Abstract
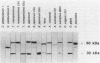
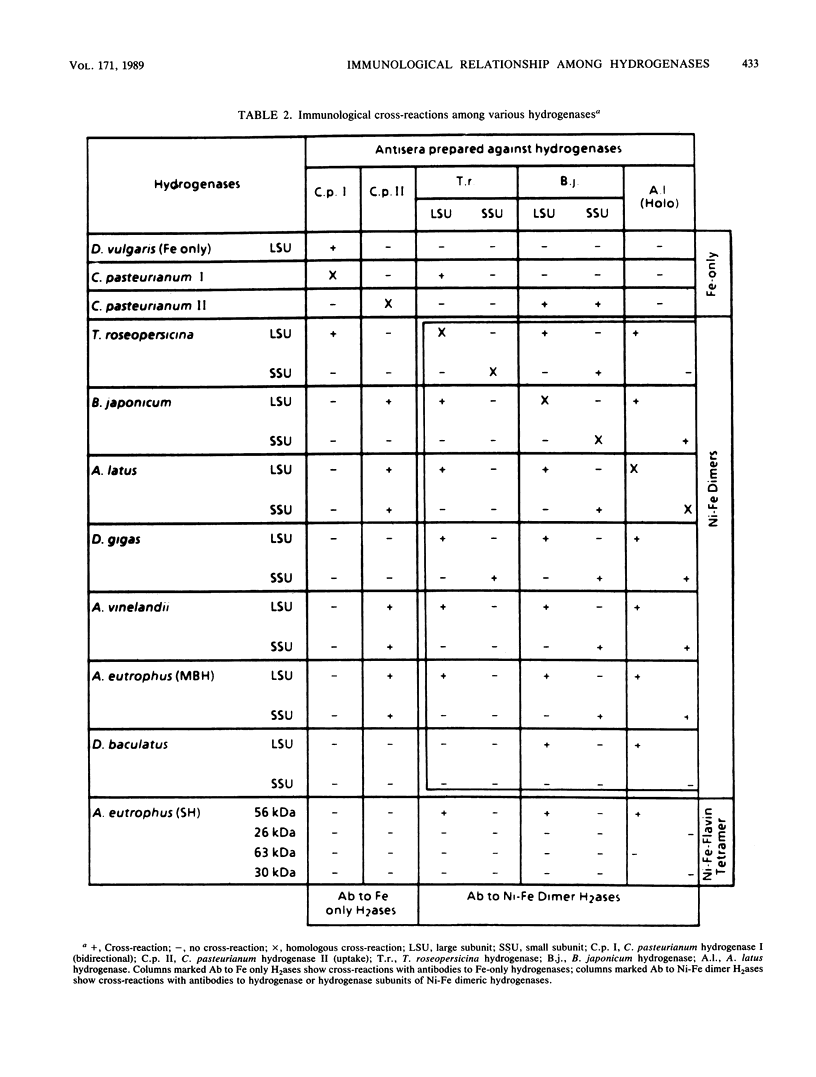
We examined the immunological cross-reactions of 11 different hydrogenase antigens with 9 different hydrogenase antibodies. Included were antibodies and antigens of both subunits of the hydrogenases of Bradyrhizobium japonicum and Thiocapsa roseopersicina. The results showed a strong relationship among the Ni-Fe dimeric hydrogenases. The two subunits of Ni-Fe dimeric hydrogenases appeared immunologically distinct: specific interactions occurred only when antibodies to the 60- and 30-kilodalton subunits reacted with the 60- and 30-kilodalton-subunit antigens. The interspecies cross-reactions suggested that at least one conserved protein region exists among the large subunits of these enzymes, whereas the small subunits are less conserved. Antibodies to the Fe-only bidirectional hydrogenase of Clostridium pasteurianum reacted with the Desulfovibrio vulgaris bidirectional hydrogenase. Surprisingly, antibodies to the clostridial uptake hydrogenase did not react with any of the Fe-only bidirectional hydrogenases but did react with several of the Ni-Fe dimeric hydrogenases. The two hydrogenases from C. pasteurianum were found to be quite different immunologically. The possible relationship of these findings to the structure and catalytic functions of hydrogenase are discussed.
Full text
PDF




Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Adams M. W., Mortenson L. E., Chen J. S. Hydrogenase. Biochim Biophys Acta. 1980 Dec;594(2-3):105–176. doi: 10.1016/0304-4173(80)90007-5. [DOI] [PubMed] [Google Scholar]
- Arp D. J., McCollum L. C., Seefeldt L. C. Molecular and immunological comparison of membrane-bound, H2-oxidizing hydrogenases of Bradyrhizobium japonicum, Alcaligenes eutrophus, Alcaligenes latus, and Azotobacter vinelandii. J Bacteriol. 1985 Jul;163(1):15–20. doi: 10.1128/jb.163.1.15-20.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Birkett C. R., Foster K. E., Johnson L., Gull K. Use of monoclonal antibodies to analyse the expression of a multi-tubulin family. FEBS Lett. 1985 Aug 5;187(2):211–218. doi: 10.1016/0014-5793(85)81244-8. [DOI] [PubMed] [Google Scholar]
- Bradford M. M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem. 1976 May 7;72:248–254. doi: 10.1016/0003-2697(76)90527-3. [DOI] [PubMed] [Google Scholar]
- Harker A. R., Zuber M., Evans H. J. Immunological homology between the membrane-bound uptake hydrogenases of Rhizobium japonicum and Escherichia coli. J Bacteriol. 1986 Feb;165(2):579–584. doi: 10.1128/jb.165.2.579-584.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hathaway G. M., Lundak T. S., Tahara S. M., Traugh J. A. Isolation of protein kinases from reticulocytes and phosphorylation of initiation factors. Methods Enzymol. 1979;60:495–511. doi: 10.1016/s0076-6879(79)60047-2. [DOI] [PubMed] [Google Scholar]
- Howard J., Shannon L. A rapid, quantitative, and highly specific assay for carbohydrate-binding proteins. Anal Biochem. 1977 May 1;79(1-2):234–239. doi: 10.1016/0003-2697(77)90398-0. [DOI] [PubMed] [Google Scholar]
- Huynh B. H., Czechowski M. H., Krüger H. J., DerVartanian D. V., Peck H. D., Jr, LeGall J. Desulfovibrio vulgaris hydrogenase: a nonheme iron enzyme lacking nickel that exhibits anomalous EPR and Mössbauer spectra. Proc Natl Acad Sci U S A. 1984 Jun;81(12):3728–3732. doi: 10.1073/pnas.81.12.3728. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kovács K. L., Tigyi G., Alfonz H. Purification of hydrogenase by fast protein liquid chromatography and by conventional separation techniques: a comparative study. Prep Biochem. 1985;15(5):321–334. doi: 10.1080/00327488508062449. [DOI] [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- LeGall J., Ljungdahl P. O., Moura I., Peck H. D., Jr, Xavier A. V., Moura J. J., Teixera M., Huynh B. H., DerVartanian D. V. The presence of redox-sensitive nickel in the periplasmic hydrogenase from Desulfovibrio gigas. Biochem Biophys Res Commun. 1982 May 31;106(2):610–616. doi: 10.1016/0006-291x(82)91154-8. [DOI] [PubMed] [Google Scholar]
- Neville D. M., Jr Molecular weight determination of protein-dodecyl sulfate complexes by gel electrophoresis in a discontinuous buffer system. J Biol Chem. 1971 Oct 25;246(20):6328–6334. [PubMed] [Google Scholar]
- Pinkwart M., Schneider K., Schlegel H. G. Purification and properties of the membrane-bound hydrogenase from N2-fixing Alcaligenes latus. Biochim Biophys Acta. 1983 Jun 29;745(3):267–278. doi: 10.1016/0167-4838(83)90058-4. [DOI] [PubMed] [Google Scholar]
- Sawers R. G., Jamieson D. J., Higgins C. F., Boxer D. H. Characterization and physiological roles of membrane-bound hydrogenase isoenzymes from Salmonella typhimurium. J Bacteriol. 1986 Oct;168(1):398–404. doi: 10.1128/jb.168.1.398-404.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schink B., Schlegel H. G. The membrane-bound hydrogenase of Alcaligenes eutrophus: II. Localization and immunological comparison with other hydrogenase systems. Antonie Van Leeuwenhoek. 1980;46(1):1–14. doi: 10.1007/BF00422224. [DOI] [PubMed] [Google Scholar]
- Schneider K., Piechulla B. Isolation and immunological characterization of the four non-identical subunits of the soluble NAD-linked hydrogenase from Alcaligenes eutrophus H16. Biochimie. 1986 Jan;68(1):5–13. doi: 10.1016/s0300-9084(86)81062-8. [DOI] [PubMed] [Google Scholar]
- Schneider K., Schlegel H. G. Purification and properties of soluble hydrogenase from Alcaligenes eutrophus H 16. Biochim Biophys Acta. 1976 Nov 8;452(1):66–80. doi: 10.1016/0005-2744(76)90058-9. [DOI] [PubMed] [Google Scholar]
- Seefeldt L. C., Arp D. J. Purification to homogeneity of Azotobacter vinelandii hydrogenase: a nickel and iron containing alpha beta dimer. Biochimie. 1986 Jan;68(1):25–34. doi: 10.1016/s0300-9084(86)81064-1. [DOI] [PubMed] [Google Scholar]
- Strandberg G. W., Wilson P. W. Formation of the nitrogen-fixing enzyme system in Azotobacter vinelandii. Can J Microbiol. 1968 Jan;14(1):25–31. doi: 10.1139/m68-005. [DOI] [PubMed] [Google Scholar]
- Teixeira M., Fauque G., Moura I., Lespinat P. A., Berlier Y., Prickril B., Peck H. D., Jr, Xavier A. V., Le Gall J., Moura J. J. Nickel-[iron-sulfur]-selenium-containing hydrogenases from Desulfovibrio baculatus (DSM 1743). Redox centers and catalytic properties. Eur J Biochem. 1987 Aug 17;167(1):47–58. doi: 10.1111/j.1432-1033.1987.tb13302.x. [DOI] [PubMed] [Google Scholar]
- Towbin H., Staehelin T., Gordon J. Electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose sheets: procedure and some applications. Proc Natl Acad Sci U S A. 1979 Sep;76(9):4350–4354. doi: 10.1073/pnas.76.9.4350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Voordouw G., Brenner S. Nucleotide sequence of the gene encoding the hydrogenase from Desulfovibrio vulgaris (Hildenborough). Eur J Biochem. 1985 May 2;148(3):515–520. doi: 10.1111/j.1432-1033.1985.tb08869.x. [DOI] [PubMed] [Google Scholar]