Biochem Sci. 2005;30:396–404. doi: 10.1016/j.tibs.2005.05.008. [DOI] [PubMed] [Google Scholar]
- 38.Simpson ER, Boyd GS. The cholesterol side-chain cleavage system of bovine adrenal cortex. Eur J Biochem. 1967;2:275–285. doi: 10.1111/j.1432-1033.1967.tb00136.x. [DOI] [PubMed] [Google Scholar]
- 39.Stocco DM. Clinical disorders associated with abnormal cholesterol transport: mutations in the steroidogenic acute regulatory protein. Mol Cell Endocrinol. 2002;191:19–25. doi: 10.1016/s0303-7207(02)00048-5. [DOI] [PubMed] [Google Scholar]
- 40.Schroeder F. Fluorescent sterols: probe molecules of membrane structure and function. Prog Lipid Res. 1984;23:97–113. doi: 10.1016/0163-7827(84)90009-2. [DOI] [PubMed] [Google Scholar]
- 41.Mukherjee S, Zha X, Tabas I, Maxfield FR. Cholesterol distribution in living cells: fluorescence imaging using dehydroergosterol as a fluorescent cholesterol analog. Biophys J. 1998;75:1915–1925. doi: 10.1016/S0006-3495(98)77632-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Schroeder F, Frolov AA, Murphy EJ, Atshaves BP, Jefferson JR, Pu L, Wood WG, Foxworth WB, Kier AB. Recent advances in membrane cholesterol domain dynamics and intracellular cholesterol trafficking. Proc Soc Exp Biol Med. 1996;213:150–177. doi: 10.3181/00379727-213-44047. [DOI] [PubMed] [Google Scholar]
- 43.Smith RJ, Green C. Fluorescence studies of protein-sterol relationships in human plasma lipoproteins. Biochem J. 1974;137:413–415. doi: 10.1042/bj1370413. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Wood WG, Schroeder F, Avdulov NA, Chochina SV, Igbavboa U. Recent advances in brain cholesterol dynamics: transport, domains, and Alzheimer's disease. Lipids. 1999;34:225–234. doi: 10.1007/s11745-999-0357-9. [DOI] [PubMed] [Google Scholar]
- 45.Schoer JK, Gallegos AM, McIntosh AL, Starodub O, Kier AB, Billheimer JT, Schroeder F. Lysosomal membrane cholesterol dynamics. Biochemistry. 2000;39:7662–7677. doi: 10.1021/bi992686h. [DOI] [PubMed] [Google Scholar]
- 46.Wustner D, Mondal M, Tabas I, Maxfield FR. Direct observation of rapid internalization and intracellular transport of sterol by macrophage foam cells. Traffic. 2005;6:396–412. doi: 10.1111/j.1600-0854.2005.00285.x. [DOI] [PubMed] [Google Scholar]
- 47.Murphy DJ, Vance J. Mechanisms of lipid-body formation. Trends Biochem Sci. 1999;24:109–115. doi: 10.1016/s0968-0004(98)01349-8. [DOI] [PubMed] [Google Scholar]
- 48.Pol A, Luetterforst R, Lindsay M, Heino S, Ikonen E, Parton RG. A caveolin dominant negative mutant associates with lipid bodies and induces intracellular cholesterol imbalance. J Cell Biol. 2001;152:1057–1070. doi: 10.1083/jcb.152.5.1057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Mullner H, Zweytick D, Leber R, Turnowsky F, Daum G. Targeting of proteins involved in sterol biosynthesis to lipid particles of the yeast Saccharomyces cerevisiae. Biochim Biophys Acta. 2004;1663:9–13. doi: 10.1016/j.bbamem.2004.03.001. [DOI] [PubMed] [Google Scholar]
- 50.Milla P, Athenstaedt K, Viola F, Oliaro-Bosso S, Kohlwein SD, Daum G, Balliano G. Yeast oxidosqualene cyclase (Erg7p) is a major component of lipid particles. J Biol Chem. 2002;277:2406–2412. doi: 10.1074/jbc.M104195200. [DOI] [PubMed] [Google Scholar]
- 51.Mo C, Milla P, Athenstaedt K, Ott R, Balliano G, Daum G, Bard M. In yeast sterol biosynthesis the 3-keto reductase protein (Erg27p) is required for oxidosqualene cyclase (Erg7p) activity. Biochim Biophys Acta. 2003;1633:68–74. doi: 10.1016/s1388-1981(03)00088-x. [DOI] [PubMed] [Google Scholar]
- 52.Leber R, Landl K, Zinser E, Ahorn H, Spok A, Kohlwein SD, Turnowsky F, Daum G. Dual localization of squalene epoxidase, Erg1p, in yeast reflects a relationship between the endoplasmic reticulum and lipid particles. Mol Biol Cell. 1998;9:375–386. doi: 10.1091/mbc.9.2.375. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.DeGrella RF, Simoni RD. Intracellular transport of cholesterol to the plasma membrane. J Biol Chem. 1982;257:14256–14262. [PubMed] [Google Scholar]
- 54.Kaplan MR, Simoni RD. Transport of cholesterol from the endoplasmic reticulum to the plasma membrane. J Cell Biol. 1985;101:446–453. doi: 10.1083/jcb.101.2.446. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Urbani L, Simoni RD. Cholesterol and vesicular stomatitis virus G protein take separate routes from the endoplasmic reticulum to the plasma membrane. J Biol Chem. 1990;265:1919–1923. [PubMed] [Google Scholar]
- 56.Heino S, Lusa S, Somerharju P, Ehnholm C, Olkkonen VM, Ikonen E. Dissecting the role of the Golgi complex and lipid rafts in biosynthetic transport of cholesterol to the cell surface. Proc Natl Acad Sci U S A. 2000;97:8375–8380. doi: 10.1073/pnas.140218797. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Baumann NA, Sullivan DP, Ohvo-Rekila H, Simonot C, Pottekat A, Klaassen Z, Beh CT, Menon AK. Transport of newly synthesized sterol to the sterol-enriched plasma membrane occurs via nonvesicular equilibration. Biochemistry. 2005;44:5816–5826. doi: 10.1021/bi048296z. [DOI] [PubMed] [Google Scholar]
- 58.Wilson DW, Wilcox CA, Flynn GC, Chen E, Kuang WJ, Henzel WJ, Block MR, Ullrich A, Rothman JE. A fusion protein required for vesicle-mediated transport in both mammalian cells and yeast. Nature. 1989;339:355–359. doi: 10.1038/339355a0. [DOI] [PubMed] [Google Scholar]
- 59.Graham TR, Emr SD. Compartmental organization of Golgi-specific protein modification and vacuolar protein sorting events defined in a yeast sec18 (NSF) mutant. J Cell Biol. 1991;114:207–218. doi: 10.1083/jcb.114.2.207. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Schnabl M, Daum G, Pichler H. Multiple lipid transport pathways to the plasma membrane in yeast. Biochim Biophys Acta. 2005;1687:130–140. doi: 10.1016/j.bbalip.2004.11.016. [DOI] [PubMed] [Google Scholar]
- 61.Li Y, Prinz WA. ABC-transporters mediate nonvesicular, raft-modulated sterol movement from the plasma membrane to the ER. J Biol Chem. 2004;279:45226–45234. doi: 10.1074/jbc.M407600200. [DOI] [PubMed] [Google Scholar]
- 62.Slotte JP, Bierman EL. Depletion of plasma-membrane sphingomyelin rapidly alters the distribution of cholesterol between plasma membranes and intracellular cholesterol pools in cultured fibroblasts. Biochem J. 1988;250:653–658. doi: 10.1042/bj2500653. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Slotte JP, Hedstrom G, Rannstrom S, Ekman S. Effects of sphingomyelin degradation on cell cholesterol oxidizability and steady-state distribution between the cell surface and the cell interior. Biochim Biophys Acta. 1989;985:90–96. doi: 10.1016/0005-2736(89)90108-9. [DOI] [PubMed] [Google Scholar]
- 64.Skiba PJ, Zha X, Maxfield FR, Schissel SL, Tabas I. The distal pathway of lipoprotein-induced cholesterol esterification, but not sphingomyelinase-induced cholesterol esterification, is energy- dependent. J Biol Chem. 1996;271:13392–13400. doi: 10.1074/jbc.271.23.13392. [DOI] [PubMed] [Google Scholar]
- 65.Juschke C, Wachter A, Schwappach B, Seedorf M. SEC18/NSF-independent, protein-sorting pathway from the yeast cortical ER to the plasma membrane. J Cell Biol. 2005;169:613–622. doi: 10.1083/jcb.200503033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Huang J, Feigenson GW. A microscopic interaction model of maximum solubility of cholesterol in lipid bilayers. Biophys J. 1999;76:2142–2157. doi: 10.1016/S0006-3495(99)77369-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Lange Y, Ye J, Steck TL. How cholesterol homeostasis is regulated by plasma membrane cholesterol in excess of phospholipids. Proc Natl Acad Sci U S A. 2004;101:11664–11667. doi: 10.1073/pnas.0404766101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Lange Y, Ye J, Steck TL. Activation of membrane cholesterol by displacement from phospholipids. J Biol Chem. 2005;280:36126–36131. doi: 10.1074/jbc.M507149200. [DOI] [PubMed] [Google Scholar]
- 69.Lange Y, Ye J, Steck TL. Scrambling of phospholipids activates red cell membrane cholesterol. Biochemistry. 2007;46:2233–2238. doi: 10.1021/bi6023397. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Wattenberg BW, Silbert DF. Sterol partitioning among intracellular membranes. Testing a model for cellular sterol distribution. J Biol Chem. 1983;258:2284–2289. [PubMed] [Google Scholar]
- 71.Chernomordik LV, Kozlov MM. Membrane hemifusion: crossing a chasm in two leaps. Cell. 2005;123:375–382. doi: 10.1016/j.cell.2005.10.015. [DOI] [PubMed] [Google Scholar]
- 72.Holthuis JC, Levine TP. Lipid traffic: floppy drives and a superhighway. Nat Rev Mol Cell Biol. 2005;6:209–220. doi: 10.1038/nrm1591. [DOI] [PubMed] [Google Scholar]
- 73.Levine T, Loewen C. Inter-organelle membrane contact sites: through a glass, darkly. Curr Opin Cell Biol. 2006;18:371–378. doi: 10.1016/j.ceb.2006.06.011. [DOI] [PubMed] [Google Scholar]
- 74.Vance JE. Phospholipid synthesis in a membrane fraction associated with mitochondria. J Biol Chem. 1989;265:7248–7256. [PubMed] [Google Scholar]
- 75.Funato K, Riezman H. Vesicular and nonvesicular transport of ceramide from ER to the Golgi apparatus in yeast. J Cell Biol. 2001;155:949–959. doi: 10.1083/jcb.200105033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Hanada K, Kumagai K, Yasuda S, Miura Y, Kawano M, Fukasawa M, Nishijima M. Molecular machinery for non-vesicular trafficking of ceramide. Nature. 2003;426:803–809. doi: 10.1038/nature02188. [DOI] [PubMed] [Google Scholar]
- 77.Achleitner G, Gaigg B, Krasser A, Kainersdorfer E, Kohlwein SD, Perktold A, Zellnig G, Daum G. Association between the endoplasmic reticulum and mitochondria of yeast facilitates interorganelle transport of phospholipids through membrane contact. Eur J Biochem. 1999;264:545–553. doi: 10.1046/j.1432-1327.1999.00658.x. [DOI] [PubMed] [Google Scholar]
- 78.Xu C, Fan J, Riekhof W, Froehlich JE, Benning C. A permease-like protein involved in ER to thylakoid lipid transfer in Arabidopsis. Embo J. 2003;22:2370–2379. doi: 10.1093/emboj/cdg234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Underwood KW, Jacobs NL, Howley A, Liscum L. Evidence for a cholesterol transport pathway from lysosomes to endoplasmic reticulum that is independent of the plasma membrane. J Biol Chem. 1998;273:4266–4274. doi: 10.1074/jbc.273.7.4266. [DOI] [PubMed] [Google Scholar]
- 80.Loewen CJ, Roy A, Levine TP. A conserved ER targeting motif in three families of lipid binding proteins and in Opi1p binds VAP. EMBO. 2003;22:2025–2035. doi: 10.1093/emboj/cdg201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Olkkonen VM, Levine TP. Oxysterol binding proteins: in more than one place at one time? Biochem Cell Biol. 2004;82:87–98. doi: 10.1139/o03-088. [DOI] [PubMed] [Google Scholar]
- 82.Roberts P, Moshitch-Moshkovitz S, Kvam E, O'Toole E, Winey M, Goldfarb DS. Piecemeal microautophagy of nucleus in Saccharomyces cerevisiae. Mol Biol Cell. 2003;14:129–141. doi: 10.1091/mbc.E02-08-0483. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Pan X, Roberts P, Chen Y, Kvam E, Shulga N, Huang K, Lemmon S, Goldfarb DS. Nucleus-vacuole junctions in Saccharomyces cerevisiae are formed through the direct interaction of Vac8p with Nvj1p. Mol Biol Cell. 2000;11:2445–2457. doi: 10.1091/mbc.11.7.2445. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Phillips SE, Vincent P, Rizzieri KE, Schaaf G, Bankaitis VA, Gaucher EA. The diverse biological functions of phosphatidylinositol transfer proteins in eukaryotes. Crit Rev Biochem Mol Biol. 2006;41:21–49. doi: 10.1080/10409230500519573. [DOI] [PubMed] [Google Scholar]
- 85.Ile KE, Schaaf G, Bankaitis VA. Phosphatidylinositol transfer proteins and cellular nanoreactors for lipid signaling. Nat Chem Biol. 2006;2:576–583. doi: 10.1038/nchembio835. [DOI] [PubMed] [Google Scholar]
- 86.Wirtz KW. Phospholipid transfer proteins in perspective. FEBS Lett. 2006;580:5436–5441. doi: 10.1016/j.febslet.2006.06.065. [DOI] [PubMed] [Google Scholar]
- 87.Stocco DM, Clark BJ. Regulation of the acute production of steroids in steroidogenic cells. Endocr Rev. 1996;17:221–244. doi: 10.1210/edrv-17-3-221. [DOI] [PubMed] [Google Scholar]
- 88.Tsujishita Y, Hurley JH. Structure and lipid transport mechanism of a StAR-related domain. Nat Struct Biol. 2000;7:408–414. doi: 10.1038/75192. [DOI] [PubMed] [Google Scholar]
- 89.Kallen CB, Billheimer JT, Summers SA, Stayrook SE, Lewis M, Strauss JF., 3rd Steroidogenic acute regulatory protein (StAR) is a sterol transfer protein. J Biol Chem. 1998;273:26285–26288. doi: 10.1074/jbc.273.41.26285. [DOI] [PubMed] [Google Scholar]
- 90.Arakane F, Sugawara T, Nishino H, Liu Z, Holt JA, Pain D, Stocco DM, Miller WL, Strauss JF., 3rd Steroidogenic acute regulatory protein (StAR) retains activity in the absence of its mitochondrial import sequence: implications for the mechanism of StAR action. Proc Natl Acad Sci U S A. 1996;93:13731–13736. doi: 10.1073/pnas.93.24.13731. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Arakane F, Kallen CB, Watari H, Foster JA, Sepuri NB, Pain D, Stayrook SE, Lewis M, Gerton GL, Strauss JF., 3rd The mechanism of action of steroidogenic acute regulatory protein (StAR). StAR acts on the outside of mitochondria to stimulate steroidogenesis. J Biol Chem. 1998;273:16339–16345. doi: 10.1074/jbc.273.26.16339. [DOI] [PubMed] [Google Scholar]
- 92.Bose H, Lingappa VR, Miller WL. Rapid regulation of steroidogenesis by mitochondrial protein import. Nature. 2002;417:87–91. doi: 10.1038/417087a. [DOI] [PubMed] [Google Scholar]
- 93.Strauss JF, 3rd, Kishida T, Christenson LK, Fujimoto T, Hiroi H. START domain proteins and the intracellular trafficking of cholesterol in steroidogenic cells. Mol Cell Endocrinol. 2003;202:59–65. doi: 10.1016/s0303-7207(03)00063-7. [DOI] [PubMed] [Google Scholar]
- 94.Miller WL. Mechanism of StAR's regulation of mitochondrial cholesterol import. Mol Cell Endocrinol. 2007;265-266:46–50. doi: 10.1016/j.mce.2006.12.002. [DOI] [PubMed] [Google Scholar]
- 95.Papadopoulos V. Peripheral-type benzodiazepine/diazepam binding inhibitor receptor: biological role in steroidogenic cell function. Endocr Rev. 1993;14:222–240. doi: 10.1210/edrv-14-2-222. [DOI] [PubMed] [Google Scholar]
- 96.Papadopoulos V, Amri H, Li H, Boujrad N, Vidic B, Garnier M. Targeted disruption of the peripheral-type benzodiazepine receptor gene inhibits steroidogenesis in the R2C Leydig tumor cell line. J Biol Chem. 1997;272:32129–32135. doi: 10.1074/jbc.272.51.32129. [DOI] [PubMed] [Google Scholar]
- 97.Alpy F, Tomasetto C. Give lipids a START: the StAR-related lipid transfer (START) domain in mammals. J Cell Sci. 2005;118:2791–2801. doi: 10.1242/jcs.02485. [DOI] [PubMed] [Google Scholar]
- 98.Soccio RE, Breslow JL. StAR-related lipid transfer (START) proteins: mediators of intracellular lipid metabolism. J Biol Chem. 2003;278:22183–22186. doi: 10.1074/jbc.R300003200. [DOI] [PubMed] [Google Scholar]
- 99.Rodriguez-Agudo D, Ren S, Hylemon PB, Redford K, Natarajan R, Del Castillo A, Gil G, Pandak WM. Human StarD5, a cytosolic StAR-related lipid binding protein. J Lipid Res. 2005;46:1615–1623. doi: 10.1194/jlr.M400501-JLR200. [DOI] [PubMed] [Google Scholar]
- 100.Rodriguez-Agudo D, Ren S, Hylemon PB, Montanez R, Redford K, Natarajan R, Medina MA, Gil G, Pandak WM. Localization of StarD5 cholesterol binding protein. J Lipid Res. 2006;47:1168–1175. doi: 10.1194/jlr.M500447-JLR200. [DOI] [PubMed] [Google Scholar]
- 101.Alpy F, Stoeckel ME, Dierich A, Escola JM, Wendling C, Chenard MP, Vanier MT, Gruenberg J, Tomasetto C, Rio MC. The steroidogenic acute regulatory protein homolog MLN64, a late endosomal cholesterol-binding protein. J Biol Chem. 2001;276:4261–4269. doi: 10.1074/jbc.M006279200. [DOI] [PubMed] [Google Scholar]
- 102.Zhang M, Liu P, Dwyer NK, Christenson LK, Fujimoto T, Martinez F, Comly M, Hanover JA, Blanchette-Mackie EJ, Strauss JF., 3rd MLN64 mediates mobilization of lysosomal cholesterol to steroidogenic mitochondria. J Biol Chem. 2002;277:33300–33310. doi: 10.1074/jbc.M200003200. [DOI] [PubMed] [Google Scholar]
- 103.Alpy F, Latchumanan VK, Kedinger V, Janoshazi A, Thiele C, Wendling C, Rio MC, Tomasetto C. Functional characterization of the MENTAL domain. J Biol Chem. 2005;280:17945–17952. doi: 10.1074/jbc.M500723200. [DOI] [PubMed] [Google Scholar]
- 104.Liscum L, Ruggiero RM, Faust JR. The intracellular transport of low density lipoprotein-derived cholesterol is defective in Niemann-Pick type C fibroblasts. J Cell Biol. 1989;108:1625–1636. doi: 10.1083/jcb.108.5.1625. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Vanier MT, Rodriguez-Lafrasse C, Rousson R, Gazzah N, Juge MC, Pentchev PG, Revol A, Louisot P. Type C Niemann-Pick disease: spectrum of phenotypic variation in disruption of intracellular LDL-derived cholesterol processing. Biochim Biophys Acta. 1991;1096:328–337. doi: 10.1016/0925-4439(91)90069-l. [DOI] [PubMed] [Google Scholar]
- 106.Pentchev PG, Brady RO, Blanchette-Mackie EJ, Vanier MT, Carstea ED, Parker CC, Goldin E, Roff CF. The Niemann-Pick C lesion and its relationship to the intracellular distribution and utilization of LDL cholesterol. Biochim Biophys Acta. 1994;1225:235–243. doi: 10.1016/0925-4439(94)90001-9. [DOI] [PubMed] [Google Scholar]
- 107.Cruz JC, Sugii S, Yu C, Chang TY. Role of Niemann-Pick type C1 protein in intracellular trafficking of low density lipoprotein-derived cholesterol. J Biol Chem. 2000;275:4013–4021. doi: 10.1074/jbc.275.6.4013. [DOI] [PubMed] [Google Scholar]
- 108.Sturley SL, Patterson MC, Balch W, Liscum L. The pathophysiology and mechanisms of NP-C disease. Biochim Biophys Acta. 2004;1685:83–87. doi: 10.1016/j.bbalip.2004.08.014. [DOI] [PubMed] [Google Scholar]
- 109.Sleat DE, Wiseman JA, El-Banna M, Price SM, Verot L, Shen MM, Tint GS, Vanier MT, Walkley SU, Lobel P. Genetic evidence for nonredundant functional cooperativity between NPC1 and NPC2 in lipid transport. Proc Natl Acad Sci U S A. 2004;101:5886–5891. doi: 10.1073/pnas.0308456101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Watari H, Blanchette-Mackie EJ, Dwyer NK, Glick JM, Patel S, Neufeld EB, Brady RO, Pentchev PG, Strauss JF., 3rd Niemann-Pick C1 protein: obligatory roles for N-terminal domains and lysosomal targeting in cholesterol mobilization. Proc Natl Acad Sci U S A. 1999;96:805–810. doi: 10.1073/pnas.96.3.805. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Patel SC, Suresh S, Kumar U, Hu CY, Cooney A, Blanchette-Mackie EJ, Neufeld EB, Patel RC, Brady RO, Patel YC, Pentchev PG, Ong WY. Localization of Niemann-Pick C1 protein in astrocytes: implications for neuronal degeneration in Niemann- Pick type C disease. Proc Natl Acad Sci U S A. 1999;96:1657–1662. doi: 10.1073/pnas.96.4.1657. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Naureckiene S, Sleat DE, Lackland H, Fensom A, Vanier MT, Wattiaux R, Jadot M, Lobel P. Identification of HE1 as the second gene of Niemann-Pick C disease. Science. 2000;290:2298–2301. doi: 10.1126/science.290.5500.2298. [DOI] [PubMed] [Google Scholar]
- 113.Okamura N, Kiuchi S, Tamba M, Kashima T, Hiramoto S, Baba T, Dacheux F, Dacheux JL, Sugita Y, Jin YZ. A porcine homolog of the major secretory protein of human epididymis, HE1, specifically binds cholesterol. Biochim Biophys Acta. 1999;1438:377–387. doi: 10.1016/s1388-1981(99)00070-0. [DOI] [PubMed] [Google Scholar]
- 114.Ko DC, Binkley J, Sidow A, Scott MP. The integrity of a cholesterol-binding pocket in Niemann-Pick C2 protein is necessary to control lysosome cholesterol levels. Proc Natl Acad Sci U S A. 2003;100:2518–2525. doi: 10.1073/pnas.0530027100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Friedland N, Liou HL, Lobel P, Stock AM. Structure of a cholesterol-binding protein deficient in Niemann-Pick type C2 disease. Proc Natl Acad Sci U S A. 2003;100:2512–2517. doi: 10.1073/pnas.0437840100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Cheruku SR, Xu Z, Dutia R, Lobel P, Storch J. Mechanism of cholesterol transfer from the Niemann-Pick type C2 protein to model membranes supports a role in lysosomal cholesterol transport. J Biol Chem. 2006;281:31594–31604. doi: 10.1074/jbc.M602765200. [DOI] [PubMed] [Google Scholar]
- 117.Kandutsch AA, Shown EP. Assay of oxysterol-binding protein in a mouse fibroblast, cell-free system. Dissociation constant and other properties of the system. J Biol Chem. 1981;256:13068–13073. [PubMed] [Google Scholar]
- 118.Im YJ, Raychaudhuri S, Prinz WA, Hurley JH. Structural mechanism for sterol sensing and transport by OSBP-related proteins. Nature. 2005;437:154–158. doi: 10.1038/nature03923. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Raychaudhuri S, Im YJ, Hurley JH, Prinz WA. Nonvesicular sterol movement from plasma membrane to ER requires oxysterol-binding protein-related proteins and phosphoinositides. J Cell Biol. 2006;173:107–119. doi: 10.1083/jcb.200510084. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Beh CT, Cool L, Phillips J, Rine J. Overlapping functions of the yeast oxysterol-binding protein homologues. Genetics. 2001;157:1117–1140. doi: 10.1093/genetics/157.3.1117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Beh CT, Rine J. A role for yeast oxysterol-binding protein homologs in endocytosis and in the maintenance of intracellular sterol-lipid distribution. J Cell Sci. 2004;117:2983–2996. doi: 10.1242/jcs.01157. [DOI] [PubMed] [Google Scholar]
- 122.Sullivan DP, Ohvo-Rekila H, Baumann NA, Beh CT, Menon AK. Sterol trafficking between the endoplasmic reticulum and plasma membrane in yeast. Biochem Soc Trans. 2006;34:356–358. doi: 10.1042/BST0340356. [DOI] [PubMed] [Google Scholar]
- 123.Roth MG. Phosphoinositides in constitutive membrane traffic. Physiol Rev. 2004;84:699–730. doi: 10.1152/physrev.00033.2003. [DOI] [PubMed] [Google Scholar]
- 124.Li X, Rivas MP, Fang M, Marchena J, Mehrotra B, Chaudhary A, Feng L, Prestwich GD, Bankaitis VA. Analysis of oxysterol binding protein homologue Kes1p function in regulation of Sec14p-dependent protein transport from the yeast Golgi complex. J Cell Biol. 2002;157:63–77. doi: 10.1083/jcb.200201037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Wang P, Duan W, Munn AL, Yang H. Molecular characterization of Osh6p, an oxysterol binding protein homolog in the yeast Saccharomyces cerevisiae. FEBS J. 2005;272:4703–4715. doi: 10.1111/j.1742-4658.2005.04886.x. [DOI] [PubMed] [Google Scholar]
- 126.Wang PY, Weng J, Anderson RGW. OSBP Is a Cholesterol-Regulated Scaffolding Protein in Control of ERK1/2 Activation. Science. 2005;307:1472–1476. doi: 10.1126/science.1107710. [DOI] [PubMed] [Google Scholar]
- 127.Perry RJ, Ridgway ND. Oxysterol-binding protein and vesicle-associated membrane protein-associated protein are required for sterol-dependent activation of the ceramide transport protein. Mol Biol Cell. 2006;17:2604–2616. doi: 10.1091/mbc.E06-01-0060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Johansson M, Rocha N, Zwart W, Jordens I, Janssen L, Kuijl C, Olkkonen VM, Neefjes J. Activation of endosomal dynein motors by stepwise assembly of Rab7-RILP-p150Glued, ORP1L, and the receptor {beta}lll spectrin. J Cell Biol. 2007;176:459–471. doi: 10.1083/jcb.200606077. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Berger AC, Vanderford TH, Gernert KM, Nichols JW, Faundez V, Corbett AH. Saccharomyces cerevisiae Npc2p is a functionally conserved homologue of the human Niemann-Pick disease type C 2 protein, hNPC2. Eukaryot Cell. 2005;4:1851–1862. doi: 10.1128/EC.4.11.1851-1862.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Seedorf U, Ellinghaus P, Roch Nofer J. Sterol carrier protein-2. Biochim Biophys Acta. 2000;1486:45–54. doi: 10.1016/s1388-1981(00)00047-0. [DOI] [PubMed] [Google Scholar]
- 131.Gallegos AM, Atshaves BP, Storey SM, Starodub O, Petrescu AD, Huang H, McIntosh AL, Martin GG, Chao H, Kier AB, Schroeder F. Gene structure, intracellular localization, and functional roles of sterol carrier protein-2. Prog Lipid Res. 2001;40:498–563. doi: 10.1016/s0163-7827(01)00015-7. [DOI] [PubMed] [Google Scholar]
- 132.Seedorf U, Raabe M, Ellinghaus P, Kannenberg F, Fobker M, Engel T, Denis S, Wouters F, Wirtz KW, Wanders RJ, Maeda N, Assmann G. Defective peroxisomal catabolism of branched fatty acyl coenzyme A in mice lacking the sterol carrier protein-2/sterol carrier protein-x gene function. Genes Dev. 1998;12:1189–1201. doi: 10.1101/gad.12.8.1189. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Keller GA, Scallen TJ, Clarke D, Maher PA, Krisans SK, Singer SJ. Subcellular localization of sterol carrier protein-2 in rat hepatocytes: its primary localization to peroxisomes. J Cell Biol. 1989;108:1353–1361. doi: 10.1083/jcb.108.4.1353. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.van Amerongen A, van Noort M, van Beckhoven JR, Rommerts FF, Orly J, Wirtz KW. The subcellular distribution of the nonspecific lipid transfer protein (sterol carrier protein 2) in rat liver and adrenal gland. Biochim Biophys Acta. 1989;1001:243–248. doi: 10.1016/0005-2760(89)90106-9. [DOI] [PubMed] [Google Scholar]
- 135.Murphy EJ, Schroeder F. Sterol carrier protein-2 mediated cholesterol esterification in transfected L-cell fibroblasts. Biochim Biophys Acta. 1997;1345:283–292. doi: 10.1016/s0005-2760(97)00003-9. [DOI] [PubMed] [Google Scholar]
- 136.Baum CL, Reschly EJ, Gayen AK, Groh ME, Schadick K. Sterol carrier protein-2 overexpression enhances sterol cycling and inhibits cholesterol ester synthesis and high density lipoprotein cholesterol secretion. J Biol Chem. 1997;272:6490–6498. doi: 10.1074/jbc.272.10.6490. [DOI] [PubMed] [Google Scholar]
- 137.Moncecchi D, Murphy EJ, Prows DR, Schroeder F. Sterol carrier protein-2 expression in mouse L-cell fibroblasts alters cholesterol uptake. Biochim Biophys Acta. 1996;1302:110–116. doi: 10.1016/0005-2760(96)00044-6. [DOI] [PubMed] [Google Scholar]
- 138.Atshaves BP, Petrescu AD, Starodub O, Roths JB, Kier AB, Schroeder F. Expression and intracellular processing of the 58 kDa sterol carrier protein-2/3-oxoacyl-CoA thiolase in transfected mouse L-cell fibroblasts. J Lipid Res. 1999;40:610–622. [PubMed] [Google Scholar]
- 139.Fuchs M, Hafer A, Munch C, Kannenberg F, Teichmann S, Scheibner J, Stange EF, Seedorf U. Disruption of the sterol carrier protein 2 gene in mice impairs biliary lipid and hepatic cholesterol metabolism. J Biol Chem. 2001;276:48058–48065. doi: 10.1074/jbc.M106732200. [DOI] [PubMed] [Google Scholar]
- 140.Atshaves BP, Starodub O, McIntosh A, Petrescu A, Roths JB, Kier AB, Schroeder F. Sterol carrier protein-2 alters high density lipoprotein-mediated cholesterol efflux. J Biol Chem. 2000;275:36852–36861. doi: 10.1074/jbc.M003434200. [DOI] [PubMed] [Google Scholar]
- 141.Gallegos AM, Atshaves BP, Storey SM, McIntosh AL, Petrescu AD, Schroeder F. Sterol carrier protein-2 expression alters plasma membrane lipid distribution and cholesterol dynamics. Biochemistry. 2001;40:6493–6506. doi: 10.1021/bi010217l. [DOI] [PubMed] [Google Scholar]
- 142.Martin S, Parton RG. Caveolin, cholesterol, and lipid bodies. Semin Cell Dev Biol. 2005;16:163–174. doi: 10.1016/j.semcdb.2005.01.007. [DOI] [PubMed] [Google Scholar]
- 143.Parton RG, Simons K. The multiple faces of caveolae. Nat Rev Mol Cell Biol. 2007;8:185–194. doi: 10.1038/nrm2122. [DOI] [PubMed] [Google Scholar]
- 144.Smart EJ, Ying Y, Donzell WC, Anderson RG. A role for caveolin in transport of cholesterol from endoplasmic reticulum to plasma membrane. J Biol Chem. 1996;271:29427–29435. doi: 10.1074/jbc.271.46.29427. [DOI] [PubMed] [Google Scholar]
- 145.Uittenbogaard A, Smart EJ. Palmitoylation of caveolin-1 is required for cholesterol binding, chaperone complex formation, and rapid transport of cholesterol to caveolae. J Biol Chem. 2000;275:25595–25599. doi: 10.1074/jbc.M003401200. [DOI] [PubMed] [Google Scholar]
- 146.Uittenbogaard A, Ying Y, Smart EJ. Characterization of a cytosolic heat-shock protein-caveolin chaperone complex. Involvement in cholesterol trafficking. J Biol Chem. 1998;273:6525–6532. doi: 10.1074/jbc.273.11.6525. [DOI] [PubMed] [Google Scholar]
- 147.Oram JF, Heinecke JW. ATP-binding cassette transporter A1: a cell cholesterol exporter that protects against cardiovascular disease. Physiol Rev. 2005;85:1343–1372. doi: 10.1152/physrev.00005.2005. [DOI] [PubMed] [Google Scholar]
- 148.Fielding PE, Nagao K, Hakamata H, Chimini G, Fielding CJ. A two-step mechanism for free cholesterol and phospholipid efflux from human vascular cells to apolipoprotein A-1. Biochemistry. 2000;39:14113–14120. doi: 10.1021/bi0004192. [DOI] [PubMed] [Google Scholar]
- 149.Wang N, Silver DL, Thiele C, Tall AR. ATP-binding cassette transporter A1 (ABCA1) functions as a cholesterol efflux regulatory protein. J Biol Chem. 2001;276:23742–23747. doi: 10.1074/jbc.M102348200. [DOI] [PubMed] [Google Scholar]
- 150.Kiss RS, Maric J, Marcel YL. Lipid efflux in human and mouse macrophagic cells: evidence for differential regulation of phospholipid and cholesterol efflux. J Lipid Res. 2005;46:1877–1887. doi: 10.1194/jlr.M400482-JLR200. [DOI] [PubMed] [Google Scholar]
- 151.Smith JD, Le Goff W, Settle M, Brubaker G, Waelde C, Horwitz A, Oda MN. ABCA1 mediates concurrent cholesterol and phospholipid efflux to apolipoprotein A-I. J Lipid Res. 2004;45:635–644. doi: 10.1194/jlr.M300336-JLR200. [DOI] [PubMed] [Google Scholar]
- 152.Gillotte KL, Davidson WS, Lund-Katz S, Rothblat GH, Phillips MC. Removal of cellular cholesterol by pre-beta-HDL involves plasma membrane microsolubilization. J Lipid Res. 1998;39:1918–1928. [PubMed] [Google Scholar]
- 153.Chen W, Wang N, Tall AR. A PEST deletion mutant of ABCA1 shows impaired internalization and defective cholesterol efflux from late endosomes. J Biol Chem. 2005;280:29277–29281. doi: 10.1074/jbc.M505566200. [DOI] [PubMed] [Google Scholar]
- 154.Klucken J, Buchler C, Orso E, Kaminski WE, Porsch-Ozcurumez M, Liebisch G, Kapinsky M, Diederich W, Drobnik W, Dean M, Allikmets R, Schmitz G. ABCG1 (ABC8), the human homolog of the Drosophila white gene, is a regulator of macrophage cholesterol and phospholipid transport. Proc Natl Acad Sci U S A. 2000;97:817–822. doi: 10.1073/pnas.97.2.817. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Wang N, Lan D, Chen W, Matsuura F, Tall AR. ATP-binding cassette transporters G1 and G4 mediate cellular cholesterol efflux to high-density lipoproteins. Proc Natl Acad Sci U S A. 2004;101:9774–9779. doi: 10.1073/pnas.0403506101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Nakamura K, Kennedy MA, Baldan A, Bojanic DD, Lyons K, Edwards PA. Expression and regulation of multiple murine ATP-binding cassette transporter G1 mRNAs/isoforms that stimulate cellular cholesterol efflux to high density lipoprotein. J Biol Chem. 2004;279:45980–45989. doi: 10.1074/jbc.M408652200. [DOI] [PubMed] [Google Scholar]
- 157.Vaughan AM, Oram JF. ABCG1 redistributes cell cholesterol to domains removable by high density lipoprotein but not by lipid-depleted apolipoproteins. J Biol Chem. 2005;280:30150–30157. doi: 10.1074/jbc.M505368200. [DOI] [PubMed] [Google Scholar]
- 158.Gelissen IC, Harris M, Rye KA, Quinn C, Brown AJ, Kockx M, Cartland S, Packianathan M, Kritharides L, Jessup W. ABCA1 and ABCG1 synergize to mediate cholesterol export to apoA-I. Arterioscler Thromb Vasc Biol. 2006;26:534–540. doi: 10.1161/01.ATV.0000200082.58536.e1. [DOI] [PubMed] [Google Scholar]
- 159.Berge KE, Tian H, Graf GA, Yu L, Grishin NV, Schultz J, Kwiterovich P, Shan B, Barnes R, Hobbs HH. Accumulation of dietary cholesterol in sitosterolemia caused by mutations in adjacent ABC transporters. Science. 2000;290:1771–1775. doi: 10.1126/science.290.5497.1771. [DOI] [PubMed] [Google Scholar]
- 160.Graf GA, Li WP, Gerard RD, Gelissen I, White A, Cohen JC, Hobbs HH. Coexpression of ATP-binding cassette proteins ABCG5 and ABCG8 permits their transport to the apical surface. J Clin Invest. 2002;110:659–669. doi: 10.1172/JCI16000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161.Graf GA, Yu L, Li WP, Gerard R, Tuma PL, Cohen JC, Hobbs HH. ABCG5 and ABCG8 are obligate heterodimers for protein trafficking and biliary cholesterol excretion. J Biol Chem. 2003;278:48275–48282. doi: 10.1074/jbc.M310223200. [DOI] [PubMed] [Google Scholar]
- 162.Yu L, Hammer RE, Li-Hawkins J, Von Bergmann K, Lutjohann D, Cohen JC, Hobbs HH. Disruption of Abcg5 and Abcg8 in mice reveals their crucial role in biliary cholesterol secretion. Proc Natl Acad Sci U S A. 2002;99:16237–16242. doi: 10.1073/pnas.252582399. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.Yu L, Li-Hawkins J, Hammer RE, Berge KE, Horton JD, Cohen JC, Hobbs HH. Overexpression of ABCG5 and ABCG8 promotes biliary cholesterol secretion and reduces fractional absorption of dietary cholesterol. J Clin Invest. 2002;110:671–680. doi: 10.1172/JCI16001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164.Wang J, Sun F, Zhang DW, Ma Y, Xu F, Belani JD, Cohen JC, Hobbs HH, Xie XS. Sterol transfer by ABCG5 and ABCG8: in vitro assay and reconstitution. J Biol Chem. 2006;281:27894–27904. doi: 10.1074/jbc.M605603200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 165.Rigotti A, Miettinen HE, Krieger M. The role of the high-density lipoprotein receptor SR-BI in the lipid metabolism of endocrine and other tissues. Endocr Rev. 2003;24:357–387. doi: 10.1210/er.2001-0037. [DOI] [PubMed] [Google Scholar]
- 166.Rigotti A, Acton SL, Krieger M. The class B scavenger receptors SR-BI and CD36 are receptors for anionic phospholipids. J Biol Chem. 1995;270:16221–16224. doi: 10.1074/jbc.270.27.16221. [DOI] [PubMed] [Google Scholar]
- 167.Rigotti A, Trigatti B, Babitt J, Penman M, Xu S, Krieger M. Scavenger receptor BI--a cell surface receptor for high density lipoprotein. Curr Opin Lipidol. 1997;8:181–188. doi: 10.1097/00041433-199706000-00009. [DOI] [PubMed] [Google Scholar]
- 168.Babitt J, Trigatti B, Rigotti A, Smart EJ, Anderson RG, Xu S, Krieger M. Murine SR-BI, a high density lipoprotein receptor that mediates selective lipid uptake, is N-glycosylated and fatty acylated and colocalizes with plasma membrane caveolae. J Biol Chem. 1997;272:13242–13249. doi: 10.1074/jbc.272.20.13242. [DOI] [PubMed] [Google Scholar]
- 169.Rodrigueza WV, Thuahnai ST, Temel RE, Lund-Katz S, Phillips MC, Williams DL. Mechanism of scavenger receptor class B type I-mediated selective uptake of cholesteryl esters from high density lipoprotein to adrenal cells. J Biol Chem. 1999;274:20344–20350. doi: 10.1074/jbc.274.29.20344. [DOI] [PubMed] [Google Scholar]
- 170.Thuahnai ST, Lund-Katz S, Williams DL, Phillips MC. Scavenger receptor class B, type I-mediated uptake of various lipids into cells. Influence of the nature of the donor particle interaction with the receptor. J Biol Chem. 2001;276:43801–43808. doi: 10.1074/jbc.M106695200. [DOI] [PubMed] [Google Scholar]
- 171.Liu B, Krieger M. Highly purified scavenger receptor class B, type I reconstituted into phosphatidylcholine/cholesterol liposomes mediates high affinity high density lipoprotein binding and selective lipid uptake. J Biol Chem. 2002;277:34125–34135. doi: 10.1074/jbc.M204265200. [DOI] [PubMed] [Google Scholar]
- 172.Yancey PG, Bortnick AE, Kellner-Weibel G, de la Llera-Moya M, Phillips MC, Rothblat GH. Importance of different pathways of cellular cholesterol efflux. Arterioscler Thromb Vasc Biol. 2003;23:712–719. doi: 10.1161/01.ATV.0000057572.97137.DD. [DOI] [PubMed] [Google Scholar]
- 173.de la Llera-Moya M, Rothblat GH, Connelly MA, Kellner-Weibel G, Sakr SW, Phillips MC, Williams DL. Scavenger receptor BI (SR-BI) mediates free cholesterol flux independently of HDL tethering to the cell surface. J Lipid Res. 1999;40:575–580. [PubMed] [Google Scholar]
- 174.Kellner-Weibel G, de La Llera-Moya M, Connelly MA, Stoudt G, Christian AE, Haynes MP, Williams DL, Rothblat GH. Expression of scavenger receptor BI in COS-7 cells alters cholesterol content and distribution. Biochemistry. 2000;39:221–229. doi: 10.1021/bi991666c. [DOI] [PubMed] [Google Scholar]
- 175.Huff MW, Pollex RL, Hegele RA. NPC1L1: evolution from pharmacological target to physiological sterol transporter. Arterioscler Thromb Vasc Biol. 2006;26:2433–2438. doi: 10.1161/01.ATV.0000245791.53245.ee. [DOI] [PubMed] [Google Scholar]
- 176.Altmann SW, Davis HR, Jr, Zhu LJ, Yao X, Hoos LM, Tetzloff G, Iyer SP, Maguire M, Golovko A, Zeng M, Wang L, Murgolo N, Graziano MP. Niemann-Pick C1 Like 1 protein is critical for intestinal cholesterol absorption. Science. 2004;303:1201–1204. doi: 10.1126/science.1093131. [DOI] [PubMed] [Google Scholar]
- 177.Garcia-Calvo M, Lisnock J, Bull HG, Hawes BE, Burnett DA, Braun MP, Crona JH, Davis HR, Jr, Dean DC, Detmers PA, Graziano MP, Hughes M, Macintyre DE, Ogawa A, O'Neill K A, Iyer SP, Shevell DE, Smith MM, Tang YS, Makarewicz AM, Ujjainwalla F, Altmann SW, Chapman KT, Thornberry NA. The target of ezetimibe is Niemann-Pick C1-Like 1 (NPC1L1) Proc Natl Acad Sci U S A. 2005;102:8132–8137. doi: 10.1073/pnas.0500269102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 178.Carstea ED, Morris JA, Coleman KG, Loftus SK, Zhang D, Cummings C, Gu J, Rosenfeld MA, Pavan WJ, Krizman DB, Nagle J, Polymeropoulos MH, Sturley SL, Ioannou YA, Higgins ME, Comly M, Cooney A, Brown A, Kaneski CR, Blanchette-Mackie EJ, Dwyer NK, Neufeld EB, Chang TY, Liscum L, Tagle DA, et al. Niemann-Pick C1 disease gene: homology to mediators of cholesterol homeostasis. Science. 1997;277:228–231. doi: 10.1126/science.277.5323.228. [DOI] [PubMed] [Google Scholar]
- 179.Kuwabara PE, Labouesse M. The sterol-sensing domain: multiple families, a unique role? Trends Genet. 2002;18:193–201. doi: 10.1016/s0168-9525(02)02640-9. [DOI] [PubMed] [Google Scholar]
- 180.Ohgami N, Ko DC, Thomas M, Scott MP, Chang CC, Chang TY. Binding between the Niemann-Pick C1 protein and a photoactivatable cholesterol analog requires a functional sterol-sensing domain. Proc Natl Acad Sci U S A. 2004;101:12473–12478. doi: 10.1073/pnas.0405255101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 181.Radhakrishnan A, Sun LP, Kwon HJ, Brown MS, Goldstein JL. Direct binding of cholesterol to the purified membrane region of SCAP: mechanism for a sterol-sensing domain. Mol Cell. 2004;15:259–268. doi: 10.1016/j.molcel.2004.06.019. [DOI] [PubMed] [Google Scholar]
- 182.Yu L, Bharadwaj S, Brown JM, Ma Y, Du W, Davis MA, Michaely P, Liu P, Willingham MC, Rudel LL. Cholesterol-regulated translocation of NPC1L1 to the cell surface facilitates free cholesterol uptake. J Biol Chem. 2006;281:6616–6624. doi: 10.1074/jbc.M511123200. [DOI] [PubMed] [Google Scholar]
- 183.Davies JP, Chen FW, Ioannou YA. Transmembrane molecular pump activity of Niemann-Pick C1 protein. Science. 2000;290:2295–2298. doi: 10.1126/science.290.5500.2295. [DOI] [PubMed] [Google Scholar]
- 184.Wilcox LJ, Balderes DA, Wharton B, Tinkelenberg AH, Rao G, Sturley SL. Transcriptional profiling identifies two members of the ATP-binding cassette transporter superfamily required for sterol uptake in yeast. J Biol Chem. 2002;277:32466–32472. doi: 10.1074/jbc.M204707200. [DOI] [PubMed] [Google Scholar]


