Abstract
To gain a better understanding of the normal characteristics of developing canine muscles, myosin heavy chain (MHC) isoform expression was analysed in the axial and limb skeletal muscles of 18 young dogs whose ages ranged from the late prenatal stage to 6 months. We compared the results of immunohistochemistry using ten monoclonal antibodies, specific to different MHC isoforms, and enzyme-histochemical reactions, which demonstrate the activity of myofibrillar ATPase, succinate dehydrogenase (SDH) and α-glycerophosphate dehydrogenase (α-GPDH). In the skeletal muscles of fetuses and neonatal dogs the developmental isoforms MHC-emb and MHC-neo were prevalent. In all muscles the primary fibres, located centrally in each muscle fascicle, strongly expressed the slow isoform MHC-I. The adult fast isoform MHC-IIa was first noted in some of the secondary fibres on fetal day 55. During the first 10 days after birth, the expression of MHC-emb declined, as did that of MHC-neo during the second and third weeks. Correspondingly, the expression of MHC-IIa, and later, of MHC-I increased in the secondary fibres. Between the sixth week and second month the expression of MHC-IIx became prominent. The slow rhomboideus muscle exhibited an early expression of the slow isoform in the secondary fibres. Our results indicate that the timing of muscle maturation depends on its activity immediately following birth. The fastest developing muscle was the diaphragm, followed by the fast muscles. A pronounced changeover from developmental to adult isoforms was noted at 4–6 weeks of age, which coincides with the increased physical activity of puppies.
Keywords: development, immunohistochemistry, mATPase, muscle fibres, myotubes
Introduction
Myosin heavy chain (MHC) is the protein primarily responsible for muscle contraction. Different isoforms of this protein have been correlated with various velocities of muscle fibre shortening (Bottinelli et al. 1994). At least nine MHC isoforms have been identified in mammalian skeletal muscles. In general, the content of a specific MHC isoform is consistent with ‘classical’ muscle fibre types determined on the basis of myofibrillar adenosine triphosphatase (mATPase) activity, i.e. I, IIA, IIB and IIX/D fibres (Pette et al. 1999). Fibres that express more than one MHC isoform simultaneously have been labelled as hybrid or polymorphic fibres. It has been established that different fast and developmental MHC isoforms are regularly coexpressed in single fibres during muscle development, regeneration, denervation and electrically induced transformations, and are the result of major changes in the hormonal state or physical training. This also occurs far more commonly in normal adult muscles than was previously believed (Schiaffino & Reggiani, 1996; Talmadge, 2000; Baldwin & Haddad, 2001; Caiozzo et al. 2003).
A very high degree of fibre polymorphism has been demonstrated in developing skeletal muscles (di Maso et al. 2000). During myogenesis it is mainly the developmental isoforms, i.e. MHC-emb and MHC-neo, that are expressed; these are gradually replaced by the adult-type isoforms during the early postnatal period (Fitzsimons & Hoh, 1981; LaFramboise et al. 1991; Schiaffino & Reggiani, 1996; Lefaucheur, 2001). It has also been reported that various slow isoforms are present in the primary myotubes as well and that they differ from the adult slow isoform (Hughes et al. 1993). MHC isoform transitions during development seem to be universal (Pette & Staron, 1997); however, more research is needed to ascertain the differences in the timing of their expression between animal species during prenatal and postnatal development.
In canine skeletal muscles, three isoforms – MHC-I, MHC-IIa and MHC-IIx – were first revealed, through denaturing polyacrylamide gel electrophoresis (SDS-PAGE), in the latissimus dorsi and quadriceps femoris muscles (Zhang et al. 1996, 1997). A small percentage of MHC-IIb, normally found only in the fast muscles of small animals, was also discovered in the laryngeal muscles (Wu et al. 1998, 2000). In our previous studies of adult dog limb and trunk muscles we confirmed the presence of three adult MHC isoforms – I, IIa and IIx – and reported a high level of hybrid phenotypes. We also proposed that the fibres expressing MHC-IIx are identical to those previously described as the IIDog type (Štrbenc et al. 2004; Smerdu et al. 2005). A detailed study of seven different trunk and limb muscles confirmed a high level of MHC-IIa and MHC-IIx coexpression (24% on average in the muscles investigated). Careful examination on a fibre-by-fibre basis combined with optical density measurements of the histochemical and immunohistochemical reactions revealed that some canine fibre types had previously been misclassified (Acevedo & Rivero, 2005).
The postnatal muscle development of dogs has been investigated in only a few muscles using the mATPase activity method. Through this method a vast number of type IIC fibres were identified in neonatal muscles or referred to as ‘undifferentiated fibres’. After a few weeks of age they differentiated into other types known in adult animals, i.e. I, IIA and IIDog (Braund & Lincoln, 1981; Latorre et al. 1993b). The only immunohistochemical study of developing trunk or limb canine muscles known to us was performed on the vastus intermedius in a study of muscular dystrophy (Lanfossi et al. 1999). In neonatal dogs (2 days old), most of the fibres expressed developmental isoforms, and the remaining few (about 7%) expressed a slow MHC isoform – these were primary fibres. About half of the secondary fibres coexpressed MHC-IIa. The number of fibres expressing adult isoforms increased, while developmental isoforms had almost disappeared by the time the dogs were 60 days old. The authors did not differentiate between the two types of developmental isoforms (Lanfossi et al. 1999).
In related species, i.e. in cats, embryonic and neonatal MHC isoforms were identified in developing limb muscles by immunohistochemistry, electrophoresis and immunoblotting (Unguez et al. 2000). At the time of birth, the majority of fibres coexpressed embryonic and adult fast isoforms; by 3 months of age the fibres became devoid of the embryonic isoforms. The greatest degree of MHC transition was noted in the fast muscle when the animals were aged between 30 and 40 days. By day 120, both slow and fast muscles exhibited the adult phenotype (Unguez et al. 2000). The successive expression of MHC-emb and MHC-neo in cats had been proposed earlier (Hoh et al. 1988a,b).
A number of hereditary and congenital non-hereditary myopathies affect young dogs but until now there was no clear insight into the fibre type composition of developing canine muscles. To evaluate the extent of pathological changes in diseased muscles, the characteristics of normal muscles should be clearly understood. Therefore, the aim of the present study was to create an outline of normal postnatal MHC isoform transitions in canine skeletal muscles using monoclonal antibodies raised against MHC isoforms. The analysis was complemented with a histochemical classification of fibre types according to their mATPase stability, and a determination of metabolic profiles by demonstrating the oxidative enzyme succinate dehydrogenase (SDH) and the glycolytic mitochondrial menadione-linked α-glycerophosphate dehydrogenase (α-GPDH). The questions that arose were (1) is there a successive expression of MHC-emb, MHC-neo and adult MHC isoforms in dogs, (2) at what time is the transition from developmental to adult morphology of muscle fibres most prominent and (3) is the timing of the transition different in functionally different muscles?
Materials and methods
Muscle samples
Prenatal muscle samples were obtained from six fetuses recovered from hysterectomies or Caesarean sections at fetal days 50, 55 and 60, and were labelled as F50, F55 and F60 (the gestational period for dogs is, on average, 63 days). Samples were recovered from two animals per day. The postnatal samples were obtained from 16 puppies, which died of natural causes or were put down due to severe trauma, on postnatal days 1 (n = 3), 3 (n = 1), 5 (n = 2), 11 (n = 1), 15 (n = 2), 22 (n = 2), 28 (n = 2), 42 (n = 1), 60 (n = 1) and 180 (n = 1). All dogs were of medium size and were offspring of German Shepherds, or mongrels of German Shepherd, Karst Shepherd, Rottweiler and Labrador Retriever parents, while the 6-month-old dog belonged to the Chow-Chow breed. The puppies had no apparent neuromuscular deficiencies. The muscles of six healthy, mature dogs (mongrels with a body mass of 25–40 kg) were included in the study for comparative purposes.
The samples were taken within 15 min after death, frozen by submersion in liquid nitrogen and stored at −80 °C. The middle portions of the following muscles were extracted: rhomboideus (p. capitis), longissimus dorsi (at the level of the last rib), diaphragm, triceps brachii (c. longum), extensor carpi radialis, sartorius (p. cranialis), semitendinosus, rectus femoris and the cranial tibial muscle. Transverse serial cryosections (10 µm) were cut on Leica CM 1800 cryostat at −17 °C, mounted on 3-aminopropyltriethoxysilane (APES)-covered slides and air-dried.
Immunohistochemistry
To demonstrate the MHC isoform expression, different monoclonal antibodies specific to MHC isoforms of different mammals were used. MHC-I was revealed with the antibody MHC-slow (diluted 1 : 40 in PBS) supplied by Novocastra Laboratories (Newcastle upon Tyne, UK). To demonstrate the expression of fast MHC isoforms, the following antibodies were used: A4.74 (diluted 1 : 40 in PBS), which is specific to MHC-IIa in humans and rodents but which also reacts with MHC-IIX in dogs (Smerdu et al. 2005); F113.15F4, which is specific to MHC-IIa and -IIb in various species (diluted 1 : 20 in PBS), both supplied by Alexis Biochemicals (San Diego, CA, USA); as well as SC-71, which is specific to MHC-IIa in rodents but also MHC-IIx in some other species (for an overview see Acevedo & Rivero, 2005). To determine MHC-IIx, negative fibres following BF-35 staining were assessed because this antibody is specific to all MHC isoforms except MHC-IIx (Schiaffino et al. 1989). For the detection of developmental isoforms, monoclonal antibodies MHC-d and MHC-n, supplied by Novocastra (diluted 1 : 20) as well as F1.652 and F158.4C10, both supplied by Alexis Biochemicals and diluted 1 : 10, were used. These four antibodies were directed against MHC-emb and MHC-neo, respectively.
Serial cryosections were air-dried, washed with PBS and then incubated with the primary antibody in a humidified box overnight at 4 °C. A Novostain Super ABC Kit (Novocastra Laboratories) was used to reveal the immunohistochemical reaction according to the manufacturer's instructions. The stained sections were then dehydrated and mounted with Synthetic Mountant (Shandon, Pittsburgh, PA, USA).
mATPase histochemistry
To determine the fibre types in canine skeletal muscles, the sections were processed for the mATPase reaction using some of the procedures described by Snow et al. (1982) and modified by Latorre et al. (1993a). The sections were incubated either in 0.1 m Na-acetate at pH 4.2, 4.3 or 4.35 or in 0.2 m Na-acetate at pH 4.4, 4.5 or 4.6 for 5 min at room temperature. For the alkaline pre-incubation, solutions of 0.1 m CaCl2, 0.07 m Na-acetate and 0.075 m Na-barbital adjusted to pH 9.8 and 10.2 were used for 15 min at room temperature. After the pre-incubation, the sections were washed and incubated in a solution of 0.1 m CaCl2, 0.07 m Na-acetate and 0.075 m Na-barbital, pH 9.45, with 1.5 mg mL−1 of ATP for 60 min (following the acid pre-incubation) or 30 min (following the alkaline pre-incubation) at 37 °C.
Metabolic profile of muscle fibres
To estimate the fibres’ basic metabolic profile, the activity of the oxidative enzyme, SDH, and the glycolytic enzyme, mitochondrial menadione-linked α-GPDH, were demonstrated as previously described by Nachlas et al. (1957) and Dubowitz & Brooke (1973), respectively.
Analysis of serial cryosections
The serial cryosections were analysed with a Nikon Microphot FXA microscope (Nikon Instruments Europe B.V., Badhoevedorp, The Netherlands) and a Lucia-G image analysing system (Laboratory Imaging Ltd, Prague, Czech Republic). The mean number of fibres expressing the MHC isoform in question was calculated from approximately 400 evaluated muscle fibres from each sample. The threshold for significant expression of the MHC isoform was set at 10% grey matter content.
Results
Morphology of neonatal skeletal fibres and their subsequent development
Considerable differences were found in the basic morphology of skeletal fibres among prenatal, perinatal and postnatal muscles. As shown in Fig. 1, large primary myotubes and smaller secondary myotubes could clearly be seen in developing muscles several days before birth. There were wide interstitial spaces with loose connective tissue. Using the mATPase staining technique, there was no difference in staining between the primary and secondary myotubes regardless of the pre-incubation media used (Fig. 1A,B). Just before birth, i.e. on fetal day 60, the typical central space inside the myotubes started to disappear, thus forming the secondary fibres (Fig. 1C) and also the primary ones after birth (day 1) (Fig. 1D). At the same time, on F60, fibre differentiation after mATPase staining appeared, with the primary myotubes/myofibres remaining bright and the secondary fibres intensively dark after acid or alkaline pre-incubations. A few moderately stained fibres could be observed in the cranial tibial extensor as early as F60. Intermediately stained fibres appeared in other muscles when the animals were between 2 and 3 weeks of age (Fig. 1F,G). The majority of the muscles could not be classified as mature (type I fibres stain dark after acid pre-incubation) until the sixth week (Fig. 1H).
Fig. 1.
The activity of mATPase after acid pre-incubation at pH 4.4 at prenatal days F50 (A), F55 (B) and F60 (C), and postnatal days 1 (D), 5 (E), 15 (F), 28 (G), 42 (H) and 62 (I) in the longissimus dorsi muscle. Arrows indicate the primary myotubes and arrowheads the secondary myotubes. In C–F the slow fibres are bright; in G, the intermediate phase, the undifferentiated fibres are the darkest; and in H and I the slow and undifferentiated fibres are dark. Scale bar, 50 µm.
During the period studied, the average diameter of the fibres increased constantly. The mean diameter of the primary fibres was on average twice that of the secondary fibres up to 6 weeks postpartum. Also, the average number of secondary fibres per fascicle increased in the neonatal period. At day F50 there were only 8–10 fibres in one primary fascicle, at F60 20–30 fibres and by the fifth day postpartum the number of fibres increased to an average of 40 in the extensor carpi radialis and up to an average of 50 in the other muscles.
Expression of embryonic and neonatal MHC isoforms
The reactivity of antibodies stated to be specific to developmental isoforms was estimated in comparison with other immunohistochemical reactions, as well as with the mATPase method in this study and is demonstrated in Table 1.
Table 1.
Specificity of antibodies directed to MHC isoforms used in our study on dog skeletal muscles
| MHC isoform | |||||
|---|---|---|---|---|---|
| Antibody | emb | neo | I (β) | IIa | IIx |
| BF-35 | + | + | + | + | – |
| SC-71 | – | – | – | + | –/+ |
| A4.74 | – | – | – | + | + |
| F113.15F4 | – | – | – | + | + |
| F158.4C10 | – | + | – | – | – |
| F1.652 | + | – | – | – | – |
| MHC-F | +* | –/+* | – | +* | + |
| MHC-S | – | – | + | – | – |
| MHC-D | + | – | – | – | – |
| MHC-N | – | – | – | – | – |
–/+, weak staining
variable staining intensity.
MHC-emb was assessed with antibodies MHC-D and F1.652. This isoform was present in all skeletal fibres of all muscles in the prenatal and perinatal period but the primary fibres exhibited a weaker reaction (Fig. 2A–D). Postnatally, the expression of this isoform diminished: obvious differences in the reactivity of the secondary fibres were first noted by day 15 in the extensor carpi radialis muscle and the diaphragm. By day 22, a weakening of fibre staining was noted in all muscles (Fig. 2E,F). At 2 months of age the expression of MHC-emb was restricted to a few solitary fibres in the longissimus dorsi, rectus femoris, sartorius and semitendinosus muscles.
Fig. 2.
Immunohistochemical reactions with the MHC-D antibody directed against MHC-emb in the diaphragm (left column) and the triceps brachii muscle (right column) on days F60 (A,B), 5 (C,D), 22 (E,F) and 42 (G,H). Note the weakening of the MHC-emb expression and the increase in fibre diameter; the most reactive fibres are, however, the smallest. Magnification 200×.
MHC-neo was strongly expressed in all fibres in the prenatal and perinatal period (Fig. 3A–D), except in the diaphragm, rhomboideus and sartorius muscles, where the primary fibres exhibited a weaker reaction with the F158.4C10 antibody after day F60. Differences in the reactivity of the secondary fibres (Fig. 3E,F) were first noted in the diaphragm on day 22. On day 28, these were also noted in other muscles, except in the longissimus dorsi and rectus femoris, which lost MHC-neo reactivity as late as day 42. The expression of MHC-neo gradually declined but solitary F158.4C10-positive fibres with a small diameter were still found at 6 months in large muscles such as the triceps brachii, rectus femoris and longissimus dorsi.
Fig. 3.
Immunohistochemical reactions with the F158.4C10 antibody directed against MHC-neo in the diaphragm (left column) and the triceps brachii muscle (right column) on days 1 (A,B), 15 (C,D), 28 (E,F) and 60 (G,H). Note the weakening of the MHC-neo expression and the increase in fibre diameter. Magnification 200×.
Expression of adult slow and fast MHC isoforms
The reactivity of the antibodies directed against the adult MHC isoforms as shown in Table 1 was assessed in our previous studies (Štrbenc et al. 2004; Smerdu et al. 2005). Additionally, the SC-71 antibody was demonstrated to have a strong affinity to MHC-IIa and an approximately 30% weaker affinity to MHC-IIx in dogs (Acevedo & Rivero, 2005).
MHC-I (MHC-β) was expressed exclusively in the primary fibres from the prenatal period up to postnatal day 15, except in the rhomboideus muscle, where some secondary slow fibres were noted as early as day F50. The number of slow fibres increased after the second postnatal week. Between days 42 and 62 their number was higher than in adult animals and in most of the muscles the proportion of slow fibres had decreased by day 180 (Fig. 4).
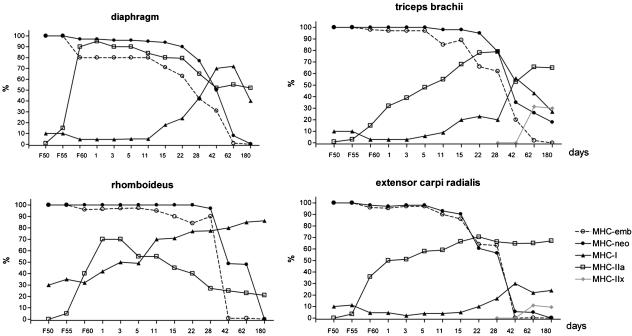
Fig. 4.
Proportion of fibres expressing different isoforms in the diaphragm, triceps brachii, rhomboideus and extensor carpi radialis muscles during postnatal development. There was a prominent switch in the high expression rates of the developmental isoforms (MHC-emb and MHC-neo) and the adult MHC-I and MHC-IIa between days 28 and 42, i.e. the fourth and sixth weeks of age. The decrease in developmental isoforms and increase in adult fast isoforms occurred earlier in the diaphragm. The line for MHC-IIa also includes hybrid IIA/IIX fibres, which react intensively with the SC-71 antibody. The strong expression of MHC-IIx (pure IIX fibres or hybrid with a predominance of MHC-IIx) was assessed by negative BF-35 staining and fibre-to-fibre comparison of SC-71 and A4.74 reactions. The rhomboideus muscle exhibited the highest percentage of slow fibres in neonates. The number of slow fibres gradually increased during postnatal development in all muscles. After the initial neonatal increase, the number of MHC-IIa fibres decreased in the slow rhomboideus muscle.
MHC-IIa was not yet expressed at day F50, but on day F55 most of the secondary fibres reacted positively with A4.74, F113.15F4 and SC-71 antibodies. The newborn animals had the most prominent expression of MHC-IIa in the diaphragm, extensor carpi radialis and cranial tibial muscle. MHC-IIa expression increased postnatally but the immunopositive fibres may have included hybrid MHC-IIa/IIx fibres as well, given that none of the antibodies used is exclusively MHC-IIa specific in dogs.
The MHC-IIx isoform was demonstrated using the principle of exclusion with the BF-35 antibody on a fibre-to-fibre basis. Fibres strongly positive with A4.74, weakly positive with SC-71 and very weak or negative with BF-35 were considered as pure IIX or hybrid IIA/IIX with prominent MHC-IIx expression. They first appeared in the longissimus dorsi, triceps brachii and cranial tibial muscle in the 2-month-old animal. The same muscles, along with the extensor carpi radialis and semitendinosus, were found to contain a higher content of MHC-IIx in adult animals.
Temporal expression of MHC isoforms in different muscles
Figure 4 compares the expression of different MHC isoforms in four selected muscles. The increase in the number of fibres expressing MHC-I or MHC-IIa correlated with diminishing reactions against developmental isoforms. The decrease in reactivity with antibodies specific to MHC-neo lagged behind the decrease in MHC-emb expression. The most prominent transition between the expression of developmental isoforms and the adult types MHC-I and MHC-IIa was noted between the fourth and sixth weeks. The general pattern of this transition was the same in all of the muscles investigated, but with some exceptions. The percentage of fibres expressing MHC-emb and MHC-neo decreased most rapidly in the diaphragm where the fastest, and simultaneous, increase in A4.74/SC-71-positive fibres occurred. A relatively rapid decrease in developmental isoforms was also noted in the extensor carpi radialis and cranial tibial muscle. In the rhomboideus muscle, the proportion of slow fibres (MHC-I) was significantly higher than in other muscles and the number of SC-71-positive fibres decreased after day 3. The triceps brachii, longissimus dorsi and rectus femoris were the slowest to gain a high expression of MHC-IIa. The IIX fibres, which are negative to BF-35, were only established in the longissimus dorsi, triceps brachii and cranial tibial muscle after the second month. Comparing the results of the immunohistochemical reactions on the serial sections, we established that in the neonatal and early postnatal periods most of the fibres were hybrid – expressing simultaneously MHC-emb, MHC-neo and MHC-IIa (Fig. 5).
Fig. 5.
Immunohistochemical demonstration of MHC isoforms in the cranial tibial muscle in a newborn puppy. Immunostaining with A4.74 for MHC-IIa/x (A), F158.4C10 for MHC-neo (B), MHC-s for MHC-I (C) and MHC-D for MHC-emb (D). All muscle fibres express developmental isoforms MHC-emb and MHC-neo. Primary fibres (p) have a weaker reaction with antibodies against MHC-emb (D) and a very strong expression of the slow isoform (C). Secondary fibres (s) have a varied expression of adult fast isoforms (A). Scale bar, 50 µm.
Comparison between MHC isoform content and histochemical characteristics of developing muscle fibres
Up to 3 weeks of age, no clear distinction between fibres could be drawn using the mATPase method, although the MHC-IIa content varied considerably. A few secondary fibres with moderate staining were observed on postnatal days 5, 11 and 15 in some muscles but a clear correlation between the mATPase staining properties and immunohistochemistry could not be drawn until after the third week. Not withstanding this, the highest activity of mATPase was noted in the fibres with the most prominent MHC-emb expression regardless of their MHC-I or MHC-IIa content (Fig. 6). The staining intensity of the primary slow fibres was similar to that of the more mature fast fibres with diminishing MHC-emb. The staining intensity of the secondary slow fibres varied from intense (coexpressing MHC-emb and MHC-I) to low (weak expression of MHC-emb). However, in older puppies, up to 50% of the fibres still expressing MHC-emb did not retain mATPase activity after acid pre-incubations, but did after the alkaline pre-incubation. At 2 months of age, mATPase fibre type classification followed the pattern previously described in adult animals (Štrbenc et al. 2004).
Fig. 6.
Muscle fibres in the cranial tibial muscle at 3 weeks of age. Activity of mATPase after pre-incubation at pH 4.5 (A); and immunohistochemical demonstration of MHC-I (B), MHC-emb (C) and MHC-neo (D). The mATPase staining is most intensive in fibres with a prominent MHC-emb expression (arrows). Primary slow fibres (I) have a medium staining intensity in the mATPase reaction; besides MHC-emb they also ceased to express MHC-neo. The mATPase staining intensity of the secondary slow fibres (arrowheads) varied according to their MHC-emb content, i.e. fibres with more prominent MHC-emb expression are darker. Scale bar, 50 µm.
The histochemical demonstration of metabolic enzymes SDH and α-GPDH revealed a weak staining in neonatal muscles but an increase in intensity thereafter. The first differences in the staining intensities among fibres were noted in the diaphragm 3 weeks postpartum. The reaction was comparable with that in adult muscles at 2 months of age. As expected, more prominent SDH staining and weaker α-GPDH was found in slow and hybrid (IIC) fibres, but a certain number of fibres expressing fast isoforms also had prominent aerobic-oxidative properties (Fig. 7).
Fig. 7.
The reaction for the glycolytic enzyme α-GPDH (A) and oxidative enzyme SDH (B), and immunohistochemical demonstration of fast fibres (MHC-IIa and MHC-IIx) (C) and slow fibres (MHC-I) (D) in the rectus femoris muscle of a 2-month-old dog. Slow fibres (I) and hybrid MHC-I/IIa (IIC) fibres have a high SDH activity and slightly weaker α-GPDH. The strongest reaction to the oxidative enzyme was noted in some fast fibres (x). Scale bar, 100 µm.
Discussion
The results of this study indicate that during development MHC isoforms in canine skeletal muscles follow the same transition pattern as reported in other animals. The immature muscle fibres intensively expressed MHC-emb and MHC-neo, which were replaced postpartum by adult isoforms, in particular MHC-IIa and MHC-I. Maturation was almost complete by 2 months postpartum, and it differed slightly among the analysed muscles. Because it is known that the degree of muscle fibre differentiation in neonates depends on the length of gravidity and physiological demands at the time of birth in a given species (Dubowitz, 1965), it was to be expected that the muscles would appear relatively immature in neonate dogs. Indeed, the degree of maturity of muscles in neonatal calves (Picard et al. 1994; Gagnière et al. 1999) was found in samples from a 6-week-old dog. The muscles of neonatal piglets are less developed than those of calves, but the developmental isoforms start to diminish on the second day postpartum (Lefaucheur et al. 1995, 2002) whereas in dogs this was first noted later, on the eleventh day postpartum. By contrast, rodent and rabbit muscles still have a fetal morphology in neonates (Gondret et al. 1996; Schiaffino & Reggiani, 1996) while in dogs we noted a rapid maturation of myotubes just before birth.
Morphology of neonatal skeletal fibres and their subsequent development
We found that the morphology of the canine muscle fibres in the perinatal period was relatively immature, and also that the number of secondary muscle fibres in primary muscle fascicles increased during this period. It was reported that, in rodents, myogenesis proceeds for a while after birth (Wigston & English, 1992), whereas in larger mammals the number of muscle fibres should be definite at the time of birth (Picard et al. 2002). However, there is a plausible presumption that by the end of gravidity in humans (Draeger et al. 1987) and cattle (Gagnière et al. 1999), the tertiary myotubes form in larger muscles. Fibres with a small diameter and late persisting neonatal isoforms (presumably the third generation) have also been described in sheep and pigs (Mascarello et al. 1992; Wilson et al. 1992; Lefaucheur et al. 1995), but not earlier than postpartum. Our finding of a fibre number increase after birth could be deceptive as very small fibres (diameter < 3 µm) could not be included in the count owing to software resolution limits. Nevertheless, a similarly small number (20–30 fibres per fascicle) was reported in neonate puppies by Braund & Lincoln (1981); and Lanfossi et al. (1999) described the appearance of very small fibres with intensive developmental isoform expression during the first month. In our study, fibres with a very small diameter and MHC-neo expression were still found at 2 and 6 months of age. Similarly, the continuing expression of MHC-neo until puberty was reported in rats and rabbits (Gondret et al. 1996; di Maso et al. 2000). The continuing myogenesis in neonate puppies could belong to a third wave of myotubes, which so far has not been proposed in carnivores. The small fibres might therefore represent a third generation of fibres that would differentiate under extrinsic signals at the beginning of puberty. The process is probably restricted to muscles with a greater mass but further studies would be necessary to confirm the existence of a third myotube generation. In view of the recent interest in the myogenic capacity of satellite or other stem cells in adult animals, it is clear that myogenesis is a continuing process and that the origin and functioning of satellite cells is not yet fully understood (for reviews see Hawke & Garry, 2001; Dhawan & Rando, 2005). Therefore, an early postnatal contribution of so-called satellite cells to the total fibre number should also be taken into consideration.
Postnatal MHC isoform transitions
In the early postnatal period the majority of muscle fibres in our study were hybrid, expressing MHC-emb, MHC-neo and MHC-IIa. This finding is in agreement with that of Lanfossi et al. (1999) and comparable with the results found in cats (Unguez et al. 2000). Results indicate that developing isoforms replace first each other (MHC-neo instead of MHC-emb) and are later replaced by adult isoforms MHC-I, MHC-IIa and MHC-IIx.
It seems that the MHC-IIx isoform starts to appear later than MHC-IIa given that secondary fibres have a comparable staining pattern with the A4.74 and SC-71 antibodies in newborns. A small number of fibres reacting strongly to the A4.74 antibody, but weakly to SC-71 (in appropriate dilutions), first appeared in the 2-month-old dog. BF-35-negative fibres also appeared in the 2-month-old animal, but were restricted to muscles that exhibit a high content of MHC-IIx in adult animals. Similarly, the MHC-IIx band was not apparent via SDS-PAGE until the second month (data not shown). The developing fibres are multiple hybrids, and developmental isoforms are likely to obscure the subtleties in staining intensities with SC-71 or BF-35 antibodies. A possibility for the future would therefore be to look for the first expression of MHC-IIx with the single-fibre electrophoresis technique.
In the perinatal period, only the primary slow fibres expressed MHC-I (2.5–7% of muscle fibres). The secondary slow fibres, developing from secondary myotubes, can follow a direct transition from MHC-neo to MHC-I or an indirect transition via MHC-IIa (Schiaffino & Reggiani, 1996). In our study, all secondary fibres expressed MHC-IIa by 11 days after birth (except in the rhomboideus), whereas only some of the secondary fibres started to express MHC-I between weeks 2 and 3 postpartum, with the fast muscles being the last to exhibit an increase in the number of slow fibres. We conclude therefore that in general the secondary slow fibres form indirectly in the sequence emb/neo → emb/neo/lla → neo/lla → neo/lla/I → I.
In comparison with what was found in studies of related species, dog muscles expressed the fast adult isoform MHC-IIa faster than cat muscles (Hoh et al. 1988a,b), although the MHC-IIa content increased significantly only after MHC-emb declined. MHC-neo was present up to 6 months in some muscles, which is much longer than studies have shown in cats. Another study of cats revealed traces of MHC-emb until day 60, but the most prominent transition between developmental and adult isoforms was reported from day 20 to day 30 (Unguez et al. 2000). In cats, MHC-IIx appeared between days 40 and 60 in fast and slow muscle, whereas in our study this isoform was difficult to confirm until the negative reaction with BF-35 was observed in certain muscles in the 2-month-old dog. It is likely that, as in cats, small amounts of MHC-IIx were previously expressed. Comparing these results, we concluded that dogs and cats have very similar patterns of MHC isoform transitions during their development. The timing was similar until the sixth week of age but thereafter the maturing of dog muscles seems to lag behind that of cat muscles. The behaviour and the exercise abilities of kittens and puppies at this age are still fairly comparable. However, the onset of puberty occurs earlier in cats and this is likely to be revealed in the maturation of muscle.
We must point out that the results of our study only indicate a general timing of MHC transitions for muscle maturation in dogs. According to anamneses, the influences of various factors on muscle development (hormones, nutrition, temperature and exercise) were regarded to be within normal limits for our samples, which all belonged to medium-sized dogs. More samples would be needed to analyse in depth the most active period noted, between the fourth and sixth weeks.
Differences among functionally different muscles
Comparative studies of rats, hamsters, guinea-pigs and rabbits have shown that the MHC isoform transition patterns are similar in different animals, but are time-specific for functionally different muscles. The developmental-to-adult isoform transitions occurred earlier in the semitendinosus muscle in rabbits and in the masseter in rats. The diaphragm showed the fastest maturation in all species studied (d’Albis et al. 1989a,b, 1991). Different timing of muscle maturation in different muscles has been reported in other animal species, such as cattle and cats (Hoh et al. 1988a,b; Picard et al. 1994). Accordingly, in our study, the diaphragm, which was fully functional at the time of birth, matured faster than other trunk and limb muscles. Also, in fast muscles, the decrease of developmental isoforms was more rapid.
The appearance of secondary slow fibres in the prenatal period was unique to the rhomboideus muscle. Such a feature has been described in the soleus muscle in rodents and cats (Butler-Browne & Whalen, 1984; Hoh et al. 1988b; Unguez et al. 2000). The number of slow fibres was increasing and the number of MHC-IIa fibres decreasing postpartum, therefore following the same transition pattern as in other muscles, as described in the slow soleus muscle by Wigston & English (1992). In our study, the rhomboideus muscle was the only relatively slow muscle in adult animals, as shown previously (Štrbenc et al. 2004), and was selected as an alternative to the non-existent soleus in dogs. Therefore, we conclude that an early distinction between slow and fast muscles can be seen in dogs as well.
Immunohistochemistry compared with mATPase and metabolic enzymes
It is well known that in fetal muscle samples the mATPase method provides odd results for fibre classification, and only the primary fibres are normally recognized as type I. We demonstrated that a small number of intermediate fibres, possible future IIA fibres, arise earlier in faster developing muscles. Braund & Lincoln (1981) noted that 1–6% of fibres remained undifferentiated as late as the tenth week and we assume that these probably correspond to the fibres from our study that had a small diameter and a persisting expression of MHC-emb and/or MHC-neo. Hyodo et al. (1999) described a ‘regular’ or adult histochemical profile of developing dog pharyngeal muscles at 9 weeks of age. Similarly, we noted a high degree of agreement between the mATPase method and the immunohistochemistry at 2 months, implying the mATPase method can be used in routine analyses after this age.
In the neonates and very young puppies, reactions demonstrating the metabolic properties of the fibres were quite weak, with the implication that the fibres are more adapted to aerobic metabolism at first. It is interesting that the differentiation of fibres according to SDH activity was quite distinctive in the 6-month-old dog, being more intense than in adult dogs. Because only one animal of this age was available for analysis, it is not clear if this was an individual or breed-dependent peculiarity. It could also suggest that the high fatigue resistance in dog muscles shown previously (Štrbenc et al. 2004) develops only after puberty.
In summary, the developmental MHC isoform transition pattern in canine skeletal muscles is similar to that in other animals. The muscle fibres of neonates express developmental isoforms, which are gradually replaced by adult isoforms and the ratio between the slow and fast isoforms generally follows the predicted adult profile of the muscle (Butler-Browne & Whalen, 1984; Hoh et al. 1988b; Harris et al. 1989; Gondret et al. 1996; Pette & Staron, 1997).
The diaphragm (which is fully functional at birth) and the fast muscles developed faster in regard to MHC transitions and metabolic profile formation. The most pronounced change from developmental to adult isoforms was noted between the fourth and sixth weeks of age. At this age puppies are weaned and begin playing energetically and investigating their surroundings, but further studies would be required to confirm the relationship between muscle maturation and the activity of animals. We also believe that further studies would indicate differences in muscle development among different breeds (early or late maturing) between 2 and 6 months of age, such as the expression of MHC-neo. Comparing our results with those from studies of other domestic animals, we have ascertained that canine skeletal muscles develop relatively slowly.
Acknowledgments
We thank Jasna Šporar, Magdalena Dobravec and Branko Travnikar for their technical assistance. This work was supported by the Ministry of Higher Education, Science and Technology, Republic of Slovenia (Grant No. P4-0053).
References
- Acevedo LM, Rivero JL. New insights into skeletal muscle fibre types in the dog with particular focus towards hybrid myosin phenotypes. Cell Tissue Res. 2005;323:283–303. doi: 10.1007/s00441-005-0057-4. [DOI] [PubMed] [Google Scholar]
- d’Albis A, Couteaux R, Janmot C, Mira JC. Myosin isoform transitions in regeneration of fast and slow muscles during postnatal development of the rat. Dev Biol. 1989a;135:320–325. doi: 10.1016/0012-1606(89)90182-6. [DOI] [PubMed] [Google Scholar]
- d’Albis A, Couteaux R, Janmot C, Roulet A. Specific programs of myosin expression in the postnatal development of rat muscles. Eur J Biochem. 1989b;183:583–590. doi: 10.1111/j.1432-1033.1989.tb21087.x. [DOI] [PubMed] [Google Scholar]
- d’Albis A, Janmot C, Couteaux R. Species- and muscle type-dependence of perinatal isomyosin transitions. Int J Dev Biol. 1991;35:53–56. [PubMed] [Google Scholar]
- Baldwin KM, Haddad F. Effects of different activity and inactivity paradigms on myosin heavy chain gene expression in striated muscle. J Appl Physiol. 2001;90:345–357. doi: 10.1152/jappl.2001.90.1.345. [DOI] [PubMed] [Google Scholar]
- Bottinelli R, Canepari M, Reggiani C, Stienen GJ. Myofibrillar ATPase activity during isometric contraction and isomyosin composition in rat single skinned mucle fibers. J Physiol. 1994;481:663–675. doi: 10.1113/jphysiol.1994.sp020472. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Braund KG, Lincoln CE. Histochemical differentiation of fiber types in neonatal canine skeletal muscle. Am J Vet Res. 1981;42:407–415. [PubMed] [Google Scholar]
- Butler-Browne GS, Whalen RG. Myosin transitions occurring during postnatal development of the rat soleus muscle. Dev Biol. 1984;102:324–334. doi: 10.1016/0012-1606(84)90197-0. [DOI] [PubMed] [Google Scholar]
- Caiozzo VJ, Baker MJ, Huang K, Chou H, Wu YZ, Baldwin KM. Single-fiber myosin heavy chain polymorphism: how many patterns and what proportions? Am J Physiol Regul Integr Comp Physiol. 2003;285:R570–R580. doi: 10.1152/ajpregu.00646.2002. [DOI] [PubMed] [Google Scholar]
- Dhawan J, Rando TA. Stem cells in postnatal myogenesis: molecular mechanisms of satellite cell quiescence, activation and replenishment. Trends Cell Biol. 2005;15:666–673. doi: 10.1016/j.tcb.2005.10.007. [DOI] [PubMed] [Google Scholar]
- Draeger A, Weeds AG, Fitzsimons RB. Primary, secondary and tertiary myotubes in developing skeletal muscle: a new approach to the analysis of human myogenesis. J Neurol Sci. 1987;81:19–43. doi: 10.1016/0022-510x(87)90181-x. [DOI] [PubMed] [Google Scholar]
- Dubowitz V. Enzyme histochemistry of skeletal muscle. Part I. Developing animal muscle. J Neurol Neurosurg Psychiatr. 1965;28:516–519. doi: 10.1136/jnnp.28.6.516. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dubowitz V, Brooke MH. Muscle biopsy: a modern approach. In: Dubowitz V, Brooke MH, editors. Major Problems in Neurology. London: Saunders; 1973. pp. 74–85. [Google Scholar]
- Fitzsimons RB, Hoh JF. Embryonic and foetal myosins in human skeletal muscle. The presence of foetal myosins in duchenne muscular dystrophy and infantile spinal muscular atrophy. J Neurol Sci. 1981;52:367–384. doi: 10.1016/0022-510x(81)90018-6. [DOI] [PubMed] [Google Scholar]
- Gagnière H, Picard B, Geay Y. Contractile differentiation of foetal cattle muscles: intermuscular variability. Reprod Nutr Dev. 1999;39:637–655. doi: 10.1051/rnd:19990510. [DOI] [PubMed] [Google Scholar]
- Gondret F, Lefaucheur L, d’Albis A, Bonneau M. Myosin isoform transitions in four rabbit muscles during postnatal growth. J Muscle Res Cell Motil. 1996;17:657–667. doi: 10.1007/BF00154060. [DOI] [PubMed] [Google Scholar]
- Harris AJ, Fitzsimons RB, McEwan JC. Neural control of the sequence of expression of myosin heavy chain isoforms in foetal mammalian muscles. Development. 1989;107:751–769. doi: 10.1242/dev.107.4.751. [DOI] [PubMed] [Google Scholar]
- Hawke TJ, Garry DJ. Myogenic satellite cells: physiology to molecular biology. J Appl Physiol. 2001;91:534–551. doi: 10.1152/jappl.2001.91.2.534. [DOI] [PubMed] [Google Scholar]
- Hoh JF, Hughes S, Chow C, Hale PT, Fitzsimons RB. Immunocytochemical and electrophoretic analyses of changes in myosin gene expression in cat posterior temporalis muscle during postnatal development. J Muscle Res Cell Motil. 1988a;9:48–58. doi: 10.1007/BF01682147. [DOI] [PubMed] [Google Scholar]
- Hoh JF, Hughes S, Hale PT, Fitzsimons RB. Immunocytochemical and electrophoretic analyses of changes in myosin gene expression in cat limb fast and slow muscles during postnatal development. J Muscle Res Cell Motil. 1988b;9:30–47. doi: 10.1007/BF01682146. [DOI] [PubMed] [Google Scholar]
- Hughes SM, Cho M, Karsch-Mizrachi I, et al. Three slow myosin heavy chains sequentially expressed in developing mammalian skeletal muscle. Dev Biol. 1993;158:183–199. doi: 10.1006/dbio.1993.1178. [DOI] [PubMed] [Google Scholar]
- Hyodo M, Yumoto E, Kawakita S, Yamagata T. Postnatal changes in the types of muscle fibre in the canine inferior pharyngeal constrictor. Acta Otolaryngol. 1999;119:843–846. doi: 10.1080/00016489950180522. [DOI] [PubMed] [Google Scholar]
- LaFramboise WA, Daood MJ, Guthrie RD, et al. Emergence of the mature myosin phenotype in the rat diaphragm muscle. Dev Biol. 1991;144:1–15. doi: 10.1016/0012-1606(91)90473-g. [DOI] [PubMed] [Google Scholar]
- Lanfossi M, Cozzi F, Bugini D, et al. Development of muscle pathology in canine X-linked muscular dystrophy. I. Delayed postnatal maturation of affected and normal muscle as revealed by myosin isoform analysis and utrophin expression. Acta Neuropathol (Berl) 1999;97:127–138. doi: 10.1007/s004010050965. [DOI] [PubMed] [Google Scholar]
- Latorre R, Gil F, Vazquez JM, Moreno F, Mascarello F, Ramirez G. Morphological and histochemical characteristics of muscle fibre types in the flexor carpi radialis of the dog. J Anat. 1993a;182:313–320. [PMC free article] [PubMed] [Google Scholar]
- Latorre R, Gil F, Ramirez G, Vazquez JM, Lopez-Albors O, Moreno F. Postnatal development of semitendinosus muscle in the dog. Anat Embryol (Berl) 1993b;188:401–407. doi: 10.1007/BF00185949. [DOI] [PubMed] [Google Scholar]
- Lefaucheur L, Edom F, Ecolan P, Butler-Browne GS. Pattern of muscle fiber type formation in the pig. Dev Dyn. 1995;203:27–41. doi: 10.1002/aja.1002030104. [DOI] [PubMed] [Google Scholar]
- Lefaucheur L. Myofiber typing and pig meat production. Slov Vet Res. 2001;38:5–28. [Google Scholar]
- Lefaucheur L, Ecolan P, Plantard L, Gueguen N. New insights into muscle fiber types in the pig. J Histochem Cytochem. 2002;50:719–730. doi: 10.1177/002215540205000513. [DOI] [PubMed] [Google Scholar]
- Mascarello F, Stecchini ML, Rowlerson A, Ballocchi E. Tertiary myotubes in postnatal growing pig muscle detected by their myosin isoform composition. J Anim Sci. 1992;70:1806–1813. doi: 10.2527/1992.7061806x. [DOI] [PubMed] [Google Scholar]
- di Maso NA, Caiozzo VJ, Baldwin KM. Single-fiber myosin heavy chain polymorphism during postnatal development: modulation by hypothyroidism. Am J Physiol Regul Integr Comp Physiol. 2000;278:R1099–R1106. doi: 10.1152/ajpregu.2000.278.4.R1099. [DOI] [PubMed] [Google Scholar]
- Nachlas MM, Tsou KC, De Souza E, Cheng CS, Seligman AM. Cytochemical demonstration of succinic dehydrogenase by the use of a new p-nitrophenyl substituted ditetrazole. J Histochem Cytochem. 1957;5:420–436. doi: 10.1177/5.4.420. [DOI] [PubMed] [Google Scholar]
- Pette D, Staron RS. Mammalian skeletal muscle fiber type transitions. Int Rev Cytol. 1997;170:143–223. doi: 10.1016/s0074-7696(08)61622-8. [DOI] [PubMed] [Google Scholar]
- Pette D, Peuker H, Staron RS. The impact of biochemical methods for single muscle fibre analysis. Acta Physiol Scand. 1999;166:261–277. doi: 10.1046/j.1365-201x.1999.00568.x. [DOI] [PubMed] [Google Scholar]
- Picard B, Robelin J, Pons F, Geay Y. Comparison of the foetal development of fibre types in four bovine muscles. J Muscle Res Cell Motil. 1994;15:473–486. doi: 10.1007/BF00122120. [DOI] [PubMed] [Google Scholar]
- Picard B, Lefaucheur L, Berri C, Duclos MJ. Muscle fibre ontogenesis in farm animal species. Reprod Nutr Dev. 2002;42:415–431. doi: 10.1051/rnd:2002035. [DOI] [PubMed] [Google Scholar]
- Schiaffino S, Gorza L, Sartore S, et al. Three myosin heavy chain isoforms in type 2 skeletal muscle fibres. J Musc Res Cell Motil. 1989;10:197–205. doi: 10.1007/BF01739810. [DOI] [PubMed] [Google Scholar]
- Schiaffino S, Reggiani C. Molecular diversity of myofibrillar proteins: gene regulation and functional significance. Physiol Rev. 1996;76:371–423. doi: 10.1152/physrev.1996.76.2.371. [DOI] [PubMed] [Google Scholar]
- Smerdu V, Štrbenc M, Meznarič-Petruša M, Fazarinc G. Identification of myosin heavy chain I, IIa and IIx in skeletal muscles of dog by an electrophoretic and immunoblotting study. Cells Tissues Organs. 2005;180:106–116. doi: 10.1159/000086751. [DOI] [PubMed] [Google Scholar]
- Štrbenc M, Smerdu V, Zupanc M, Tozon N, Fazarinc G. Pattern of myosin heavy chain isoforms in different fibre types of canine trunk and limb skeletal muscles. Cells Tissues Organs. 2004;176:178–186. doi: 10.1159/000077034. [DOI] [PubMed] [Google Scholar]
- Talmadge RJ. Myosin heavy chain isoform expression following reduced neuromuscular activity: potential regulatory mechanisms. Muscle Nerve. 2000;23:661–679. doi: 10.1002/(sici)1097-4598(200005)23:5<661::aid-mus3>3.0.co;2-j. [DOI] [PubMed] [Google Scholar]
- Unguez GA, Talmadge RJ, Roy RR, Dalponte D, Edgerton VR. Distinct myosin heavy chain isoform transitions in developing slow and fast cat hindlimb muscles. Cells Tissues Organs. 2000;167:138–152. doi: 10.1159/000016777. [DOI] [PubMed] [Google Scholar]
- Wigston DJ, English AW. Fiber-type proportions in mammalian soleus muscle during postnatal development. J Neurobiol. 1992;23:61–70. doi: 10.1002/neu.480230107. [DOI] [PubMed] [Google Scholar]
- Wilson SJ, McEwan JC, Sheard PW, Harris AJ. Early stages of myogenesis in a large mammal: formation of successive generations of myotubes in sheep tibialis cranialis muscle. J Muscle Res Cell Motil. 1992;13:534–550. doi: 10.1007/BF01737996. [DOI] [PubMed] [Google Scholar]
- Wu YZ, Baker MJ, Crumley RL, Blanks RH, Caiozzo VJ. A new concept in laryngeal muscle: multiple myosin isoform types in single muscle fibers of the lateral cricoarytenoid. Otolaryngol Head Neck Surg. 1998;118:86–94. doi: 10.1016/S0194-5998(98)70380-8. [DOI] [PubMed] [Google Scholar]
- Wu YZ, Crumley RL, Caiozzo VJ. Are hybrid fibers a common motif of canine laryngeal muscles? Single-fiber analyses of myosin heavy-chain isoform composition. Arch Otolaryngol Head Neck Surg. 2000;126:865–873. doi: 10.1001/archotol.126.7.865. [DOI] [PubMed] [Google Scholar]
- Zhang KM, Hu P, Wang SW, et al. Salbutamol changes the molecular and mechanical properties of canine skeletal muscle. J Physiol. 1996;496:211–220. doi: 10.1113/jphysiol.1996.sp021678. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang KM, Wright LD, Hu P, Spratt JA, Wechsler AS, Briggs FN. Induction of molecular and mechanical transformations in canine skeletal muscle by chronic neuromuscular stimulation. J Muscle Res Cell Motil. 1997;18:81–90. doi: 10.1023/a:1018685001214. [DOI] [PubMed] [Google Scholar]
- Snow DH, Billeter R, Mascarello F, Carpene E, Rowlerson A, Jenny E. No classical IIB fibres in dog skeletal muscle. Histochem. 1982;75:53–65. doi: 10.1007/BF00492533. [DOI] [PubMed] [Google Scholar]