Abstract
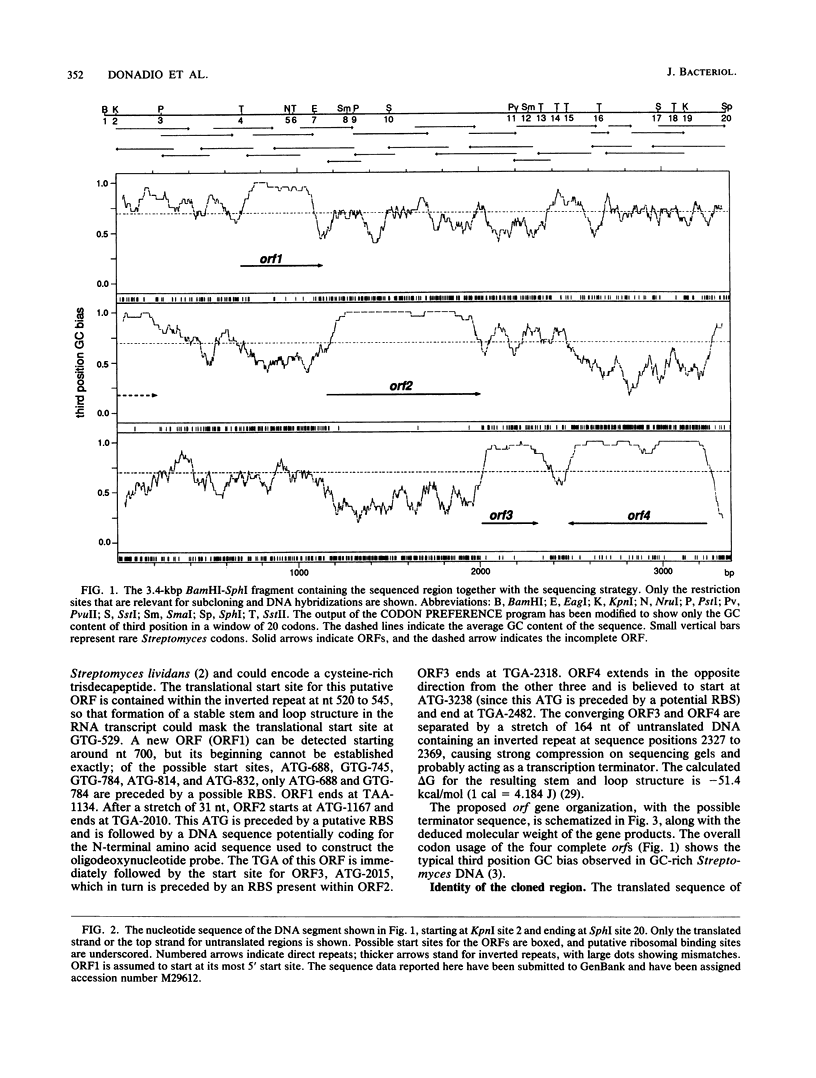
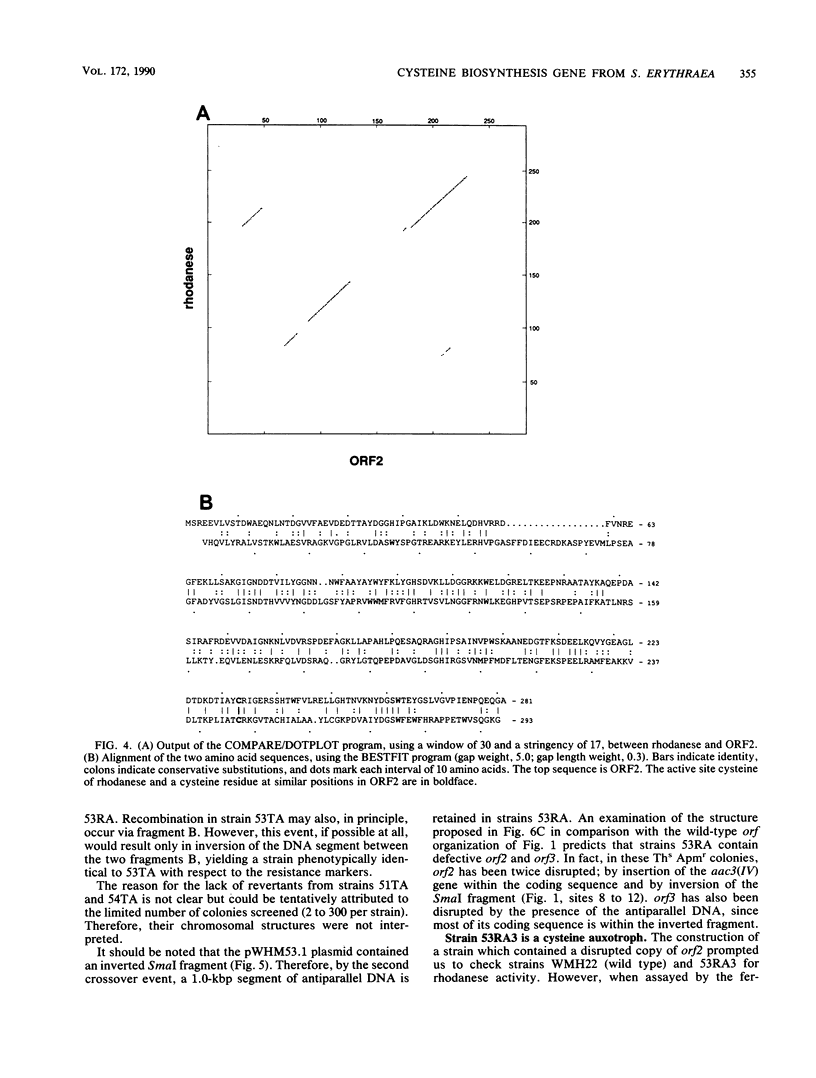
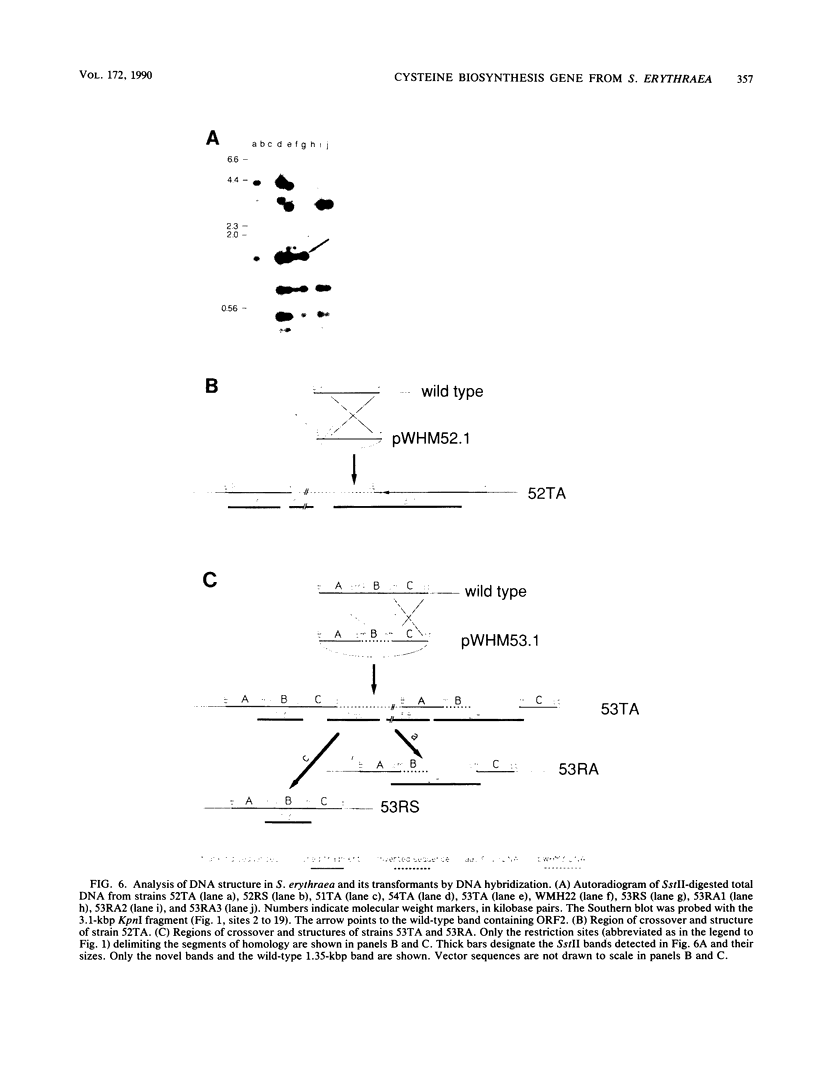
A 3,373-base-pair DNA segment from a clone fortuitously isolated from Saccharopolyspora erythraea by hybridization to an oligodeoxynucleotide probe was sequenced. Computer-assisted analysis of the nucleotide sequence reveals three closely linked Streptomyces open reading frames plus a fourth converging on the others. The deduced product of one of them, ORF2, shows considerable similarity to bovine liver rhodanese. orf2, and the closely linked orf3 located just downstream of it, were disrupted by insertion of an apramycin resistance cassette into the orf2 coding sequence along with inversion of the fragment carrying most of orf2 and orf3 via two successive recombinational events in the wild-type strain. The mutant strain thus created contains wild-type levels of rhodanese activity but cannot grow on minimal medium. It is a cysteine auxotroph, capable of utilizing efficiently only thiosulfate among the inorganic sulfur sources tested. orf2 has been designated cysA. The possible role of the rhodaneselike cysA gene product in thiosulfate formation is discussed.
Full text
PDF






Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Alexander K., Volini M. Properties of an Escherichia coli rhodanese. J Biol Chem. 1987 May 15;262(14):6595–6604. [PubMed] [Google Scholar]
- Bibb M. J., Cohen S. N. Gene expression in Streptomyces: construction and application of promoter-probe plasmid vectors in Streptomyces lividans. Mol Gen Genet. 1982;187(2):265–277. doi: 10.1007/BF00331128. [DOI] [PubMed] [Google Scholar]
- Bibb M. J., Findlay P. R., Johnson M. W. The relationship between base composition and codon usage in bacterial genes and its use for the simple and reliable identification of protein-coding sequences. Gene. 1984 Oct;30(1-3):157–166. doi: 10.1016/0378-1119(84)90116-1. [DOI] [PubMed] [Google Scholar]
- Devereux J., Haeberli P., Smithies O. A comprehensive set of sequence analysis programs for the VAX. Nucleic Acids Res. 1984 Jan 11;12(1 Pt 1):387–395. doi: 10.1093/nar/12.1part1.387. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gribskov M., Burgess R. R. Sigma factors from E. coli, B. subtilis, phage SP01, and phage T4 are homologous proteins. Nucleic Acids Res. 1986 Aug 26;14(16):6745–6763. doi: 10.1093/nar/14.16.6745. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Henikoff S. Unidirectional digestion with exonuclease III creates targeted breakpoints for DNA sequencing. Gene. 1984 Jun;28(3):351–359. doi: 10.1016/0378-1119(84)90153-7. [DOI] [PubMed] [Google Scholar]
- Hutchinson C. R. The impact of genetic engineering on the commercial production of antibiotics by Streptomyces and related bacteria. Appl Biochem Biotechnol. 1987 Sep-Dec;16:169–190. doi: 10.1007/BF02798365. [DOI] [PubMed] [Google Scholar]
- Katz E., Thompson C. J., Hopwood D. A. Cloning and expression of the tyrosinase gene from Streptomyces antibioticus in Streptomyces lividans. J Gen Microbiol. 1983 Sep;129(9):2703–2714. doi: 10.1099/00221287-129-9-2703. [DOI] [PubMed] [Google Scholar]
- Katz L., Brown D., Boris K., Tuan J. Expression of the macrolide-lincosamide-streptogramin-B-resistance methylase gene, ermE, from Streptomyces erythraeus in Escherichia coli results in N6-monomethylation and N6,N6-dimethylation of ribosomal RNA. Gene. 1987;55(2-3):319–325. doi: 10.1016/0378-1119(87)90291-5. [DOI] [PubMed] [Google Scholar]
- Kern B. A., Inamine E. Cystathionine gamma-lyase activity in the cephamycin C producer Streptomyces lactamdurans. J Antibiot (Tokyo) 1981 May;34(5):583–589. doi: 10.7164/antibiotics.34.583. [DOI] [PubMed] [Google Scholar]
- LEINWEBER F. J., MONTY K. J. CYSTEINE BIOSYNTHESIS IN NEUROSPORA CRASSA. I. THE METABOLISM OF SULFITE, SULFIDE, AND CYSTEINESULFINIC ACID. J Biol Chem. 1965 Feb;240:782–787. [PubMed] [Google Scholar]
- Langridge J., Langridge P., Bergquist P. L. Extraction of nucleic acids from agarose gels. Anal Biochem. 1980 Apr;103(2):264–271. doi: 10.1016/0003-2697(80)90266-3. [DOI] [PubMed] [Google Scholar]
- Meinkoth J., Wahl G. Hybridization of nucleic acids immobilized on solid supports. Anal Biochem. 1984 May 1;138(2):267–284. doi: 10.1016/0003-2697(84)90808-x. [DOI] [PubMed] [Google Scholar]
- Messing J. New M13 vectors for cloning. Methods Enzymol. 1983;101:20–78. doi: 10.1016/0076-6879(83)01005-8. [DOI] [PubMed] [Google Scholar]
- Ohtsuka E., Matsuki S., Ikehara M., Takahashi Y., Matsubara K. An alternative approach to deoxyoligonucleotides as hybridization probes by insertion of deoxyinosine at ambiguous codon positions. J Biol Chem. 1985 Mar 10;260(5):2605–2608. [PubMed] [Google Scholar]
- POSTGATE J. R. The examination of sulphur auxotrophs: a warning. J Gen Microbiol. 1963 Mar;30:481–484. doi: 10.1099/00221287-30-3-481. [DOI] [PubMed] [Google Scholar]
- Poustka A., Rackwitz H. R., Frischauf A. M., Hohn B., Lehrach H. Selective isolation of cosmid clones by homologous recombination in Escherichia coli. Proc Natl Acad Sci U S A. 1984 Jul;81(13):4129–4133. doi: 10.1073/pnas.81.13.4129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Richardson M. A., Kuhstoss S., Solenberg P., Schaus N. A., Rao R. N. A new shuttle cosmid vector, pKC505, for streptomycetes: its use in the cloning of three different spiramycin-resistance genes from a Streptomyces ambofaciens library. Gene. 1987;61(3):231–241. doi: 10.1016/0378-1119(87)90187-9. [DOI] [PubMed] [Google Scholar]
- Russell J., Weng L., Keim P. S., Heinrikson R. L. The covalent structure of bovine liver rhodanese. Isolation and partial structural analysis of cyanogen bromide fragements and the complete sequence of the enzyme. J Biol Chem. 1978 Nov 25;253(22):8102–8108. [PubMed] [Google Scholar]
- Shafiee A., Hutchinson C. R. Macrolide antibiotic biosynthesis: isolation and properties of two forms of 6-deoxyerythronolide B hydroxylase from Saccharopolyspora erythraea (Streptomyces erythreus). Biochemistry. 1987 Sep 22;26(19):6204–6210. doi: 10.1021/bi00393a037. [DOI] [PubMed] [Google Scholar]
- Tinoco I., Jr, Borer P. N., Dengler B., Levin M. D., Uhlenbeck O. C., Crothers D. M., Bralla J. Improved estimation of secondary structure in ribonucleic acids. Nat New Biol. 1973 Nov 14;246(150):40–41. doi: 10.1038/newbio246040a0. [DOI] [PubMed] [Google Scholar]
- Vara J., Lewandowska-Skarbek M., Wang Y. G., Donadio S., Hutchinson C. R. Cloning of genes governing the deoxysugar portion of the erythromycin biosynthesis pathway in Saccharopolyspora erythraea (Streptomyces erythreus). J Bacteriol. 1989 Nov;171(11):5872–5881. doi: 10.1128/jb.171.11.5872-5881.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vieira J., Messing J. The pUC plasmids, an M13mp7-derived system for insertion mutagenesis and sequencing with synthetic universal primers. Gene. 1982 Oct;19(3):259–268. doi: 10.1016/0378-1119(82)90015-4. [DOI] [PubMed] [Google Scholar]
- Weber J. M., Losick R. The use of a chromosome integration vector to map erythromycin resistance and production genes in Saccharopolyspora erythraea (Streptomyces erythraeus). Gene. 1988 Sep 7;68(2):173–180. doi: 10.1016/0378-1119(88)90019-4. [DOI] [PubMed] [Google Scholar]
- Weber J. M., Schoner B., Losick R. Identification of a gene required for the terminal step in erythromycin A biosynthesis in Saccharopolyspora erythraea (Streptomyces erythreus). Gene. 1989 Feb 20;75(2):235–241. doi: 10.1016/0378-1119(89)90269-2. [DOI] [PubMed] [Google Scholar]
- Weber J. M., Wierman C. K., Hutchinson C. R. Genetic analysis of erythromycin production in Streptomyces erythreus. J Bacteriol. 1985 Oct;164(1):425–433. doi: 10.1128/jb.164.1.425-433.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weng L., Heinrikson R. L., Westley J. Active site cysteinyl and arginyl residues of rhodanese. A novel formation of disulfide bonds in the active site promoted by phenylglyoxal. J Biol Chem. 1978 Nov 25;253(22):8109–8119. [PubMed] [Google Scholar]
- Westley J. Rhodanese. Adv Enzymol Relat Areas Mol Biol. 1973;39:327–368. doi: 10.1002/9780470122846.ch5. [DOI] [PubMed] [Google Scholar]
- Westley J. Thiosulfate: cyanide sulfurtransferase (rhodanese). Methods Enzymol. 1981;77:285–291. doi: 10.1016/s0076-6879(81)77039-3. [DOI] [PubMed] [Google Scholar]
- Yamamoto H., Maurer K. H., Hutchinson C. R. Transformation of Streptomyces erythraeus. J Antibiot (Tokyo) 1986 Sep;39(9):1304–1313. doi: 10.7164/antibiotics.39.1304. [DOI] [PubMed] [Google Scholar]
- Zalacain M., González A., Guerrero M. C., Mattaliano R. J., Malpartida F., Jiménez A. Nucleotide sequence of the hygromycin B phosphotransferase gene from Streptomyces hygroscopicus. Nucleic Acids Res. 1986 Feb 25;14(4):1565–1581. doi: 10.1093/nar/14.4.1565. [DOI] [PMC free article] [PubMed] [Google Scholar]