Abstract
Background
The optimal dose of protein substitute has not been determined in children with phenylketonuria (PKU).
Aim
To determine if a lower dose of protein substitute could achieve the same or better degree of blood phenylalanine control when compared to the dosage recommended by the UK MRC.1
Methods
In a six week randomised, crossover study, two doses of protein substitute (Protocol A: 2 g/kg/day of protein equivalent; Protocol B: 1.2 g/kg/day protein equivalent) were compared in 25 children with well controlled PKU aged 2–10 years (median 6 years). Each dose of protein substitute was taken for 14 days, with a 14 day washout period in between. Twice daily blood samples (fasting pre‐breakfast and evening, at standard times) for plasma phenylalanine were taken on day 8–14 of each protocol. The median usual dose of protein substitute was 2.2 g/kg/day (range 1.5–3.1 g/kg/day).
Results
When compared with control values, median plasma phenylalanine on the low dose of protein substitute increased at pre‐breakfast by 301 μmol/l (95% CI 215 to 386) and in the evening by 337 μmol/l (95% CI 248 to 431). On the high dose of protein substitute, plasma phenylalanine concentrations remained unchanged when compared to control values. However, wide variability was seen between subjects.
Conclusions
A higher dosage of protein substitute appeared to contribute to lower blood phenylalanine concentrations in PKU, but it did have a variable and individual impact and may have been influenced by the carbohydrate (+/− fat) content of the protein substitute.
Keywords: phenylketonuria, phenylalanine, protein substitute, energy intake
Phenylalanine‐free protein substitute is fundamental in the dietary management of PKU. It is based on L‐amino acids, supplemented with tyrosine, and may contain added carbohydrate, fat, vitamins, and minerals. In PKU, it supplies 80–85% of protein requirements. The MRC working group1 recommended that all children treated for PKU should receive a protein substitute. They suggested that the dose could be important, as it is likely to influence blood phenylalanine control. However, there is ongoing debate regarding precise protein substitute requirements in PKU. So far, available data is contradictory and insufficient to help assemble rational recommendations on actual dosage.
There are two schools of thought on protein substitute dosage. In the UK, the MRC1 guidelines recommend generous quantities of amino acid. It is suggested that children over 2 years should be maintained on a level of 2 g/kg/day. This is higher than the normal protein requirements.2 A high dose of protein substitute may lead to an increase in nitrogen retention and improve phenylalanine tolerance.3,4 Administration of protein equivalent in the form of L‐amino acids may be poorly utilised5,6 and a higher intake may be necessary to compensate for ineffective utilisation.
The second view states that there is no reason why children with PKU should have different protein requirements from other children without PKU.7 This is the preferred view of some centres in the USA8 and other European countries. For children between the ages of 1–10 years, normal protein requirements are relatively low (1.0–1.2 g/kg/day;9,2,10 table 1).
Table 1 Dietary reference values for protein (g/day).
If protein equivalent from protein substitute is given to provide normal requirements only and acceptable blood phenylalanine control is achieved, UK patients could significantly reduce the quantity of protein substitute they have to take each day. Protein substitute is unpalatable and expensive and it may be argued that lower doses of protein substitute may improve patient acceptance and compliance.
The aim of this randomised controlled study was therefore to investigate if a lower dose of protein substitute could achieve the same or better level of blood phenylalanine control when compared to the dosage recommended by the MRC.1 Two different doses of protein substitute were compared in a group of well controlled children with PKU.
Methods
Subjects
Twenty five children with well controlled PKU from the Birmingham Children's Hospital clinic were recruited. All children followed a strict low phenylalanine diet comprising: (1) a dietary phenylalanine allocation using a 50 mg phenylalanine exchange system (50 mg = 1 g protein); (2) a phenylalanine‐free protein substitute; and (3) low phenylalanine foods, e.g. most fruits, many vegetables, and special low protein products, or so called “free” foods permitted in usual quantities. There were three inclusion criteria: (1) maintenance of at least 70% plasma phenylalanine concentrations (weekly or fortnightly according to age group) within ranges recommended by the MRC Working Group1* in the six months before entering the study; (2) age 2–10 years; and (3) parental ability to take skin puncture blood specimens.
Fourteen girls and 11 boys, with a median age of 6 years (range 2–10 years) fulfilled these criteria and agreed to participate. The median number of 50 mg phenylalanine exchanges allocated was 5.5 per day (range 3−14) with an equivalent median natural protein intake of 5.5 g daily (table 2). Subjects continued to take their usual brand of protein substitute throughout the study.
Table 2 Age and natural protein intake of subjects.
| Subject no. | Age (y) | Number of 50 mg phenylalanine exchanges | Total daily natural protein intake (g) |
|---|---|---|---|
| 1 | 10 | 6 | 11.2 |
| 2 | 7 | 9 | 13 |
| 3 | 7 | 6 | 14 |
| 4 | 7 | 5 | 6 |
| 5 | 2 | 3 | 6 |
| 6 | 5 | 5 | 7 |
| 7 | 6 | 5 | 7 |
| 8 | 5 | 6 | 12 |
| 9 | 9 | 5 | 8.6 |
| 10 | 6 | 9 | 12 |
| 11 | 2 | 3.5 | 5 |
| 12 | 10 | 6 | 10 |
| 13 | 6 | 8 | 10 |
| 14 | 10 | 14 | 21 |
| 15 | 6 | 6 | 9 |
| 16 | 3 | 6 | 8 |
| 17 | 8 | 5.5 | 10 |
| 18 | 5 | 5.5 | 8 |
| 19 | 5 | 5 | 7 |
| 20 | 7 | 5.5 | 13 |
| 21 | 4 | 3 | 10.5 |
| 22 | 4 | 3 | 7 |
| 23 | 4 | 3 | 7 |
| 24 | 2 | 3.5 | 5 |
| 25 | 10 | 7 | 14 |
This was taken either as a paste or a drink. Eight subjects used XP Maxamaid (SHS International), eight PKU Gel (Vitaflo), two Minaphlex (SHS International), two Phlexy 10 Drink Mix (SHS International), two PKU Express (Vitaflo), one Aminogran Food Supplement powder (UCB), one Aminogran Food Supplement tablets (UCB), and one Pam 2 (SHS International). The composition of the different protein substitutes is given in table 3. The median dose of protein substitute was 2.2 g/kg/day (range 1.5–3.1 g/kg/day) at the start of the study.
Table 3 Nutritional composition of protein substitutes.
| Protein substitute | Company | Protein equivalent (g/100 g) | Carbohydrate (g/100 g) | Fat (g/100 g) | Energy (kcal/100 g) |
|---|---|---|---|---|---|
| Aminogran Food Supplement powder | UCB | 97.2 (amino acid) | – | – | 400 |
| Aminogran PKU Tablets | UCB | 97.2 (amino acid) | Neg | 0.19 | 264 |
| Minaphlex | SHS International | 29 | 38 | 13.5 | 390 |
| PAM 2 | SHS International | 77.5 | Nil added | Nil added | 310 |
| Phlexy 10 drink mix | SHS International | 42 | 44 | – | 343 |
| PKU Express | Vitaflo | 60 | 15 | <0.5 | 302 |
| PKU Gel | Vitaflo | 42 | 43 | <0.5 | 342 |
| XP Maxamaid | SHS International | 25 | 51 | <0.5 | 309 |
The local research ethics committee (LREC) approved the study. Informed consent was obtained from all parents and assent from all competent children.
Study design
In a six week randomised, crossover, prospective study, two dosages of protein substitute were given and blood phenylalanine concentrations compared. Each dosage of protein substitute was taken for 14 days, with a 14 day washout period in between. The protein substitute was taken three or four times (according to usual practice) throughout the day at standard times for each individual subject.
Protocol A: Children were given 2 g/kg/body weight per day of protein equivalent from protein substitute (high dose of protein substitute) for 14 days.
Protocol B: Protein substitute dosage was administered according to UK dietary reference values,2 i.e. 1.2 g/kg/day of protein equivalent for children (low dose of protein substitute) for 14 days.
Washout period: Patients took their usual type and dose of protein substitute for 14 days. The median control dose of protein equivalent from substitute was 2.2 g/kg/day (range 1.5 g/kg/day to 3.1 g/kg/day).
The randomisation order was generated in blocks of four. Each subject's randomisation order was retained in a sealed envelope until after the subjects or carers had signed the consent form.
Assessment of plasma phenylalanine concentrations
Twice daily finger prick blood samples (fasting pre‐breakfast and pre‐evening meal at standard times) were taken on day 8–14 of each trial period by parents/carers. Twice daily blood samples were collected for three days during a baseline and washout period; median phenylalanine concentrations were estimated from morning and evening blood samples. Twice daily blood samples for plasma phenylalanine were taken due to expected diurnal variation in plasma phenylalanine concentrations.11 Daily bloods were collected between day 8 and day 14 of each trial period to determine any day to day variability in protein substitute dosages. Median phenylalanine concentrations were estimated from morning and evening blood samples for each subject.
Parents or carers had been previously taught how to do thumb skin puncture blood specimens at home. Blood specimens were collected from the home and taken to the laboratory within eight hours of collection of the final specimen by the principal investigator. All blood specimens were centrifuged on receipt and resulting plasma samples stored at –20°C until analysed. Plasma phenylalanine concentrations were measured by high performance liquid chromatography (HPLC).
Assessment of protein substitute and dietary intake
As patients were unlikely to change their protein substitute to one brand or type of protein substitute, subjects took their usual type of protein substitute recommended by the dietician. The protein substitute was taken in three or four dosages at standard timings throughout the day for each individual subject. Patients also followed the same individualised, standard menu between days 8–14 of each protocol. In the first protocol the subjects were randomised to, parents or carers were asked to weigh and record their entire usual food intake between day 8 and day 14. They then repeated the same menu between day 8 and day 14 of the second protocol and documented all food intake. All food and drink was weighed using Salter scales (accurate to 5 g), which parents/carers were instructed on how to use.
Nutritional analysis was calculated using the Microdiet computer program based on McCance and Widdowson's “The Composition of Foods”12 with supplementary data provided by manufacturers.
Statistics
Non‐parametric tests for crossover studies (adapted from Wilcoxon signed rank tests) were used to detect differences in plasma phenylalanine concentrations between the two trial periods.
A power calculation indicated that 25 subjects should detect a 30% difference in blood phenylalanine concentrations between the two study protocols at a power of 90%.
Results
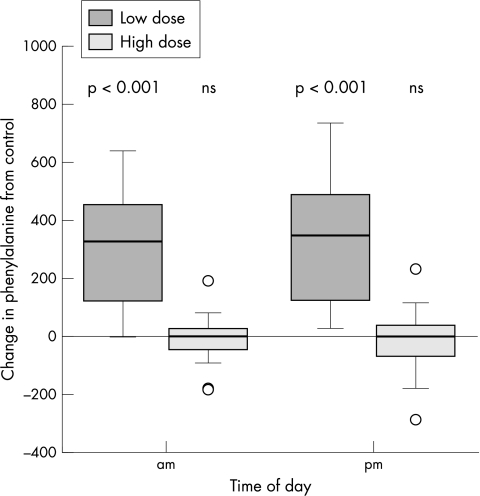
The higher dosage of protein substitute was associated with lower blood phenylalanine concentrations in PKU (table 4). When compared with control values, median plasma phenylalanine on the low dose of protein substitute increased at pre‐breakfast by 301 μmol/l (95% CI 215 to 386) (p < 0.001) and in the evening by 337 μmol/l (95% CI 248 to 431) (p < 0.001) (fig 1). On the high dose of protein substitute, when compared to control values, the median plasma phenylalanine concentrations remained unchanged (they decreased at pre‐breakfast by 4.5 μmol/l (95% CI −34 to 23) and in the evening by 6 μmol/l (95% CI −46 to 31).
Table 4 Differences in blood phenylalanine concentrations between the two protocols.
| Subject no. | Median morning phe levels on low dose of protein substitute (µmol/l) | Range of phe levels (µmol/l) | Median evening phe levels on low dose of protein substitute (µmol/l) | Range of phe levels (µmol/l) | Median morning phe levels on high dose of protein substitute (µmol/l) | Range of phe levels (µmol/l) | Median evening phe levels on high dose of protein substitute (µmol/l) | Range of phe levels (µmol/l) | Median difference in morning blood phe levels between low and high dose of protein substitute | Median difference in evening blood phe levels between low and high dose of protein substitute | % EAR2 on low dose of protein substitute | Energy difference per kg/day/body weight between low and high dose of protein substitute |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 200 | 90–360 | 240 | 60–310 | 100 | 60–110 | 40 | 30–100 | 100 | 200 | 106 | 3 |
| 2 | 250 | 200–320 | 220 | 180–320 | 180 | 130–250 | 170 | 140–210 | 70 | 50 | 117 | 4 |
| 3 | 460 | 370–480 | 410 | 210–430 | 290 | 210–350 | 270 | 190–350 | 170 | 140 | 96 | 6 |
| 4 | 530 | 460–550 | 510 | 440–590 | 200 | 100–310 | 170 | 60–350 | 330 | 340 | 121 | 8 |
| 5 | 420 | 300–570 | 330 | 180–430 | 220 | 200–290 | 30 | <30–70 | 200 | 300 | 81 | 12 |
| 6 | 770 | 700–1000 | 720 | 670–970 | 170 | 130–200 | 90 | 50–140 | 600 | 630 | 69 | 12 |
| 7 | 650 | 480–770 | 620 | 510–690 | 390 | 190–560 | 510 | 420–530 | 260 | 110 | 95 | 11 |
| 8 | 720 | 540–780 | 650 | 440–760 | 230 | 210–320 | 190 | 160–330 | 490 | 460 | 95 | 9 |
| 9 | 630 | 550–670 | 550 | 480–610 | 210 | 150–240 | 50 | 40–70 | 420 | 500 | 92 | 6 |
| 10 | 400 | 350–470 | 390 | 360–450 | 230 | 190–380 | 230 | 150–350 | 170 | 160 | 87 | 9 |
| 11 | 180 | 150–350 | 130 | 40–200 | 170 | 160–260 | 90 | 30–150 | 10 | 40 | 78 | 7 |
| 12 | 530 | 480–580 | 520 | 500–620 | 120 | 90–210 | 150 | 30–270 | 410 | 370 | 86 | 7 |
| 13 | 390 | 300–430 | 370 | 320–420 | 180 | 130–210 | 120 | 110–180 | 210 | 250 | 79 | 5 |
| 14 | 530 | 490–560 | 540 | 480–570 | 270 | 260–310 | 260 | 170–290 | 260 | 280 | 109 | 4 |
| 15 | 370 | 340–400 | 360 | 320–380 | 150 | 140–170 | 100 | 70–140 | 220 | 260 | 84 | 7 |
| 16 | 570 | 450–640 | 560 | 420–600 | 270 | 210–290 | 140 | 90–290 | 300 | 420 | 80 | 9 |
| 17 | 680 | 600–750 | 690 | 540–730 | 220 | 160–350 | 100 | 80–170 | 460 | 590 | 112 | 10 |
| 18 | 910 | 810–1000 | 920 | 830–940 | 310 | 190–400 | 280 | 220–300 | 600 | 640 | 108 | 12 |
| 19 | 460 | 450–530 | 400 | 360–440 | 140 | 140–290 | 115 | 70–270 | 320 | 285 | 70 | 3 |
| 20 | 260 | 210–330 | 250 | 170–300 | 180 | 130–230 | 140 | 100–190 | 80 | 110 | 114 | 6 |
| 21 | 785 | 730–930 | 825 | 790–990 | 160 | 110–190 | 120 | 90–150 | 625 | 705 | 96 | 11 |
| 22 | 440 | 400–580 | 420 | 310–560 | 190 | 140–560 | 260 | 50–460 | 250 | 160 | 67 | 12 |
| 23 | 600 | 540–670 | 570 | 470–660 | 360 | 200–450 | 390 | 210–400 | 240 | 180 | 69 | 12 |
| 24 | 770 | 630–860 | 740 | 660–820 | 310 | 150–520 | 360 | 120–420 | 460 | 380 | 68 | 9 |
| 25 | 140 | 100–180 | 90 | 40–140 | 120 | 40–140 | 26 | <30–180 | 20 | 64 | 151 | 3 |
phe, phenylalanine.
Figure 1 Change in blood phenylalanine concentrations from control values on high and low dose of protein substitute.
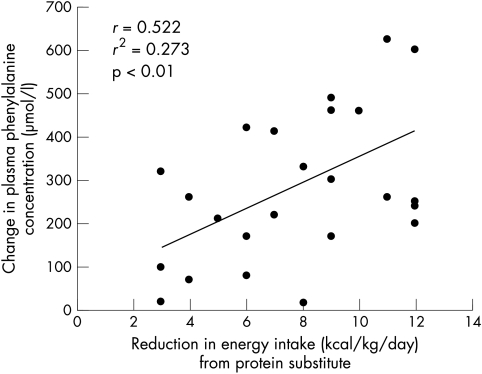
There was wide variability in changes in blood phenylalanine concentrations between individual subjects. It was not correlated with age, phenylalanine tolerance, or total energy intake. However, there was a reduction in energy intake during administration of the lower dose of protein substitute in protocol B (median reduction 8 kcal/kg/day; range 3–12 kcal/kg/day) and this was positively correlated with blood phenylalanine concentrations (r = 0.522; r2 = 0.273, p < 0.01) (fig 2). Children who took protein substitute with a higher energy content (higher carbohydrate and fat) had the greatest reductions in energy intake.
Figure 2 Relation between reduction in energy intake from protein substitute in the two phases of the study and changes in blood phenylalanine concentrations.
Discussion
This is the first randomised, controlled trial to identify that a lower dose of protein equivalent provided by a protein substitute, similar to the safe levels of protein intake recommend by the FAO/WHO/UNU10 is associated with an increase in blood phenylalanine concentrations. Other studies have observed that lower protein substitute intake adversely affects overall blood phenylalanine control and phenylalanine tolerance but this was in smaller or uncontrolled studies. Duran et al13 observed in six patients with PKU that compliance with protein substitute intake following supervision in hospital led to a significant reduction in blood phenylalanine control. Acosta and Yannicelli3 showed in 25 infants with PKU given an infant protein substitute containing high quantities of protein equivalent (3.12 g of protein per 100 kcal), that dietary phenylalanine tolerance improved and blood phenylalanine concentrations were lower. In a longitudinal study, Kindt et al4 fed two groups of eight children with PKU different quantities of total protein. One group adhered to the US Recommended Dietary Amounts (median intake 2.0 g/kg/day)14 and the other group to the lower safe levels (median intake 1.46 g/kg/day) recommended by the Joint FAO/WHO Expert Committee.15 The RDA group tolerated additional phenylalanine (3–5 mg/kg body weight/day).
Two other studies do not support a high intake of protein substitute. Clemens et al16 gave 10 patients, aged 15–37 years, three different levels of protein equivalent (1.5 g/kg; 0.75 g/kg; and 0.65 g/kg) from a protein substitute consisting of phenylalanine‐free essential amino acids and tyrosine only for three months each. For all three levels of protein intake, there was no sign of amino acid imbalance or catabolism. On the basis of these results, Clemens suggested that as long as phenylalanine‐free essential amino acids and tyrosine are given, the overall quantity of the protein substitute may be reduced. However, all these patients were on more natural protein than younger children with PKU, and intake varied between 9 and 26 g/day. Prince et al8 found, in a group of patients aged 4–10 years, the actual amino acid intake from protein substitute decreased from only 0.9 g/kg body weight/day to as little as 0.4 g/kg body weight/day in children over 4.2 years of age. Growth and nutritional biochemistry were not adversely affected, although mean serum phenylalanine increased from 380 to 480 µmol/l. The authors suggested that the increase in serum phenylalanine was just a reflection of increasing patient age, but equally it could have related to reduced intake of protein substitute.
What is already known on this topic
Phenylalanine‐free protein substitute provides 80–85% of protein requirements in PKU
The optimal dosage of protein substitute has not been determined in PKU
In our study, changes in dosage of protein substitute appeared to have an immediate effect on plasma phenylalanine concentrations. The exact mechanism for the higher blood phenylalanine concentrations on a lower dose of protein substitute is unclear. It may be explained by increased catabolism associated with either a lower amino acid intake or a reduction in energy consumed from carbohydrate or fat added to the protein substitute; the influence of energy on protein utilisation and nitrogen balance is well recognised.17 It is generally believed that stimulation of protein deposition occurs in response to intakes of dietary energy as well as protein, with dietary carbohydrate more effective than fat.18 Individual subject phenylalanine tolerance (a possible indicator of disease severity) did not appear to influence the results, although further mutation analysis work has been conducted on this group of patients.
The variable energy content of the different protein substitutes appeared to be an important factor affecting the results. This was controlled for in the study as recruitment would have been difficult if all the patients had been asked to standardise their protein substitute to one type. A disparate group of protein substitutes was therefore used. However, although all subjects had an increase in blood phenylalanine concentrations on lower doses of protein substitutes, subjects consuming protein substitutes with added carbohydrate (+/− fat) at baseline had greater reductions in energy intake on the lower doses and had the greater corresponding increases in blood phenylalanine concentrations. Illsinger et al19 recently showed in subjects with PKU that phenylalanine concentrations decreased with energy supplementation. In contrast, MacDonald et al20 showed that the use of a low carbohydrate protein substitute (15 g/100 g) did not appear to adversely affect phenylalanine control in the majority of a group of 23 teenagers and adults with PKU.
In conclusion, a high dose of protein substitute appeared to lower blood phenylalanine concentrations in PKU. However, it did have a variable and individual impact on overall phenylalanine control. Phenylalanine control may have been influenced by inadvertently increasing the intake of exogenous carbohydrate (+/− fat) added to the protein substitutes on the higher dosage. To further explore the effect of protein substitute dosage, additional controlled studies maintaining a constant intake of carbohydrate and fat are necessary. However, the results of this study do suggest that dosage of protein substitute may have an important role in overall blood phenylalanine control.
What this study adds
This is the first randomised controlled trial to show that a higher dose of protein substitute (as recommended by the MRC in 1993) appears to lower blood phenylalanine concentrations
The impact of the dosage of protein substitute is variable but its effect is enhanced by an increasing carbohydrate (+/− fat) intake
Acknowledgements
We wish to thank all the patients and their carers who took part in this study.
Footnotes
Children 0–5 years maintained blood phenylalanine concentrations at 120−360 µmol/l; school age children 120−480 µmol/l.
Funding: Birmingham Children's Hospital Research Foundation
Competing interests: The principle author receives a grant from the BCH Research Foundation to pay for blood sampling and materials investigating alternative methods of estimating phenylalanine exchanges in PKU. The principle author also receives research grants from SHS International to pay for blood analysis for a project on the efficacy of a new vitamin and mineral tablet for use in PKU and from Vitaflo International to pay for blood analysis for a project in very long chain fatty acid disorders.
References
- 1.Medical Research Council Working Party on Phenylketonuria Recommendations on the dietary management of phenylketonuria. Arch Dis Child 199368426–427. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Department of Health Dietary reference values for food, energy and nutrients for the United Kingdom. Report on Health and Social Subjects. No. 41. London: HMSO, 1991 [PubMed]
- 3.Acosta P B, Yannicelli S. Protein intake affects phenylalanine requirements and growth of infants with phenylketonuria. Acta Paediatr 199483(suppl 407)66–67. [DOI] [PubMed] [Google Scholar]
- 4.Kindt E, Motzfeldt K, Halvorsen S.et al Is phenylalanine requirements in infants and children related to protein intake? Br J Nutr 198451435–442. [DOI] [PubMed] [Google Scholar]
- 5.Woolf L I. Nutrition in relation to phenylketonuria. Proceedings of the Nutrition Society 19622121–28. [DOI] [PubMed] [Google Scholar]
- 6.Jones B J M, Lees R, Andrews J.et al Comparison of an elemental enteral and polymeric diet in patients with normal gastrointestinal function. Gut 19832478–84. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Przyrembel H. Recommendations for protein and amino acid intake in phenylketonuria patients. Eur J Pediatr 1996155S130–S131. [DOI] [PubMed] [Google Scholar]
- 8.Prince A P, McMurray M P, Buist N R M. Treatment products and approaches for phenylketonuria: improved palatability and flexibility demonstrate safety, efficacy and acceptance in US clinical trials. J Inher Metab Dis 199720486–498. [DOI] [PubMed] [Google Scholar]
- 9. EC Scientific Committee for Food Report (31st series). Nutrient and energy intakes for the European Community. Luxembourg: Office for Official Publications of the European Communities, 1993
- 10.World Health Organisation Energy and protein requirements. Report of a Joint FAO/WHO/UNU Expert Consultation. WHO Technical Report Series. No. 724. Geneva: WHO, 1985 [PubMed]
- 11.MacDonald A, Rylance G, Asplin D.et al Does a single plasma phenylalanine predict quality of control in phenylketonuria? Arch Dis Child 199878122–126. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Roe M A, Finglas P M, Church S M. (Food Standards Agency and Institute of Food Research). McCance and Widdowson's “The composition of foods”. Cambridge: Royal Society of Chemistry and Foods Standards Agency, 2002
- 13.Duran G P, Rohr F J, Slonim A.et al Necessity of complete intake of phenylalanine‐free amino acid mixture for metabolic control of phenylketonuria. J Am Diet Assoc 1999991559–1563. [DOI] [PubMed] [Google Scholar]
- 14.National Research Council, Food and Nutrition Board, Commission on Life Sciences Recommended dietary allowances, 9th edn. Washington, DC: National Academy Press, 1980
- 15.World Health Organisation Energy and protein requirements. Report of a Joint FAO/WHO/UNU Expert Consultation. WHO Technical Report Series. No. 522. Geneva: WHO, 1973
- 16.Clemens P C, Heddrich‐Ellerbrok M, Wachtel U.et al Plasma amino acids in adolescents and adults with phenylketonuria on three different levels of protein intake. Acta Paediatr 199180577–580. [DOI] [PubMed] [Google Scholar]
- 17.Millward D J, Rivers J P W. The nutritional role of indispensable amino acids and the metabolic basis for their requirements. Eur J Clin Nutr 198842367–393. [PubMed] [Google Scholar]
- 18.Munro H N, Thomson W S T. The influence of glucose on amino acid metabolism. Metabolism 19532354–361. [PubMed] [Google Scholar]
- 19.Illsinger S, Lucke T, Meyer U.et al Branched chain amino acids as a parameter for catabolism in treated phenylketonuria. Amino Acids 20052845–50. [DOI] [PubMed] [Google Scholar]
- 20.MacDonald A, Lilburn M, Cochrane B.et al A new, low volume protein substitute for teenagers and adults with PKU. J Inherit Metab Dis 200427127–135. [DOI] [PubMed] [Google Scholar]