Abstract
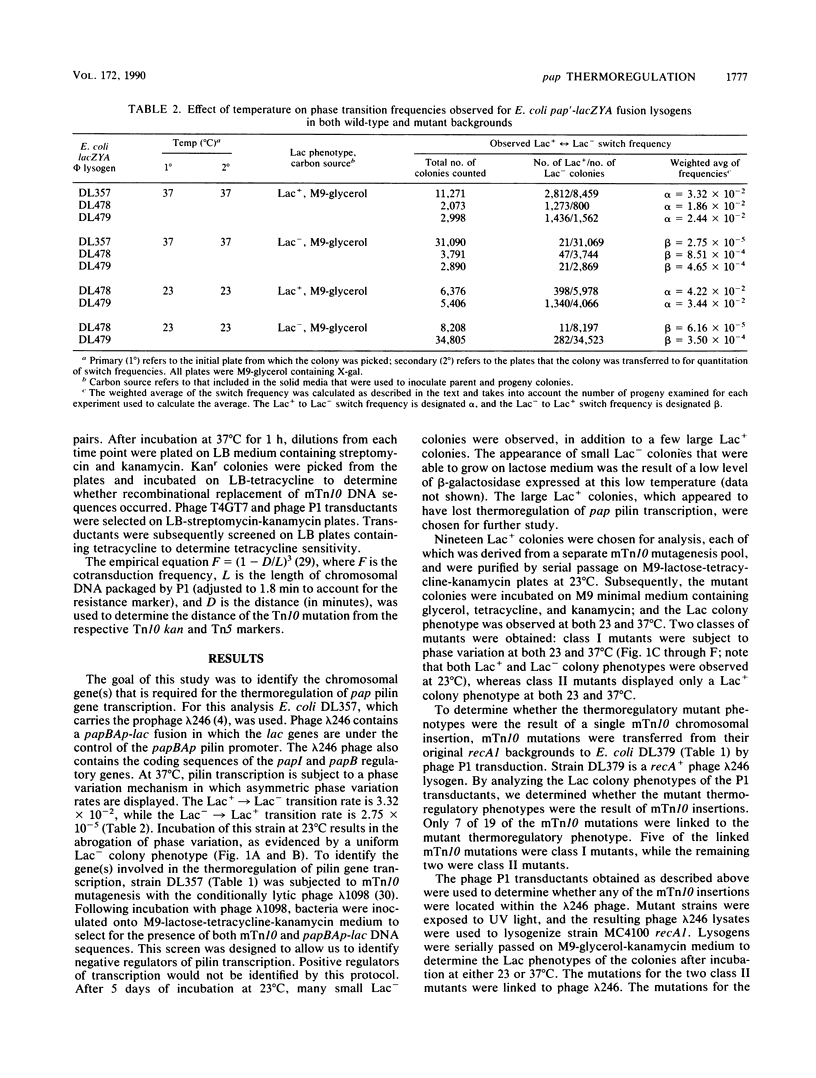
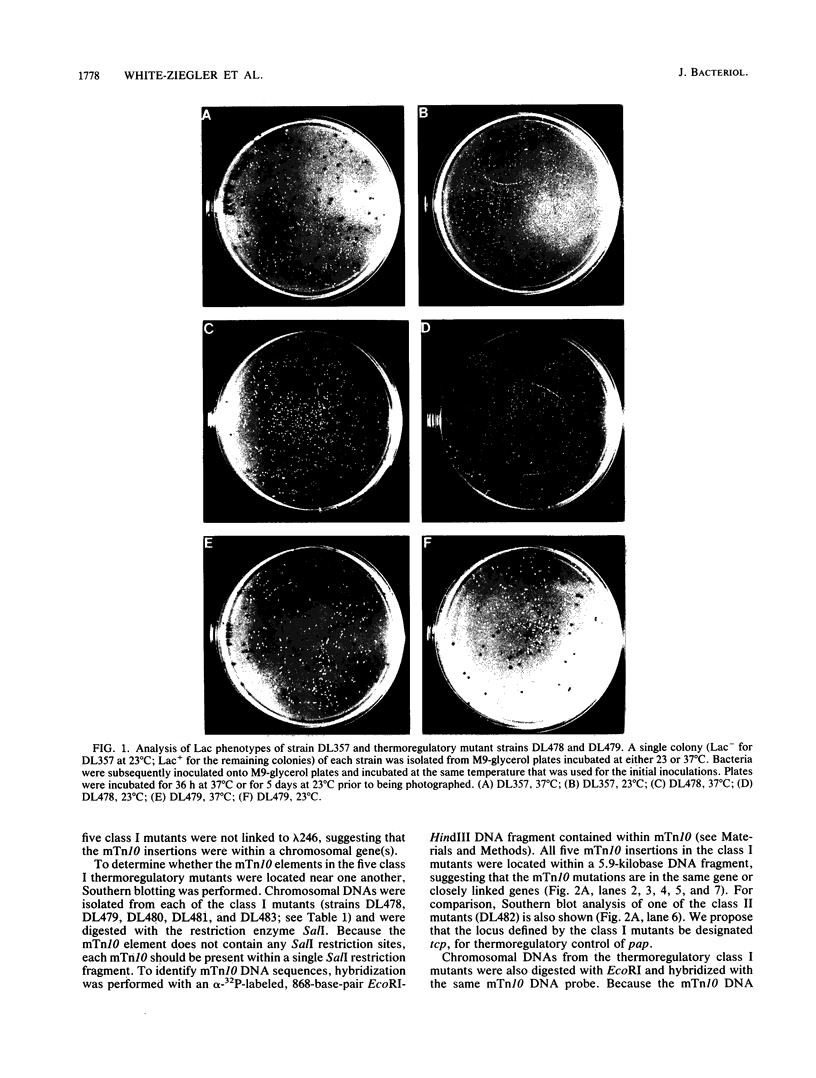
We previously showed, using a single-copy papBAp-lac fusion (previously designated papBA-lac), that pyelonephritis-associated pili (pap) pilin gene transcription is subject to both phase variation and thermoregulatory control mechanisms (L. B. Blyn, B. A. Braaten, C. A. White-Ziegler, D. H. Rolfson, and D. A. Low, EMBO J. 8:613-620, 1989). At 37 degrees C, Escherichia coli strains carrying the papBAp-lac fusion displayed both Lac+ and Lac- colony phenotypes. In contrast, at 23 degrees C, colonies displayed a uniform Lac- phenotype, suggesting that pilin was not transcribed at this temperature. In this study, a strain carrying the papBAp-lac fusion was subjected to mini-Tn10 (mTn10) mutagenesis to isolate mutants that could initiate transcription of pilin at the nonpermissive temperature. Two classes of thermoregulatory mutants were identified in which the mTn10 mutation was linked to the mutant phenotype. Class I mutants displayed a phase variation phenotype at both 37 degrees C and 23 degrees C, whereas class II mutants displayed a uniform Lac+ colony phenotype at both temperatures. Preliminary analysis of these mutants showed that the mTn10 insertions in the class I mutants were chromosomally located, whereas the mTn10 insertions in the class II mutants were located within the papBAp-lac fusion phage. Southern blot analysis of the class I mutants demonstrated that mTn10 was present in the same 5.9-kilobase SalI DNA fragment in each mutant. Two of the class I mTn10 mutations were mapped to approximately 23.4 min on the E. coli K-12 chromosome. The locus defined by the class I mTn10 mutations was designated tcp, for thermoregulatory control of pap. Analysis of phase transition rates of the class I mutants showed that the phase-off (Lac-)----phase-on (Lac+) transition rates were higher than those observed with the nonmutant E. coli strain.
Full text
PDF





Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Aricó B., Miller J. F., Roy C., Stibitz S., Monack D., Falkow S., Gross R., Rappuoli R. Sequences required for expression of Bordetella pertussis virulence factors share homology with prokaryotic signal transduction proteins. Proc Natl Acad Sci U S A. 1989 Sep;86(17):6671–6675. doi: 10.1073/pnas.86.17.6671. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Blyn L. B., Braaten B. A., White-Ziegler C. A., Rolfson D. H., Low D. A. Phase-variation of pyelonephritis-associated pili in Escherichia coli: evidence for transcriptional regulation. EMBO J. 1989 Feb;8(2):613–620. doi: 10.1002/j.1460-2075.1989.tb03416.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bortolussi R., Ferrieri P., Quie P. G. Influence of growth temperature of Escherichia coli on K1 capsular antigen production and resistance to opsonization. Infect Immun. 1983 Mar;39(3):1136–1141. doi: 10.1128/iai.39.3.1136-1141.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Båga M., Göransson M., Normark S., Uhlin B. E. Processed mRNA with differential stability in the regulation of E. coli pilin gene expression. Cell. 1988 Jan 29;52(2):197–206. doi: 10.1016/0092-8674(88)90508-9. [DOI] [PubMed] [Google Scholar]
- Casadaban M. J., Cohen S. N. Analysis of gene control signals by DNA fusion and cloning in Escherichia coli. J Mol Biol. 1980 Apr;138(2):179–207. doi: 10.1016/0022-2836(80)90283-1. [DOI] [PubMed] [Google Scholar]
- Casadaban M. J. Transposition and fusion of the lac genes to selected promoters in Escherichia coli using bacteriophage lambda and Mu. J Mol Biol. 1976 Jul 5;104(3):541–555. doi: 10.1016/0022-2836(76)90119-4. [DOI] [PubMed] [Google Scholar]
- Cornelis G., Sluiters C., de Rouvroit C. L., Michiels T. Homology between virF, the transcriptional activator of the Yersinia virulence regulon, and AraC, the Escherichia coli arabinose operon regulator. J Bacteriol. 1989 Jan;171(1):254–262. doi: 10.1128/jb.171.1.254-262.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Evans D. G., Evans D. J., Jr, Tjoa W. Hemagglutination of human group A erythrocytes by enterotoxigenic Escherichia coli isolated from adults with diarrhea: correlation with colonization factor. Infect Immun. 1977 Nov;18(2):330–337. doi: 10.1128/iai.18.2.330-337.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guinée P. A., Jansen W. H. Behavior of Escherichia coli K antigens K88ab, K88ac, and K88ad in immunoelectrophoresis, double diffusion, and hemagglutination. Infect Immun. 1979 Mar;23(3):700–705. doi: 10.1128/iai.23.3.700-705.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Göransson M., Forsman K., Uhlin B. E. Regulatory genes in the thermoregulation of Escherichia coli pili gene transcription. Genes Dev. 1989 Jan;3(1):123–130. doi: 10.1101/gad.3.1.123. [DOI] [PubMed] [Google Scholar]
- Hromockyj A. E., Maurelli A. T. Identification of an Escherichia coli gene homologous to virR, a regulator of Shigella virulence. J Bacteriol. 1989 May;171(5):2879–2881. doi: 10.1128/jb.171.5.2879-2881.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Idigbe E. O., Parton R., Wardlaw A. C. Rapidity of antigenic modulation of Bordetella pertussis in modified Hornibrook medium. J Med Microbiol. 1981 Nov;14(4):409–418. doi: 10.1099/00222615-14-4-409. [DOI] [PubMed] [Google Scholar]
- Koomey J. M., Gill R. E., Falkow S. Genetic and biochemical analysis of gonococcal IgA1 protease: cloning in Escherichia coli and construction of mutants of gonococci that fail to produce the activity. Proc Natl Acad Sci U S A. 1982 Dec;79(24):7881–7885. doi: 10.1073/pnas.79.24.7881. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Low D., Robinson E. N., Jr, McGee Z. A., Falkow S. The frequency of expression of pyelonephritis-associated pili is under regulatory control. Mol Microbiol. 1987 Nov;1(3):335–346. doi: 10.1111/j.1365-2958.1987.tb01940.x. [DOI] [PubMed] [Google Scholar]
- Maurelli A. T., Blackmon B., Curtiss R., 3rd Temperature-dependent expression of virulence genes in Shigella species. Infect Immun. 1984 Jan;43(1):195–201. doi: 10.1128/iai.43.1.195-201.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maurelli A. T., Sansonetti P. J. Identification of a chromosomal gene controlling temperature-regulated expression of Shigella virulence. Proc Natl Acad Sci U S A. 1988 Apr;85(8):2820–2824. doi: 10.1073/pnas.85.8.2820. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Melton A. R., Weiss A. A. Environmental regulation of expression of virulence determinants in Bordetella pertussis. J Bacteriol. 1989 Nov;171(11):6206–6212. doi: 10.1128/jb.171.11.6206-6212.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nagy L. K., Mackenzie T., Pickard D. J., Dougan G. Effects of immune colostrum on the expression of a K88 plasmid encoded determinant: role of plasmid stability and influence of phenotypic expression of K88 fimbriae. J Gen Microbiol. 1986 Sep;132(9):2497–2503. doi: 10.1099/00221287-132-9-2497. [DOI] [PubMed] [Google Scholar]
- Norgren M., Normark S., Lark D., O'Hanley P., Schoolnik G., Falkow S., Svanborg-Edén C., Båga M., Uhlin B. E. Mutations in E coli cistrons affecting adhesion to human cells do not abolish Pap pili fiber formation. EMBO J. 1984 May;3(5):1159–1165. doi: 10.1002/j.1460-2075.1984.tb01945.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Normark S., Lark D., Hull R., Norgren M., Båga M., O'Hanley P., Schoolnik G., Falkow S. Genetics of digalactoside-binding adhesin from a uropathogenic Escherichia coli strain. Infect Immun. 1983 Sep;41(3):942–949. doi: 10.1128/iai.41.3.942-949.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- O'Hanley P., Low D., Romero I., Lark D., Vosti K., Falkow S., Schoolnik G. Gal-Gal binding and hemolysin phenotypes and genotypes associated with uropathogenic Escherichia coli. N Engl J Med. 1985 Aug 15;313(7):414–420. doi: 10.1056/NEJM198508153130704. [DOI] [PubMed] [Google Scholar]
- Portnoy D. A., Wolf-Watz H., Bolin I., Beeder A. B., Falkow S. Characterization of common virulence plasmids in Yersinia species and their role in the expression of outer membrane proteins. Infect Immun. 1984 Jan;43(1):108–114. doi: 10.1128/iai.43.1.108-114.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roosendaal B., van Bergen en Henegouwen P. M., de Graaf F. K. Subcellular localization of K99 fimbrial subunits and effect of temperature on subunit synthesis and assembly. J Bacteriol. 1986 Mar;165(3):1029–1032. doi: 10.1128/jb.165.3.1029-1032.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roy C. R., Miller J. F., Falkow S. The bvgA gene of Bordetella pertussis encodes a transcriptional activator required for coordinate regulation of several virulence genes. J Bacteriol. 1989 Nov;171(11):6338–6344. doi: 10.1128/jb.171.11.6338-6344.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Singer M., Baker T. A., Schnitzler G., Deischel S. M., Goel M., Dove W., Jaacks K. J., Grossman A. D., Erickson J. W., Gross C. A. A collection of strains containing genetically linked alternating antibiotic resistance elements for genetic mapping of Escherichia coli. Microbiol Rev. 1989 Mar;53(1):1–24. doi: 10.1128/mr.53.1.1-24.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Way J. C., Davis M. A., Morisato D., Roberts D. E., Kleckner N. New Tn10 derivatives for transposon mutagenesis and for construction of lacZ operon fusions by transposition. Gene. 1984 Dec;32(3):369–379. doi: 10.1016/0378-1119(84)90012-x. [DOI] [PubMed] [Google Scholar]
- Young K. K., Edlin G. Physical and genetical analysis of bacteriophage T4 generalized transduction. Mol Gen Genet. 1983;192(1-2):241–246. doi: 10.1007/BF00327673. [DOI] [PubMed] [Google Scholar]