Abstract
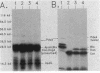
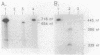
We report that pdxA, which is required for de novo biosynthesis of pyridoxine (vitamin B6) and pyridoxal phosphate, belongs to an unusual, multifunctional operon. The pdxA gene was cloned in the same 3.5-kilobase BamHI-EcoRI restriction fragment that contains ksgA, which encodes the 16S rRNA modification enzyme m6(2)A methyltransferase, and apaH, which encodes diadenosine tetraphosphatase (ApppA hydrolase). Previously, Blanchin-Roland et al. showed that ksgA and apaH form a complex operon (Mol. Gen. Genet. 205:515-522, 1986). The pdxA gene was located on recombinant plasmids by subcloning, complementation, and insertion mutagenesis, and chromosomal insertions at five positions upstream from ksgA inactivated pdxA function. DNA sequence analysis and minicell translation experiments demonstrated that pdxA encoded a 35.1-kilodalton polypeptide and that the stop codon of pdxA overlapped the start codon of ksgA by 2 nucleotides. The translational start codon of pdxA was tentatively assigned based on polypeptide size and on the presence of a unique sequence that was also found near the translational start of PdxB. This conserved sequence may play a role in translational control of certain pyridoxine biosynthetic genes. RNase T2 mapping of chromosomal transcripts confirmed that pdxA and ksgA were members of the same complex operon, yet about half of ksgA transcripts arose in vivo under some culture conditions from an internal promoter mapped near the end of pdxA. Transcript analysis further suggested that pdxA is not the first gene in the operon. These structural features support the idea that pyridoxine-biosynthetic genes are members of complex operons, perhaps to interweave coenzyme biosynthesis genetically with other metabolic processes. The results are also considered in terms of ksgA expression.
Full text
PDF










Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Andrésson O. S., Davies J. E. Genetic organization and restriction enzyme cleavage map of the ksgA-pdxA region of the Escherichia coli chromosome. Mol Gen Genet. 1980;179(1):211–216. doi: 10.1007/BF00268465. [DOI] [PubMed] [Google Scholar]
- Arps P. J., Marvel C. C., Rubin B. C., Tolan D. A., Penhoet E. E., Winkler M. E. Structural features of the hisT operon of Escherichia coli K-12. Nucleic Acids Res. 1985 Jul 25;13(14):5297–5315. doi: 10.1093/nar/13.14.5297. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Arps P. J., Winkler M. E. An unusual genetic link between vitamin B6 biosynthesis and tRNA pseudouridine modification in Escherichia coli K-12. J Bacteriol. 1987 Mar;169(3):1071–1079. doi: 10.1128/jb.169.3.1071-1079.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Arps P. J., Winkler M. E. Structural analysis of the Escherichia coli K-12 hisT operon by using a kanamycin resistance cassette. J Bacteriol. 1987 Mar;169(3):1061–1070. doi: 10.1128/jb.169.3.1061-1070.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bartlett D. H., Matsumura P. Identification of Escherichia coli region III flagellar gene products and description of two new flagellar genes. J Bacteriol. 1984 Nov;160(2):577–585. doi: 10.1128/jb.160.2.577-585.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Björk G. R. E. coli ribosomal protein operons: the case of the misplaced genes. Cell. 1985 Aug;42(1):7–8. doi: 10.1016/s0092-8674(85)80093-3. [DOI] [PubMed] [Google Scholar]
- Blanchin-Roland S., Blanquet S., Schmitter J. M., Fayat G. The gene for Escherichia coli diadenosine tetraphosphatase is located immediately clockwise to folA and forms an operon with ksgA. Mol Gen Genet. 1986 Dec;205(3):515–522. doi: 10.1007/BF00338091. [DOI] [PubMed] [Google Scholar]
- Bochner B. R., Lee P. C., Wilson S. W., Cutler C. W., Ames B. N. AppppA and related adenylylated nucleotides are synthesized as a consequence of oxidation stress. Cell. 1984 May;37(1):225–232. doi: 10.1016/0092-8674(84)90318-0. [DOI] [PubMed] [Google Scholar]
- Bognar A. L., Osborne C., Shane B. Primary structure of the Escherichia coli folC gene and its folylpolyglutamate synthetase-dihydrofolate synthetase product and regulation of expression by an upstream gene. J Biol Chem. 1987 Sep 5;262(25):12337–12343. [PubMed] [Google Scholar]
- Castilho B. A., Olfson P., Casadaban M. J. Plasmid insertion mutagenesis and lac gene fusion with mini-mu bacteriophage transposons. J Bacteriol. 1984 May;158(2):488–495. doi: 10.1128/jb.158.2.488-495.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Costa R. H., Lai E., Darnell J. E., Jr Transcriptional control of the mouse prealbumin (transthyretin) gene: both promoter sequences and a distinct enhancer are cell specific. Mol Cell Biol. 1986 Dec;6(12):4697–4708. doi: 10.1128/mcb.6.12.4697. [DOI] [PMC free article] [PubMed] [Google Scholar]
- DEMPSEY W. B., CHRISTENSEN H. N. The specific binding of pyridoxal 5'-phosphate to bovine plasma albumin. J Biol Chem. 1962 Apr;237:1113–1120. [PubMed] [Google Scholar]
- DEMPSEY W. B. CONTROL OF PYRIDOXINE BIOSYNTHESIS IN ESCHERICHIA COLI. J Bacteriol. 1965 Aug;90:431–437. doi: 10.1128/jb.90.2.431-437.1965. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dempsey W. B. Characterization of pyridoxine auxotrophs of Escherichia coli: P1 transduction. J Bacteriol. 1969 Mar;97(3):1403–1410. doi: 10.1128/jb.97.3.1403-1410.1969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dempsey W. B. Control of vitamin B 6 biosynthesis in Escherichia coli. J Bacteriol. 1971 Oct;108(1):415–421. doi: 10.1128/jb.108.1.415-421.1971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dempsey W. B., Ito H. Characterization of pyridoxine auxotrophs of Escherichia coli: serine and pdxF mutants. J Bacteriol. 1970 Nov;104(2):658–667. doi: 10.1128/jb.104.2.658-667.1970. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Duncan K., Coggins J. R. The serC-aro A operon of Escherichia coli. A mixed function operon encoding enzymes from two different amino acid biosynthetic pathways. Biochem J. 1986 Feb 15;234(1):49–57. doi: 10.1042/bj2340049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hedrick J. L., Fischer E. H. On the role of pyridoxal 5'-phosphate in phosphorylase. I. Absence of classical vitamin B6--dependent enzymatic activities in muscle glycogen phosphorylase. Biochemistry. 1965 Jul;4(7):1337–1343. doi: 10.1021/bi00883a018. [DOI] [PubMed] [Google Scholar]
- Henikoff S., Wallace J. C. Detection of protein similarities using nucleotide sequence databases. Nucleic Acids Res. 1988 Jul 11;16(13):6191–6204. doi: 10.1093/nar/16.13.6191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hughes K. T., Roth J. R. Conditionally transposition-defective derivative of Mu d1(Amp Lac). J Bacteriol. 1984 Jul;159(1):130–137. doi: 10.1128/jb.159.1.130-137.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marvel C. C., Arps P. J., Rubin B. C., Kammen H. O., Penhoet E. E., Winkler M. E. hisT is part of a multigene operon in Escherichia coli K-12. J Bacteriol. 1985 Jan;161(1):60–71. doi: 10.1128/jb.161.1.60-71.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mechulam Y., Fromant M., Mellot P., Plateau P., Blanchin-Roland S., Fayat G., Blanquet S. Molecular cloning of the Escherichia coli gene for diadenosine 5',5'''-P1,P4-tetraphosphate pyrophosphohydrolase. J Bacteriol. 1985 Oct;164(1):63–69. doi: 10.1128/jb.164.1.63-69.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Messing J., Crea R., Seeburg P. H. A system for shotgun DNA sequencing. Nucleic Acids Res. 1981 Jan 24;9(2):309–321. doi: 10.1093/nar/9.2.309. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nomura M., Gourse R., Baughman G. Regulation of the synthesis of ribosomes and ribosomal components. Annu Rev Biochem. 1984;53:75–117. doi: 10.1146/annurev.bi.53.070184.000451. [DOI] [PubMed] [Google Scholar]
- Nonet M. L., Marvel C. C., Tolan D. R. The hisT-purF region of the Escherichia coli K-12 chromosome. Identification of additional genes of the hisT and purF operons. J Biol Chem. 1987 Sep 5;262(25):12209–12217. [PubMed] [Google Scholar]
- Plateau P., Fromant M., Blanquet S. Heat shock and hydrogen peroxide responses of Escherichia coli are not changed by dinucleoside tetraphosphate hydrolase overproduction. J Bacteriol. 1987 Aug;169(8):3817–3820. doi: 10.1128/jb.169.8.3817-3820.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sanger F., Coulson A. R. The use of thin acrylamide gels for DNA sequencing. FEBS Lett. 1978 Mar 1;87(1):107–110. doi: 10.1016/0014-5793(78)80145-8. [DOI] [PubMed] [Google Scholar]
- Sanger F., Nicklen S., Coulson A. R. DNA sequencing with chain-terminating inhibitors. Proc Natl Acad Sci U S A. 1977 Dec;74(12):5463–5467. doi: 10.1073/pnas.74.12.5463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sarmientos P., Cashel M. Carbon starvation and growth rate-dependent regulation of the Escherichia coli ribosomal RNA promoters: differential control of dual promoters. Proc Natl Acad Sci U S A. 1983 Nov;80(22):7010–7013. doi: 10.1073/pnas.80.22.7010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shine J., Dalgarno L. The 3'-terminal sequence of Escherichia coli 16S ribosomal RNA: complementarity to nonsense triplets and ribosome binding sites. Proc Natl Acad Sci U S A. 1974 Apr;71(4):1342–1346. doi: 10.1073/pnas.71.4.1342. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tadera K., Mori E., Yagi F., Kobayashi A., Imada K., Imabeppu M. Isolation and structure of a minor metabolite of pyridoxine in seedlings of Pisum sativum L. J Nutr Sci Vitaminol (Tokyo) 1985 Aug;31(4):403–408. doi: 10.3177/jnsv.31.403. [DOI] [PubMed] [Google Scholar]
- Tani Y., Dempsey W. B. Glycolaldehyde is a precursor of pyridoxal phosphate in Escherichia coli B. J Bacteriol. 1973 Oct;116(1):341–345. doi: 10.1128/jb.116.1.341-345.1973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Uyeda K. Reaction of phosphofructokinase with maleic anhydride, succinic anhydride, and pyridoxal 5'-phosphate. Biochemistry. 1969 Jun;8(6):2366–2373. doi: 10.1021/bi00834a017. [DOI] [PubMed] [Google Scholar]
- Winans S. C., Elledge S. J., Krueger J. H., Walker G. C. Site-directed insertion and deletion mutagenesis with cloned fragments in Escherichia coli. J Bacteriol. 1985 Mar;161(3):1219–1221. doi: 10.1128/jb.161.3.1219-1221.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yanisch-Perron C., Vieira J., Messing J. Improved M13 phage cloning vectors and host strains: nucleotide sequences of the M13mp18 and pUC19 vectors. Gene. 1985;33(1):103–119. doi: 10.1016/0378-1119(85)90120-9. [DOI] [PubMed] [Google Scholar]
- de Boer H. A., Gilbert S. F., Nomura M. DNA sequences of promoter regions for rRNA operons rrnE and rrnA in E. coli. Cell. 1979 May;17(1):201–209. doi: 10.1016/0092-8674(79)90308-8. [DOI] [PubMed] [Google Scholar]
- van Buul C. P., van Knippenberg P. H. Nucleotide sequence of the ksgA gene of Escherichia coli: comparison of methyltransferases effecting dimethylation of adenosine in ribosomal RNA. Gene. 1985;38(1-3):65–72. doi: 10.1016/0378-1119(85)90204-5. [DOI] [PubMed] [Google Scholar]
- van Gemen B., Koets H. J., Plooy C. A., Bodlaender J., Van Knippenberg P. H. Characterization of the ksgA gene of Escherichia coli determining kasugamycin sensitivity. Biochimie. 1987 Aug;69(8):841–848. doi: 10.1016/0300-9084(87)90210-0. [DOI] [PubMed] [Google Scholar]