Abstract
Elucidation of the underlying pathogenic mechanisms leading to apoptosis of neurons and oligodendrocytes and activation of microglia and astrocytes in different neurodegenerative and neuroinflammatory disorders remains a challenge in neuroscience. In order to overcome the challenge and find out therapeutic remedies, it is important to study live and death processes in each and every cell type of the brain. Here we present a protocol of isolating highly purified microglia, astrocytes, oligodendrocytes, and neurons, all four major cell types of the CNS, from the same human fetal brain tissue. As found in vivo, these primary neurons and oligodendroglia underwent apoptosis and cell death in response to neurodegenerative challenges. On the other hand, astroglia, and microglia, cells that do not die in neurodegenerative brains, became activated after inflammatory challenge. The availability of highly purified human brain cells will increase the possibility of developing therapies for different neurodegenerative disorders.
Keywords: Human fetal tissue, Neurons, Oligodendroglia, Microglia, Astroglia, Neurodegeneration
Introduction
Brain cells are mainly divided into two groups–glia and neurons. While neurons form the basis of the so-called supercomputing system of the brain, glial cells carry out diverse functions that are necessary for the proper development and function of the complex nervous system. For example, axons are surrounded by myelin that enables axons to conduct impulses between the brain and other parts of the body. This myelin is synthesized by oligodendroglia in the CNS. Astroglia, the major glial cells in the CNS, have many important functions, such as structural support, food supply, water balance, ion homeostasis, regulation of neurotransmitters, detoxification of ammonia, organizing information network, and release of neuropeptides and neurotrophins. On the other hand, microglia, the other important glial population, keeps the CNS microenvironment clean by scavenging any debris or dead cell bodies. Additionally, when immune responses are generated within the CNS or from outside the CNS, microglia, being the primary CNS immune cells, receive and pass on that response to other cells [20, 21]. Under physiological condition, the immune response usually ends up with a logical conclusion leading to the development of a better neuroimmune system.
However, as part of our life, we experience stress, trauma, infection, injury, degeneration, etc. and come in contact with various toxic substances. Although there are four major cell types in the CNS, under inflammatory and degenerative stress conditions, only neurons and oligodendroglia succumb to cell death. On the other hand, astroglia and microglia do not die but undergo activation and gliosis under the same condition. In multiple sclerosis, the most common human demyelinating disease, mainly oligodendroglia die while microglia and astroglia undergo activation. Similarly, in neurodegenerative disorders such as, Alzheimer's disease (AD), Parkinson's disease, HIV-associated dementia, mainly neurons die while microglia and astroglia undergo activation and gliosis [15, 22].
Despite intense investigations in past decades, neither underlying pathogenic mechanisms are fully understood nor is effective therapy available to halt these neurodegenerative and neuroinflammatory disorders. Therefore, understanding degenerative processes in neurons and oligodendroglia and/or activation processes in astroglia and microglia is an important area of neuroscience research. In recent years, a number of protocols on the preparation of mixed primary cultures of brain cells and separation of a particular brain cell type from the whole primary cell culture have been established [1, 2, 5, 11, 12]. However, it is not known how to utilize the same tissue for the isolation of all four major cell types of the CNS. For example, a single fetal tissue has been used to isolate only neurons [19]. In some cases, the purity of fetal neurons is also below 80% [19]. Here we describe a facile and reproducible method of isolating highly purified neurons, oligodendroglia, astroglia, and microglia from the same fetal brain tissue. Moreover, we provide examples of the feasibility of manipulating these cultures under neurodegenerative challenges.
Materials and methods
Reagents
Fetal bovine serum (FBS), Hank's balanced salt solution (HBSS), trypsin and DMEM/F-12 were from Mediatech (USA). Neurobasal media and B27 supplement were purchased from Invitrogen (San Diego, CA, USA). Recombinant human TNF-α was obtained from R&D (USA). HIV-1 coat protein gp120 (expressed in Chinese hamster ovary cells; strain, HIV-1 MN) was obtained from US Biological (Swampscott, MA, USA). Human Aβ peptides 1-42 and 42–1 were obtained from Bachem Bioscience (Philadelphia, PA, USA). Antibodies against myelin basic protein (MBP) were purchased from Sigma (St. Louis, MO, USA). Antibodies against CD11b, 2′,3′-cyclic nucleotide 3′-phosphodiesterase (CNPase) and myelin oligodendrocyte glycoprotein (MOG) were obtained from Chemicon International (Temecula, CA, USA). Antibodies against glial fibrillary acidic protein (GFAP) and microtubule-associated protein-2 (MAP-2) were purchased from Santa Cruz Biotechnology (Santa Cruz, CA, USA).
Isolation of human primary neurons
Human primary neurons were prepared as described previously [23] with some modifications [8, 9]. All of the experimental protocols were reviewed and approved by the Institutional Review Board of the University of Nebraska Medical Center and the Rush University Medical Center. Briefly, 11- to 17-week-old fetal brains obtained from the Human Embryology Laboratory (University of Washington, Seattle, WA, USA) were dissociated by trituration and trypsinization (0.25% trypsin in PBS at 37°C for 15 min). The trypsin was inactivated with 10% heat-inactivated FBS (Mediatech, Washington, DC, USA). The dissociated cells were filtered successively through 380 and 140 μm meshes (Sigma) and pelleted by centrifugation. The cell pellet was washed once with PBS and once with Neurobasal media containing 2% B27 and 1% antibiotic-antimycotic mixture (Sigma). In the first step, neurons were enriched by allowing the cells (3 × 106/ml) to adhere to poly-d-lysine-coated plates or coverslips for 5 min (Fig. 1). Nonadherent cells containing microglia, astrocytes, and oligodendrocytes were removed and used for the isolation of other cells (Fig. 1). Adherent cells (mostly neurons) received the same Neurobasal media and were further treated with 10 μM cytosine arabinoside (AraC) for 10 days to prevent proliferation of dividing cells. More than 98% of this preparation was positive for MAP-2, a marker for neurons.
Fig. 1.
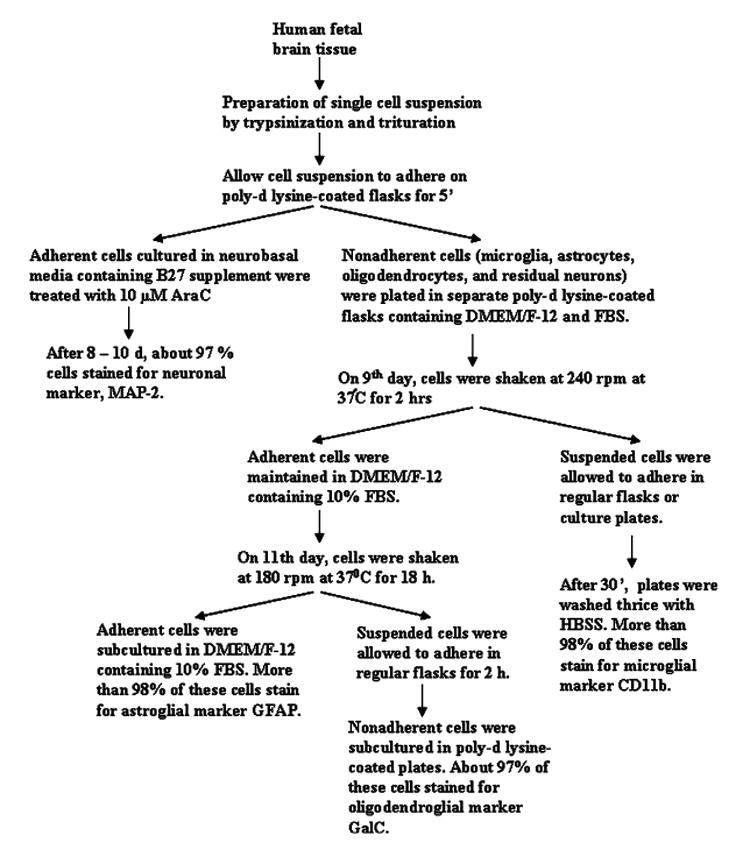
Flowcharts representing the entire isolation process
Isolation of human primary microglia
The remaining nonadherent suspension enriched with mixed glial population were centrifuged for 10 min at 1,500 rpm and then suspended in DMEM supplemented with 10% heat inactivated FBS (complete medium). Cells were plated on poly-d-lysine precoated 75 cm2 flasks and incubated at 37°C with 5% CO2 in air. Culture medium was changed after 3 days and every 3 days thereafter. On ninth day these mixed glial cultures were subjected to shaking to take advantage of the selective cell adhesion properties of the different glial subtypes as previously described by Giulian and Baker [7]. The cultures were placed on a rotary shaker at 240 rpm at 37°C for 2 h to remove loosely attached microglia. The cell suspensions were placed on uncoated culture plates for 30 min followed by removal of nonadherent cells by washing (Fig. 1). Adherent cells were cultured in DMEM/F-12 containing 10% FBS. More than 98% of these cells stained for microglial marker CD11b.
Isolation of human primary oligodendrocytes
The oligodendrocytes were obtained after shaking the remaining cell culture for 18 h at 190 rpm on eleventh day (Fig. 1). To purify oligodendrocytes from astrocytes and remaining microglia, the detached cell suspension was plated in non-coated tissue culture dishes (2 × 106 cells/100 mm) for 60 min at 37°C. This step was repeated twice to minimize the contamination [10]. The nonadhering cells were seeded into poly-d-lysine-coated culture plates in complete medium at 37°C with 5% CO2 in air. More than 97% of these cells stained for oligodendroglial marker GalC.
Isolation of human primary astrocytes
The attached cells remaining after removal of oligodendrocytes were primarily astrocytes. These cells were trypsinized and subcultured in complete media at 37°C with 5% CO2 in air to yield more viable and healthy cells [13, 17]. More than 98% of these cells obtained by this method were found to be positive for GFAP, a marker for astrocytes.
Phagocytosis assay
Human primary microglia were stimulated with 1 μg/ml LPS for 3 h. After 3 h of stimulation with LPS, latex beads (0.8-μm size, Sigma) were added to wells containing cultured cells at 1 μl beads/ml concentration. After 90 min of incubation, cultures were rinsed several times with PBS and fixed in 4% paraformaldehyde in 0.1 M phosphate buffer. After fixation, the phagocytosis of latex beads by microglia was examined under Nikon phase contrast microscope.
Immunostaining of GFAP, CD11b, MOG, MBP, CNPase, and MAP-2
Immunostaining was performed as described earlier [3, 10, 18]. Briefly, coverslips containing 200–300 cells/mm2 were fixed with 4% paraformaldehyde for 15 min, followed by treatment with cold ethanol (−20°C) for 5 min and two rinses in PBS. Samples were blocked with 3% BSA in PBS containing Tween 20 (PBST) for 30 min and incubated in PBST containing 1% BSA and rabbit anti-CD11b (1:50), mouse anti-GFAP (1:50), goat anti-MOG (1:50), rat or mouse anti-MBP (1:50), rabbit anti CNPase (1:50), goat anti-MAP-2 (1:70). After three washes in PBST (15 min each), slides were further incubated with Cy5, CY3, and CY2 (Jackson ImmunoResearch, West Grove, PA, USA). For negative controls, a set of culture slides was incubated under similar conditions without the primary antibodies. The samples were mounted and observed under a Bio-Rad (Hercules, CA, USA) MRC1024ES confocal laser-scanning microscope.
Semi-quantitative real-time polymerase chain reaction (RT-PCR) analysis
Total RNA was isolated from human oligodendrocytes and mouse cerebral tissues using RNA-Easy Qiagen kit following manufactures protocol. To remove any contaminating genomic DNA, total RNA was digested with DNase. Semi-quantitative RT-PCR was carried out as described earlier [6, 10] using oligo(dT)12–18 as primer and MMLV reverse transcriptase (Clontech, Heidelberg, Germany) in a 20-μl reaction mixture. The resulting cDNA was appropriately diluted, and diluted cDNA was amplified using Titanium Taq polymerase and the following primers. TNF-α: Sense, 5′-CTG AGT CGG TCA CCC TTC TCC AGC T-3′;antisense: 5′-CCC GAG TGA CAA GCC TGT AGC CCA T-3′;GAPDH: sense, 5′-GTT GTC ATT GTT GTG AGC CG -3;antisense, 5′-CAT CAC AGC CAT GAT GTT GC -3′; Amplified products were electrophoresed on a 1.8% agarose gels and visualized by ethidium bromide staining. Message for the GAPDH (glyceraldehyde-3-phosphate dehydrogenase) gene was used to ascertain that an equivalent amount of cDNA was synthesized from different samples.
Fragment end labeling of DNA
Fragmented DNA was detected in situ by the terminal deoxynucleotidyltransferase-mediated binding of 3′-OH ends of DNA fragments generated in response to either fibrillar Aβ1–42 or gp120 using a commercially available kit (TdT FragEL™) from Calbiochem as described earlier [8, 9]. Briefly, cover slips were treated with 20 μg/ml proteinase K for 15 min at room temperature and washed prior to terminal deoxynucleotidyltransferase staining.
Cell viability measurement
Mitochondrial activity was measured with the 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay (Sigma) as described earlier [8, 9].
Lactate dehydrogenase measurement
The activity of lactate dehydrogenase (LDH) was measured using the direct spectrophotometric assay using an assay kit from Sigma as described earlier [8, 9].
Statistics
Data are expressed as means ± SD-values. Statistical comparisons were made using Student's t test (The SAS system, Caly, NC, USA). Differences between means were considered significant when P < 0.05.
Results
Neuronal culture
After 1 day of plating, we added Ara C to suppress the growth of glial cells [16]. This Ara C-treated neuronal culture was fed with B27 media that contained vitamin, hormone, fatty acids, and antioxidant that specifically supported the growth of embryonic neurons [4]. The presence of antioxidant in the medium reduces oxidative damage to membranes, protein and DNA. Isolated neurons showed differentiated form and this neuronal morphology could be maintained over several weeks in culture. We used antibodies against MAP-2, characteristic marker of neurons, to immunostain our purified neuronal culture. It is clear from double-labeled immunofluorescence results in Fig. 2a that primary neuronal culture was almost devoid of CD11b-containing microglia and GalC-containing oligodendroglia. However, sometimes, we found one GFAP-positive astrocyte out of fifty MAP-2-positive neurons (Fig. 2a).
Fig. 2.
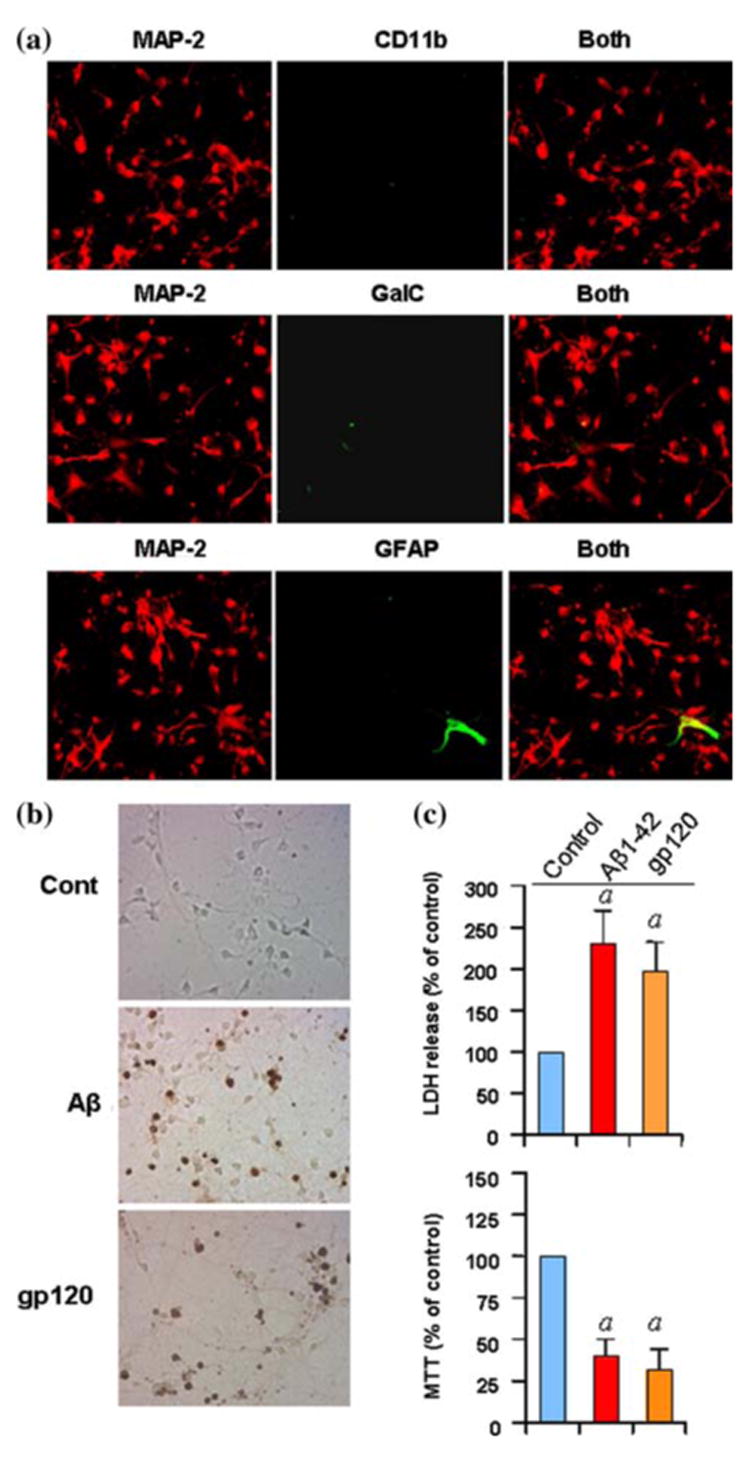
To examine the purity of neurons, cells were double-immunolabeled with MAP-2 and either CD11b, GalC or GFAP and observed under a confocal laser-scanning microscope (a). Human primary neurons were incubated with either 1 μM fibrillar Aβ peptides or 200pM of gp120 for 6 h in Neurobasal medium containing 2% B27-AO followed by TUNEL. Digital images were collected under bright-field setting using a ×40 objective (b). After 18 h of stimulation with either 1 μM fibrillar Aβ peptides or 200pM of gp120, cell viability was examined by the metabolism of MTT and the release of LDH. Values obtained from the control group served as 100%, and data obtained in other groups were calculated as percent of control accordingly (c). Results are mean ± SD of three different experiments. ap < 0.001 versus control
Next we examined if these neurons respond to any neurodegenerative insult. Therefore, these cells were challenged with fibrillar Aβ1-42 peptides (etiological reagent for AD) and gp120 (one of the etiological reagents for HIV-associated dementia) followed by monitoring of apoptosis and cell death. As expected, both Aβ1-42 peptides and gp120 induced apoptosis (Fig. 2b), increased LDH release and decreased MTT metabolism (Fig. 2c), thereby simulating the in vivo condition found in different neurodegenerative diseases.
Microglial culture
Following the method outlined in Fig. 1, we have been able to isolate highly purified microglia. We employed double-label immunofluorescence to check contamination by other cells. As evident in Fig. 3a, we did not detect other cells in this microglial preparation. Rarely did we observe GFAP-positive astrocyte in this preparation. Microglial activation is one of the hallmarks of various neurodegenerative disorders. During activation, microglia exhibit phagocytosis property, express various proinflammatory molecules, ramify, and produce excessive amount of CD11b. As seen in Fig. 3b, after LPS stimulation, human fetal microglia phagocytosed higher amount of latex beads per cell compared to unstimulated control. In response to LPS, this microglial preparation also expressed TNF-α mRNA (Fig. 3c) and exhibited large ameboid-like structure with higher expression of CD11b (Fig. 3d).
Fig. 3.
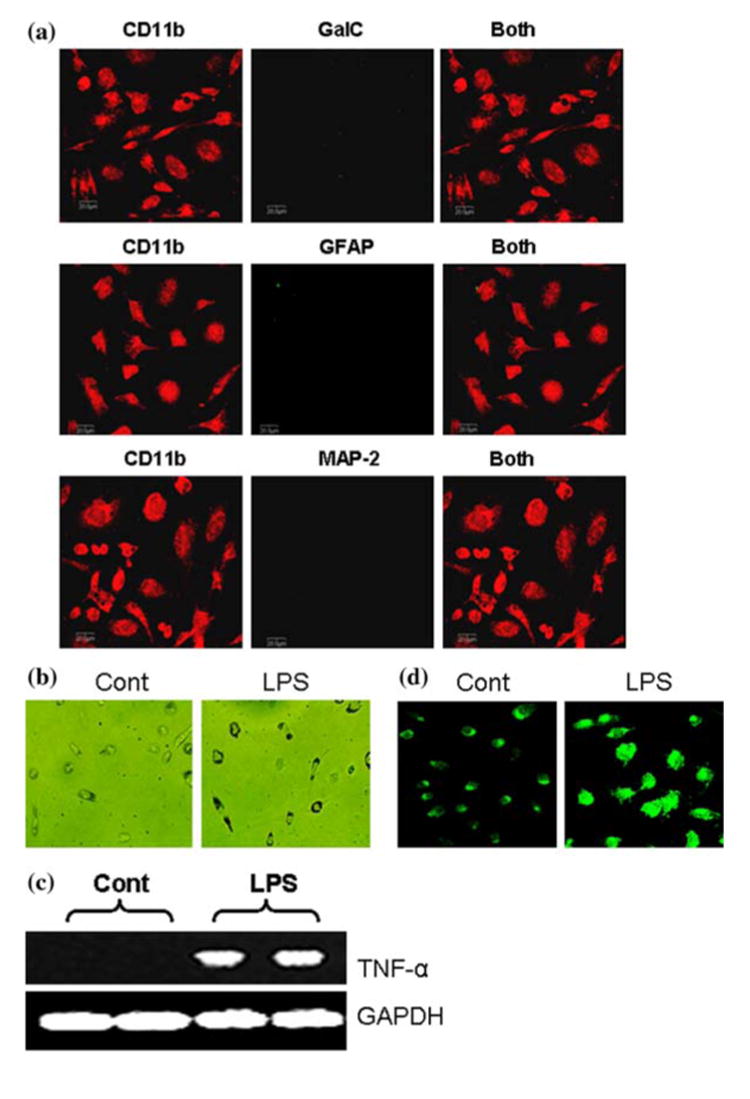
To examine the purity of microglia, cells were double-immunolabeled with CD11b and either GalC, GFAP or MAP-2 and observed under a confocal laser-scanning microscope (a). Microglial cells were either stimulated with LPS (1 μg/ml) in serum free condition or treated with serum free media alone and after 3 h cells were exposed to latex beads for 90 min at 37°C to monitor phagocytosis (b). Microglia were treated with LPS under serum-free condition. After 6 h of treatment, total RNA was analyzed for the expression of TNF-α by semi-quantitative RT-PCR as described under “Materials and Methods” (c). Microglia were treated with LPS and after 24 h of treatment, cells were immunostained with antibodies against CD11b (d). Results are representative of three separate experiments
Oligodendroglial culture
As mentioned under “Materials and Methods” and in the Fig. 1, mixed glia cultures were shaken on eleventh day of culture to obtain oligodendroglia. To purify oligodendroglia from astrocytes and microglia further, cells were allowed to adhere in uncoated plates for 2 h. By double-labeling immunofluorescence, we examined purity of these cells. These cells were positive for GalC (Fig. 4a), MBP, MOG, PLP, and CNPase [10]. However, we were unable to detect any CD11b-positive microglia and MAP-2-positive neurons in this oligodendroglial preparation (Fig. 4a). Rarely did we observe one or two GFAP-positive astrocytes in this preparation (Fig. 4a).
Fig. 4.
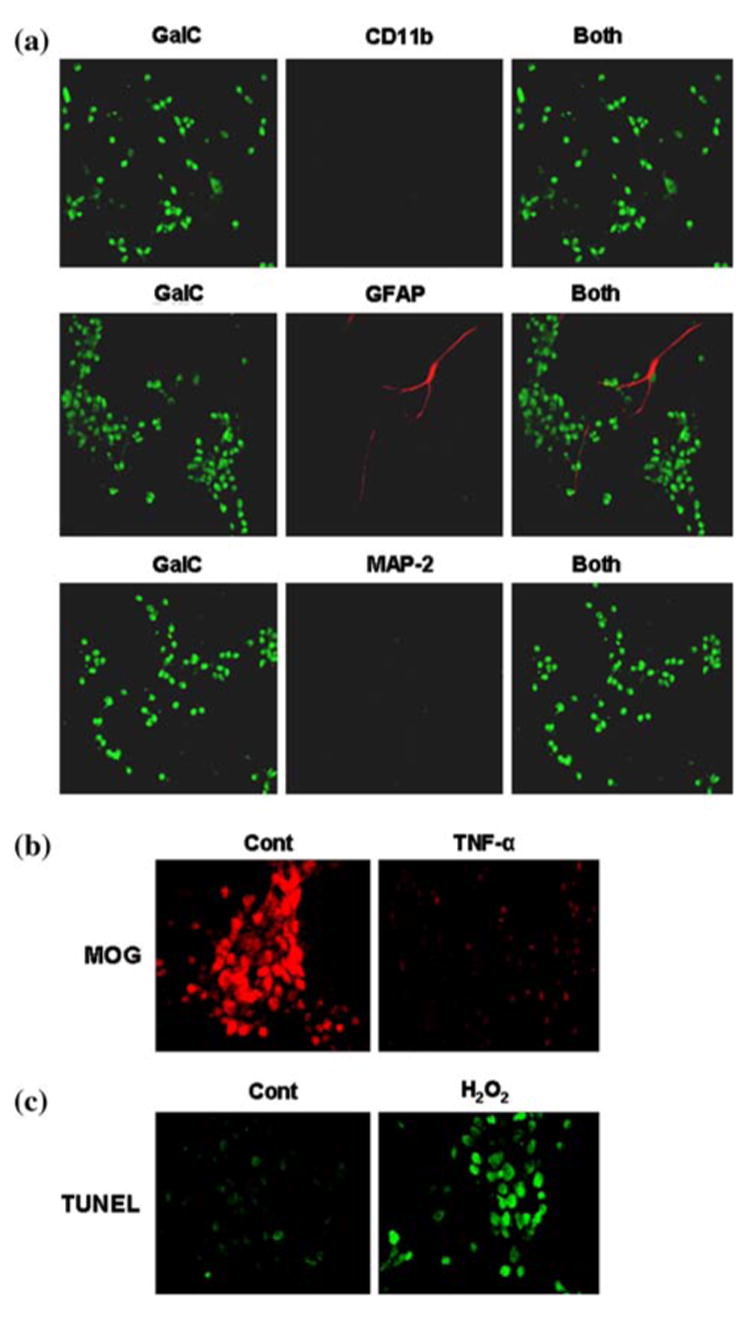
To examine the purity of oligodendrocytes, cells were double-immunolabeled with GalC and either CD11b, GFAP or MAP-2 and observed under a confocal laser-scanning microscope (a). Differentiated oligodendrocytes were treated with 30 ng/ml of TNF-α and after 24 h of treatment, cells were immunostained with antibodies against MOG (b). Human primary oligodendrocytes were incubated with 300 μM of H2O2 for 6 h in serum free media followed by TUNEL (c). Results are representative of three separate experiments
Next we examined if these oligodendroglia showed any responsiveness to proinflammatory cytokine. Control oligodendroglia expressed MOG abundantly (Fig. 4b). However, TNF-α markedly reduced the expression of MOG protein in human fetal oligodendroglia (Fig. 4b). Oxidative stress plays an important role in the loss of oligodendroglia in MS. Consistently, we observed that H2O2 markedly induced apoptosis in primary oligodendroglia (Fig. 4c). These results suggest that these highly purified oligodendroglia may be used to study the demyelination process observed in MS and other demyelinating disorders.
Astrocyte culture
Human fetal astrocytes obtained by this method showed typical astrocyte morphology and expressed the astrocytic marker GFAP and were found to be more than 98% pure (Fig. 5a). We did not see any GalC-positive oligodendroglia and MAP-2-positive neurons in this astroglial preparation (Fig. 5a). Rarely did we observe the presence of CD11b-positive microglia in this preparation. One of the hallmarks of various neurodegenerative disorders is the activation of astroglia. During activation, astroglia express higher amount of proinflammatory molecules and GFAP. We therefore, examined if this astroglial preparation respond to any inflammatory challenge by displaying increase in proinflammatory molecule and GFAP. Expectedly, bacterial LPS induced the expression of TNF-α (a proinflammatory cytokine) mRNA (Fig. 5b) and increased the expression of GFAP protein (Fig. 5c) in human fetal astroglia.
Fig. 5.
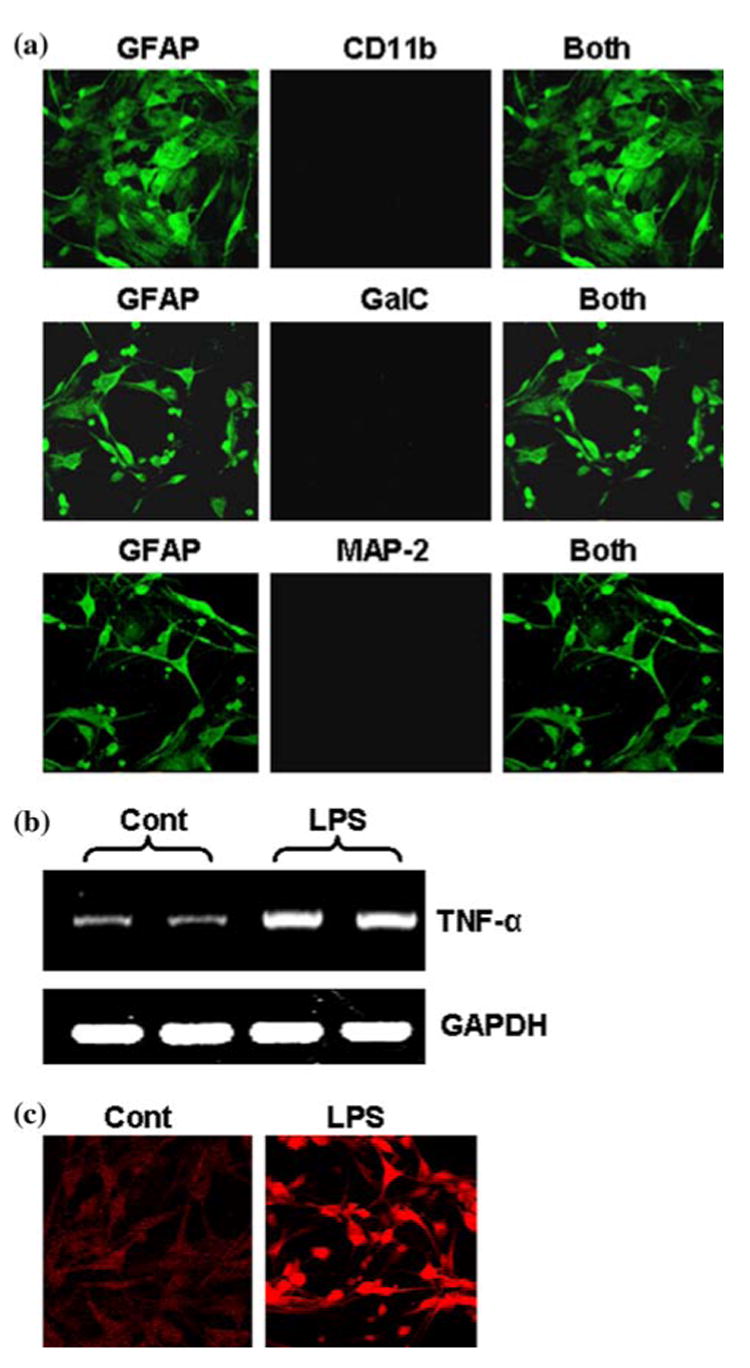
To examine the purity of astrocytes, cells were double-immunolabeled with GFAP and either CD11b, GalC or MAP-2 and observed under a confocal laser-scanning microscope (a). Astrocytes were treated with LPS under serum-free condition. After 6 h of treatment, total RNA was analyzed for the expression of TNF-α by semi-quantitative RT-PCR (b). Astrocytes were treated with 1 μg /ml of LPS and after 24 h of treatment, cells were immunostained with antibodies against GFAP (c). Results are representative of three separate experiments
Discussion
To characterize metabolic disturbances occurring during CNS pathologies, it is imperative to develop experimental condition mimicking the diseased condition at a cellular level. In the present study we have successfully developed a method that generates large quantities of highly purified and viable cultures of neurons, microglia, oligodendrocytes, and astrocytes from the same human fetal brain tissue. These culture systems offer several advantages over mixed primary cultures and single cell type primary culture. First, our highly purified culture will help us to interpret data in the absence of any interactions from other cell types as seen with mixed in vitro primary cultures. Second, the interactions between disease-causing agents and particular cells can be studied effectively with enriched cultures. For example, in AD and HIV-associated dementia (HAD), neurons undergo apoptosis [14]. We have observed that etiological reagents of AD and HAD induce apoptosis in human fetal neurons. Third, these enriched cultures will decrease the ambiguity of interpreting the cellular target of different drugs in contrast to mixed primary culture that often precludes identification of specific cellular targets. Fourth, these enriched populations will also help us in controlling the environmental condition of each cell type thereby allowing us to maintain a uniform condition in all the experiments and promoting a greater reproducibility of the data. Fifth, the interactions among different types of cells in the same environment can be studied using co-culture technique. Lastly, since all the cell types were obtained from the same human fetal tissue at the same stage of development, standardized interpretations of the data is possible. Although some protocols are available to isolate these cells [1, 2, 5, 7, 11], our protocol has a major advantage over the others in obtaining all four cell types from the same tissue preparation.
In summary, we have provided detailed methods of isolation and cultivation of highly purified neurons, astrocytes, microglia, and oligodendrocytes from the same human fetal brain tissue. These cells express their characteristic cell surface markers and perform their functions as they do under physiological and pathophysiological conditions. However, many diseases occur only in matured neurons and glial cells. Therefore, the in vitro situation of human fetal cells in culture may not truly resemble the in vivo condition observed during different pathological states. Nevertheless, our pure cell culture protocol will definitely provide investigators another option while exploring and modulating life and death signaling pathways at the cellular level.
Acknowledgments
The authors would like to thank Dr. You Zhou of the University of Nebraska at Lincoln for his help in microscopy.
This study was supported by grants from NIH (NS39940 and NS48923) and National Multiple Sclerosis Society (RG3422A1).
Contributor Information
Malabendu Jana, Department of Neurological Sciences, Rush University Medical Center, 1735 West Harrison St, Cohn Res Bldg, Suite 320, Chicago, IL 60612, USA.
Arundhati Jana, Department of Neurological Sciences, Rush University Medical Center, 1735 West Harrison St, Cohn Res Bldg, Suite 320, Chicago, IL 60612, USA.
Utpal Pal, Department of Veterinary Medicine, University of Maryland, College Park, MD 20742, USA.
Kalipada Pahan, Department of Neurological Sciences, Rush University Medical Center, 1735 West Harrison St, Cohn Res Bldg, Suite 320, Chicago, IL 60612, USA; Section of Neuroscience, Department of Oral Biology, University of Nebraska Medical Center College of Dentistry, Lincoln, NE 68583, USA.
References
- 1.Armstrong RC. Methods. 3. Vol. 16. San Diego, CA: 1998. Isolation and characterization of immature oligodendrocyte lineage cells; pp. 282–292. [DOI] [PubMed] [Google Scholar]
- 2.Barnea A, Roberts J. An improved method for dissociation and aggregate culture of human fetal brain cells in serum-free medium. Brain Res. 1999;4(2):156–164. doi: 10.1016/s1385-299x(99)00015-x. [DOI] [PubMed] [Google Scholar]
- 3.Brahmachari S, Fung YK, Pahan K. Induction of glial fibrillary acidic protein expression in astrocytes by nitric oxide. J Neurosci. 2006;26(18):4930–4939. doi: 10.1523/JNEUROSCI.5480-05.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Brewer GJ. Serum-free B27/neurobasal medium supports differentiated growth of neurons from the striatum, substantia nigra, septum, cerebral cortex, cerebellum, and dentate gyrus. J Neurosci Res. 1995;42(5):674–683. doi: 10.1002/jnr.490420510. [DOI] [PubMed] [Google Scholar]
- 5.Cheepsunthorn P, Radov L, Menzies S, Reid J, Connor JR. Characterization of a novel brain-derived microglial cell line isolated from neonatal rat brain. Glia. 2001;35(1):53–62. doi: 10.1002/glia.1070. [DOI] [PubMed] [Google Scholar]
- 6.Dasgupta S, Jana M, Zhou Y, Fung YK, Ghosh S, Pahan K. Antineuroinflammatory effect of NF-kappaB essential modifier-binding domain peptides in the adoptive transfer model of experimental allergic encephalomyelitis. J Immunol. 2004;173(2):1344–1354. doi: 10.4049/jimmunol.173.2.1344. [DOI] [PubMed] [Google Scholar]
- 7.Giulian D, Baker TJ. Characterization of ameboid microglia isolated from developing mammalian brain. J Neurosci. 1986;6(8):2163–2178. doi: 10.1523/JNEUROSCI.06-08-02163.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Jana A, Pahan K. Fibrillar amyloid-beta peptides kill human primary neurons via NADPH oxidase-mediated activation of neutral sphingomyelinase. Implications for Alzheimer's disease. J Biol Chem. 2004a;279(49):51451–51459. doi: 10.1074/jbc.M404635200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Jana A, Pahan K. Human immunodeficiency virus type 1 gp120 induces apoptosis in human primary neurons through redox-regulated activation of neutral sphingomyelinase. J Neurosci. 2004b;24(43):9531–9540. doi: 10.1523/JNEUROSCI.3085-04.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Jana M, Pahan K. Redox regulation of cytokine-mediated inhibition of myelin gene expression in human primary oligodendrocytes. Free radical biology & medicine. 2005;39(6):823–831. doi: 10.1016/j.freeradbiomed.2005.05.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Kay AR, Wong RK. Isolation of neurons suitable for patch-clamping from adult mammalian central nervous systems. J Neurosci Methods. 1986;16(3):227–238. doi: 10.1016/0165-0270(86)90040-3. [DOI] [PubMed] [Google Scholar]
- 12.Lee SC, Liu W, Brosnan CF, Dickson DW. Characterization of primary human fetal dissociated central nervous system cultures with an emphasis on microglia. Lab Invest J Tech Methods Pathol. 1992;67(4):465–476. [PubMed] [Google Scholar]
- 13.Liu X, Jana M, Dasgupta S, Koka S, He J, Wood C, Pahan K. Human immunodeficiency virus type 1 (HIV-1) tat induces nitric-oxide synthase in human astroglia. J Biol Chem. 2002;277(42):39312–39319. doi: 10.1074/jbc.M205107200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Mattson MP, Haughey NJ, Nath A. Cell death in HIV dementia. Cell Death Differ. 2005;12 1:893–904. doi: 10.1038/sj.cdd.4401577. [DOI] [PubMed] [Google Scholar]
- 15.McMillian MK, Thai L, Hong JS, O'Callaghan JP, Pennypacker KR. Brain injury in a dish: a model for reactive gliosis. Trends Neurosci. 1994;17(4):138–142. doi: 10.1016/0166-2236(94)90086-8. [DOI] [PubMed] [Google Scholar]
- 16.Oorschot DE, Jones DG. Non-neuronal cell proliferation in tissue culture: implications for axonal regeneration in the central nervous system. Brain Res. 1986;368(1):49–61. doi: 10.1016/0006-8993(86)91041-3. [DOI] [PubMed] [Google Scholar]
- 17.Pahan K, Jana M, Liu X, Taylor BS, Wood C, Fischer SM. Gemfibrozil, a lipid-lowering drug, inhibits the induction of nitric-oxide synthase in human astrocytes. J Biol Chem. 2002;277(48):45984–45991. doi: 10.1074/jbc.M200250200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Roy A, Fung YK, Liu X, Pahan K. Up-regulation of microglial CD11b expression by nitric oxide. J Biol Chem. 2006;281(21):14971–14980. doi: 10.1074/jbc.M600236200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Ryan LA, Peng H, Erichsen DA, Huang Y, Persidsky Y, Zhou Y, Gendelman HE, Zheng J. TNF-related apoptosis-inducing ligand mediates human neuronal apoptosis: links to HIV-1-associated dementia. J Neuroimmunol. 2004;148(1–2):127–139. doi: 10.1016/j.jneuroim.2003.11.019. [DOI] [PubMed] [Google Scholar]
- 20.Saha RN, Pahan K. Tumor necrosis factor-alpha at the crossroads of neuronal life and death during HIV-associated dementia. J Neurochem. 2003;86(5):1057–1071. doi: 10.1046/j.1471-4159.2003.01942.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Saha RN, Pahan K. Regulation of inducible nitric oxide synthase gene in glial cells. Antioxid Redox Signal. 2006;8(5–6):929–947. doi: 10.1089/ars.2006.8.929. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Vila M, Jackson-Lewis V, Guegan C, Wu DC, Teismann P, Choi DK, Tieu K, Przedborski S. The role of glial cells in Parkinson's disease. Curr Opin Neurol. 2001;14(4):483–489. doi: 10.1097/00019052-200108000-00009. [DOI] [PubMed] [Google Scholar]
- 23.Zhang Y, McLaughlin R, Goodyer C, LeBlanc A. Selective cytotoxicity of intracellular amyloid beta peptide 1-42 through p53 and bax in cultured primary human neurons. J Cell Biol. 2002;156(3):519–529. doi: 10.1083/jcb.200110119. [DOI] [PMC free article] [PubMed] [Google Scholar]


