Abstract
Cross-sectional analyses show that an index of aging-associated health/well-being deficits, called the “frailty index”, can characterize the aging process in humans. This study provides support for such characterization from a longitudinal analysis of the frailty index properties. The data are from the National Long Term Care Survey assessed longitudinally health and functioning of the U.S. elderly in the period 1982 to 1999. In cross-sectional analysis, the frailty index exhibits accelerated increase with age till oldest-old ages (95+), with possible deceleration thereafter. Longitudinal analysis confirms the accelerated accumulation of deficits in aging individuals. The time-dynamics of the frailty index is affected by two sex-sensitive processes: i) selection of robust individuals, resulting in a decline of the mean frailty index with age, and ii) accumulation of deficits associated with physiological aging and its interaction with environment, which results in an accelerated increase of individual frailty index prior to death irrespective of chronological age. Current frailty index levels in individuals are more predictive of death than the index past values. Longitudinal analysis provides strong evidence that the cumulative index of health/well-being deficits can characterize aging-associated processes in humans and predict death better than chronological age during short-term periods.
Keywords: Frailty index, aging, health, longevity, sex differences
INTRODUCTION
Recently, the concept of frailty as a characteristic of aging-associated processes has attracted considerable research attention (Ferrucci et al., 2003; Fisher, 2005; Fried et al., 2004; Kulminski et al., 2007a; Kulminski et al., 2007b; Kulminski et al., 2006; Levers et al., 2006; Mitnitski et al., 2005; Mitnitski et al., 2001; Puts et al., 2005; Rockwood et al., 2000; Rockwood et al., 2006; Woo et al., 2006; Yashin et al., 2007a; 2007b). Frailty is usually found to be associated with a high risk of death (Ferrucci et al., 2003; Mitnitski et al., 2005; Puts et al., 2005), pure health status, and hospitalization (Fried et al., 2001; Newman et al., 2001; Woo et al., 2006). Despite recent advances, understanding the etiology and pathophysiology of frailty is still limited (Ferrucci et al., 2003). Moreover, the boundaries of the frailty syndrome, which can have medical, functional, and social components (Fisher, 2005), even are uncertain. This prevents the development of a clear and widely acceptable definition of frailty (Bortz, 2002; Lally and Crome, 2007; Rockwood et al., 2005). Nevertheless, there is an agreement that frailty is a state of an organism with non-specific vulnerability to stressors, which results from decreased physiological reserves, and deregulation of multiple physiologic systems (Fried et al., 2004). In addition, new ideas concerning the physiological nature of frailty are emerging which increase our understanding of this phenomenon. One promising hypothesis is that frailty is associated with degradation/decline of the neuroendocrine and immune functions of an organism (Vanitallie, 2003). Such changes can result in an age-related decline of the organism's allostatic capacity, which can be associated with an age-related decline in stress-resistance (Kulminski et al., 2006; Yashin et al., 2007b) and/or increasing time of recovery from stress (e.g., due to decline in physiological complexity (Lipsitz, 2004)).
Hypotheses about the physiological origin of frailty permit various specific criteria of frailty (Ferrucci et al., 2003). One of them was suggested by Mitnitski and Rockwood and is based on tracking health/well-being disorders (called deficits, i.e., signs, symptoms, impairments, abnormal laboratory tests, etc.) accumulated by individuals during their life course (Mitnitski et al., 2004; Rockwood et al., 2000). According to this concept, the frailty index (FI) – calculated as the proportion of deficits in an individual (Mitnitski et al., 2004) – depends to a large extent on how deficits are accumulated and to much less extent on which deficits are included in the FI construction. Therefore, the underlying paradigm of the FI is that it can characterize frailty by measuring a wide set of health disorders, which could reflect aging-associated physiological changes in an individual. In other words, a frail person will suffer from more distinct health problems (of any type!) than a non-frail counterpart. The validity of such the FI has been examined in a number of studies (Goggins et al., 2005; Kulminski et al., 2007a; Kulminski et al., 2007b; Kulminski et al., 2006; Mitnitski et al., 2005; Rockwood et al., 2006; Woo et al., 2006; Yashin et al., 2007a; 2007b). According to these studies, the FI constitutes a broad view on frailty (Fisher, 2005).
Despite the fact that the FI concept has been applied in various studies, few of these studies (Kulminski et al., 2007a; Mitnitski et al., 2007) tested this conceptualization as a characteristic of the aging process in a longitudinal context. This paper seeks to address this underdeveloped element. Specifically, we apply the Rockwood-Mitnitski approach to test the ability of the FI to characterize the aging process in humans focusing on a comparison of results from cross-sectional and longitudinal analyses in a sample of the U.S. elderly individuals using data from the National Long Term Care Survey (NLTCS).
METHODS
We analyze the data from the 1982, 1984, 1989, 1994, and 1999 NLTCS and linked Medicare Vital statistics files which provide information on death until August, 2003. The NLTCS uses a sample of individuals drawn from national Medicare enrollment files. The longitudinal design of the study minimizes bias in longitudinal estimates, which is also reduced by the high (95%) response rates. To complete the NLTCS a two-stage interviewing process was used. A screening interview assessing chronic disability was given to all members of the sample. A detailed interview was given to i) those who reported at least one chronic (90+ days) impairment in (instrumental) activities of daily living (I)ADL, ii) institutionalized individuals, and iii) those who received a detailed interview in a prior survey. In 1994 and 1999 the NLTCS design was changed by adding the “healthy” supplement for which the individuals were designated to receive a detailed interview even if screened initially as non-institutional and unimpaired. For each new survey, a cohort sample of about 5,000 persons ages 65−69 was added to the surviving sample to replace the deaths occurring since the prior survey and to ensure that the new sample is representative of the entire U.S. elderly population (65+). Such a procedure ensures a valid longitudinal and cross sectional design for the survey (for further details see (Manton et al., 2006)).
Following (Mitnitski et al., 2001) we define the FI as an unweighted count of the number of deficits divided by the total number of all potential deficits considered for a person. For this study we considered the following set of 32 deficits, which is similar to that assessed from the Canadian Study of Health and Aging (Mitnitski et al., 2001), i.e., difficulty with eating, dressing, walk around, getting in/out bed, getting bath, toileting, using telephone, going out, shopping, cooking, light house work, taking medicine, managing money, arthritis, Parkinson's disease, glaucoma, diabetes, stomach problem, history of heart attack, hypertension, history of stroke, flu, broken hip, broken bones, trouble with bladder/bowels, dementia, self-rated health, as well as problems with vision, hearing, ear, teeth, and feet. To assess the robustness of the FI so defined, we randomly replaced certain deficits from this list by other deficits (e.g., headaches, trouble sleeping, difficulty with doing laundry, indicators of physical performance, mental and emotional problems). In all cases the results were qualitatively similar, which indicates the relative independence of the of the FI concept of the nature of particular deficits used (Fisher, 2005; Mitnitski et al., 2004; Rockwood et al., 2000). Consequently, throughout this study we retained only the original definition of the FI on the basis of the listed 32 deficits.
RESULTS
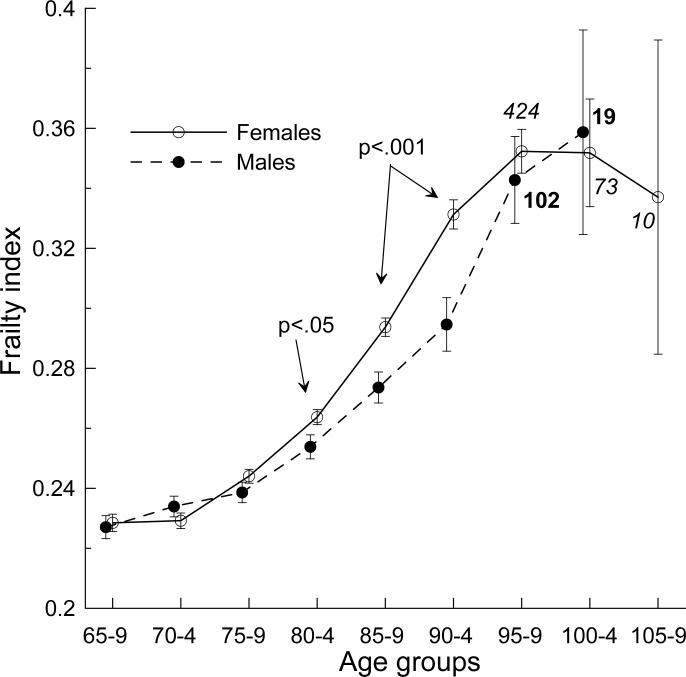
To be consistent with the NLTCS design across waves and deficits included into the FI definition, we focus on a sample of non-institutionalized respondents who were selected using the same procedures in all waves (i.e., we excluded the “healthy” supplement in the community survey and institutionalized persons). First we assess the cross-sectional FI age patterns. Since they are qualitatively similar across the NLTCS waves (Kulminski et al., 2006), we pooled data for all waves (that provides 26,712 observations) and grouped them into five-year age groups to increase the statistical power for the estimates (Figure 1).
Figure 1.
FI age patterns for males and females. P-values show significant differences between estimates for males and females in the respective age groups. The patterns are slightly offset for the sake of clarity. Bars show +/−Standard Error (SE). Italic (bold) numbers indicate sample size of females (males) at extreme ages.
All FI age patterns exhibit nonlinear relationship with age. At younger ages, we observe slow increase of the mean FI. It accelerates at old ages and tends to decelerate at extremely old ages. In contrast to previous studies (Goggins et al., 2005; Mitnitski et al., 2005), we do not observe consistent differences in the FI between sexes for all age groups. Statistically significant differences are found only for the 80−84 (p<.05) and 85−94 (p<.001) age groups.
Next, we begin the longitudinal analyses by calculating the mean FI for participants of the 1982, 1984, or 1989 NLTCS (Table 1). It is evident that the mean FI for the sex-unstratified samples remains nearly constant (the 1982 survey) or exhibits slight increases (the 1984 and 1989 surveys). And there are pronounced sex-specific dynamics: the FI for males tends to initially decrease and then exhibits a tendency for growth while for females it always increases.
Table 1.
Longitudinal changes of the FI for participants of the 1982, 1984, and 1989 NLTCS and for groups A and B (i.e., groups comprised of survived individuals participating in three consecutive surveys; see text) for males and females combined (M&F), males (M), and females (F). Standard Errors (SEs) are shown in parentheses.
| NLTCS | |||||||
|---|---|---|---|---|---|---|---|
| Sample | Sex | N | 1982 | 1984 | 1989 | 1994 | 1999 |
| M&F | .273 (.002) | .266 (.002) | .268 (.004) | .269 (.006) | .274 (.010) | ||
| 1982 | M | .266 (.003) | .257 (.004) | .254 (.007) | .236 (.011) | .243 (.018)* | |
| F | .277 (.002) | .271 (.003) | .275 (.004) | .283 (.007) | .289 (.012)* | ||
| M&F | .257 (.002) | .259 (.003) | .261 (.005) | .266 (.008) | |||
| 1984 | M | .251 (.003)* | .248 (.005)* | .234 (.009) | .244 (.015)# | ||
| F | .261 (.002)* | .263 (.003)* | .272 (.005) | .276 (.009)# | |||
| M&F | .252 (.002) | .253 (.004) | .263 (.006) | ||||
| 1989 | M | .241 (.004) | .231 (.007) | .234 (.010) | |||
| F | .258 (.003) | .263 (.004) | .276 (.007) | ||||
| M&F | 1129 | .180 (.003) | .201 (.004) | .261 (.005) | |||
| A | M | 328 | .167 (.006)* | .178 (.006) | .234 (.009) | ||
| F | 801 | .185 (.004)* | .211 (.004) | .272 (.005) | |||
| M&F | 749 | .177 (.004) | .201 (.005) | .263 (.006) | |||
| B | M | 224 | .168 (.008)# | .175 (.008) | .234 (.010) | ||
| F | 525 | .181 (.004)# | .212 (.006) | .276 (.007) | |||
Note that NLTCS is the National Long Term Care Survey; p<.01,
p<.05,
p>.05 for differences between sexes.
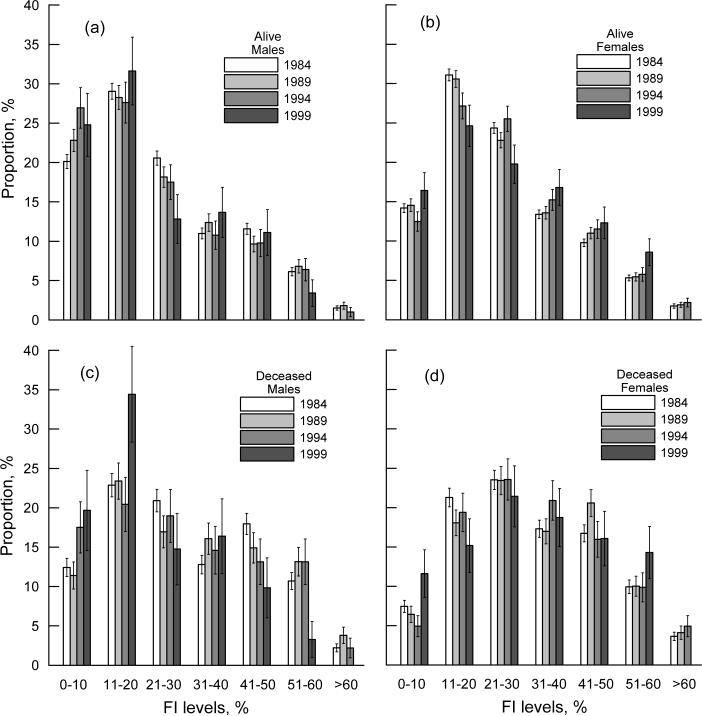
To uncover possible changes in the FI frequency patterns in these samples over time, we plotted the frequency distribution of the surviving (Figures 2a,b) and dying (Figures 2c,d) males and females from the 1984 survey (for other surveys the results are similar). For surviving individuals, we observe positively skewed (gamma-like) patterns (Figures 2a,b). For males, the skewness (shape asymmetry) of the frequency distributions increases from 1984 to 1999 as 0.672 (SE, 0.054), 0.717 (0.082), 0.747 (0.141), 0.883 (0.224), while, for females, it decreases as 0.728 (0.039), 0.678 (0.056), 0.601 (0.091), 0.411 (0.149). In contrast, the FI patterns for deceased individuals are bell-shaped and approximately normal. In fact, our analysis reveals a smooth transition from a nearly symmetric distribution for individuals who died within one year after their last interview to a gamma-like distribution for those who died during a longer follow-up period, e.g., skewness for the entire 1984 survey sample changes as −0.049 (SE: 0.086), 0.108 (0.058), 0.261 (0.041), 0.433 (0.031), and 0.545 (0.028) for 1, 2, 4, 8, and 12 years of follow-up, respectively. Furthermore, the FI-specific frequency patterns for individuals died during the same time period after an interview are similar (Figures 2c,d). Note that the estimates for males for the 1999 NLTCS are of relatively low precision due to the small numbers of individuals for each FI level (e.g., the maximum number of males who died is 21 for the 11−20% interval of the FI). We also note that similar differences in shapes of the frequency patterns were observed among diseased and non-diseased individuals (Mitnitski et al., 2001).
Figure 2.
(a,b) FI-specific frequency patterns for male (a) and female (b) participants of the 1984 NLTCS surviving to the date of the interview (year is given in the figure). (c,d) The same as in (a,b) but for individuals who died within four years after the interview date. Bars show +/−SE.
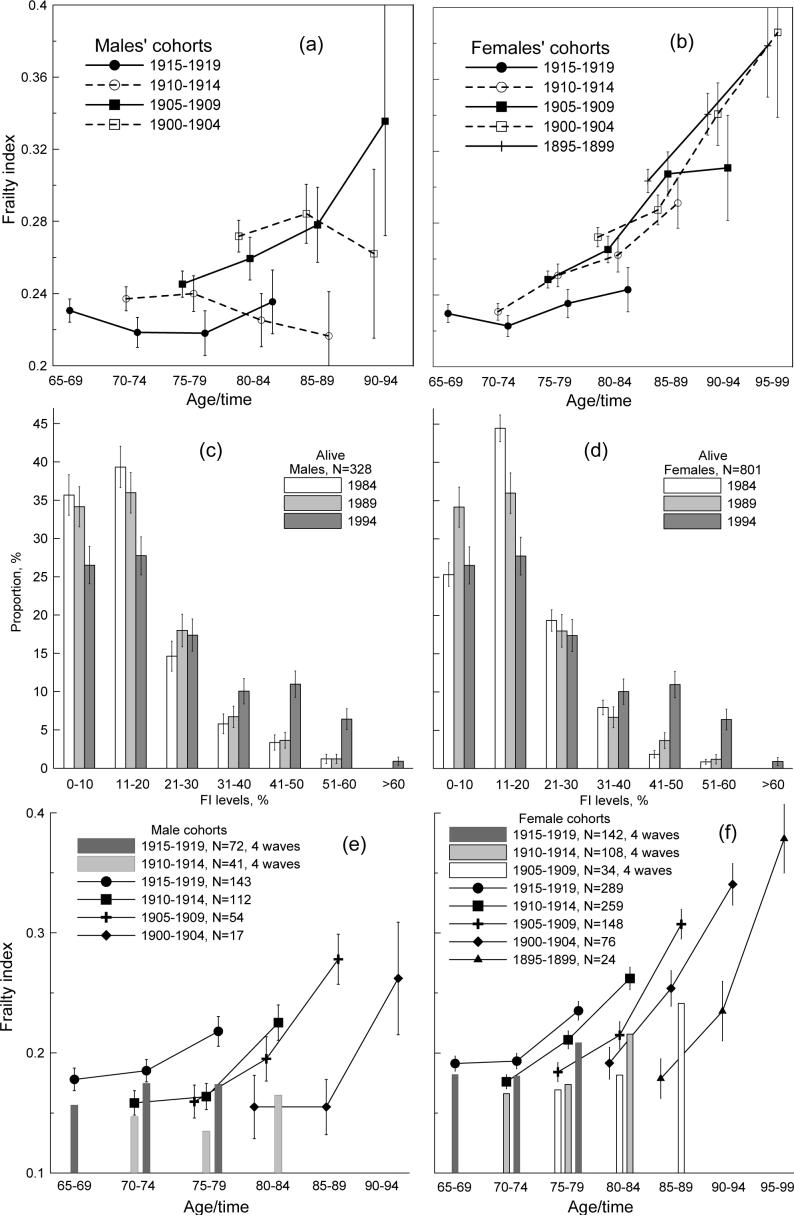
Figures 3a,b depict the age-specific evolution of the mean FI for the five-year birth cohorts starting from the 1984 NLTCS. The FI dynamics undergoes noticeable changes over the cohorts. For the two youngest male cohorts (1915−1919 and 1910−1914), we observe nearly flat temporal patterns. For the 1905−1909 cohort, the FI consistently increases. But the older cohorts have considerably smaller sample sizes, which results in large SEs that decreases the significance of the estimates. For females, the nearly flat pattern is seen only for the 1915−1919 cohort. For older female cohorts, the mean FI basically increases.
Figure 3.
(a,b) Mean FI for selected 5-year birth cohorts (shown in the figure) of males (a) and females (b) in the 1984, 1989, 1994, and 1999 surveys. (c,d) FI-specific frequency patterns for male (c) and female (d) participants of the 1984, 1989, and 1994 NLTCSs. (e,f) FI dynamics (conditional on survivors) for male and female 5-year birth cohorts participating in three (line plots) and four (bar charts) consecutive surveys starting from the 1984 NLTCS. Thin bars show +/−SE.
Further insights into connections between the FI and aging-associated processes can be obtained by considering conditional dynamics, i.e., we focus only on those individuals who were alive and participated in several surveys. We distinguish two groups comprised of participants of the 1984−1994 (group A) and the 1989−1999 (group B) surveys. Table 1 shows that the FI increases over time for male and female sample as well as for each sex. It is also seen that the FI increase accelerates between subsequent surveys with larger acceleration for males. Quantitatively, acceleration is provided by the FI second derivative, which, for the difference scheme, becomes,
where j is an integer and Δt is a time interval between surveys. Since we compare acceleration at the same time intervals, the denominator can be dropped. Then, from Table 1 for group A the acceleration parameter for males is (0.234−(2×0.178)+0.167) = 0.045 , while for females we obtain (0.272−(2×0.211)+0.185) = 0.035 . For group B we obtain 0.052 for males and 0.033 for females. The larger acceleration for males is also seen from the ratios of the relative FI increase between two surveys: for males it is 4.78 (A) and 8.09 (B) while for females it is 2.06 (A) and 1.76 (B). Our analysis shows no FI time trends for samples A and B.
Analysis of the FI frequency patterns for surviving males and females (Figures 3c,d) shows that they have large positive skewness, especially for males. Therefore, surviving males have to be of better health than females.
Figures 3e,f (lines) shows age-specific dynamics of the FI for group A, i.e., conditional on surviving individuals. An acceleration of FI increases is seen for each five-year age group. Thus, we explicitly show that inferences from cross-sectional studies of nonlinear increase of the FI with age are corroborated by (age-specific) longitudinal analysis. We also distinguish individuals participating in four consecutive surveys, i.e., 1984, 1989, 1994, and 1999 (128 males and 298 females). Bars in Figures 3e,f show the mean FIs for the first three surveys for two cohorts (born in 1915−1919 and 1910−1914) for males and three cohorts (1915−1919, 1910−1914, and 1905−1909) for females. In all cases, participants of the four surveys have smaller mean FIs than those of three surveys. Moreover, these patterns become flatter, especially for males. This is attributed to the fact that 90.5% of males (181) and 72.5% of females (365) among those who did not participate in 1999 NLTCS from the participants of the 1984, 1989, and 1994 NLTCS died during the period between 1994 and 1999 years.
Finally, we performed multivariate Cox (hazard) regression analysis to assess association of the FI with death. We tested static (current FI) and dynamic measures of frailty. The latter can be assessed by using two equivalent strategies. First, we can define difference between current and past FI (FI_P) values for each individual. A second strategy, which will be used below, is simply to use the FI_P as an independent covariate. To ensure that our predictions are unbiased by the non-equidistant survey intervals, we focused on samples of the 1989, 1994, and 1999 NLTCS participants. Since the results were qualitatively similar for each sample, we pooled data into one sample to increase the statistical power and presented the results for the pooled sample of participants of these surveys. We performed our analyses separately for males and females.
The Cox regressions with two covariates, i.e., either age and FI or age and FI_P, show that both FI and FI_P are significantly associated with death for both sexes. However, the Cox regression with three covariates (age, FI, and FI_P) reveals that the FI is significantly predictive of death (see Table 2), while the FI_P is not (not shown). The lack of statistical significance of FI_P in the presence of FI may be due to the fact that the FI and FI_P are strongly correlated – the correlation coefficients are 0.57 (males) and 0.56 (females). This indicates that individuals who have higher initial values of the FI also are more likely to have larger changes in the FI – which is entirely consistent with the growth patterns described above.
Table 2.
Cox multivariate proportional hazard regression models for 1- and 2-year follow-up periods for death events for all ages and for distinct age groups. Numbers show the logarithm of the relative risks, ln(RR), for a 1% increase of the Frailty Index (FI). Covariates included age, current Frailty Index (FI) value and the Frailty Index in the preceding survey (FI_P). Numbers in parentheses show Standard Errors of the ln(RR).
| Observation period, yrs. | All ages | Age groups | ||||
|---|---|---|---|---|---|---|
| Sex | 65−74 | 75−84 | 85−94 | 95+ | ||
| Males | 1 | .039 (.004) | .045 (.011) | .043 (.006) | .026 (.008) | -- |
| 2 | .031 (.003) | .039 (.007) | .035 (.004) | .020 (.006) | -- | |
| Females | 1 | .041 (.004) | .068 (.012) | .042 (.005) | .039 (.006) | -- |
| 2 | .034 (.002) | .052 (.008) | .036 (.004) | .030 (.004) | .019* (.009) | |
Note that p<.001;
p<.05.
We also found that FI_P is insignificant for specific age groups. The FI-attributed Relative Risk (RR) declines with age for males and females (Table 2). Therefore, such behavior can be characteristic of more fundamental processes associated with senescence and can be attributed to a superposition of two processes. One is the increase of the baseline hazard with aging (Yashin et al., 2006). The other is the process of aging-sensitive adaptation to the accumulated deficits. The arguments in support of the second process follow from a comparison of the age-specific RR for both sexes (Table 2). In particular, we observe that the risk of death for females is larger for the 65−74 and 85−94 groups than for males of the same age. For the 75−84 group, the risks are similar. The changes in the RR between the sexes may be due to fundamental differences between males and females, e.g., by sex-specific aging-sensitive adaptation mechanisms. However, this possibility requires additional study.
DISCUSSION AND CONCLUSIONS
A number of recent cross-sectional studies examined the FI age patterns in various national settings (Goggins et al., 2005; Kulminski et al., 2007b; Kulminski et al., 2006; Mitnitski and Rockwood, 2006; Mitnitski et al., 2005; Rockwood et al., 2006; Woo et al., 2006). General conclusions from these studies are that the FI increases with age, this increase is, generally, nonlinear, i.e., accelerates with age, and the FI predicts adverse events (death, institutionalization, hospitalization). Such properties are characteristic of a marker of aging processes (Karasik et al., 2005). Consequently, the FI can be considered as a measure of aging and aging-associated processes. In this study, we provided further support toward this view from both cross-sectional and longitudinal analyses of the FI.
We show that the FI age patterns in the sample of the U.S. males and females exhibit increase with age and that this increase is nonlinear, i.e., with acceleration at younger-advanced ages and possible deceleration at extreme ages. Leveling-off of the FI age patterns is more convincing for females for whom the sample size is relatively large at extreme ages (Figure 1). Unlike other studies (Goggins et al., 2005; Mitnitski et al., 2005), we found that these patterns are sex-insensitive at younger and extreme ages, a finding that can be attributed to high proportion of disabled individuals (∼80%) in the sample. For other ages the FI for females tends to be larger than for males (Figure 1) while males have large chances to die (Kulminski et al., 2007b; Mitnitski et al., 2005). It is worth noting that a similar situation for males compared to females is observed when global health is assessed using a single self-rated measure of health (Deeg and Kriegsman, 2003; Idler and Benyamini, 1997).
Our longitudinal analysis reveals a strong impact of sex on the FI dynamics. This can result in a relatively flat longitudinal pattern for the sex-unstratified sample while the male and female FIs exhibiting pronounced dynamics of opposite tendencies (Table 1). Therefore, sex-specific analysis of the FI is crucial. The presence of these opposite tendencies as well as opposite changes in the shape of the FI frequency patterns over time (Figures 2a,b) for males and females suggest that there are at least two competing processes in variation of the FI with time. One of these is the cohort effect of selective survival of robust individuals with low FIs, which results in a decline of the mean FI (Table 1) and an increase of the proportion of surviving males with lower FI values (Figures 2a,b). The increase of the FI with time for females suggest the presence of the processes associated with physiological changes loaded by environmental exposures in aging organisms, which result in growth of individual frailty and, consequently, a larger mean FI. These observations suggest that females are more tolerant to accumulated deficits than males.
Further insights follow from the analysis of the sex- and age-specific FI dynamics (Figures 3a,b). For males, we observe that in younger cohorts (1915−1919 and 1910−1914) the mean FI is nearly constant, while in older cohort (1905−1909) it clearly increases with time. Therefore, the FI for males as well as for females can also characterize aging-associated physiological processes. The implication is that the balance of the selection and physiological processes can be age dependent. This conclusion is also supported by the analysis of FI age-sensitivity in female cohorts, which shows that younger females accumulate deficits at a lower rate than older females.
Hypotheses on the association of the FI with aging-related processes can be directly tested by estimating changes in the FI over time in the same individuals, i.e., by excluding the selection effect. This test shows that both males and females accumulate deficits over time (Table 1). We also found that they accumulate deficits at an accelerated pace that is larger for males. These effects are also seen for more homogeneous age groups (Figures 3e,f). The larger acceleration for males provides further support for our conclusion that females are more tolerant to accumulated deficits than males. The fact that males surviving for longer time periods have flatter time patterns than those surviving for shorter time periods (Figures 3e,f) also suggests that males accumulate deficits near their time of death. For females, this process is extended over longer time periods prior to death. The larger acceleration prior to death is seen independently of chronological age – which indicates connection between the FI and aging process. This is in concordance with our early analysis, which, particularly, shows that time-to-death is better correlated with the FI than with the chronological age (Kulminski et al., 2007b). Since humans can experience assimilative/accommodative (by changing their behavior, (Boerner, 2004)) and/or physiological (Patten et al., 1998; Ward and Cohen, 2004) adaptation to their declining health status, such differences in deficit accumulation in males and females can be due to a larger adaptation potential in females than in males. This may be associated with biological differences in sex-specific aging processes. A possible basis for physiological adaptation could be lower average metabolism rates in females. On acquiring a health disorder a female's organism then has a larger array of choices for its localization and, respectively, for a more specific (and, consequently, more effective) response. Acceleration of the aging process can be also affected by the socio-economical status and interrelated demographic processes (e.g., education, widowhood). This problem, however, deserves a separate analysis.
Finally, we found that the rate of increase in the FI or, equivalently, the past values of the FI are not predictive of deaths occurring during short-term periods (e.g., 1−2 years) in multivariate Cox regression model which includes age, past and current FIs as covariates. The current frailty status appears to be a better predictor of death than this state in the past independently of sex.
In sum, all the above findings suggest that the cumulative index of health/well-being deficits can usefully characterize the aging-associated processes in humans. Furthermore, the FI appears to be a convenient and useful indicator of aging suitable for large-scale population-based studies of health, well-being, and survival.
Acknowledgments
The research reported in this paper was supported by the grants 5UO1-AG-007198-18, 1R01 AG028259-01, 1R01-AG-027019-01 and 5P01-AG-008761-16 from the National Institute on Aging. We also thank two anonymous referees for their useful suggestions which allowed us to essentially improve the manuscript.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- Boerner K. Adaptation to disability among middle-aged and older adults: the role of assimilative and accommodative coping. J Gerontol B Psychol Sci Soc Sci. 2004;59:P35–42. doi: 10.1093/geronb/59.1.p35. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bortz WM. A conceptual framework of frailty: a review. J Gerontol A Biol Sci Med Sci. 2002;57:M283–288. doi: 10.1093/gerona/57.5.m283. 2nd. [DOI] [PubMed] [Google Scholar]
- Deeg DJ, Kriegsman DM. Concepts of self-rated health: specifying the gender difference in mortality risk. Gerontologist. 2003;43:376–386. doi: 10.1093/geront/43.3.376. discussion 372−375. [DOI] [PubMed] [Google Scholar]
- Ferrucci L, Guralnik JM, Cavazzini C, Bandinelli S, Lauretani F, Bartali B, Repetto L, Longo DL. The frailty syndrome: a critical issue in geriatric oncology. Crit Rev Oncol Hematol. 2003;46:127–137. doi: 10.1016/s1040-8428(02)00177-4. [DOI] [PubMed] [Google Scholar]
- Fisher AL. Just what defines frailty? J Am Geriatr Soc. 2005;53:2229–2230. doi: 10.1111/j.1532-5415.2005.00510.x. [DOI] [PubMed] [Google Scholar]
- Fried LP, Ferrucci L, Darer J, Williamson JD, Anderson G. Untangling the concepts of disability, frailty, and comorbidity: implications for improved targeting and care. J Gerontol A Biol Sci Med Sci. 2004;59:255–263. doi: 10.1093/gerona/59.3.m255. [DOI] [PubMed] [Google Scholar]
- Fried LP, Tangen CM, Walston J, Newman AB, Hirsch C, Gottdiener J, Seeman T, Tracy R, Kop WJ, Burke G, McBurnie MA. Frailty in older adults: evidence for a phenotype. J Gerontol A Biol Sci Med Sci. 2001;56:M146–156. doi: 10.1093/gerona/56.3.m146. [DOI] [PubMed] [Google Scholar]
- Goggins WB, Woo J, Sham A, Ho SC. Frailty index as a measure of biological age in a Chinese population. J Gerontol A Biol Sci Med Sci. 2005;60:1046–1051. doi: 10.1093/gerona/60.8.1046. [DOI] [PubMed] [Google Scholar]
- Idler EL, Benyamini Y. Self-rated health and mortality: a review of twenty-seven community studies. J Health Soc Behav. 1997;38:21–37. [PubMed] [Google Scholar]
- Karasik D, Demissie S, Cupples LA, Kiel DP. Disentangling the genetic determinants of human aging: biological age as an alternative to the use of survival measures. J Gerontol A Biol Sci Med Sci. 2005;60:574–587. doi: 10.1093/gerona/60.5.574. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kulminski A, Ukraintseva S, Akushevich I, Arbeev K, Yashin A. Cumulative Index of Health Deficiencies as a Characteristic of Long Life. J Am Geriatr Soc. 2007a doi: 10.1111/j.1532-5415.2007.01155.x. in press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kulminski A, Yashin A, Arbeev K, Akushevich I, Ukraintseva S, Land K, Manton K. Cumulative index of health disorders as an indicator of aging-associated processes in the elderly: Results from analyses of the National Long Term Care Survey. Mech Ageing Dev. 2007b;128:250–258. doi: 10.1016/j.mad.2006.12.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kulminski A, Yashin A, Ukraintseva S, Akushevich I, Arbeev K, Land K, Manton K. Accumulation of health disorders as a systemic measure of aging: Findings from the NLTCS data. Mech Ageing Dev. 2006;127:840–848. doi: 10.1016/j.mad.2006.08.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lally F, Crome P. Understanding frailty. Postgrad Med J. 2007;83:16–20. doi: 10.1136/pgmj.2006.048587. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Levers MJ, Estabrooks CA, Ross Kerr JC. Factors contributing to frailty: literature review. J Adv Nurs. 2006;56:282–291. doi: 10.1111/j.1365-2648.2006.04021.x. [DOI] [PubMed] [Google Scholar]
- Lipsitz LA. Physiological complexity, aging, and the path to frailty. Sci Aging Knowledge Environ. 2004;2004:pe16. doi: 10.1126/sageke.2004.16.pe16. [DOI] [PubMed] [Google Scholar]
- Manton KG, Gu X, Lamb VL. Change in chronic disability from 1982 to 2004/2005 as measured by long-term changes in function and health in the U.S. elderly population. Proc Natl Acad Sci U S A. 2006;103:18374–18379. doi: 10.1073/pnas.0608483103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mitnitski A, Bao L, Skoog I, Rockwood K. A cross-national study of transitions in deficit counts in two birth cohorts: Implications for modeling ageing. Exp Gerontol. 2007;42:241–246. doi: 10.1016/j.exger.2006.10.001. [DOI] [PubMed] [Google Scholar]
- Mitnitski A, Rockwood K. Decrease in the relative heterogeneity of health with age: a cross-national comparison. Mech Ageing Dev. 2006;127:70–72. doi: 10.1016/j.mad.2005.09.007. [DOI] [PubMed] [Google Scholar]
- Mitnitski A, Song X, Rockwood K. The estimation of relative fitness and frailty in community-dwelling older adults using self-report data. J Gerontol A Biol Sci Med Sci. 2004;59:M627–632. doi: 10.1093/gerona/59.6.m627. [DOI] [PubMed] [Google Scholar]
- Mitnitski A, Song X, Skoog I, Broe GA, Cox JL, Grunfeld E, Rockwood K. Relative fitness and frailty of elderly men and women in developed countries and their relationship with mortality. J Am Geriatr Soc. 2005;53:2184–2189. doi: 10.1111/j.1532-5415.2005.00506.x. [DOI] [PubMed] [Google Scholar]
- Mitnitski AB, Mogilner AJ, Rockwood K. Accumulation of deficits as a proxy measure of aging. ScientificWorldJournal. 2001;1:323–336. doi: 10.1100/tsw.2001.58. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Newman AB, Gottdiener JS, McBurnie MA, Hirsch CH, Kop WJ, Tracy R, Walston JD, Fried LP. Associations of subclinical cardiovascular disease with frailty. J Gerontol A Biol Sci Med Sci. 2001;56:M158–166. doi: 10.1093/gerona/56.3.m158. [DOI] [PubMed] [Google Scholar]
- Patten RD, Udelson JE, Konstam MA. Ventricular remodeling and its prevention in the treatment of heart failure. Curr Opin Cardiol. 1998;13:162–167. [PubMed] [Google Scholar]
- Puts MT, Lips P, Deeg DJ. Sex differences in the risk of frailty for mortality independent of disability and chronic diseases. J Am Geriatr Soc. 2005;53:40–47. doi: 10.1111/j.1532-5415.2005.53008.x. [DOI] [PubMed] [Google Scholar]
- Rockwood K, Hogan DB, MacKnight C. Conceptualisation and measurement of frailty in elderly people. Drugs Aging. 2000;17:295–302. doi: 10.2165/00002512-200017040-00005. [DOI] [PubMed] [Google Scholar]
- Rockwood K, Mitnitski A, Song X, Steen B, Skoog I. Long-term risks of death and institutionalization of elderly people in relation to deficit accumulation at age 70. J Am Geriatr Soc. 2006;54:975–979. doi: 10.1111/j.1532-5415.2006.00738.x. [DOI] [PubMed] [Google Scholar]
- Rockwood K, Song X, MacKnight C, Bergman H, Hogan DB, McDowell I, Mitnitski A. A global clinical measure of fitness and frailty in elderly people. Cmaj. 2005;173:489–495. doi: 10.1503/cmaj.050051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vanitallie TB. Frailty in the elderly: contributions of sarcopenia and visceral protein depletion. Metabolism. 2003;52:22–26. doi: 10.1016/s0026-0495(03)00297-x. [DOI] [PubMed] [Google Scholar]
- Ward NS, Cohen LG. Mechanisms underlying recovery of motor function after stroke. Arch Neurol. 2004;61:1844–1848. doi: 10.1001/archneur.61.12.1844. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Woo J, Goggins W, Sham A, Ho SC. Public health significance of the frailty index. Disabil Rehabil. 2006;28:515–521. doi: 10.1080/09638280500215867. [DOI] [PubMed] [Google Scholar]
- Yashin AI, Akushevich IV, Arbeev KG, Akushevich L, Ukraintseva SV, Kulminski A. Insights on aging and exceptional longevity from longitudinal data: novel findings from the Framingham Heart Study. Age. 2006;28:363–374. doi: 10.1007/s11357-006-9023-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yashin AI, Arbeev KG, Kulminski A, Akushevich I, Akushevich L, Ukraintseva SV. Cumulative index of elderly disorders and its dynamic contribution to mortality and longevity. Rejuvenation Res. 2007a;10:75–86. doi: 10.1089/rej.2006.0500. [DOI] [PubMed] [Google Scholar]
- Yashin AI, Arbeev KG, Kulminski A, Akushevich I, Akushevich L, Ukraintseva SV. Health decline, aging and mortality: how are they related? Biogerontology. 2007b doi: 10.1007/s10522-006-9073-3. [DOI] [PMC free article] [PubMed] [Google Scholar]