Abstract
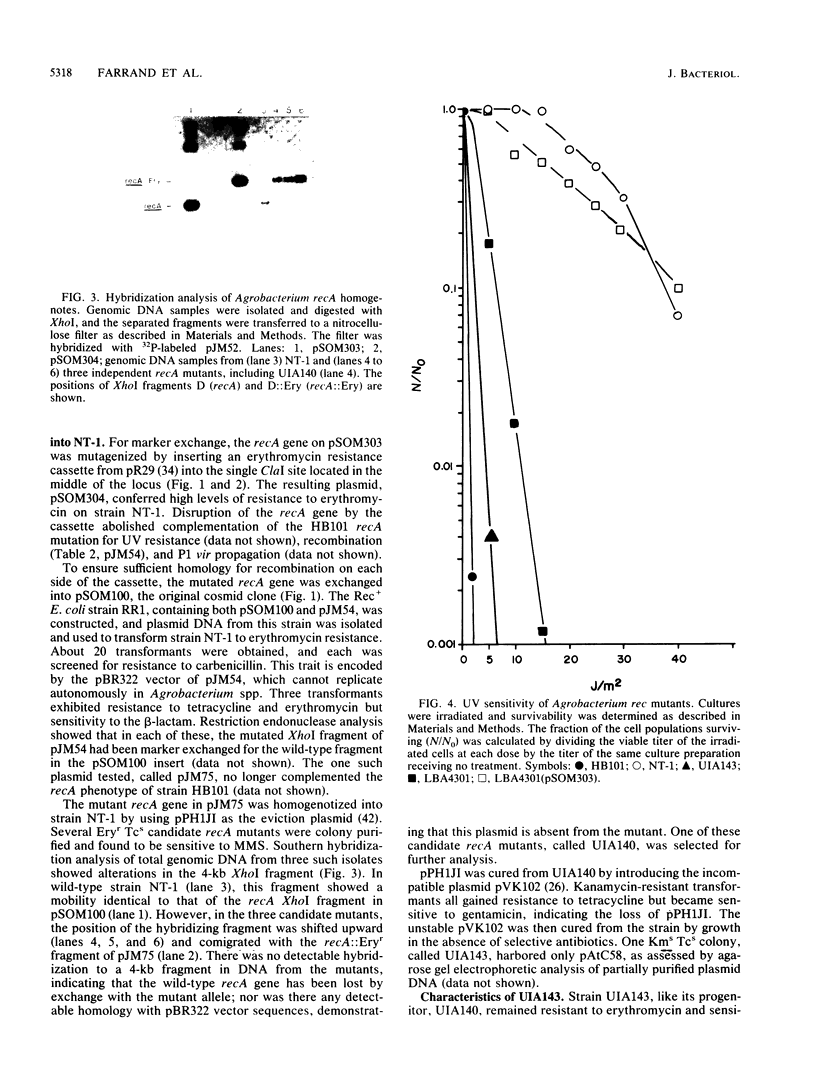
Clones encoding the recA gene of Agrobacterium tumefaciens C58 were isolated from a cosmid bank by complementation of an Escherichia coli recA mutation. Subcloning and mutagenesis with the lacZ fusion transposon Tn3HoHo1 located the Agrobacterium recA gene to a 1.3-kilobase segment of DNA. beta-Galactosidase expression from the fusions established the direction in which the gene was transcribed. The gene restored homologous recombination as well as DNA repair functions in E. coli recA mutants. Similar complementation of DNA repair functions was observed in the UV-induced Rec- Agrobacterium mutant, LBA4301. The Agrobacterium recA gene was disrupted by insertion of a cassette encoding resistance to erythromycin, and the mutated gene was marker exchanged into the chromosome of strain NT-1. The resulting strain, called UIA143, was sensitive to UV irradiation and methanesulfonic acid methyl ester and unable to carry out homologous recombination functions. The mutation was stable and had no effect on other genetic properties of the Agrobacterium strain, including transformability and proficiency as a conjugal donor or recipient. Furthermore, strain UIA143 became tumorigenic upon introduction of a Ti plasmid, indicating that tumor induction is independent of recA functions. Sequence homology was detected between the recA genes of strain C58 and E. coli as well as with DNA isolated from agrobacteria representing the three major biochemically differentiated biovars of this genus. In some cases, biovar-specific restriction fragment length polymorphisms were apparent at the recA locus.
Full text
PDF






Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Better M., Helinski D. R. Isolation and characterization of the recA gene of Rhizobium meliloti. J Bacteriol. 1983 Jul;155(1):311–316. doi: 10.1128/jb.155.1.311-316.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bolivar F., Rodriguez R. L., Greene P. J., Betlach M. C., Heyneker H. L., Boyer H. W., Crosa J. H., Falkow S. Construction and characterization of new cloning vehicles. II. A multipurpose cloning system. Gene. 1977;2(2):95–113. [PubMed] [Google Scholar]
- Chilton M. D., Currier T. C., Farrand S. K., Bendich A. J., Gordon M. P., Nester E. W. Agrobacterium tumefaciens DNA and PS8 bacteriophage DNA not detected in crown gall tumors. Proc Natl Acad Sci U S A. 1974 Sep;71(9):3672–3676. doi: 10.1073/pnas.71.9.3672. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cohen G. Electron microscopy study of early lytic replication forms of bacteriophage P1 DNA. Virology. 1983 Nov;131(1):159–170. doi: 10.1016/0042-6822(83)90542-1. [DOI] [PubMed] [Google Scholar]
- Dessaux Y., Tempé J., Farrand S. K. Genetic analysis of mannityl opine catabolism in octopine-type Agrobacterium tumefaciens strain 15955. Mol Gen Genet. 1987 Jun;208(1-2):301–308. doi: 10.1007/BF00330457. [DOI] [PubMed] [Google Scholar]
- Ditta G., Stanfield S., Corbin D., Helinski D. R. Broad host range DNA cloning system for gram-negative bacteria: construction of a gene bank of Rhizobium meliloti. Proc Natl Acad Sci U S A. 1980 Dec;77(12):7347–7351. doi: 10.1073/pnas.77.12.7347. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Douglas C. J., Halperin W., Nester E. W. Agrobacterium tumefaciens mutants affected in attachment to plant cells. J Bacteriol. 1982 Dec;152(3):1265–1275. doi: 10.1128/jb.152.3.1265-1275.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Douglas C. J., Staneloni R. J., Rubin R. A., Nester E. W. Identification and genetic analysis of an Agrobacterium tumefaciens chromosomal virulence region. J Bacteriol. 1985 Mar;161(3):850–860. doi: 10.1128/jb.161.3.850-860.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ellis J. G., Kerr A., Tempé J., Petit A. Arginine catabolism: a new function of both octopine and nopaline Ti-plasmids of Agrobacterium. Mol Gen Genet. 1979 Jun 20;173(3):263–269. doi: 10.1007/BF00268636. [DOI] [PubMed] [Google Scholar]
- Farrand S. K., Slota J. E., Shim J. S., Kerr A. Tn5 insertions in the agrocin 84 plasmid: the conjugal nature of pAgK84 and the locations of determinants for transfer and agrocin 84 production. Plasmid. 1985 Mar;13(2):106–117. doi: 10.1016/0147-619x(85)90063-0. [DOI] [PubMed] [Google Scholar]
- Figurski D. H., Helinski D. R. Replication of an origin-containing derivative of plasmid RK2 dependent on a plasmid function provided in trans. Proc Natl Acad Sci U S A. 1979 Apr;76(4):1648–1652. doi: 10.1073/pnas.76.4.1648. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gill J. F., Deretic V., Chakrabarty A. M. Overproduction and assay of Pseudomonas aeruginosa phosphomannose isomerase. J Bacteriol. 1986 Aug;167(2):611–615. doi: 10.1128/jb.167.2.611-615.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goldberg I., Mekalanos J. J. Cloning of the Vibrio cholerae recA gene and construction of a Vibrio cholerae recA mutant. J Bacteriol. 1986 Mar;165(3):715–722. doi: 10.1128/jb.165.3.715-722.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hamada S. E., Farrand S. K. Diversity among B6 strains of Agrobacterium tumefaciens. J Bacteriol. 1980 Mar;141(3):1127–1133. doi: 10.1128/jb.141.3.1127-1133.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hayman G. T., Farrand S. K. Characterization and mapping of the agrocinopine-agrocin 84 locus on the nopaline Ti plasmid pTiC58. J Bacteriol. 1988 Apr;170(4):1759–1767. doi: 10.1128/jb.170.4.1759-1767.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hickman M. J., Orser C. S., Willis D. K., Lindow S. E., Panopoulos N. J. Molecular cloning and biological characterization of the recA gene from Pseudomonas syringae. J Bacteriol. 1987 Jun;169(6):2906–2910. doi: 10.1128/jb.169.6.2906-2910.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hirsch P. R., Beringer J. E. A physical map of pPH1JI and pJB4JI. Plasmid. 1984 Sep;12(2):139–141. doi: 10.1016/0147-619x(84)90059-3. [DOI] [PubMed] [Google Scholar]
- Holsters M., de Waele D., Depicker A., Messens E., van Montagu M., Schell J. Transfection and transformation of Agrobacterium tumefaciens. Mol Gen Genet. 1978 Jul 11;163(2):181–187. doi: 10.1007/BF00267408. [DOI] [PubMed] [Google Scholar]
- Hynes M. F., Simon R., Pühler A. The development of plasmid-free strains of Agrobacterium tumefaciens by using incompatibility with a Rhizobium meliloti plasmid to eliminate pAtC58. Plasmid. 1985 Mar;13(2):99–105. doi: 10.1016/0147-619x(85)90062-9. [DOI] [PubMed] [Google Scholar]
- Iyer V. N., Klee H. J., Nester E. W. Units of genetic expression in the virulence region of a plant tumor-inducing plasmid of Agrobacterium tumefaciens. Mol Gen Genet. 1982;188(3):418–424. doi: 10.1007/BF00330043. [DOI] [PubMed] [Google Scholar]
- Klapwijk P. M., van Beelen P., Schilperoort R. A. Isolation of a recombination deficient Agrobacterium tumefaciens mutant. Mol Gen Genet. 1979 Jun 7;173(2):171–175. doi: 10.1007/BF00330307. [DOI] [PubMed] [Google Scholar]
- Knauf V. C., Nester E. W. Wide host range cloning vectors: a cosmid clone bank of an Agrobacterium Ti plasmid. Plasmid. 1982 Jul;8(1):45–54. doi: 10.1016/0147-619x(82)90040-3. [DOI] [PubMed] [Google Scholar]
- Kokjohn T. A., Miller R. V. Molecular cloning and characterization of the recA gene of Pseudomonas aeruginosa PAO. J Bacteriol. 1985 Aug;163(2):568–572. doi: 10.1128/jb.163.2.568-572.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Koomey J. M., Falkow S. Cloning of the recA gene of Neisseria gonorrhoeae and construction of gonococcal recA mutants. J Bacteriol. 1987 Feb;169(2):790–795. doi: 10.1128/jb.169.2.790-795.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Matthysse A. G. Characterization of nonattaching mutants of Agrobacterium tumefaciens. J Bacteriol. 1987 Jan;169(1):313–323. doi: 10.1128/jb.169.1.313-323.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Morrison D. A., Trombe M. C., Hayden M. K., Waszak G. A., Chen J. D. Isolation of transformation-deficient Streptococcus pneumoniae mutants defective in control of competence, using insertion-duplication mutagenesis with the erythromycin resistance determinant of pAM beta 1. J Bacteriol. 1984 Sep;159(3):870–876. doi: 10.1128/jb.159.3.870-876.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Olexy V. M., Bird T. J., Grieble H. G., Farrand S. K. Hospital isolates of Serratia marcescens transferring ampicillin, carbenicillin, and gentamicin resistance to other gram-negative bacteria including Pseudomonas aeruginosa. Antimicrob Agents Chemother. 1979 Jan;15(1):93–100. doi: 10.1128/aac.15.1.93. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Owttrim G. W., Coleman J. R. Molecular cloning of a recA-like gene from the cyanobacterium Anabaena variabilis. J Bacteriol. 1987 May;169(5):1824–1829. doi: 10.1128/jb.169.5.1824-1829.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Resnick D., Nelson D. R. Cloning and characterization of the Aeromonas caviae recA gene and construction of an A. caviae recA mutant. J Bacteriol. 1988 Jan;170(1):48–55. doi: 10.1128/jb.170.1.48-55.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rogowsky P. M., Close T. J., Chimera J. A., Shaw J. J., Kado C. I. Regulation of the vir genes of Agrobacterium tumefaciens plasmid pTiC58. J Bacteriol. 1987 Nov;169(11):5101–5112. doi: 10.1128/jb.169.11.5101-5112.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ruvkun G. B., Ausubel F. M. A general method for site-directed mutagenesis in prokaryotes. Nature. 1981 Jan 1;289(5793):85–88. doi: 10.1038/289085a0. [DOI] [PubMed] [Google Scholar]
- Sciaky D., Montoya A. L., Chilton M. D. Fingerprints of Agrobacterium Ti plasmids. Plasmid. 1978 Feb;1(2):238–253. doi: 10.1016/0147-619x(78)90042-2. [DOI] [PubMed] [Google Scholar]
- Slota J. E., Farrand S. K. Genetic isolation and physical characterization of pAgK84, the plasmid responsible for agrocin 84 production. Plasmid. 1982 Sep;8(2):175–186. doi: 10.1016/0147-619x(82)90055-5. [DOI] [PubMed] [Google Scholar]
- Stachel S. E., An G., Flores C., Nester E. W. A Tn3 lacZ transposon for the random generation of beta-galactosidase gene fusions: application to the analysis of gene expression in Agrobacterium. EMBO J. 1985 Apr;4(4):891–898. doi: 10.1002/j.1460-2075.1985.tb03715.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Van Larebeke N., Engler G., Holsters M., Van den Elsacker S., Zaenen I., Schilperoort R. A., Schell J. Large plasmid in Agrobacterium tumefaciens essential for crown gall-inducing ability. Nature. 1974 Nov 8;252(5479):169–170. doi: 10.1038/252169a0. [DOI] [PubMed] [Google Scholar]
- Watson B., Currier T. C., Gordon M. P., Chilton M. D., Nester E. W. Plasmid required for virulence of Agrobacterium tumefaciens. J Bacteriol. 1975 Jul;123(1):255–264. doi: 10.1128/jb.123.1.255-264.1975. [DOI] [PMC free article] [PubMed] [Google Scholar]