Abstract
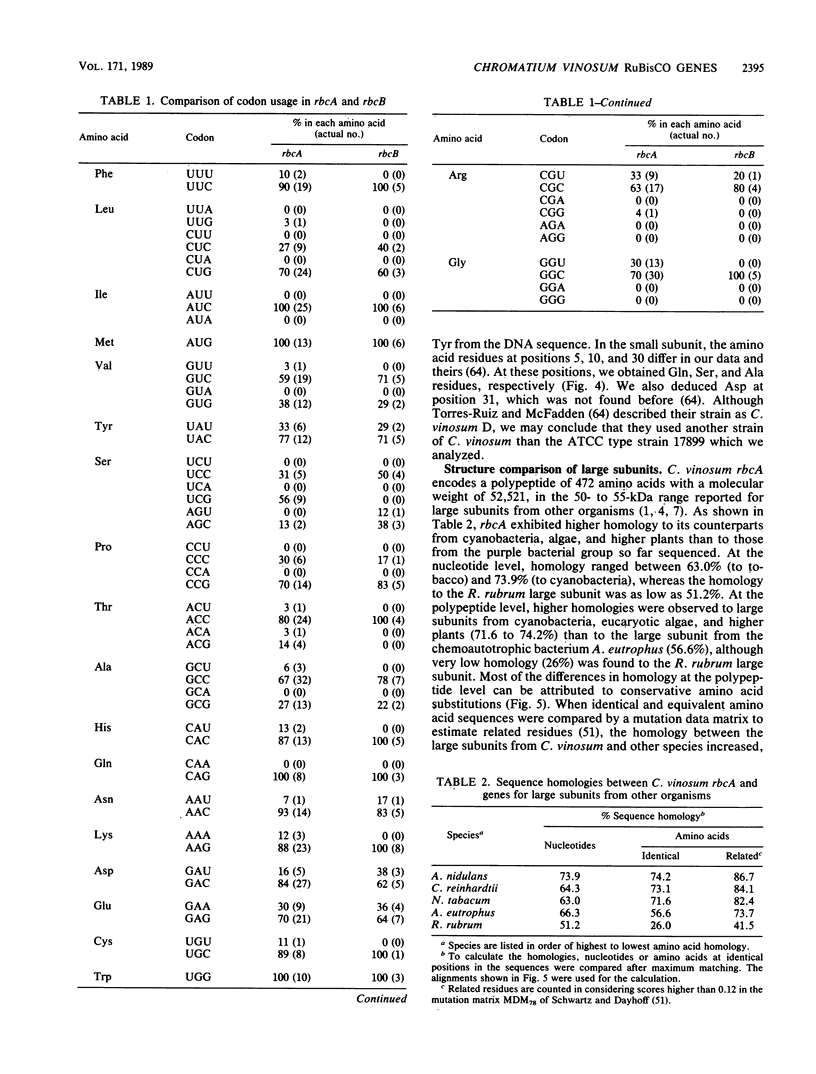
Two sets of genes for the large and small subunits of ribulose 1,5-bisphosphate carboxylase/oxygenase (RuBisCO) were detected in the photosynthetic purple sulfur bacterium Chromatium vinosum by hybridization analysis with RuBisCO gene probes, cloned by using the lambda Fix vector, and designated rbcL-rbcS and rbcA-rbcB. rbcL and rbcA encode the large subunits, and rbcS and rbcB encode the small subunits. rbcL-rbcS was the same as that reported previously (A. M. Viale, H. Kobayashi, T. Takabe, and T. Akazawa, FEBS Lett. 192:283-288, 1985). A DNA fragment bearing rbcA-rbcB was subcloned in plasmid vectors and sequenced. We found that rbcB was located 177 base pairs downstream of the rbcA coding region, and both genes were preceded by plausible procaryotic ribosome-binding sites. rbcA and rbcD encoded polypeptides of 472 and 118 amino acids, respectively. Edman degradation analysis of the subunits of RuBisCO isolated from C. vinosum showed that rbcA-rbcB encoded the enzyme present in this bacterium. The large- and small-subunit polypeptides were posttranslationally processed to remove 2 and 1 amino acid residues from their N-termini, respectively. Among hetero-oligomeric RuBisCOs, the C. vinosum large subunit exhibited higher homology to that from cyanobacteria, eucaryotic algae, and higher plants (71.6 to 74.2%) than to that from the chemolithotrophic bacterium Alcaligenes eutrophus (56.6%). A similar situation has been observed for the C. vinosum small subunit, although the homology among small subunits from different organisms was lower than that among the large subunits.
Full text
PDF








Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Andersen K., Caton J. Sequence analysis of the Alcaligenes eutrophus chromosomally encoded ribulose bisphosphate carboxylase large and small subunit genes and their gene products. J Bacteriol. 1987 Oct;169(10):4547–4558. doi: 10.1128/jb.169.10.4547-4558.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Andersen K., Wilke-Douglas M. Construction and use of a gene bank of Alcaligenes eutrophus in the analysis of ribulose bisphosphate carboxylase genes. J Bacteriol. 1984 Sep;159(3):973–978. doi: 10.1128/jb.159.3.973-978.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Andersen K., Wilke-Douglas M. Genetic and physical mapping and expression in Pseudomonas aeruginosa of the chromosomally encoded ribulose bisphosphate carboxylase genes of Alcaligenes eutrophus. J Bacteriol. 1987 May;169(5):1997–2004. doi: 10.1128/jb.169.5.1997-2004.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Blombäck B., Blombäck M., Edman P., Hessel B. Human fibrinopeptides. Isolation, characterization and structure. Biochim Biophys Acta. 1966 Feb 28;115(2):371–396. doi: 10.1016/0304-4165(66)90437-5. [DOI] [PubMed] [Google Scholar]
- Buchanan B. B., Sirevåg R. Ribulose 1,5-diphosphate carboxylase and Cholorobium thiosulfatophilum. Arch Microbiol. 1976 Aug;109(1-2):15–19. doi: 10.1007/BF00425107. [DOI] [PubMed] [Google Scholar]
- Chapman M. S., Suh S. W., Curmi P. M., Cascio D., Smith W. W., Eisenberg D. S. Tertiary structure of plant RuBisCO: domains and their contacts. Science. 1988 Jul 1;241(4861):71–74. doi: 10.1126/science.3133767. [DOI] [PubMed] [Google Scholar]
- Deaton M. A., Bock S. E., Zwick H., Freeman J. A. Common properties shared by growth-associated proteins of the regenerating optic nerve of goldfish (C. auratus). Neurosci Lett. 1988 Feb 29;85(2):267–271. doi: 10.1016/0304-3940(88)90363-1. [DOI] [PubMed] [Google Scholar]
- Dron M., Rahire M., Rochaix J. D. Sequence of the chloroplast DNA region of Chlamydomonas reinhardii containing the gene of the large subunit of ribulose bisphosphate carboxylase and parts of its flanking genes. J Mol Biol. 1982 Dec 25;162(4):775–793. doi: 10.1016/0022-2836(82)90547-2. [DOI] [PubMed] [Google Scholar]
- Goldschmidt-Clermont M., Rahire M. Sequence, evolution and differential expression of the two genes encoding variant small subunits of ribulose bisphosphate carboxylase/oxygenase in Chlamydomonas reinhardtii. J Mol Biol. 1986 Oct 5;191(3):421–432. doi: 10.1016/0022-2836(86)90137-3. [DOI] [PubMed] [Google Scholar]
- Gutteridge S., Sigal I., Thomas B., Arentzen R., Cordova A., Lorimer G. A site-specific mutation within the active site of ribulose-1,5-bisphosphate carboxylase of Rhodospirillum rubrum. EMBO J. 1984 Dec 1;3(12):2737–2743. doi: 10.1002/j.1460-2075.1984.tb02204.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hartman F. C., Larimer F. W., Mural R. J., Machanoff R., Soper T. S. Essentiality of Glu-48 of ribulose bisphosphate carboxylase/oxygenase as demonstrated by site-directed mutagenesis. Biochem Biophys Res Commun. 1987 Jun 30;145(3):1158–1163. doi: 10.1016/0006-291x(87)91558-0. [DOI] [PubMed] [Google Scholar]
- Hartman F. C., Soper T. S., Niyogi S. K., Mural R. J., Foote R. S., Mitra S., Lee E. H., Machanoff R., Larimer F. W. Function of Lys-166 of Rhodospirillum rubrum ribulosebisphosphate carboxylase/oxygenase as examined by site-directed mutagenesis. J Biol Chem. 1987 Mar 15;262(8):3496–3501. [PubMed] [Google Scholar]
- Herndon C. S., Hartman F. C. 2-(4-Bromoacetamido)anilino-2-deoxypentitol 1,5-bisphosphate, a new affinity label for ribulose bisphosphate carboxylase/oxygenase from Rhodospirillum rubrum. Determination of reaction parameters and characterization of an active site peptide. J Biol Chem. 1984 Mar 10;259(5):3102–3110. [PubMed] [Google Scholar]
- Igarashi Y., McFadden B. A., el-Gul T. Active site histidine in spinach ribulosebisphosphate carboxylase/oxygenase modified by diethyl pyrocarbonate. Biochemistry. 1985 Jul 16;24(15):3957–3962. doi: 10.1021/bi00336a024. [DOI] [PubMed] [Google Scholar]
- Jones W. J., Nagle D. P., Jr, Whitman W. B. Methanogens and the diversity of archaebacteria. Microbiol Rev. 1987 Mar;51(1):135–177. doi: 10.1128/mr.51.1.135-177.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jordan D. B., Chollet R. Subunit dissociation and reconstitution of ribulose-1,5-bisphosphate carboxylase from Chromatium vinosum. Arch Biochem Biophys. 1985 Feb 1;236(2):487–496. doi: 10.1016/0003-9861(85)90651-4. [DOI] [PubMed] [Google Scholar]
- Klintworth R., Husemann M., Salnikow J., Bowien B. Chromosomal and plasmid locations for phosphoribulokinase genes in Alcaligenes eutrophus. J Bacteriol. 1985 Nov;164(2):954–956. doi: 10.1128/jb.164.2.954-956.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kobayashi H., Akazawa T. Biosynthetic mechanism of ribulose-1,5-bisphosphate carboxylase in the purple photosynthetic bacterium, Chromatium vinosum. II. Biosynthesis of constituent subunits. Arch Biochem Biophys. 1982 Apr 1;214(2):540–549. doi: 10.1016/0003-9861(82)90058-3. [DOI] [PubMed] [Google Scholar]
- Kobayashi H., Akazawa T. Biosynthetic mechanism of ribulose-1,5-bisphosphate carboxylase in the purple photosynthetic bacterium, Chromatium vinosum. Arch Biochem Biophys. 1982 Apr 1;214(2):531–539. doi: 10.1016/0003-9861(82)90057-1. [DOI] [PubMed] [Google Scholar]
- Kobayashi H., Takabe T., Nishimura M., Akazawa T. Role of the large and small subunits of ribulose-1,5-bisphosphate carboxylase in the activation by CO2 and Mg2+. J Biochem. 1979 Apr;85(4):923–930. doi: 10.1093/oxfordjournals.jbchem.a132424. [DOI] [PubMed] [Google Scholar]
- Konings R. N., Verhoeven E. J., Peeters B. P. pKUN, vectors for the separate production of both DNA strands of recombinant plasmids. Methods Enzymol. 1987;153:12–34. doi: 10.1016/0076-6879(87)53045-2. [DOI] [PubMed] [Google Scholar]
- Kyte J., Doolittle R. F. A simple method for displaying the hydropathic character of a protein. J Mol Biol. 1982 May 5;157(1):105–132. doi: 10.1016/0022-2836(82)90515-0. [DOI] [PubMed] [Google Scholar]
- Lorimer G. H. Ribulosebisphosphate carboxylase: amino acid sequence of a peptide bearing the activator carbon dioxide. Biochemistry. 1981 Mar 3;20(5):1236–1240. doi: 10.1021/bi00508a028. [DOI] [PubMed] [Google Scholar]
- Mazur B. J., Chui C. F. Sequence of a genomic DNA clone for the small subunit of ribulose bis-phosphate carboxylase-oxygenase from tobacco. Nucleic Acids Res. 1985 Apr 11;13(7):2373–2386. doi: 10.1093/nar/13.7.2373. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nierzwicki-Bauer S. A., Curtis S. E., Haselkorn R. Cotranscription of genes encoding the small and large subunits of ribulose-1,5-bisphosphate carboxylase in the cyanobacterium Anabaena 7120. Proc Natl Acad Sci U S A. 1984 Oct;81(19):5961–5965. doi: 10.1073/pnas.81.19.5961. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Niyogi S. K., Foote R. S., Mural R. J., Larimer F. W., Mitra S., Soper T. S., Machanoff R., Hartman F. C. Nonessentiality of histidine 291 of Rhodospirillum rubrum ribulose-bisphosphate carboxylase/oxygenase as determined by site-directed mutagenesis. J Biol Chem. 1986 Aug 5;261(22):10087–10092. [PubMed] [Google Scholar]
- Oyaizu H., Debrunner-Vossbrinck B., Mandelco L., Studier J. A., Woese C. R. The green non-sulfur bacteria: a deep branching in the eubacterial line of descent. Syst Appl Microbiol. 1987;9:47–53. doi: 10.1016/s0723-2020(87)80055-3. [DOI] [PubMed] [Google Scholar]
- Platt T. Termination of transcription and its regulation in the tryptophan operon of E. coli. Cell. 1981 Apr;24(1):10–23. doi: 10.1016/0092-8674(81)90496-7. [DOI] [PubMed] [Google Scholar]
- Robison P. D., Tabita F. R. Modification of ribulose bisphosphate carboxylase from Rhodospirillum rubrum with tetranitromethane. Biochem Biophys Res Commun. 1979 May 14;88(1):85–91. doi: 10.1016/0006-291x(79)91699-1. [DOI] [PubMed] [Google Scholar]
- Russel M., Kidd S., Kelley M. R. An improved filamentous helper phage for generating single-stranded plasmid DNA. Gene. 1986;45(3):333–338. doi: 10.1016/0378-1119(86)90032-6. [DOI] [PubMed] [Google Scholar]
- Sanger F., Nicklen S., Coulson A. R. DNA sequencing with chain-terminating inhibitors. Proc Natl Acad Sci U S A. 1977 Dec;74(12):5463–5467. doi: 10.1073/pnas.74.12.5463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schloss J. V., Stringer C. D., Hartman F. C. Identification of essential lysyl and cysteinyl residues in spinach ribulosebisphosphate carboxylase/oxygenase modified by the affinity label N-bromoacetylethanolamine phosphate. J Biol Chem. 1978 Aug 25;253(16):5707–5711. [PubMed] [Google Scholar]
- Schneider G., Lindqvist Y., Brändén C. I., Lorimer G. Three-dimensional structure of ribulose-1,5-bisphosphate carboxylase/oxygenase from Rhodospirillum rubrum at 2.9 A resolution. EMBO J. 1986 Dec 20;5(13):3409–3415. doi: 10.1002/j.1460-2075.1986.tb04662.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shinozaki K., Sugiura M. The gene for the small subunit of ribulose-1,5-bisphosphate carboxylase/oxygenase is located close to the gene for the large subunit in the cyanobacterium Anacystis nidulans 6301. Nucleic Acids Res. 1983 Oct 25;11(20):6957–6964. doi: 10.1093/nar/11.20.6957. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shinozaki K., Sugiura M. The nucleotide sequence of the tobacco chloroplast gene for the large subunit of ribulose-1,5-bisphosphate carboxylase/oxygenase. Gene. 1982 Nov;20(1):91–102. doi: 10.1016/0378-1119(82)90090-7. [DOI] [PubMed] [Google Scholar]
- Shinozaki K., Yamada C., Takahata N., Sugiura M. Molecular cloning and sequence analysis of the cyanobacterial gene for the large subunit of ribulose-1,5-bisphosphate carboxylase/oxygenase. Proc Natl Acad Sci U S A. 1983 Jul;80(13):4050–4054. doi: 10.1073/pnas.80.13.4050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Staehelin L. A., Golecki J. R., Drews G. Supramolecular organization of chlorosomes (chlorobium vesicles) and of their membrane attachment sites in Chlorobium limicola. Biochim Biophys Acta. 1980 Jan 4;589(1):30–45. doi: 10.1016/0005-2728(80)90130-9. [DOI] [PubMed] [Google Scholar]
- Starlinger P. DNA rearrangements in procaryotes. Annu Rev Genet. 1977;11:103–126. doi: 10.1146/annurev.ge.11.120177.000535. [DOI] [PubMed] [Google Scholar]
- Tabita F. R. Molecular and cellular regulation of autotrophic carbon dioxide fixation in microorganisms. Microbiol Rev. 1988 Jun;52(2):155–189. doi: 10.1128/mr.52.2.155-189.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Terzaghi B. E., Laing W. A., Christeller J. T., Petersen G. B., Hill D. F. Ribulose 1,5-bisphosphate carboxylase. Effect on the catalytic properties of changing methionine-330 to leucine in the Rhodospirillum rubrum enzyme. Biochem J. 1986 May 1;235(3):839–846. doi: 10.1042/bj2350839. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Torres-Ruiz J., McFadden B. A. The nature of L8 and L8S8 forms of ribulose bisphosphate carboxylase/oxygenase from Chromatium vinosum. Arch Biochem Biophys. 1987 Apr;254(1):63–68. doi: 10.1016/0003-9861(87)90081-6. [DOI] [PubMed] [Google Scholar]
- Valle E., Kobayashi H., Akazawa T. Transcriptional regulation of genes for plant-type ribulose-1,5-bisphosphate carboxylase/oxygenase in the photosynthetic bacterium, Chromatium vinosum. Eur J Biochem. 1988 May 2;173(3):483–489. doi: 10.1111/j.1432-1033.1988.tb14024.x. [DOI] [PubMed] [Google Scholar]
- Viale A. M., Kobayashi H., Takabe T., Akazawa T. Expression of genes for subunits of plant-type RuBisCO from Chromatium and production of the enzymically active molecule in Escherichia coli. FEBS Lett. 1985 Nov 18;192(2):283–288. doi: 10.1016/0014-5793(85)80126-5. [DOI] [PubMed] [Google Scholar]
- Weisberg R. A., Adhya S. Illegitimate recombination in bacteria and bacteriophage. Annu Rev Genet. 1977;11:451–473. doi: 10.1146/annurev.ge.11.120177.002315. [DOI] [PubMed] [Google Scholar]
- Woese C. R. Bacterial evolution. Microbiol Rev. 1987 Jun;51(2):221–271. doi: 10.1128/mr.51.2.221-271.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Woese C. R., Stackebrandt E., Macke T. J., Fox G. E. A phylogenetic definition of the major eubacterial taxa. Syst Appl Microbiol. 1985;6:143–151. doi: 10.1016/s0723-2020(85)80047-3. [DOI] [PubMed] [Google Scholar]
- Yanisch-Perron C., Vieira J., Messing J. Improved M13 phage cloning vectors and host strains: nucleotide sequences of the M13mp18 and pUC19 vectors. Gene. 1985;33(1):103–119. doi: 10.1016/0378-1119(85)90120-9. [DOI] [PubMed] [Google Scholar]
- Zimmerman C. L., Appella E., Pisano J. J. Rapid analysis of amino acid phenylthiohydantoins by high-performance liquid chromatography. Anal Biochem. 1977 Feb;77(2):569–573. doi: 10.1016/0003-2697(77)90276-7. [DOI] [PubMed] [Google Scholar]