Abstract
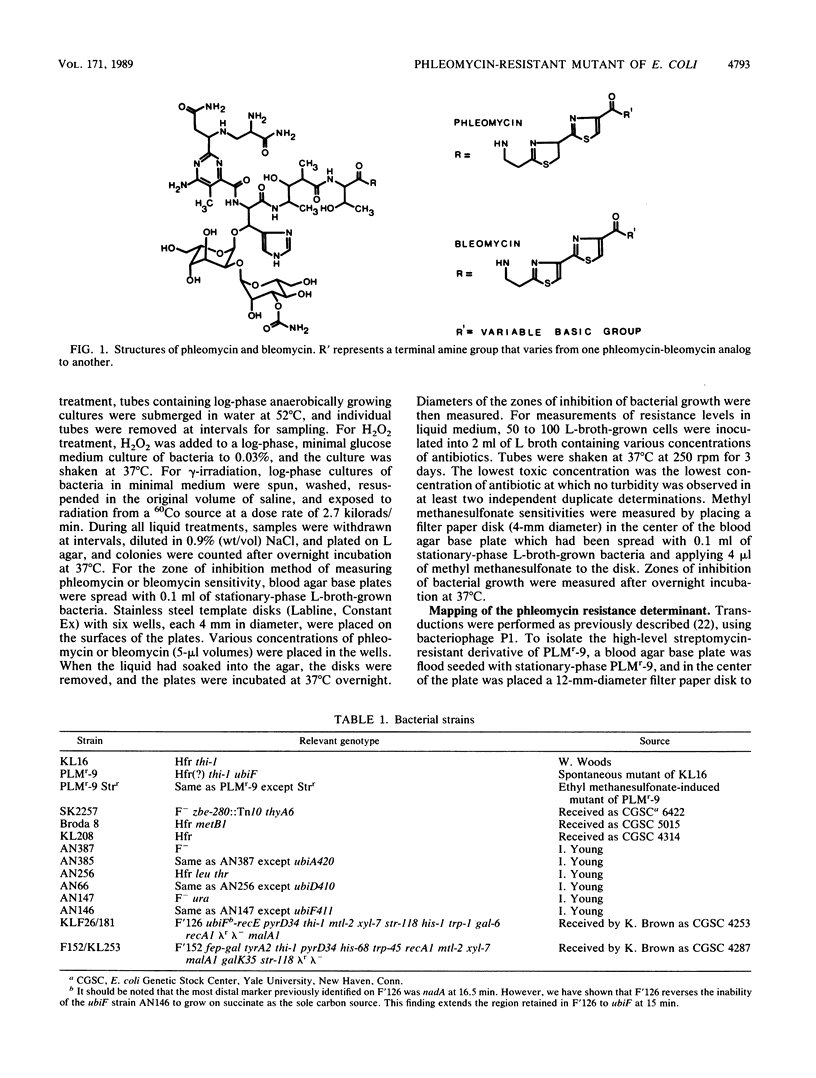
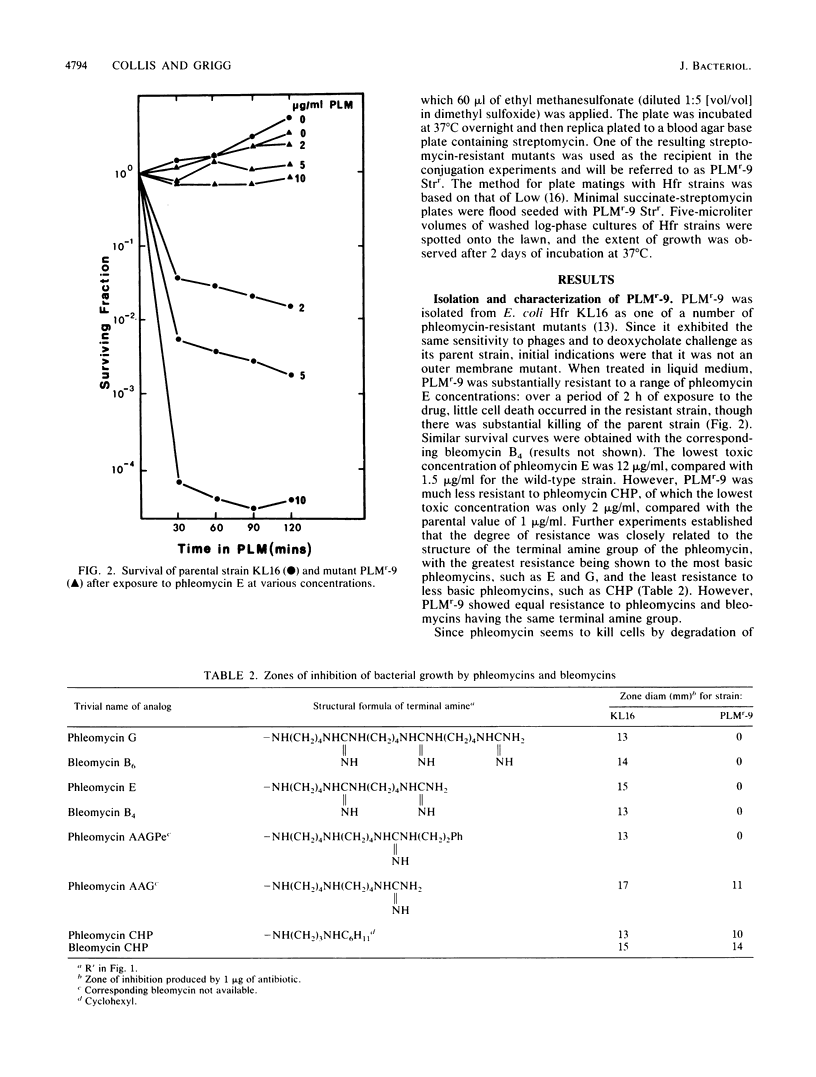
A mutant of Escherichia coli, selected for resistance to the antibiotic and antitumor agent phleomycin, has been characterized, and the phleomycin resistance determinant has been identified. The mutant is equally resistant to bleomycins. The resistance to phleomycin is strongly dependent on the nature of the C-terminal amine of the drug, with the greatest resistance being shown to phleomycins and bleomycins with the most basic terminal amines. The mutation also confers resistance to the lethal effects of heating at 52 degrees C. Other characteristics of the phleomycin-resistant strain include a slow growth rate, an inability to grow on succinate as the sole carbon source (Suc- phenotype), cross resistance to aminoglycoside antibiotics, and a slight sensitivity to hydrogen peroxide, methyl methanesulfonate, and gamma-irradiation. Some of these characteristics, together with mapping data, suggested that the phleomycin resistance and Suc- determinant probably lies within the ubiF gene coding for an enzyme effecting a step in the biosynthesis of ubiquinone. The phenotypes of known mutants defective in this and other steps of the ubiquinone pathway were found to be closely similar to those of the original phleomycin-resistant strain.
Full text
PDF




Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Berry D. E., Kilkuskie R. E., Hecht S. M. DNA damage induced by bleomycin in the presence of dibucaine is not predictive of cell growth inhibition. Biochemistry. 1985 Jun 18;24(13):3214–3219. doi: 10.1021/bi00334a021. [DOI] [PubMed] [Google Scholar]
- Bridges B. A., Ashwood-Smith M. J., Munson R. J. Correlation of bacterial sensitivities to ionizing radiation and mild heating. J Gen Microbiol. 1969 Sep;58(1):115–124. doi: 10.1099/00221287-58-1-115. [DOI] [PubMed] [Google Scholar]
- Bryan L. E., Kwan S. Roles of ribosomal binding, membrane potential, and electron transport in bacterial uptake of streptomycin and gentamicin. Antimicrob Agents Chemother. 1983 Jun;23(6):835–845. doi: 10.1128/aac.23.6.835. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bryan L. E., Van Den Elzen H. M. Effects of membrane-energy mutations and cations on streptomycin and gentamicin accumulation by bacteria: a model for entry of streptomycin and gentamicin in susceptible and resistant bacteria. Antimicrob Agents Chemother. 1977 Aug;12(2):163–177. doi: 10.1128/aac.12.2.163. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cox G. B., Gibson F. Studies on electron transport and energy-linked reactions using mutants of Escherichia coli. Biochim Biophys Acta. 1974 Apr 30;346(1):1–25. doi: 10.1016/0304-4173(74)90010-x. [DOI] [PubMed] [Google Scholar]
- Damper P. D., Epstein W. Role of the membrane potential in bacterial resistance to aminoglycoside antibiotics. Antimicrob Agents Chemother. 1981 Dec;20(6):803–808. doi: 10.1128/aac.20.6.803. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Davies J., Smith D. I. Plasmid-determined resistance to antimicrobial agents. Annu Rev Microbiol. 1978;32:469–518. doi: 10.1146/annurev.mi.32.100178.002345. [DOI] [PubMed] [Google Scholar]
- Downie J. A., Cox G. B. Sequence of b cytochromes relative to ubiquinone in the electron transport chain of Escherichia coli. J Bacteriol. 1978 Feb;133(2):477–484. doi: 10.1128/jb.133.2.477-484.1978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Freedman J. H., Horwitz S. B., Peisach J. Reduction of copper(II)-bleomycin: a model for in vivo drug activity. Biochemistry. 1982 Apr 27;21(9):2203–2210. doi: 10.1021/bi00538a032. [DOI] [PubMed] [Google Scholar]
- Grigg G. W., Collis C. M. Phleomycin and bleomycin resistance in Escherichia coli. J Antibiot (Tokyo) 1989 Mar;42(3):482–485. doi: 10.7164/antibiotics.42.482. [DOI] [PubMed] [Google Scholar]
- Grigg G. W. Induction of DNA breakdown and death in Escherichia coli by phleomycin. Its association with dark-repair processes. Mol Gen Genet. 1969;104(1):1–11. doi: 10.1007/BF00277357. [DOI] [PubMed] [Google Scholar]
- Lee P. C., Bochner B. R., Ames B. N. AppppA, heat-shock stress, and cell oxidation. Proc Natl Acad Sci U S A. 1983 Dec;80(24):7496–7500. doi: 10.1073/pnas.80.24.7496. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Low B. Rapid mapping of conditional and auxotrophic mutations in Escherichia coli K-12. J Bacteriol. 1973 Feb;113(2):798–812. doi: 10.1128/jb.113.2.798-812.1973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Newton N. A., Cox G. B., Gibson F. Function of ubiquinone in Escherichia coli: a mutant strain forming a low level of ubiquinone. J Bacteriol. 1972 Jan;109(1):69–73. doi: 10.1128/jb.109.1.69-73.1972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nikaido H., Vaara M. Molecular basis of bacterial outer membrane permeability. Microbiol Rev. 1985 Mar;49(1):1–32. doi: 10.1128/mr.49.1.1-32.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Privalle C. T., Fridovich I. Induction of superoxide dismutase in Escherichia coli by heat shock. Proc Natl Acad Sci U S A. 1987 May;84(9):2723–2726. doi: 10.1073/pnas.84.9.2723. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sedgwick S. G., Bridges B. A. Evidence for indirect production of DNA strand scissions during mild heating of Escherichia coli. J Gen Microbiol. 1972 Jun;71(1):191–193. doi: 10.1099/00221287-71-1-191. [DOI] [PubMed] [Google Scholar]
- Sleigh M. J., Grigg G. W. The mechanism of sensitivity to phleomycin in growing Escherichia coli cells. Biochem J. 1976 Apr 1;155(1):87–99. doi: 10.1042/bj1550087. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Taber H. W., Mueller J. P., Miller P. F., Arrow A. S. Bacterial uptake of aminoglycoside antibiotics. Microbiol Rev. 1987 Dec;51(4):439–457. doi: 10.1128/mr.51.4.439-457.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wallace B. J., Young I. G. Aerobic respiration in mutants of Escherichia coli accumulating quinone analogues of ubiquinone. Biochim Biophys Acta. 1977 Jul 7;461(1):75–83. doi: 10.1016/0005-2728(77)90070-6. [DOI] [PubMed] [Google Scholar]
- Wallace B. J., Young I. G. Role of quinones in electron transport to oxygen and nitrate in Escherichia coli. Studies with a ubiA- menA- double quinone mutant. Biochim Biophys Acta. 1977 Jul 7;461(1):84–100. doi: 10.1016/0005-2728(77)90071-8. [DOI] [PubMed] [Google Scholar]
- Waring M. J. DNA modification and cancer. Annu Rev Biochem. 1981;50:159–192. doi: 10.1146/annurev.bi.50.070181.001111. [DOI] [PubMed] [Google Scholar]
- Woodcock E., Grigg G. W. Repair of thermally induced DNA breakage in Escherichia coli. Nat New Biol. 1972 May 17;237(72):76–79. doi: 10.1038/newbio237076a0. [DOI] [PubMed] [Google Scholar]
- Yamamori T., Yura T. Genetic control of heat-shock protein synthesis and its bearing on growth and thermal resistance in Escherichia coli K-12. Proc Natl Acad Sci U S A. 1982 Feb;79(3):860–864. doi: 10.1073/pnas.79.3.860. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yamamoto K., Hutchinson F. Response to bleomycin of Escherichia coli mutants deficient in DNA repair. J Antibiot (Tokyo) 1979 Nov;32(11):1181–1185. doi: 10.7164/antibiotics.32.1181. [DOI] [PubMed] [Google Scholar]
- Young I. G., McCann L. M., Stroobant P., Gibson F. Characterization and genetic analysis of mutant strains of Escherichia coli K-12 accumulating the biquinone precursors 2-octaprenyl-6-methoxy-1,4-benzoquinone and 2-octaprenyl-3-methyl-6-methoxy-1,4-benzoquinone. J Bacteriol. 1971 Mar;105(3):769–778. doi: 10.1128/jb.105.3.769-778.1971. [DOI] [PMC free article] [PubMed] [Google Scholar]



