Abstract
Background
Sublinical myocardial dysfunction occurs in a significant number of patients with type 2 diabetes. Assessment of ventricular long‐axis function by measuring mitral annular velocities using tissue Doppler echocardiography (TDE) is thought to provide a more sensitive index of systolic and diastolic function. We hypothesised that augmentation of left ventricular (LV) longitudinal contraction and relaxation during exercise would be blunted in patients with type 2 diabetes.
Methods
Mitral annular systolic (S′) and early diastolic (E′) velocities were measured at rest and during supine bicycle exercise (25 W, 3 min increments) in 53 patients (27 male, mean age 53±14 years) with type 2 diabetes and 53 subjects with age and gender‐matched control. None had echocardiographic evidence of resting or inducible myocardial ischaemia.
Results
There were no significant differences in mitral inflow velocities at rest between the two groups. E′ and S′ at rest were also similar between the groups. However, S′ (7.1±1.3 vs 8.3±1.8 cm/s at 25 W, p = 0.0021; 8.1±1.5 vs 9.1±2.0 cm/s at 50 W, p = 0.026) and E′ (8.5±2.3 vs 9.9±3.1 cm/s at 25 W, p = 0.054; 9.1±2.1 vs 10.9±2.5 cm/s at 50 W, p = 0.0093) during exercise were significantly lower in patients with diabetes compared with controls. Longitudinal systolic and diastolic function reserve indices were significantly lower in patients with diabetes compared with that of controls (systolic index, 0.6±0.70 vs 1.2±1.5 cm/s at 25 W, p = 0.029; 1.2±1.2 vs 2.1±1.6 cm/s at 50 W, p = 0.009; diastolic index, 1.9±1.2 vs 2.5±2.2 cm/s at 25 W, p = 0.07; 2.3±1.3 vs 3.2±2.2 cm/s at 50 W, p = 0.031).
Conclusion
In conclusion, unlike resting mitral inflow and annular velocities, changes of systolic and diastolic velocities of the mitral annulus during exercise were significantly reduced in patients with type 2 diabetes compared with the control group. The assessment of LV longitudinal functional reserve with exercise using TDE appears to be helpful in identifying early myocardial dysfunction in patients with type 2 diabetes.
Cardiovascular disease is the most common cause of death in patients with type 2 diabetes.1 Patients with diabetes are more likely to develop heart failure, and their outcome with heart failure is worse than that of patients without diabetes. The spectrum of diabetic heart disease involves a progression from the normal heart, to subclinical left ventricular (LV) diastolic and systolic dysfunction, followed by clinically overt symptomatic heart failure. Notably, subclinical LV dysfunction is common in patients with diabetes,2 and the detection of subclinical LV dysfunction in these patients may provide an approach for identifying high‐risk individuals who may benefit from earlier and more active intervention to prevent heart failure.3 Although overt LV diastolic and systolic dysfunction can be readily identified by conventional diagnostic techniques including echocardiography, the initial stage of myocardial dysfunction may be concealed by various compensatory mechanisms.4 Previous studies using sophisticated echocardiographic techniques such as tissue Doppler, strain, strain rate and ultrasonic tissue characterisation have shown abnormal myocardial function in patients with diabetes but without overt heart disease.2,5,6,7,8,9,10 However, all of these observations were made in the resting state or non‐physiological inotropic stimulation. A useful and physiological technique for evaluating ventricular performance involves the measurement of the circulatory changes that occur during exercise. In patients with diastolic dysfunction, already impaired myocardial relaxation does not augment as much as seen in normal individuals during exercise.11,12,13 Nagueh et al showed in a canine model that E′ increases with increased transmitral gradient in subjects with normal myocardial relaxation, whereas it remained unchanged in subjects with diastolic dysfunction.14 A similar result has been shown in human beings.15 However, the response of mitral annular systolic and diastolic velocities to exercise in patients with type 2 diabetes has not been explored previously. We hypothesised that augmentation of LV longitudinal contraction and relaxation during exercise would be blunted in patients with type 2 diabetes. Therefore, the purpose of this study was to evaluate resting LV longitudinal function and functional reserve during exercise in patients with type 2 diabetes using mitral annular velocities by tissue Doppler echocardiography (TDE).
Methods
Study population
Mitral inflow and septal mitral annular velocities were measured at rest and during graded supine bicycle exercise (25 W, 3‐min increments) in 53 patients (27 male, mean age 53±14 years) with type 2 diabetes and 53 subjects with age‐and gender‐matched control. The patients with diabetes had no symptoms or signs of heart disease, were in sinus rhythm and had a normal resting 12‐lead ECG. Exclusion criteria were significant atrial or ventricular arrhythmia, significant valvular diseases (⩾moderate severity), significant coronary artery disease, prior history of myocardial infarction, depressed LV systolic function (ejection fraction <50%, or any regional wall motion abnormality), pericardial disease and unable to exercise. The control subjects were considered to have a low probability of coronary artery disease on clinical grounds and had a normal exercise stress echocardiogram. Since this study aims at examining diabetes as an independent disease contributing preclinical myocardial dysfunction, patients who have coexisting hypertension were excluded. Study approval was obtained from the Internal Review Board of Yonsei University College of Medicine.
Two‐dimensional and exercise Doppler echocardiography (diastolic stress echocardiography)16
Standard two‐dimensional measurements (left ventricular diastolic and systolic dimensions, ventricular septum and posterior wall thickness, left atrial volume, left ventricular outflow tract) were obtained with the patient in the left lateral position. LV ejection fraction (EF) was calculated by the modified Quinones method.17 After obtaining the rest images from the standard parasternal and apical views, a multistage supine bicycle exercise testing was performed with a variable load bicycle ergometer (Medical Positioning, Kansas City, MO). Patients pedalled at constant speed beginning at a workload of 25 W with an increment of 25 W every 3 min. Echocardiography was performed using a GE Vingmed System 7 ultrasound system with 2.5‐MHz transducer during rest, each stage of exercise, and recovery in the sequence described as follows. From the apical window, a 1–2‐mm pulsed Doppler sample volume was placed at the mitral valve tip, and mitral flow velocities from 5 to 10 cardiac cycles were recorded. The mitral inflow velocities were traced, and the following variables were obtained: peak velocity of early (E) and late (A) filling, and deceleration time of the E wave velocity. Tricuspid regurgitant jet velocity was also obtained to estimate pulmonary artery systolic pressure using continuous‐wave Doppler, if measurable. Mitral annular velocity was measured by TDE using the pulsed‐wave Doppler mode. The filter was set to exclude high‐frequency signals, and the Nyquist limit was adjusted to a range of 15–20 cm/s. Gain and sample volume were minimised to allow for a clear tissue signal with minimal background noise. Early diastolic (E′) and systolic (S′) velocities of the mitral annulus were measured from the apical 4‐chamber view with a 2–5‐mm sample volume placed at the septal corner of the mitral annulus. These measurements were performed at baseline, at each stage of exercise, and recovery in the same sequence. All data were stored digitally, and measurements were made at completion of each study. Two‐dimensional echocardiographic images from apical views at rest and during peak exercise were acquired, digitised, recorded and analysed. Both digitised and videotaped images were used for the wall motion analysis.
LV diastolic and systolic longitudinal function reserve index were calculated using a new formula as follows. The underlying concept for the design of new index was to express the change of E′ or S′ with exercise relative to E′ or S′ at baseline:
where ΔE′ is the change of E′ from baseline to 25 W or 50 W of exercise, and E′base is early diastolic mitral annular velocity at rest.
where ΔS′ is the change of S′ from baseline to 25 W or 50 W of exercise, and S′base is systolic mitral annular velocity at rest.
Statistical analysis
Continuous variables were summarised as a mean ± standard deviation. Categorical variables were summarised as a percentage of the group total. Differences in Doppler indices between rest and peak exercise were compared with repeated measures ANOVA. Statistical significance was defined as <0.05.
Results
Table 1 shows the clinical characteristics of patients with diabetes. Patients with diabetes had a mean duration from the diagnosis of disease of 12.6±8.4 years (range, 2–31 years). Sixteen patients were on insulin, 17 on sulphonylureas, 13 on metformin, and 7 on thiazolidinediones. Ten (19%) had mild retinopathy, 10 (19%) had mild peripheral neuropathy, and 2 (4%) had renal impairment (creatinine >2.0 mg/dl). The mean HbA1c level in the patients with diabetes was 8.3±1.4% (range, 6–11%). Forty‐three patients had no albuminuria; 5 with microalbuminuria; and 5 with macroalbuminuria. Exercise duration was significantly shorter in patients with diabetes compared with that of the control (510±192 vs 593±193 s, p = 0.0296).
Table 1 Clinical characteristics of patients with diabetes.
| Duration of diabetes (years) | 12.6±8.4 (2–31) |
| Renal impairment | 2 |
| Retinopathy | 10 |
| Peripheral neuropathy | 10 |
| HbA1c (%) | 8.3±1.4 (6–11) |
| Insulin | 16 |
| Metformin | 13 |
| Sulfonylurea | 17 |
| Thiazolidinediones | 7 |
Echocardiographic findings
There were no significant differences in LV end‐diastolic, end‐systolic dimensions, wall thickness, left atrial volume index and EF between the groups. There were no significant differences in mitral inflow velocities (E, A, E/A, DT) between the two groups (table 2). TR velocity was also similar between the groups.
Table 2 Baseline echocardiographic findings.
| Diabetes (n = 53) | Control (n = 53) | p Value | |
|---|---|---|---|
| LVEDD (mm) | 49±4 | 49±5 | 0.62 |
| LVESD (mm) | 32±4 | 32±5 | 0.69 |
| Ejection fraction (%) | 66±7 | 67±7 | 0.64 |
| IVS (mm) | 9±2 | 9±2 | 0.67 |
| PW (mm) | 9±2 | 9±2 | 0.67 |
| LAVI (ml/m2) | 20±5 | 22±9 | 0.071 |
| E (m/s) | 0.67±0.13 | 0.62±0.15 | 0.12 |
| A (m/s) | 0.66±0.16 | 0.66±0.19 | 0.87 |
| E/A | 1.1±0.3 | 1.0±0.4 | 0.53 |
| DT (ms) | 193±39 | 202±44 | 0.29 |
| TR velocity (m/s) | 2.1±0.3 | 2.2±0.2 | 0.18 |
| E′ (cm/s) | 6.6±2.1 | 6.7±2.3 | 0.73 |
| E/E′ | 10.9±3.4 | 9.7±2.5 | 0.036 |
| S′ | 6.5±1.1 | 6.8±1.1 | 0.22 |
| Longitudinal diastolic function reserve index (25 W) | 1.9±1.2 | 2.5±2.2 | 0.07 |
| Longitudinal diastolic function reserve index (50 W) | 2.3±1.3 | 3.2±2.2 | 0.031 |
| Longitudinal systolic function reserve index (25 W) | 0.6±0.7 | 1.2±1.5 | 0.029 |
| Longitudinal systolic function reserve index (50 W) | 1.2±1.2 | 2.1±1.6 | 0.009 |
A, peak velocity of diastolic filling during atrial contraction; DT, deceleration time; E, peak velocity of early diastolic filling; E′ early diastolic mitral annular velocity; IVS, interventricular septal thickness; LAVI, left atrial volume index; LVEDD, left ventricular end‐diastolic dimension; LVESD, left ventricular end‐systolic dimension; PW, posterior wall thickness; S′, systolic mitral annular velocity; TR, tricuspid regurgitation.
Haemodynamic response to exercise
The effect of supine bicycle exercise on heart rate and blood pressure is shown in table 3. In both groups, heart rate, systolic blood pressure and diastolic blood pressure were increased after exercise compared with those at rest. However, no significant differences were observed in the changes of heart rate, systolic and diastolic blood pressures at rest or during exercise between the groups.
Table 3 Haemodynamic response to exercise.
| Diabetes (n = 53) | Control (n = 53) | p Value | |
|---|---|---|---|
| HR at rest (beat/min) | 67±9 | 65±11 | 0.31 |
| HR at 25 W (beat/min) | 93±11 | 98±16 | 0.072 |
| HR at 50 W (beat/min) | 105±15 | 109±17 | 0.22 |
| Systolic BP (mm Hg) at rest | 116±17 | 121±15 | 0.14 |
| Systolic BP (mm Hg) at 25 W | 146±21 | 143±21 | 0.49 |
| Systolic BP (mm Hg) at 50 W | 158±26 | 152±20 | 0.24 |
| Diastolic BP (mm Hg) at rest | 70±8 | 73±9 | 0.06 |
| Diastolic BP (mm Hg) at 25 W | 82±9 | 82±12 | 0.94 |
| Diastolic BP (mm Hg) at 50 W | 86±10 | 86±10 | 0.93 |
BP, blood pressure; HR, heart rate.
LV longitudinal diastolic function at rest and with exercise
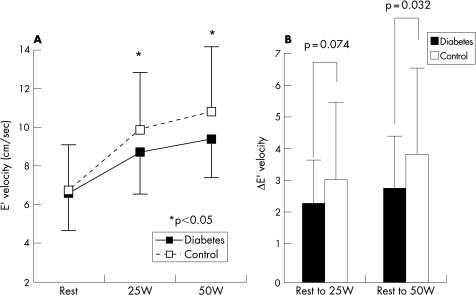
E′ at rest (6.6±2.1 vs 6.7±2.3 cm/s, p = 0.73) was similar between the groups. E′ showed a progressive increase from resting to graded exercise in both groups. However, E′ during exercise was significantly lower in patients with diabetes compared with those of the controls (8.7±2.4 vs 9.9±3.2 cm/s at 25 W, p = 0.048; 9.4±2.4 vs 10.8±3.3 cm/s at 50 W, p = 0.034). In addition, the magnitude of change of E′ with exercise was significantly smaller in patients with diabetes compared with control (2.3±1.4 vs 3.0±2.5 cm/s from base to 25 W, p = 0.074; 2.8±1.6 vs 3.9±2.7 cm/s from base to 50 W, p = 0.032) (fig 1). The longitudinal diastolic function reserve index was significantly lower in patients with diabetes compared with that of controls (1.9±1.2 vs 2.5±2.2 cm/s at 25 W, p = 0.07; 2.3±1.3 vs 3.2±2.2 cm/s at 50 W, p = 0.031).
Figure 1 Left panel: change of E′ from rest to exercise. Note that an increase in E′ with exercise was less pronounced in patients with diabetes. Right panel: the change of E′ with exercise was significantly smaller in patients with diabetes compared with the control.
LV longitudinal systolic function at rest and with exercise
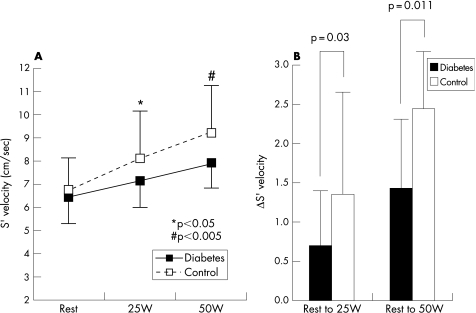
S′ at rest was similar between the groups. S′ showed a progressive increase from resting to graded exercise in both groups. However, S′ during exercise was significantly lower in patients with diabetes compared with that of the control (7.2±1.2 vs 8.1±2.1 cm/s at 25 W, p = 0.012; 7.9±1.4 vs 9.2±2.1 cm/s at 50 W, p = 0.0019). Additionally, the increment of S′ with exercise was significantly smaller in patients with diabetes compared with the control (0.7±0.8 vs 1.4±1.7 cm/s from base to 25 W, p = 0.03; 1.4±1.5 vs 2.4±1.9 cm/s from base to 50 W, p = 0.011) (fig 2). The longitudinal systolic function reserve index was significantly lower in patients with diabetes compared with that of controls (0.6±0.70 vs 1.2±1.5 cm/s at 25 W, p = 0.029; 1.2±1.2 vs 2.1±1.6 cm/s at 50 W, p = 0.009).
Figure 2. Left panel: change of S′ from rest to exercise. Note that an increase in S′ with exercise was blunted in patients with diabetes. Right panel: the increment of S′ with exercise was significantly smaller in patients with diabetes compared with control.
Discussion
The principal and novel findings of this study were: (1) LV longitudinal diastolic functional reserve, assessed with the change of E′ during exercise, is abnormal in patients with diabetes without overt heart disease; (2) even though EF and S′ at rest were similar between the groups, S′ during exercise and the magnitude of increment of S′ during exercise were significantly smaller in patients with diabetes, suggesting that their longitudinal contractile reserve is reduced. The present study is the first to demonstrate abnormal LV longitudinal functional reserve during exercise in patients with diabetes using exercise TDE. The strength and the significance of the present study reside in the use of low‐level exercise TDE in early detection of myocardial dysfunction in patients with diabetes without overt heart disease. We also proposed new indices of LV longitudinal systolic and diastolic functional reserve by expressing the change of E′ or S′ with exercise relative to E′ or S′ at baseline. We believe these novel indices are helpful to quantify the LV longitudinal functional reserve in various groups of patients with myocardial functional abnormalities.
The importance of assessing detailed information of LV myocardial performance is essential in understanding the development of heart failure and may provide the opportunity to initiate therapeutic intervention at an early stage. The recent use of sensitive and less load‐dependent techniques such as strain, strain rate and myocardial velocity by TDE has demonstrated the occurrence of both systolic and diastolic abnormalities as a marker of subclinical LV dysfunction in patients with diabetes. However, a useful technique for evaluating ventricular performance involves the measurement of the circulatory changes that occur during exercise. The potential value of stressing LV to assess its performance is emphasised by the fact that the normal ranges of LV end‐diastolic pressure, cardiac index, and ventricular stroke work in the resting state are wide, with values that frequently overlap those seen in patients with ventricular dysfunction. In patients with normal systolic and diastolic myocardial velocities at rest, they may reveal abnormal or blunted LV longitudinal functional reserve by exercise TDE. In this regard, using exercise TDE may be a promising, noninvasive diagnostic modality for early detection of subclinical LV dysfunction in patients with diabetes.
LV longitudinal diastolic function and functional reserve in patients with diabetes
Experimental models have shown disturbances of relaxation both in vitro and in vivo. In humans, diastolic inflow patterns are frequently abnormal, with inferred disturbances of myocardial relaxation or myocardial compliance. Diastolic dysfunction has also been identified in diabetic subjects without evidence of other causes of heart disease. However, these findings have not been uniform. The normal physiological response to exercise is a faster relaxation rate induced by increased sympathetic tone and an exaggerated LV suction effect. Isotonic treadmill exercise produces a downward shift of the early diastolic portion of the LV pressure–volume loop, which augments early diastolic transmitral pressure gradient, increases early mitral flow and sustains LV filling volumes despite a reduced filling time without a marked increase in left atrial pressure.18 It has been shown that E′ was inversely correlated with the time constant of isovolumic relaxation (τ). The primary mechanism for the increase in E′ during exercise may be increased sympathetic tone and resultant faster myocardial relaxation. In a recent study, E′ was unchanged with increased transmitral gradient in dogs with diastolic dysfunction, whereas &'E′ was increased in dogs with normal tau.14 A similar result has been shown in human beings.15 These findings are consistent with normal physiological augmentation of myocardial relaxation in healthy subjects. Although E′ was previously shown to be reduced in patients with diabetes, the response of E′ to exercise, which can be considered as LV longitudinal diastolic functional reserve, was not examined. The current study demonstrated that the augmentation of myocardial relaxation during exercise, assessed with TDE, is blunted in patients with diabetes. Based on our observation, LV longitudinal diastolic functional reserve in patients with diabetes can be considered as abnormal.
LV longitudinal systolic function and functional reserve in patients with diabetes
Although EF is a simple yet powerful parameter used as an estimate of systolic function, it is influenced by preload, afterload, and contractility and it only examines the difference in cavity area at systole and end‐diastole. Thus, it does not necessarily reflect the contractile state of the myocardium. Moreover, EF, if measured by M‐mode echo in a parasternal short axis image, mostly reflects LV circumferential fibre shortening along the LV short axis . However, previous studies have shown that a significant proportion of LV systolic contraction occurs along the long axis, and thus measurement of the ventricular long‐axis velocities, which can be quantified by measuring S′ by TDE, is thought to provide a more sensitive index of systolic function than EF. A previous study has shown that patients with diabetes without overt heart disease demonstrated evidence of systolic dysfunction and increased myocardial reflectivity.2 In our study, we further demonstrated, despite similar resting EF and S′ between patients with diabetes and controls, that S′ during exercise and the magnitude of increment of S′ with exercise were significantly smaller in patients with diabetes, suggesting that their longitudinal contractile reserve is abnormal. Therefore, the assessment of longitudinal function should be incorporated for the comprehensive evaluation of LV systolic function and maybe the better parameter for earlier detection of LV systolic dysfunction. The results of this study add important new information regarding the assessment of myocardial function in patients with diabetes, where longitudinal contractile reserve assessed by TDE may be useful to identify subtle abnormalities of systolic longitudinal function in patients with normal EF.
Study limitations
In this study, the effects of coronary microvascular disease could not be excluded. It has been suggested that microvascular disease, which is common in patients with diabetes, leading to reduced coronary flow reserve with angiographically normal coronary arteries can be associated with a supernormal LV function during exercise.19 Although none of the patients developed exercise‐induced myocardial ischaemia by ECG or wall motion abnormality, potential influence of coronary microvascular disease cannot be excluded. Although it did not reach statistical significance, the heart rates achieved during exercise in diabetics was lower than those of the control group. The positive effect of heart rate on contractility (force–frequency relation and the Bowditch effect) was therefore lower in patients with diabetes and could be one of the reasons for the lower values during exercise.
Clinical implications
Early detection and treatment of subclinical ventricular dysfunction are suggested as an effective strategy to prevent or delay the onset of heart failure. Such a strategy would mandate the effective diagnostic approach for the detection of preclinical ventricular dysfunction in these patients. Although the recently published revised ACC/AHA guidelines20 broadened the scope of therapeutic guidelines to those of preventive‐therapeutic guidelines by including patients who are at risk of developing heart failure and thus emphasised that the detection and treatment of subclinical LV dysfunction are important, no recommendations were available regarding how to detect subclinical LV dysfunction. Therefore, the challenge of recognising subclinical LV dysfunction in a noninvasive way still remains. The novel concept of the assessment of LV longitudinal functional reserve can be used for evaluation of myocardial function in patients with diabetes, since patients with diabetes are an important group of individuals with subclinical myocardial dysfunction.
Acknowledgements
This work was supported by the Korea Science and Engineering Foundation (KOSEF) grant funded by the Korea government (M10642120001‐06N4212‐00110).
Abbreviations
A - peak velocity of diastolic filling during atrial contraction
E - peak velocity of early diastolic filling
E′ - early diastolic mitral annular velocity
EF - ejection fraction
LV - left ventricular
S′ - systolic mitral annular velocity
TDE - tissue Doppler echocardiography
Footnotes
Competing interests: None.
References
- 1.Tang W H, Young J B. Cardiomyopathy and heart failure in diabetes. Endocrinol Metab Clin North Am 2001301031–1046. [DOI] [PubMed] [Google Scholar]
- 2.Fang Z Y, Yuda S, Anderson V.et al Echocardiographic detection of early diabetic myocardial disease. J Am Coll Cardiol 200341611–617. [DOI] [PubMed] [Google Scholar]
- 3.Struthers A D, Morris A D. Screening for and treating left‐ventricular abnormalities in diabetes mellitus: a new way of reducing cardiac deaths. Lancet 20023591430–1432. [DOI] [PubMed] [Google Scholar]
- 4.Fang Z Y, Schull‐Meade R, Leano R.et al Screening for heart disease in diabetic subjects. Am Heart J 2005149349–354. [DOI] [PubMed] [Google Scholar]
- 5.Marwick T H. Tissue Doppler imaging for evaluation of myocardial function in patients with diabetes mellitus. Curr Opin Cardiol 200419442–446. [DOI] [PubMed] [Google Scholar]
- 6.Di Bello V, Talarico L, Picano E.et al Increased echodensity of myocardial wall in the diabetic heart: an ultrasound tissue characterization study. J Am Coll Cardiol 1995251408–1415. [DOI] [PubMed] [Google Scholar]
- 7.Hansen A, Johansson B L, Wahren J.et al C‐peptide exerts beneficial effects on myocardial blood flow and function in patients with type 1 diabetes. Diabetes 2002513077–3082. [DOI] [PubMed] [Google Scholar]
- 8.Andersen N H, Poulsen S H, Eiskjaer H.et al Decreased left ventricular longitudinal contraction in normotensive and normoalbuminuric patients with Type II diabetes mellitus: a Doppler tissue tracking and strain rate echocardiography study. Clin Sci (Lond) 200310559–66. [DOI] [PubMed] [Google Scholar]
- 9.Fang Z Y, Najos‐Valencia O, Leano R.et al Patients with early diabetic heart disease demonstrate a normal myocardial response to dobutamine. J Am Coll Cardiol 200342446–453. [DOI] [PubMed] [Google Scholar]
- 10.Vinereanu D, Nicolaides E, Tweddel A C.et al Subclinical left ventricular dysfunction in asymptomatic patients with Type II diabetes mellitus, related to serum lipids and glycated haemoglobin. Clin Sci (Lond) 2003105591–599. [DOI] [PubMed] [Google Scholar]
- 11.Liu C P, Ting C T, Lawrence W.et al Diminished contractile response to increased heart rate in intact human left ventricular hypertrophy. Systolic versus diastolic determinants. Circulation 1993881893–1906. [DOI] [PubMed] [Google Scholar]
- 12.Mulieri L A, Hasenfuss G, Leavitt B.et al Altered myocardial force–frequency relation in human heart failure. Circulation 1992851743–1750. [DOI] [PubMed] [Google Scholar]
- 13.Gwathmey J K, Warren S E, Briggs G M.et al Diastolic dysfunction in hypertrophic cardiomyopathy. Effect on active force generation during systole. J Clin Invest 1991871023–1031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Nagueh S F, Sun H, Kopelen H A.et al Hemodynamic determinants of the mitral annulus diastolic velocities by tissue Doppler. J Am Coll Cardiol 200137278–285. [DOI] [PubMed] [Google Scholar]
- 15.Ha J W, Lulic F, Bailey K R.et al Effects of treadmill exercise on mitral inflow and annular velocities in healthy adults. Am J Cardiol 200391114–115. [DOI] [PubMed] [Google Scholar]
- 16.Ha J W, Oh J K, Pellikka P A.et al Diastolic stress echocardiography: a novel noninvasive diagnostic test for diastolic dysfunction using supine bicycle exercise Doppler echocardiography. J Am Soc Echocardiogr 20051863–68. [DOI] [PubMed] [Google Scholar]
- 17.Quinones M A, Waggoner A D, Reduto L A.et al A new, simplified and accurate method for determining ejection fraction with two‐dimensional echocardiography. Circulation 198164744–753. [DOI] [PubMed] [Google Scholar]
- 18.Cheng C P, Igarashi Y, Little W C. Mechanism of augmented rate of left ventricular filling during exercise. Circ Res 1992709–19. [DOI] [PubMed] [Google Scholar]
- 19.Picano E, Palinkas A, Amyot R. Diagnosis of myocardial ischemia in hypertensive patients. J Hypertens 2001191177–1183. [DOI] [PubMed] [Google Scholar]
- 20.Hunt S A, Baker D W, Chin M H.et al ACC/AHA Guidelines for the Evaluation and Management of Chronic Heart Failure in the Adult: Executive Summary A Report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Committee to Revise the 1995 Guidelines for the Evaluation and Management of Heart Failure). Circulation 20011042996–3007. [DOI] [PubMed] [Google Scholar]