Abstract
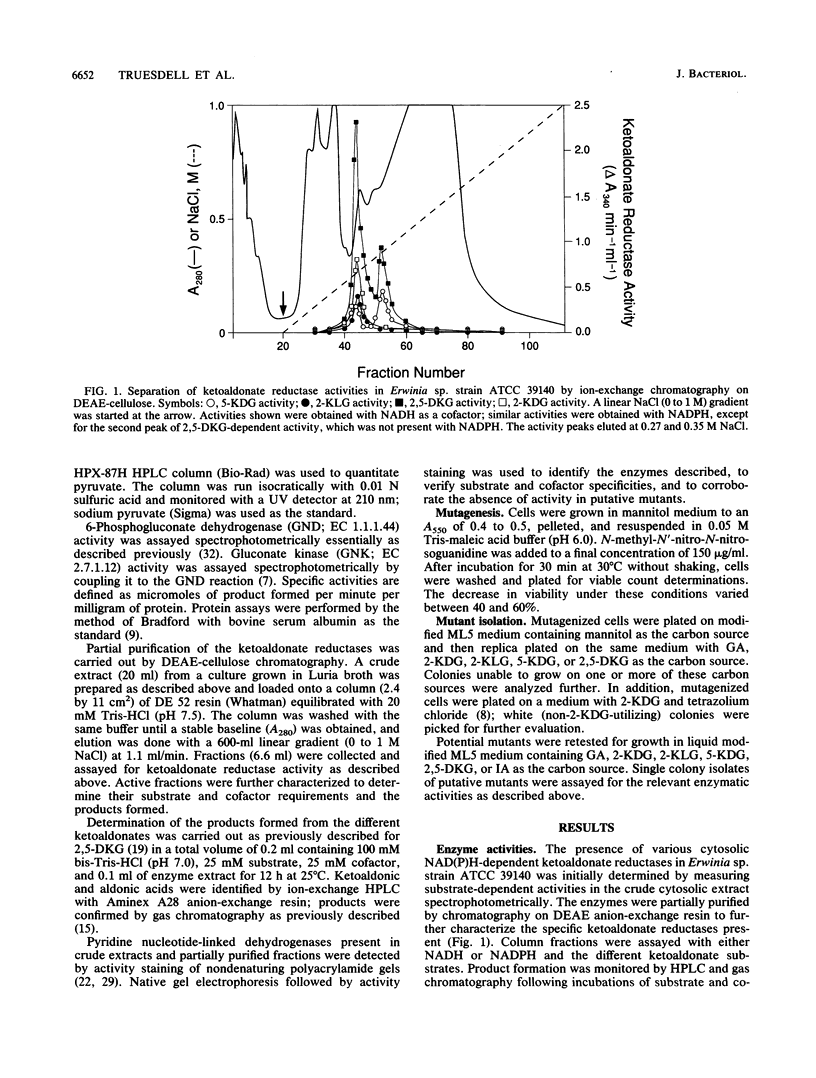
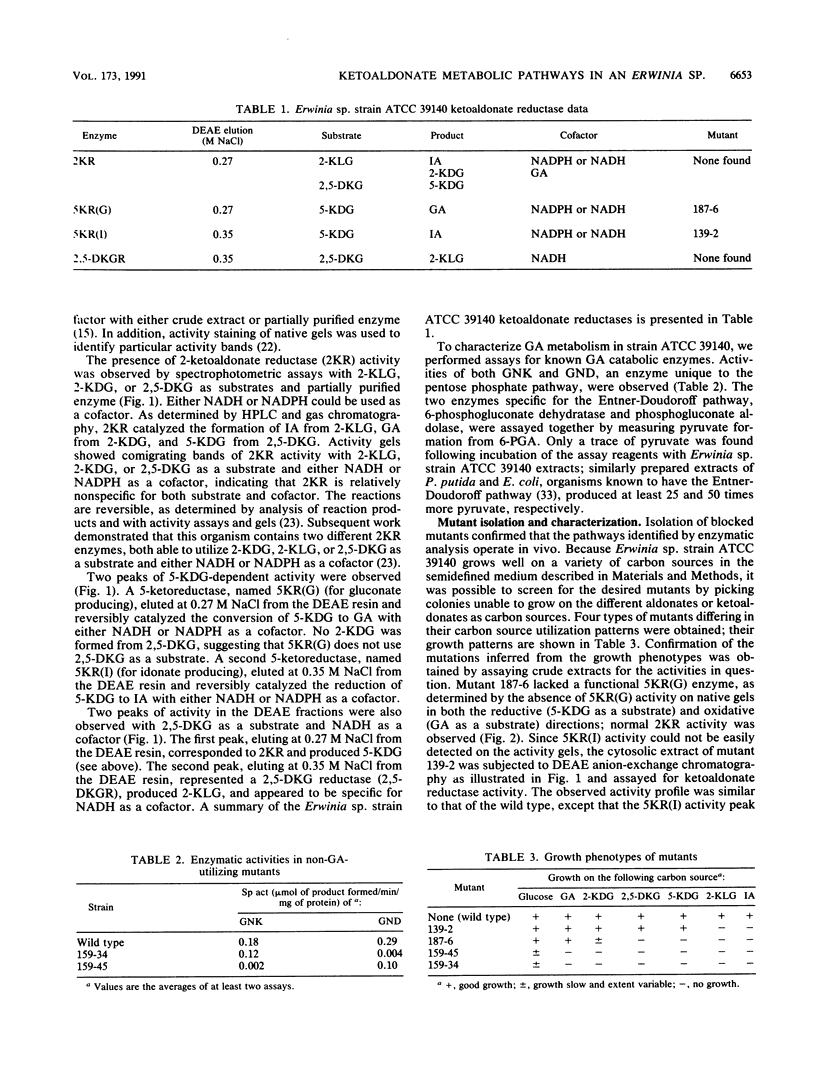
The pathways involved in the metabolism of ketoaldonic acids by Erwinia sp. strain ATCC 39140 have been investigated by use of a combination of enzyme assays and isolation of bacterial mutants. The catabolism of 2,5-diketo-D-gluconate (2,5-DKG) to gluconate can proceed by two separate NAD(P)H-dependent pathways. The first pathway involves the direct reduction of 2,5-DKG to 5-keto-D-gluconate, which is then reduced to gluconate. The second pathway involves the consecutive reduction of 2,5-DKG to 2-keto-L-gulonate and L-idonic acid, which is then oxidized to 5-keto-D-gluconate, which is then reduced to gluconate. Gluconate, which can also be produced by the NAD(P)H-dependent reduction of 2-keto-D-gluconate, is phosphorylated to 6-phosphogluconate and further metabolized through the pentose phosphate pathway. No evidence was found for the existence of the Entner-Doudoroff pathway in this strain.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Allenza P., Lessie T. G. Pseudomonas cepacia mutants blocked in the Entner-Doudoroff pathway. J Bacteriol. 1982 Jun;150(3):1340–1347. doi: 10.1128/jb.150.3.1340-1347.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Anderson S., Marks C. B., Lazarus R., Miller J., Stafford K., Seymour J., Light D., Rastetter W., Estell D. Production of 2-Keto-L-Gulonate, an Intermediate in L-Ascorbate Synthesis, by a Genetically Modified Erwinia herbicola. Science. 1985 Oct 11;230(4722):144–149. doi: 10.1126/science.230.4722.144. [DOI] [PubMed] [Google Scholar]
- Bochner B. R., Savageau M. A. Generalized indicator plate for genetic, metabolic, and taxonomic studies with microorganisms. Appl Environ Microbiol. 1977 Feb;33(2):434–444. doi: 10.1128/aem.33.2.434-444.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bradford M. M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem. 1976 May 7;72:248–254. doi: 10.1016/0003-2697(76)90527-3. [DOI] [PubMed] [Google Scholar]
- DE LEY J., STOUTHAMER A. J. The mechanism and localization of hexonate metabolism in Acetobacter suboxydans and Acetobacter melanogenum. Biochim Biophys Acta. 1959 Jul;34:171–183. doi: 10.1016/0006-3002(59)90245-8. [DOI] [PubMed] [Google Scholar]
- DE LEY J., VANDAMME J. The metabolism of sodium 2-keto-D-gluconate by micro-organisms. J Gen Microbiol. 1955 Apr;12(2):162–171. doi: 10.1099/00221287-12-2-162. [DOI] [PubMed] [Google Scholar]
- Grindley J. F., Payton M. A., van de Pol H., Hardy K. G. Conversion of Glucose to 2-Keto-l-Gulonate, an Intermediate in l-Ascorbate Synthesis, by a Recombinant Strain of Erwinia citreus. Appl Environ Microbiol. 1988 Jul;54(7):1770–1775. doi: 10.1128/aem.54.7.1770-1775.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lazarus R. A., Seymour J. L. Determination of 2-keto-L-gulonic and other ketoaldonic and aldonic acids produced by ketogenic bacterial fermentation. Anal Biochem. 1986 Sep;157(2):360–366. doi: 10.1016/0003-2697(86)90638-x. [DOI] [PubMed] [Google Scholar]
- Lessie T. G., Phibbs P. V., Jr Alternative pathways of carbohydrate utilization in pseudomonads. Annu Rev Microbiol. 1984;38:359–388. doi: 10.1146/annurev.mi.38.100184.002043. [DOI] [PubMed] [Google Scholar]
- Miller J. V., Estell D. A., Lazarus R. A. Purification and characterization of 2,5-diketo-D-gluconate reductase from Corynebacterium sp. J Biol Chem. 1987 Jul 5;262(19):9016–9020. [PubMed] [Google Scholar]
- Roberts B. K., Midgley M., Dawes E. A. The metabolism of 2-oxogluconate by Pseudomonas aeruginosa. J Gen Microbiol. 1973 Oct;78(2):319–329. doi: 10.1099/00221287-78-2-319. [DOI] [PubMed] [Google Scholar]
- Seymour J. L., Lazarus R. A. Native gel activity stain and preparative electrophoretic method for the detection and purification of pyridine nucleotide-linked dehydrogenases. Anal Biochem. 1989 May 1;178(2):243–247. doi: 10.1016/0003-2697(89)90632-5. [DOI] [PubMed] [Google Scholar]
- Susor W. A., Penhoet E., Rutter W. J. Fructose-diphosphate aldolase, pyruvate kinase, and pyridine nucleotide-linked activities after electrophoresis. Methods Enzymol. 1975;41:66–73. doi: 10.1016/s0076-6879(75)41017-5. [DOI] [PubMed] [Google Scholar]
- Veronese F. M., Boccù E., Fontana A. 6-Phosphogluconate dehydrogenase from Bacillus stearothermophilus. Methods Enzymol. 1982;89(Pt 500):282–291. doi: 10.1016/s0076-6879(82)89051-4. [DOI] [PubMed] [Google Scholar]
- Vicente M., Cánovas J. L. Glucolysis in Pseudomonas putida: physiological role of alternative routes from the analysis of defective mutants. J Bacteriol. 1973 Nov;116(2):908–914. doi: 10.1128/jb.116.2.908-914.1973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Whiting P. H., Midgley M., Dawes E. A. The role of glucose limitation in the regulation of the transport of glucose, gluconate and 2-oxogluconate, and of glucose metabolism in Pseudomonas aeruginosa. J Gen Microbiol. 1976 Feb;92(2):304–310. doi: 10.1099/00221287-92-2-304. [DOI] [PubMed] [Google Scholar]