Abstract
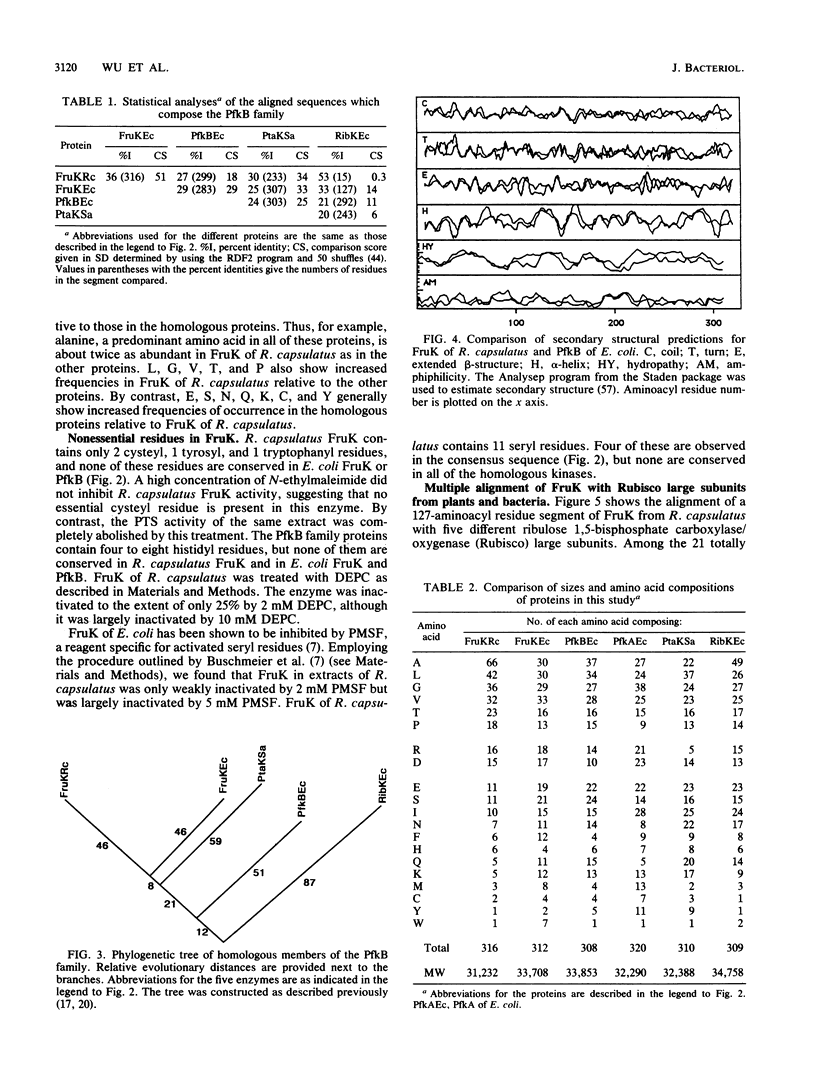
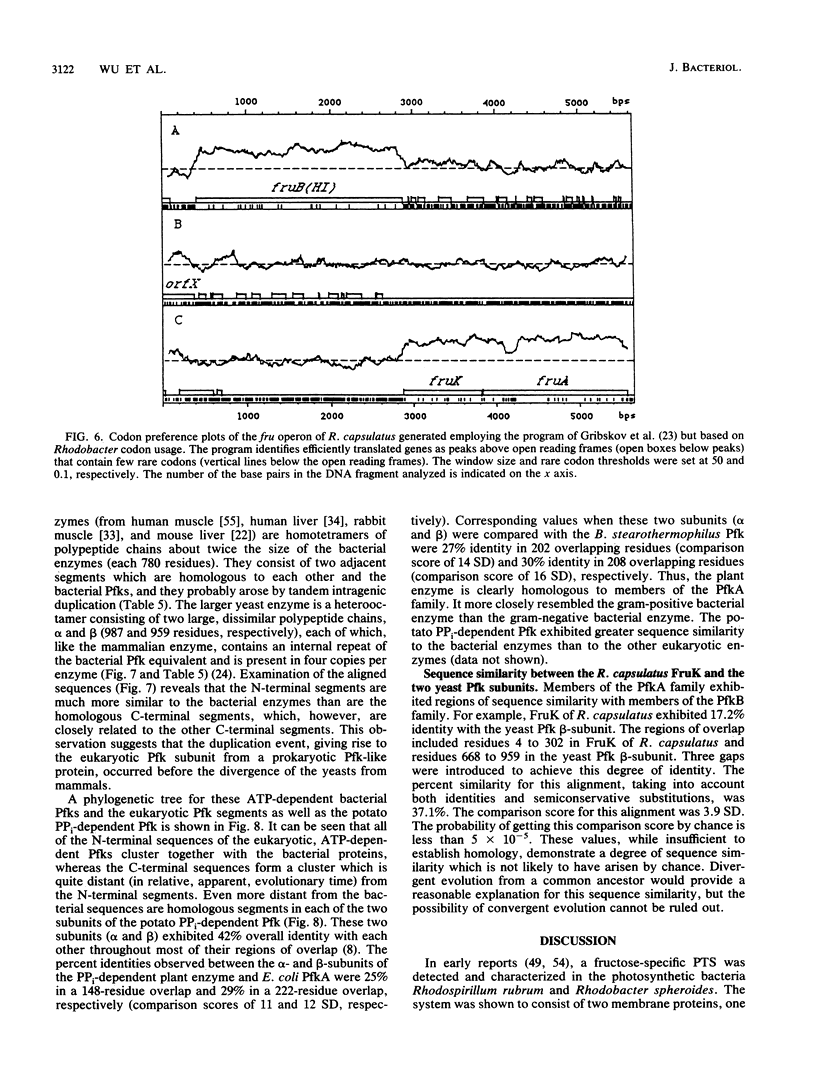
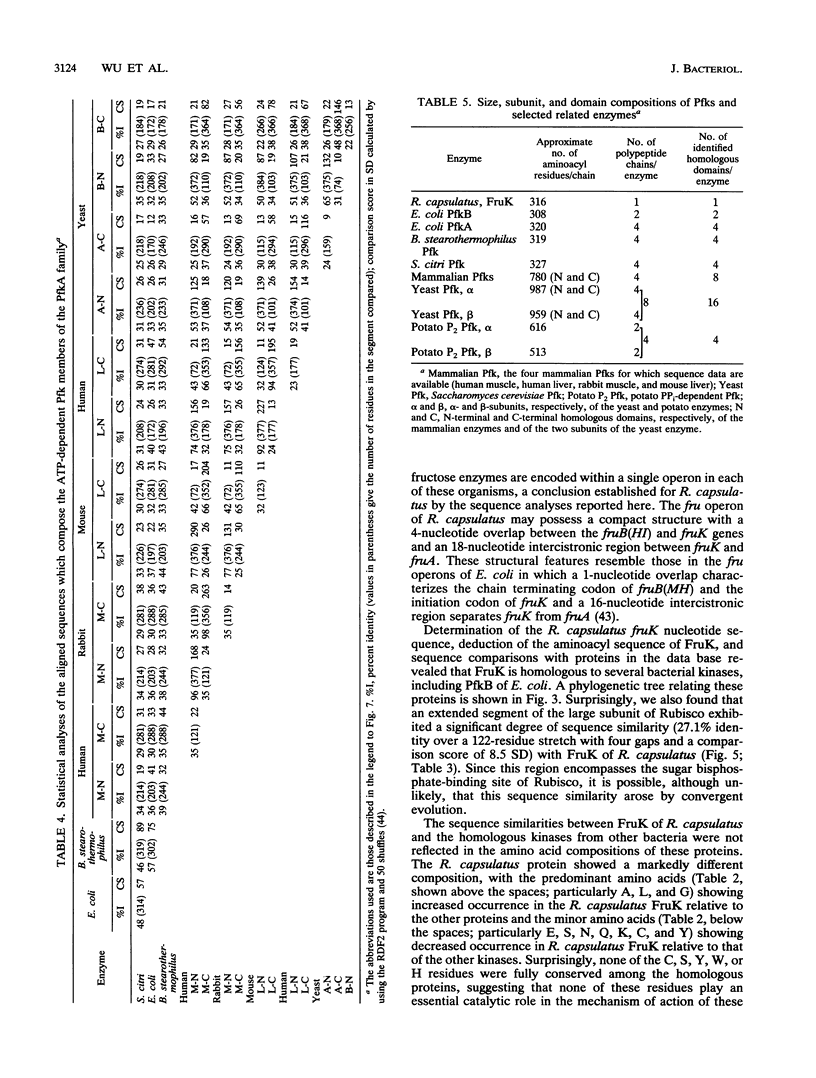
The fruK gene encoding fructose-1-phosphate kinase (FruK), located within the fructose (fru)-catabolic operon of Rhodobacter capsulatus, was sequenced. FruK of R. capsulatus (316 amino acids; molecular weight = 31,232) is the same size as and is homologous to FruK of Escherichia coli, phosphofructokinase B (PfkB) of E. coli, phosphotagatokinase of Staphylococcus aureus, and ribokinase of E. coli. These proteins therefore make up a family of homologous proteins, termed the PfkB family. A phylogenetic tree for this new family was constructed. Sequence comparisons plus chemical inactivation studies suggested the lack of involvement of specific residues in catalysis. Although the Rhodobacter FruK differed markedly from the other enzymes within the PfkB family with respect to amino acid composition, these enzymes exhibited similar predicted secondary structural features. A large internal segment of the Rhodobacter FruK was found to be similar in sequence to the domain bearing the sugar bisphosphate-binding region of the large subunit of ribulose 1,5-bisphosphate carboxylase/oxygenase of plants and bacteria. Proteins of the PfkB family did not exhibit statistically significant sequence identity with PfkA of E. coli. PfkA, however, is homologous to other prokaryotic and eukaryotic ATP- and PPi-dependent Pfks (the PfkA family). These eukaryotic, ATP-dependent enzymes each consist of a homotetramer (mammalian) or a heterooctamer (yeasts), with each subunit containing an internal duplication of the size of the entire PfkA protein of E. coli. In some of these enzymes, additional domains are present. A phylogenetic tree was constructed for the PfkA family and revealed that the bacterial enzymes closely resemble the N-terminal domains of the eukaryotic enzyme subunits whereas the C-terminal domains have diverged more extensively. The PPi-dependent Pfk of potato is only distantly related to the ATP-dependent enzymes. On the basis of their similar functions, sizes, predicted secondary structures, and sequences, we suggest that the PfkA and PfkB families share a common evolutionary origin.
Full text
PDF







Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Aldrich J., Cherney B., Merlin E., Palmer J. Sequence of the rbcL gene for the large subunit of ribulose bisphosphate carboxylase-oxygenase from alfalfa. Nucleic Acids Res. 1986 Dec 9;14(23):9535–9535. doi: 10.1093/nar/14.23.9535. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Aldrich J., Cherney B., Merlin E., Palmer J. Sequence of the rbcL gene for the large subunit of ribulose bisphosphate carboxylase-oxygenase from petunia. Nucleic Acids Res. 1986 Dec 9;14(23):9534–9534. doi: 10.1093/nar/14.23.9534. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Andersen K., Caton J. Sequence analysis of the Alcaligenes eutrophus chromosomally encoded ribulose bisphosphate carboxylase large and small subunit genes and their gene products. J Bacteriol. 1987 Oct;169(10):4547–4558. doi: 10.1128/jb.169.10.4547-4558.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Babul J. Phosphofructokinases from Escherichia coli. Purification and characterization of the nonallosteric isozyme. J Biol Chem. 1978 Jun 25;253(12):4350–4355. [PubMed] [Google Scholar]
- Birnboim H. C. A rapid alkaline extraction method for the isolation of plasmid DNA. Methods Enzymol. 1983;100:243–255. doi: 10.1016/0076-6879(83)00059-2. [DOI] [PubMed] [Google Scholar]
- Blangy D., Buc H., Monod J. Kinetics of the allosteric interactions of phosphofructokinase from Escherichia coli. J Mol Biol. 1968 Jan 14;31(1):13–35. doi: 10.1016/0022-2836(68)90051-x. [DOI] [PubMed] [Google Scholar]
- Carlisle S. M., Blakeley S. D., Hemmingsen S. M., Trevanion S. J., Hiyoshi T., Kruger N. J., Dennis D. T. Pyrophosphate-dependent phosphofructokinase. Conservation of protein sequence between the alpha- and beta-subunits and with the ATP-dependent phosphofructokinase. J Biol Chem. 1990 Oct 25;265(30):18366–18371. [PubMed] [Google Scholar]
- Carter P., Bedouelle H., Winter G. Improved oligonucleotide site-directed mutagenesis using M13 vectors. Nucleic Acids Res. 1985 Jun 25;13(12):4431–4443. doi: 10.1093/nar/13.12.4431. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chevalier C., Saillard C., Bové J. M. Organization and nucleotide sequences of the Spiroplasma citri genes for ribosomal protein S2, elongation factor Ts, spiralin, phosphofructokinase, pyruvate kinase, and an unidentified protein. J Bacteriol. 1990 May;172(5):2693–2703. doi: 10.1128/jb.172.5.2693-2703.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Daldal F. Molecular cloning of the gene for phosphofructokinase-2 of Escherichia coli and the nature of a mutation, pfkB1, causing a high level of the enzyme. J Mol Biol. 1983 Aug 5;168(2):285–305. doi: 10.1016/s0022-2836(83)80019-9. [DOI] [PubMed] [Google Scholar]
- Daldal F. Nucleotide sequence of gene pfkB encoding the minor phosphofructokinase of Escherichia coli K-12. Gene. 1984 Jun;28(3):337–342. doi: 10.1016/0378-1119(84)90151-3. [DOI] [PubMed] [Google Scholar]
- Daniels G. A., Drews G., Saier M. H., Jr Properties of a Tn5 insertion mutant defective in the structural gene (fruA) of the fructose-specific phosphotransferase system of Rhodobacter capsulatus and cloning of the fru regulon. J Bacteriol. 1988 Apr;170(4):1698–1703. doi: 10.1128/jb.170.4.1698-1703.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dayhoff M. O., Barker W. C., Hunt L. T. Establishing homologies in protein sequences. Methods Enzymol. 1983;91:524–545. doi: 10.1016/s0076-6879(83)91049-2. [DOI] [PubMed] [Google Scholar]
- Dills S. S., Apperson A., Schmidt M. R., Saier M. H., Jr Carbohydrate transport in bacteria. Microbiol Rev. 1980 Sep;44(3):385–418. doi: 10.1128/mr.44.3.385-418.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Doolittle R. F., Feng D. F. Nearest neighbor procedure for relating progressively aligned amino acid sequences. Methods Enzymol. 1990;183:659–669. doi: 10.1016/0076-6879(90)83043-9. [DOI] [PubMed] [Google Scholar]
- Feng D. F., Doolittle R. F. Progressive alignment and phylogenetic tree construction of protein sequences. Methods Enzymol. 1990;183:375–387. doi: 10.1016/0076-6879(90)83025-5. [DOI] [PubMed] [Google Scholar]
- French B. A., Chang S. H. Nucleotide sequence of the phosphofructokinase gene from Bacillus stearothermophilus and comparison with the homologous Escherichia coli gene. Gene. 1987;54(1):65–71. doi: 10.1016/0378-1119(87)90348-9. [DOI] [PubMed] [Google Scholar]
- Gehnrich S. C., Gekakis N., Sul H. S. Liver (B-type) phosphofructokinase mRNA. Cloning, structure, and expression. J Biol Chem. 1988 Aug 25;263(24):11755–11759. [PubMed] [Google Scholar]
- Gribskov M., Devereux J., Burgess R. R. The codon preference plot: graphic analysis of protein coding sequences and prediction of gene expression. Nucleic Acids Res. 1984 Jan 11;12(1 Pt 2):539–549. doi: 10.1093/nar/12.1part2.539. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Heinisch J., Ritzel R. G., von Borstel R. C., Aguilera A., Rodicio R., Zimmermann F. K. The phosphofructokinase genes of yeast evolved from two duplication events. Gene. 1989 May 30;78(2):309–321. doi: 10.1016/0378-1119(89)90233-3. [DOI] [PubMed] [Google Scholar]
- Hellinga H. W., Evans P. R. Mutations in the active site of Escherichia coli phosphofructokinase. Nature. 1987 Jun 4;327(6121):437–439. doi: 10.1038/327437a0. [DOI] [PubMed] [Google Scholar]
- Hellinga H. W., Evans P. R. Nucleotide sequence and high-level expression of the major Escherichia coli phosphofructokinase. Eur J Biochem. 1985 Jun 3;149(2):363–373. doi: 10.1111/j.1432-1033.1985.tb08934.x. [DOI] [PubMed] [Google Scholar]
- Hope J. N., Bell A. W., Hermodson M. A., Groarke J. M. Ribokinase from Escherichia coli K12. Nucleotide sequence and overexpression of the rbsK gene and purification of ribokinase. J Biol Chem. 1986 Jun 15;261(17):7663–7668. [PubMed] [Google Scholar]
- Kanehisa M. I. Los Alamos sequence analysis package for nucleic acids and proteins. Nucleic Acids Res. 1982 Jan 11;10(1):183–196. doi: 10.1093/nar/10.1.183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Knaff D. B. Structure and regulation of ribulose-1,5-bisphosphate carboxylase/oxygenase. Trends Biochem Sci. 1989 May;14(5):159–160. doi: 10.1016/0968-0004(89)90264-8. [DOI] [PubMed] [Google Scholar]
- Kotlarz D., Buc H. Regulatory properties of phosphofructokinase 2 from Escherichia coli. Eur J Biochem. 1981 Jul;117(3):569–574. doi: 10.1111/j.1432-1033.1981.tb06375.x. [DOI] [PubMed] [Google Scholar]
- Kotlarz D., Buc H. Two Escherichia coli fructose-6-phosphate kinases. Preparative purification, oligomeric structure and immunological studies. Biochim Biophys Acta. 1977 Sep 15;484(1):35–48. doi: 10.1016/0005-2744(77)90111-5. [DOI] [PubMed] [Google Scholar]
- Kyte J., Doolittle R. F. A simple method for displaying the hydropathic character of a protein. J Mol Biol. 1982 May 5;157(1):105–132. doi: 10.1016/0022-2836(82)90515-0. [DOI] [PubMed] [Google Scholar]
- Lee C. P., Kao M. C., French B. A., Putney S. D., Chang S. H. The rabbit muscle phosphofructokinase gene. Implications for protein structure, function, and tissue specificity. J Biol Chem. 1987 Mar 25;262(9):4195–4199. [PubMed] [Google Scholar]
- Levanon D., Danciger E., Dafni N., Bernstein Y., Elson A., Moens W., Brandeis M., Groner Y. The primary structure of human liver type phosphofructokinase and its comparison with other types of PFK. DNA. 1989 Dec;8(10):733–743. doi: 10.1089/dna.1989.8.733. [DOI] [PubMed] [Google Scholar]
- Lolkema J. S., Robillard G. T. Phosphoenolpyruvate-dependent fructose phosphotransferase system in Rhodopseudomonas sphaeroides. The coupling between transport and phosphorylation in inside-out vesicles. Eur J Biochem. 1985 Feb 15;147(1):69–75. doi: 10.1111/j.1432-1033.1985.tb08720.x. [DOI] [PubMed] [Google Scholar]
- Lolkema J. S., ten Hoeve-Duurkens R. H., Robillard G. T. The phosphoenolpyruvate-dependent fructose-specific phosphotransferase system in Rhodopseudomonas sphaeroides. Energetics of the phosphoryl group transfer from phosphoenolpyruvate to fructose. Eur J Biochem. 1986 Jan 15;154(2):387–393. doi: 10.1111/j.1432-1033.1986.tb09410.x. [DOI] [PubMed] [Google Scholar]
- Messing J. New M13 vectors for cloning. Methods Enzymol. 1983;101:20–78. doi: 10.1016/0076-6879(83)01005-8. [DOI] [PubMed] [Google Scholar]
- Needleman S. B., Wunsch C. D. A general method applicable to the search for similarities in the amino acid sequence of two proteins. J Mol Biol. 1970 Mar;48(3):443–453. doi: 10.1016/0022-2836(70)90057-4. [DOI] [PubMed] [Google Scholar]
- O'Neill M. C., Chiafari F. Escherichia coli promoters. II. A spacing class-dependent promoter search protocol. J Biol Chem. 1989 Apr 5;264(10):5531–5534. [PubMed] [Google Scholar]
- O'Neill M. C. Consensus methods for finding and ranking DNA binding sites. Application to Escherichia coli promoters. J Mol Biol. 1989 May 20;207(2):301–310. doi: 10.1016/0022-2836(89)90256-8. [DOI] [PubMed] [Google Scholar]
- O'Neill M. C. Escherichia coli promoters. I. Consensus as it relates to spacing class, specificity, repeat substructure, and three-dimensional organization. J Biol Chem. 1989 Apr 5;264(10):5522–5530. [PubMed] [Google Scholar]
- Orchard L. M., Kornberg H. L. Sequence similarities between the gene specifying 1-phosphofructokinase (fruK), genes specifying other kinases in Escherichia coli K12, and lacC of Staphylococcus aureus. Proc Biol Sci. 1990 Nov 22;242(1304):87–90. doi: 10.1098/rspb.1990.0108. [DOI] [PubMed] [Google Scholar]
- Pearson W. R., Lipman D. J. Improved tools for biological sequence comparison. Proc Natl Acad Sci U S A. 1988 Apr;85(8):2444–2448. doi: 10.1073/pnas.85.8.2444. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Poorman R. A., Randolph A., Kemp R. G., Heinrikson R. L. Evolution of phosphofructokinase--gene duplication and creation of new effector sites. 1984 May 31-Jun 6Nature. 309(5967):467–469. doi: 10.1038/309467a0. [DOI] [PubMed] [Google Scholar]
- Postma P. W., Lengeler J. W. Phosphoenolpyruvate:carbohydrate phosphotransferase system of bacteria. Microbiol Rev. 1985 Sep;49(3):232–269. doi: 10.1128/mr.49.3.232-269.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Riley M., Anilionis A. Evolution of the bacterial genome. Annu Rev Microbiol. 1978;32:519–560. doi: 10.1146/annurev.mi.32.100178.002511. [DOI] [PubMed] [Google Scholar]
- Rosey E. L., Stewart G. C. The nucleotide sequence of the lacC and lacD genes of Staphylococcus aureus. Nucleic Acids Res. 1989 May 25;17(10):3980–3980. doi: 10.1093/nar/17.10.3980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saier M. H., Jr Bacterial phosphoenolpyruvate: sugar phosphotransferase systems: structural, functional, and evolutionary interrelationships. Bacteriol Rev. 1977 Dec;41(4):856–871. doi: 10.1128/br.41.4.856-871.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saier M. H., Jr Evolution of permease diversity and energy-coupling mechanisms: an introduction. Res Microbiol. 1990 Mar-Apr;141(3):281–286. doi: 10.1016/0923-2508(90)90001-7. [DOI] [PubMed] [Google Scholar]
- Saier M. H., Jr, Feucht B. U., Roseman S. Phosphoenolpyruvate-dependent fructose phosphorylation in photosynthetic bacteria. J Biol Chem. 1971 Dec 25;246(24):7819–7821. [PubMed] [Google Scholar]
- Sharma P. M., Reddy G. R., Vora S., Babior B. M., McLachlan A. Cloning and expression of a human muscle phosphofructokinase cDNA. Gene. 1989 Apr 15;77(1):177–183. doi: 10.1016/0378-1119(89)90372-7. [DOI] [PubMed] [Google Scholar]
- Shinozaki K., Sugiura M. The nucleotide sequence of the tobacco chloroplast gene for the large subunit of ribulose-1,5-bisphosphate carboxylase/oxygenase. Gene. 1982 Nov;20(1):91–102. doi: 10.1016/0378-1119(82)90090-7. [DOI] [PubMed] [Google Scholar]
- Staden R. The current status and portability of our sequence handling software. Nucleic Acids Res. 1986 Jan 10;14(1):217–231. doi: 10.1093/nar/14.1.217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thornburgh B. N., Wu L. L., Griffin C. C. Phosphofructokinase from Escherichia coli: further evidence for identical subunits. Can J Biochem. 1978 Aug;56(8):836–838. doi: 10.1139/o78-127. [DOI] [PubMed] [Google Scholar]
- White D. W., Jacobson G. R. Molecular cloning of the C-terminal domain of Escherichia coli D-mannitol permease: expression, phosphorylation, and complementation with C-terminal permease deletion proteins. J Bacteriol. 1990 Mar;172(3):1509–1515. doi: 10.1128/jb.172.3.1509-1515.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu L. F., Saier M. H., Jr Nucleotide sequence of the fruA gene, encoding the fructose permease of the Rhodobacter capsulatus phosphotransferase system, and analyses of the deduced protein sequence. J Bacteriol. 1990 Dec;172(12):7167–7178. doi: 10.1128/jb.172.12.7167-7178.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu L. F., Tomich J. M., Saier M. H., Jr Structure and evolution of a multidomain multiphosphoryl transfer protein. Nucleotide sequence of the fruB(HI) gene in Rhodobacter capsulatus and comparisons with homologous genes from other organisms. J Mol Biol. 1990 Jun 20;213(4):687–703. doi: 10.1016/S0022-2836(05)80256-6. [DOI] [PubMed] [Google Scholar]
- Zurawski G., Whitfeld P. R., Bottomley W. Sequence of the gene for the large subunit of ribulose 1,5-bisphosphate carboxylase from pea chloroplasts. Nucleic Acids Res. 1986 May 12;14(9):3975–3975. doi: 10.1093/nar/14.9.3975. [DOI] [PMC free article] [PubMed] [Google Scholar]



