Abstract
The development of allergy is related to differences in the intestinal microbiota. Therefore, it is suggested that the immune responses induced by different genera of bacteria might be regulated through adaptive as well as innate immunity. In this study, we examined whether antigen-specific immune responses were affected by stimulation with the different genera of intestinal bacteria in vitro. Mesenteric lymph node (MLN) cells isolated from germ-free ovalbumin (OVA)-specific T cell receptor transgenic (OVA-Tg) mice were stimulated with OVA and intestinal bacteria. Cecal contents from conventional mice but not germ-free mice could induce OVA-specific cytokine production. Among the murine intestinal bacteria, Bacteroides acidofaciens (BA) enhanced OVA-specific IFN-γ and IL-10 production while Lactobacillusjohnsonii (LA) increased OVA-specific IL-10 production only. The expression of cell surface molecules and cytokine production by antigen-presenting cells (APCs) from germ-free Balb/c mice were analyzed. BA increased the expression of MHC II and co-stimulatory molecules on APCs compared with LA. BA increased IL-6 and IL-10 production but induced less IL-12p40 than LA. To examine the effects of prior stimulation of APCs by intestinal bacteria on the induction of antigen-specific immune responses, cytokine production was determined following co-culture with OVA, CD4+ T cells from OVA-Tg mice, and APCs which were pre-stimulated with the bacteria or not. APCs pre-stimulated with LA did not enhance OVA-specific cytokine production while BA stimulated OVA-specific IL-10 production. These results suggest that the prior stimulation of intestinal immunocytes by Lactobacillus might regulate excessive antigen-specific cytokine responses via APCs when compared with prior stimulation by Bacteroides.
Keywords: Lactobacillus, Bacteroides, Germ-free mice, Antigen-specific immune response, Antigen-presenting cells
Introduction
The hygiene hypothesis suggests that the increase in allergic disease is caused by a decreased exposure to childhood infections. It is held that the maturation of the intestinal tissues and the immune system in the gut is enhanced by the microbial environment in the intestine, particularly through stimulation by commensal bacteria colonizing the intestine. Bjorksten et al. (1999) have shown that Swedish and Estonian children with allergy display Bifidobacterium and Lactobacillus less often and have a higher count of aerobic bacteria when compared with non-allergic children. A study in Finland showed that allergic sensitized children had more clostridia and fewer bifidobacteria than non-allergic subjects (Kalliomaki et al. 2001). These data suggested the differences in the gut microbiota relate to the development of allergy and have a crucial role in the normal maturation of the immune system. However, it remains unclear how intestinal bacteria modulate adaptive immune responses.
As many as a hundred trillion bacteria of more than 500 different species colonize the gut. In the murine intestinal microbiota, Bacteroides colonize early in the newborn period and are the most prominent component (Kononen et al. 1992). Lactobacillus are a major component of the commensals of the small and large intestinal tracts of animals and are frequently used as probiotics (Ahrne et al. 1998). Bacteria can be divided into gram-positive and gram-negative according to their cell wall structures and constituents. Gram-positive bacteria have a cell wall containing thick layers of peptidoglycan (PGN) with chains of lipotechoic acid (LTA), while the cell wall of gram-negative bacteria is composed of a thinner PGN layer and an outer membrane containing lipopolysaccharide (LPS). Hessle et al. (2000) reported that gram-positive bacteria were, in general, more potent IL-12 and less potent IL-10 inducers in monocytes than gram-negative bacteria. Generally, bacteria might be recognized by the innate immune system with subsequent modulation of adaptive immunity to specific-antigens including allergens. The innate immune cells such as macrophages (Mϕs), dendritic cells (DCs) and others express pattern recognition receptors (PRRs) that recognize pathogen associated molecular patterns (PAMPs) including the bacterial components. Toll-like receptors (TLRs), which are one of the PRRs, are preferentially expressed on antigen-presenting cells (APCs). TLR2 can recognize LTA and PGN from gram-positive bacterial components and TLR4 can recognize LPS from gram-negative bacteria. The interaction between PAMPs and TLRs leads to the expression of co-stimulatory molecules and cytokine production by APCs (Raymond and Wilkie 2005), which leads to adaptive immunity. APCs play an important role in antigen presentation, T-cell activation, and cytokine regulation. Antigen presentation to, and co-stimulation of, T cells would be influenced by MHC and B7 (CD80 and CD86) expression, while T cell activation and differentiation would be affected by the cytokines produced by APCs. Therefore, APCs stimulated by different bacteria might have the differential ability to modulate antigen-specific T cell responses dependent on the microbacteria.
We have previously established germ-free OVA23-3 mice to examine the direct effect of intestinal bacteria on antigen-specific immune responses (Fujioka et al. 2004). OVA23-3 mice are one of the ovalbumin (OVA)-specific T cell receptor transgenic (OVA-Tg) mice and are transduced with T cell receptors that specifically recognize the MHC class II molecule I-Ad bound to the peptide residue 323–339 of OVA (Shida et al. 2000; Nakajima-Adachi et al. 2006). When we prepared intestinal immune cells derived from germ-free or conventional mice, we demonstrated that the immunocytes from germ-free mice showed higher IFN-γ and IL-10 production in response to OVA stimulation compared with those from conventional mice. Therefore, constant stimulation of intestinal immune cells by intestinal bacteria may induce down-regulation of OVA-specific cytokine responses.
In this study, we used an ex vivo experimental system with immune cells derived from germ-free OVA23-3 mice to examine whether stimulation by different genera of intestinal bacteria induced different immunomodulatory effects on the antigen-specific immune responses.
Materials and methods
Mice
Germ-free OVA23-3 mice; OVA specific T cell receptor transgenic mice or Balb/c mice (8–13-weeks old) were used in the experiments. These mice were bred and maintained in a sterile vinyl isolator in the animal facilities at the University of Tokyo until just before sacrifice. Germ-free Balb/c mice were purchased from Japan Clea Inc. (Tokyo, Japan). All animal experiments were carried out in accordance with the ‘Guidelines for the Care and Use of Laboratory Animals’ by the College of Bioresource Sciences, Nihon University, and similar guidelines formulated by the University of Tokyo.
Bacteria preparation
Ceca were removed from conventional, germ-free Balb/c, gnotobiotic segmented filamentous bacteria (SFB)-associated or 46 strains of clostridia-associated mice (Umesaki et al. 1999; Itoh and Mitsuoka 1985), and opened to collect and suspend the cecal contents in phosphate-buffered saline (PBS). The residual tissue was removed by filtration through a cell strainer (100-μm Nylon, BD Falcon) and washed by centrifugation. 3 strains of Lactobacillus (LA: L. johnsonii 129, originally identified as L. acidophilus; LF: L. reuteri 106, originally identified as L. fermentum; LM: L. murinus 91, originally identified as L. murinum) (Itoh et al. 1983; Park and Itoh 2005), 3 strains of Bacteroides (unidentified, defined as 1, 7, 11) derived from murine intestinal microbiota were cultured in MRS broth, GAM broth and nutrient broth, respectively. Cultured bacteria were harvested by centrifugation and washed with PBS. To kill the bacteria, the bacterial suspensions and cecum suspensions were exposed to UV-light for 15 min. Death of these bacteria was confirmed because the bacteria in the preparation could not form colonies when these suspensions were cultured on plates. The bacterial suspensions were lyophilized and stored in the frozen state until use for co-culture with immune cells.
Cell preparation
Mesenteric lymph nodes (MLNs) were removed from germ-free Balb/c or OVA23-3 mice, cut into small fragments and incubated for 50 min at 37 °C with RPMI 1640 medium containing 10% FCS, 1 mg/ml collagenase D (Roche Diagnostics, Indianapolis, IN, USA) and 0.1 mg/ml Dnase I (Roche Diagnostics). After centrifugation, the cell pellets were incubated for 10 min at 37 °C with Hanks’ balanced salt solution (HBSS; Ca, Mg free, Sigma) containing 5 mM EDTA and 5% FCS. Single-cell suspensions were filtered through a cell strainer (70-μm Nylon, BD Falcon, Franklin Lakes, NJ, USA), washed and re-suspended with FCS-RPMI medium. CD4+ cells were positively isolated by using anti-mouse CD4 mAb conjugated magnetic microbeads and magnetic cell sorting (MACS; Miltenyi Biotec, Bergisch Gladbach, Germany) according to the manufacture’s protocol. Thy1.2 (CD90.2) − cells were negatively isolated by using anti-mouse Thy1.2 mAb conjugated magnetic microbeads and MACS. Purity of CD4+ cells and Thy1.2− was > 90% by flow cytometry.
Cell culture
Whole MLN cells (2.5 × 106 cells) from OVA23-3 mice were added to 48-well flat-bottom plates and cultured in the absence or the presence of OVA (250 μg/ml) and intestinal bacteria (50 μg/ml) in 1 ml of RPMI 1640 medium containing 5% FCS final concentration. Two groups of culture conditions, representing different periods of bacterial stimulation, were employed in our experiments. In pre-stimulation, MLN cells and bacteria were co-cultured for 24 h, the supernatants removed by centrifugation (1,300 rpm, 5 min) and OVA subsequently added. We confirmed that bacteria were left in the culture after centrifugation. In simultaneous stimulation, MLN cells were co-cultured with bacteria and OVA. The supernatants were collected 48 h later for cytokine analysis.
MLN CD4+ cells (5 × 105 cells) from OVA23-3 mice were similarly cultured in 48-well flat-bottom plates in the absence or the presence of OVA and intestinal bacteria with MLN Thy1.2− cell (2 × 106 cells) from Balb/c mice as APCs. In the pre-stimulation condition, Thy1.2− cells and bacteria were co-cultured for 24 h, and then the supernatants were removed by centrifugation, and CD4+ cells and OVA were added. In the simultaneous stimulation condition, CD4+ cells, OVA and bacteria were added into Thy1.2− cells simultaneously.
Cytokine production
Cytokines in the cell-culture supernatants were assayed by sandwich ELISA. For IFN-γ and IL-6 measurement, purified rat anti-mouse IFN-γ (R4-6A2), IL-6 (MP5-20F3) monoclonal antibodies (BD Pharmingen, San Diego, CA, USA), and biotinylated rat anti-mouse IFN-γ (XMG1.2), and IL-6 (MP5-32C11) monoclonal antibodies (BD Pharmingen) were used. IL-10 and IL-12p40 were measured by using BD OptEIA mouse ELISA set (BD Pharmingen) following the manufacturer’s instructions. Transforming growth factor (TGF)-β1 was measured by using the TGFβ1 E®max ImmunoAssay System (Promega, Madison, WI, USA) following the manufacturer’s instructions.
FACS analysis
For analyzing the expression of cell surface molecules, MLN Thy1.2− cells (2.5 × 106 cells) from GF Balb/c mice were cultured in 48-well flat-bottom plates and stimulated with 50 μg/ml of either bacterial preparation for 24 h. The cultured cells were collected and washed with FACS buffer (HBSS containing 1% FCS and 0.02% NaH3). The cells were pre-treated for 20 min on ice with anti-mouse CD16/CD32 (2.4G2) (BD Pharmingen) to block Fc receptors (Fcγ III/II receptor). Following washing by centrifugation, the cells were stained for 20 min on ice with FITC-labeled anti-Thy1.2 (53-2.1) and PE-conjugated anti-I-Ad MHC class II (AMS-32.1), CD80 (16-10A1), CD86 (GL1) (BD Pharmingen) or PE-conjugated mouse IgG2b, κ, with hamster IgG2, κ, or Rat IgG2a, κ, as matched-isotype control antibodies, respectively. Cells were then washed and resuspended with FACS buffer. Flow cytometric analysis of the immune cells was performed on a FACS Canto using Flowjo software (Tree star, Inc., Ashland, OR, USA).
Statistics analysis
Data are represented as mean ± SD. Differences were examined by one-way analysis of variance (ANOVA), and significant differences found between cultures were further evaluated by Tukey’s test (SPSS Ver. 13.0, SPSS, Chicago, IL, USA). Differences were considered to be statistically significant for values of p < 0.05.
Results
Intestinal bacteria from the cecum in conventional mice can modulate OVA-specific cytokine response
As the majority of the intestinal bacteria reside in the cecum in mice, we first examined whether the bacteria in the murine cecum had the ability to modulate antigen-specific immune responses. MLN cells from germ-free OVA23-3 mice were stimulated with OVA and cecal contents from conventional or germ-free mice. The cecal contents from conventional mice induced OVA-specific IFN-γ and IL-10 production by MLN cells, although those from germ-free mice could not induce these cytokines (Fig. 1). This suggests that intestinal bacteria in the cecal contents from conventional mice have the ability to modulate antigen-specific cytokine responses. Conventional cecal contents increased IL-6 and IL-12p40 production by MLN cells stimulated both with and without OVA (Fig. 1). Thus, this enhancement of IL-6 and IL-12p40 production by MLN cells was not affected by OVA stimulation, indicating that it was independent of T cell responses. In contrast, germ-free cecal contents could not induce these cytokines. These data suggests that bacteria in the cecum can activate the innate immune response.
Fig. 1.
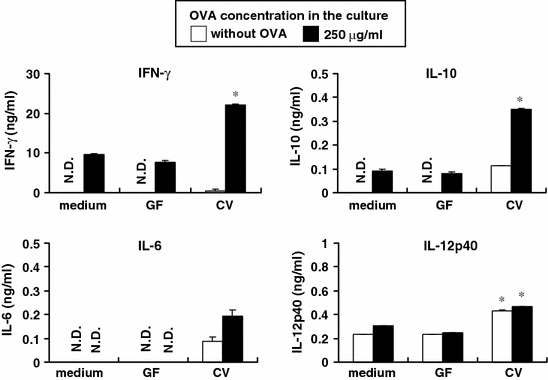
Cecal contents derived from conventional but not germ-free mice can induce OVA-specific cytokine production. MLN lymphocytes were isolated from germ-free OVA23-3 mice, and 2.5 × 106 cells were stimulated with or without OVA (250 μg/ml) and cecal contents (50 μg/ml) from conventional (CV) or germ-free (GF) mice for 48 h. Cytokines (IFN-γ, IL-6, IL-10 and IL-12p40) secreted in the supernatants were assayed by ELISA. All data represent mean ± S.D. *Significant differences from non-stimulated cells (medium) to cells stimulated with bacterial preparation (p < 0.05). Data shown are representative of 2 independent experiments
Modulation of OVA-specific immune responses by intestinal bacteria was affected by the different genera of bacteria
To examine whether different genera of bacteria in the murine microbiota have different modulatory effects, MLN cells were stimulated with either Lactobacillus, Bacteroides, SFB or clostridia (Fig. 2). Three strains of the genus of Bacteroides, which represent the most dominant gram-negative bacteria in murine intestines, induced OVA-specific IFN-γ and IL-10 production by MLN cells. Three strains of the genus of Lactobacillus, the dominant gram-positive bacteria, induced OVA-specific IL-10 production but not IFN-γ production. Specifically, LA was the strongest IL-10 inducer among the bacterial preparations tested in this study. SFB weakly induced OVA-specific cytokine production, and clostridia strongly induced OVA-specific cytokine production as did Bacteroides. IL-6 and IL-12p40 production by MLN cells were also independent on OVA stimulation. IL-6 production was strongly stimulated by Bacteroides and clostridia, and IL-12p40 production was strongly stimulated by Lactobacillus (Fig. 2–2). These results suggest that different genera of bacteria have different modulatory effects on the antigen-specific cytokine responses. Therefore, we selected LA in the genus of Lactobacillus, as the dominant gram-positive bacteria, and BA1 in the genus of Bacteroides as the dominant gram-negative bacteria, for use in subsequent experiments. Furthermore, BA1 was identified as Bacteroides acidofacience (BA).
Fig. 2.
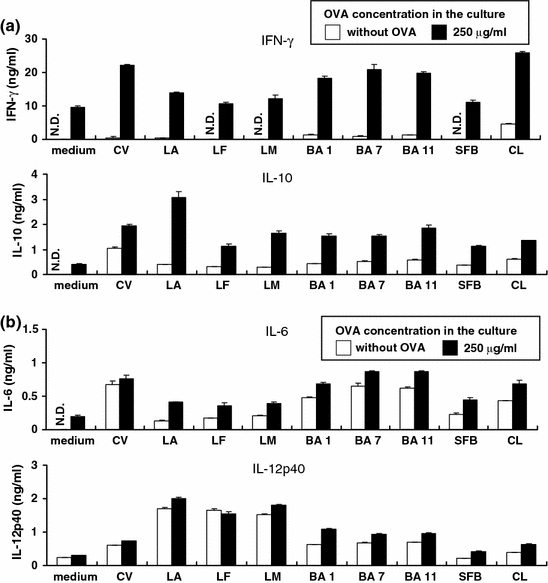
Different genera of intestinal bacteria modulate different patterns of OVA-specific cytokine responses. Bacterial stimulations are shown under the graphs. Abbreviations of the bacterial contents: CV, conventional cecal contents; LA, L. johnsonii; LF, L. reuteri; LM, L. murinus; BA1, BA7, and BA11 which are undefined Bacteroides strains; SFB which is the cecal contents derived from SFB-monoassociated mice, CL which is the cecal contents derived from clostridia-associated mice. MLN lymphocytes were isolated from germ-free OVA23-3 mice, and the cells (2.5 × 106 cells) were stimulated with or without OVA (250 μg/ml) and murine intestinal bacterial preparation (50 μg/ml) for 48 h. Cytokines (IFN-γ, IL-10 (a), IL-6, IL-12p40 (b)) secreted in the supernatants were assayed by ELISA. All data represent mean ± S.D. Data shown are representative of 3 independent experiments
BA induces the expression of co-stimulatory molecules on APCs more effectively than LA
Appropriate antigen presentation to T cells requires co-stimulation by APCs, and this is influenced by MHC II and B7 (CD80 and CD86) expression. Furthermore, it has been shown that signaling through TLRs increases MHC II and co-stimulatory molecules such as CD86 (Raymond and Wilkie 2005). Therefore, we examined the effects of different intestinal bacterial stimulation on the expression of co-stimulatory molecules on APCs (Fig. 3). Both BA and LA increased the expression of MHC II and CD86 on APCs, with BA being more effective than LA. BA increased CD80 expression markedly. On the other hand, LA induced expression of CD86 only and not CD80. Thus, these results suggest that BA may induce more effective antigen presentation than LA. Furthermore, to elucidate the modulation of APCs by different genera of intestinal bacteria, we examined cytokine production profiles of APCs upon stimulation with BA or LA. LA induced higher levels of IL-12p40 production and lower levels of IL-6 and IL-10 production compared with BA (Fig. 4). The regulatory cytokine TGF-β1 production was not affected by the either bacterial stimulation. These results suggest that LA and BA have different abilities to modulate APCs.
Fig. 3.
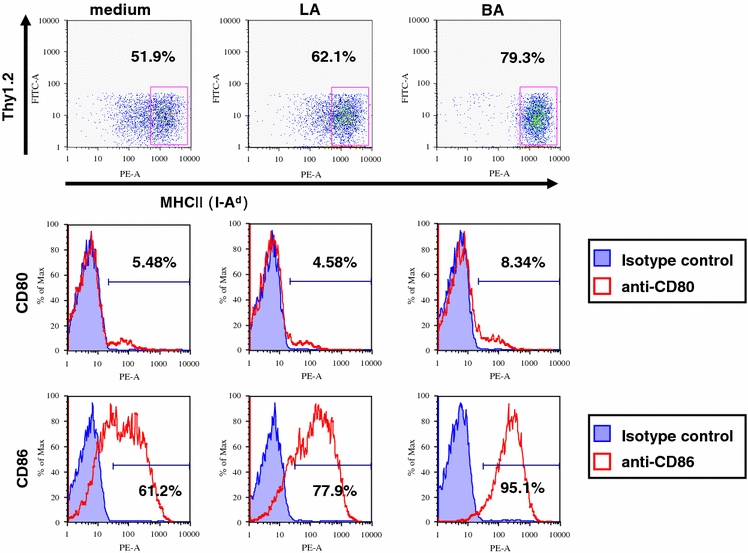
Bacteroides induces the expression of MHC and co-stimulatory molecules by APCs more effectively than Lactobacillus. Thy1.2− cells were isolated from MLN cells of germ-free Balb/c mice by using MACS. These cells (2.5 × 106 cells) were stimulated with LA or BA (50 μg/ml) for 24 h. The surface phenotype (MHC II (I-Ad), CD80, CD86) of the cells was analyzed by flow cytometry. Data shown are representative of 4 independent experiments
Fig. 4.
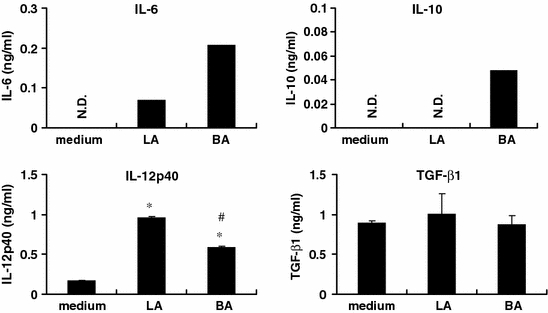
Lactobacillus induces more IL-12p40 and less IL-6 and IL-10 production by APCs than Bacteroides. Thy1.2− cells were isolated from MLN cells of germ-free Balb/c mice by using MACS. These cells (2.5 × 106 cells) were stimulated with LA or BA (50 μg/ml) for 24 h. The supernatants were assayed for cytokines (IL-6, IL-10, IL-12p40 and TGF-β) by ELISA. All data represent mean ± S.D. *Significant differences between non-stimulated cells (medium) and cells stimulated with bacteria (p < 0.05). #Significant differences between cells stimulated with LA and the cells stimulated with BA (p < 0.05). Data represent the typical results of 3 independent experiments
Pre-stimulation of MLN cells with LA do not markedly induce OVA-specific cytokine production compared with BA
Intestinal bacteria colonize the intestinal tract and so constantly stimulate intestinal immune systems. Intestinal immune cells from germ-free OVA23-3 mice showed higher IFN-γ and IL-10 production in response to OVA stimulation than that of conventional mice (our unpublished observation). Therefore, constant stimulation of intestinal immune cells by intestinal bacteria may induce down-regulation of OVA-specific cytokine responses. We then examined how prior stimulation with intestinal bacteria affected the OVA-specific cytokine response. MLN cells were pre-stimulated with intestinal bacteria for 24 h before co-culture with OVA (Fig. 5). Pre-stimulation with LA did not enhance OVA-specific IFN-γ and IL-10 production, although simultaneous stimulation with LA and OVA strongly increased OVA-specific IL-10 production. The pre-stimulation with BA also did not enhance OVA-specific IFN-γ production, but enhanced OVA-specific IL-10 production. The level of IL-10 production was the same following simultaneous stimulation or pre-stimulation with BA. Thus, these results suggested that MLN cells pre-stimulated with LA did not markedly enhance OVA-specific cytokine responses compared with BA.
Fig. 5.
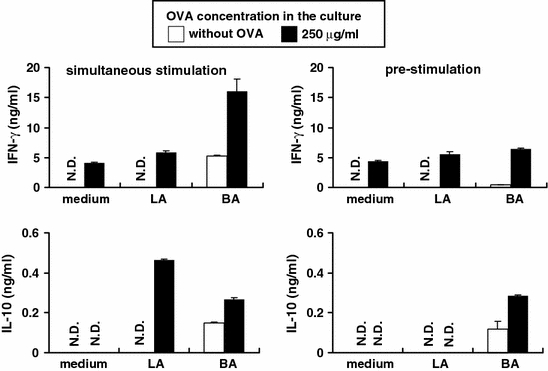
Pre-stimulation of MLN cells with LA does not induce OVA-specific cytokine production markedly compared with pre-stimulation with BA. MLN cells were isolated from germ-free OVA23-3 mice. In the pre-stimulation group, these cells and intestinal bacteria were co-cultured for 24 h and then the supernatants were removed by centrifugation. OVA was added for a further stimulation of 48 h. In the simultaneous stimulation, MLN cells were co-cultured with intestinal bacteria and OVA for 48 h. Final concentration of intestinal bacteria (LA or BA) in the culture was 50 μg/ml, and OVA was added at 250 μg/ml. The supernatants were assayed for cytokines (IFN-γ, IL-10) by ELISA. All data represent mean ± S.D. Data shown are representative of 3 independent experiments
APCs pre-stimulated with LA do not enhance OVA-specific cytokine production by CD4+ T cells
We have shown that Lactobacillus and Bacteroides differentially modulate the expression of co-stimulatory molecules by APCs, which are needed for appropriate antigen-presentation to T cells. Therefore, we focused on the different abilities of Lactobacillus and Bacteroides to modulate OVA-specific T cell responses through APCs. When APCs were stimulated with intestinal bacteria and OVA simultaneously, both LA and BA induced OVA-specific IL-10 and IFN-γ production by CD4+ T cells. However, pre-stimulation of APCs with LA did not enhance OVA-specific IFN-γ and IL-10 production compared with non-bacterial stimulation (Fig. 6). IFN-γ levels following pre-stimulation with BA were lower than those following simultaneous stimulation with BA. However, IL-10 levels were not different between pre-stimulation and simultaneous stimulation with BA. These data suggest that pre-stimulation of APCs with LA did not enhance OVA-specific cytokine responses by CD4+ T cells over that with BA, thus implying that the different immunomodulatory effects of LA and BA on OVA-specific T cell responses were modulated through APCs.
Fig. 6.
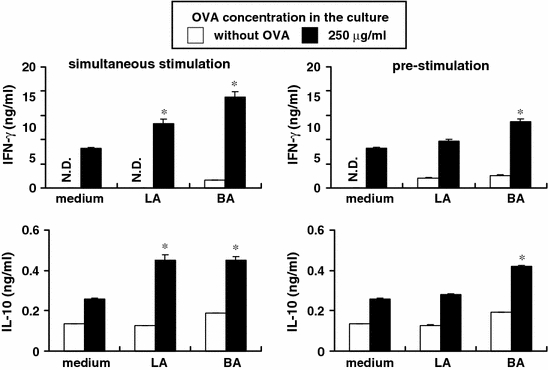
APCs pre-stimulated with LA do not enhance OVA-specific cytokine production by CD4+ T cells. MLN CD4+ cells (5 × 105 cells) from OVA23-3 mice were cultured in 48-well flat-bottom plates in the absence or the presence of OVA and intestinal bacteria with MLN Thy1.2− cell (2 × 106 cells) from Balb/c mice as APCs. In the pre-stimulation condition, Thy1.2− cells and intestinal bacteria were co-cultured for 24 h, and then the supernatants were removed by centrifugation, and CD4+ cells and OVA were added. In the simultaneous stimulation, CD4+ cells, OVA and intestinal bacteria were added to the Thy1.2− cell culture simultaneously. Final concentration of intestinal bacteria (LA or BA) in the culture was 50 μg/ml, and OVA was added at 250 μg/ml. The supernatants were assayed for cytokines (IFN-γ, IL-10) by ELISA. All data represent mean ± S.D. *Significant differences between non-stimulated cells (medium) and the cells stimulated with bacteria (p < 0.05). Data shown are representative of 3 independent experiments
Discussion
Many reports have shown that intestinal commensal bacteria contribute to the development of the tissues and functions of the intestinal immune systems. However, it is unclear how intestinal bacterial stimulation can modulate the antigen-specific immune responses that cause allergy. In this study, we used an ex vivo experimental system with immune cells derived from germ-free OVA23-3 mice to examine whether different genera of intestinal bacteria have different immunomodulatory effects on antigen-specific immune responses.
Most intestinal bacteria in mice reside in their cecum. To examine the immunomodulatory effect of intestinal bacteria on the antigen-specific immune response, MLN cells were stimulated with cecal contents from conventional or germ-free mice and OVA. Cecal contents from conventional mice enhanced OVA-specific IFN-γ and IL-10 production, suggesting that bacteria in the cecum had the ability to modulate antigen-specific cytokine responses. These immunomodulatory effects may be due to the various bacterial components and their bacterial metabolites found in the cecum. When MLN cells were co-cultured with intestinal bacteria and OVA together, the cells enhanced IFN-γ, IL-6, IL-10 and IL-12p40 production compared to non-stimulation (medium only). The pattern of cytokine production could be divided into OVA-dependent (IFN-γ, IL-10) and independent (IL-6, IL-12p40) responses. This suggests that IFN-γ and IL-10 production were T cell-dependent response and were modified by stimulation with intestinal bacteria, on the other hand, IL-6 and IL-12p40 production were T cell-independent response in the culture. More than 500 species of bacteria reside in the intestinal tract and it seems that the balance of microbiota affects not only the innate immune system but also the adaptive immune system consisting of both humoral and cellular immunity. Therefore, it seems that the different genera of bacteria may differentially modulate the antigen-specific immune responses. We examined the ability of various intestinal bacteria derived from mice to cause immunomodulation. We used bacteria or cecal contents derived from gnotobiotic mice that are reported to have immunologic activity among the murine microbiota. The genus of Bacteroides is one of the earlier-colonizing bacteria in the newborn and is the most prominent component of the gut microbiota. Three strains of Bacteroides induced OVA-specific IFN-γ and IL-10 production by MLN cells. Lactobacillus is a major component of the commensal microbiota of the small and large intestinal tract of mice and is frequently used as a probiotic. Three strains of Lactobacillus induced OVA-specific IL-10 production but not IFN-γ production. Interestingly, LA was the strongest IL-10 inducer among the bacterial preparations tested in this study. SFB colonize the small intestine of a wide range of animals and can promote an IgA response (Umesaki et al. 1999). However, SFB have a weakly enhancing effect on OVA-specific cytokine production. Consistent with our findings, it was reported that the association of germ-free mice with SFB did not enhance OVA-specific IgA responses to those seen in oral booster immunized OVA in vivo (Snel et al. 1997). Thus, SFB may not have modulatory effects on the antigen-specific immune response while the bacteria do contribute to the induction of an IgA response. Clostridia are dominant in the large intestine and are able to normalize cecal size (Itoh and Mitsuoka 1985) and to increase IgA producing cells in the colon when associated with germ-free mice (Umesaki et al. 1999). Clostridia induced OVA-specific IFN-γ and IL-10 production as effectively as Bacteroides. However, we have to consider that the preparation of clostridia was a mixture of 46 strains from the bacterial cecal contents of gnotobiotic mice, so it is difficult for us to identify the most effective bacterial strain and component. At least these data showed that the dominant gram-negative Bacteroides species and gram-positive Lactobacillus species differentially modulated OVA-specific cytokine production by MLN cells. Differences in immunomodulation of the OVA-specific cytokine response by different strains of bacteria invited two hypotheses, first: modulation of T cells via interaction of APCs with intestinal bacteria, the second: direct modulation of T cells by intestinal bacteria. Cytokines produced by APCs were modulated by stimulation with different intestinal bacteria when immune cells derived from OVA23-3 mice were stimulated with OVA and intestinal bacteria. Consequently, we focused on whether different genera of bacteria modulated APCs to prime T cell responses differentially.
Stimulation of T cells by APCs plays an important role in antigen presentation, T-cell activation, and cytokine regulation. Antigen presentation to, and co-stimulation of, T cells would be influenced by expression of MHC and B7 (CD80 and CD86), while activation and differentiation of T cells would be affected by the cytokines produced by APCs. We examined the expression of cell surface molecules and cytokine production by APCs stimulated with Lactobacillus or Bacteroides. Both Lactobacillus and Bacteroides increased MHC II and co-stimulatory molecules on APCs. It has been reported that Lactobacillus (Christensen et al. 2002) or probiotic mixture VSL#3 (Drakes et al. 2004) induce up-regulation of MHC II and CD86 expression by DCs. In the same way, a polysaccharide from Bacteroides increased the expression of these molecules by DCs (Wang et al. 2006). APCs used in this study were a mixture of various cell types such as DCs, Mϕs and B cells. Bacteroides was a stronger inducer of these molecules than Lactobacillus. These results suggested that Bacteroides could activate APCs and might induce more effective antigen-presentation to T cells. Furthermore, Lactobacillus and Bacteroides induced a different cytokine profile in APCs. Lactobacillus strongly induced IL-12p40 while Bacteroides induced IL-10 and IL-6 production. Consistent with our study, Hessle et al. (2000) found that gram-positive bacteria were, in general, more potent IL-12 and less potent IL-10 inducers than gram-negative bacteria. It has been reported that T cell differentiation depends on the phenotype and the pattern of cytokine production by DC which are one type of APCs and the most effective at antigen-priming of naïve T cells. It has been postulated that intestinal DC expressing a default level of CD86 produce little IL-12 (favoring induction of a regulatory response), whereas active immunity is induced by stimulated DCs, which express high levels of CD86 with either low production of IL-12 (favoring a Th2 response) or high-level IL-12 production (favoring a Th1 response) (Kalinski et al. 1999; Chen et al. 2000). In our study, the expression of cell surface molecules and cytokine production by APCs stimulated with Lactobacillus were shown to be different to those stimulated by Bacteroides, resulting in an altered ability of APCs to prime T cells. We speculate that Lactobacillus might not induce stronger presentation and co-stimulation than Bacteroides, contributing to induction of nonactive T cell response, although Lactobacillus led to produce higher level of IL-12p40 by APCs.
The difference in immunomodulation between Lactobacillus and Bacteroides may attribute to recognition of their different bacterial components by APCs. Lactobacillus is a gram-positive bacterium which mainly contain LTA acid and PGN in its cell wall components, which are recognized by TLR2, while, Bacteroides are gram-negative bacteria which mainly contain LPS, which is recognized by TLR4. Therefore, the stimulation by Lactobacillus may induce different intracellular signaling of APCs from those of Bacteroides. Activation of both TLR2 and TLR4 by each microbial component induces to MyD88 pathway to activate transcriptional factors. LPS-mediated signaling but not TLR2 signaling also mediates MyD88-independent pathways. It has been reported that LPS-mediated TLR4 signaling via MyD88-dependent pathway is essential for the production of inflammatory cytokines. MyD88-independent pathway has been shown to induce maturation of DCs including the increased expression of co-stimulatory molecules (Kaisho et al. 2001). TLR4 signaling has been reported to be able to induce MyD88−/− DC to support Th2 cell differentiation (Kaisho et al. 2002). Thus, the balance of intracellular signaling between TLR2 and TLR4 or other TLRs might affect the difference of immunomodulation between Lactobacillus and Bacteroides.
It would appear that intestinal bacteria can constantly stimulate intestinal immune systems as the bacteria colonize and reside in the gut of host animals. Intestinal immune cells from germ-free OVA23-3 mice showed higher IFN-γ and IL-10 production in response to OVA stimulation than those of conventional mice (our unpublished observation). Therefore, constant stimulation of intestinal immune cells by intestinal bacteria may induce down-regulation of the OVA-specific cytokine response. We examined whether prior stimulation of APCs with intestinal bacteria could regulate T cell responses. Compared with simultaneous stimulation, pre-stimulation of APCs with Lactobacillus or Bacteroides did not enhance OVA-specific cytokine production (IFN-γ). These data suggest that APCs can control T cell responses following prior stimulation with intestinal bacteria. This phenomenon may be partially explained by LPS tolerance. LPS tolerance is seen when pre-exposure to LPS causes a reduced response to subsequent host responses. Thus, this tolerance is considered a host response to prevent excessive inflammation. In addition, it has been reported that microbial components such as lipopeptides (TLR2 ligands) can induce LPS tolerance (Sato et al. 2002). Both LPS- and lipopeptides-induced LPS tolerance blocked MyD88-dependent pathway. Furthermore, previous reports have shown that MyD88-knockout mice are deficient in T cell activation (Pasare and Medzhitov 2004). Compared with APCs stimulated by intestinal bacteria simultaneously, APCs pre-stimulated by intestinal bacteria might down-regulate intracellular signaling such as MyD88-dependent pathway, resulting in a lowered production of cytokines in the culture. Thus, we speculate that the cells pre-stimulated with intestinal bacteria might not induce more excessive OVA-specific cytokine production than those receiving simultaneous stimulation. Specifically, APCs pre-stimulated with Lactobacillus did not induce enhanced OVA-specific IL-10 and IFN-γ production whereas Bacteroides enhanced OVA-specific IL-10 production. These data suggested that Lactobacillus induce a lesser antigen-specific cytokine response in vitro when compared with Bacteroides. Differences in cell wall components between Lactobacillus and Bacteroides may affect differential modulation of intracellular signaling on APCs. However, it has recently been shown that T cells express various types of TLRs, indicating the possibility that the T cell response was modulated by intestinal bacteria directly (Kabelitz 2007). Furthermore, we used APCs which were mixture of various cell types such as DCs, Mϕs and B cells. It has reported that Mϕs expressed higher level of TLR2 transcripts and produced more IL-10 and less IL-12 responding to TLR ligands or bacteria than DCs (Werling et al. 2004). Thus, it remains possible that the target immune cells for Lactobacillus were different from those for Bacteroides, inducing different immunomodulatory effect on antigen-specific cytokine response.
Considering our results using the ex vivo experimental system with germ-free animals, we can speculate the differential immunomodulatory effects by Lactobacillus and Bacteroides in vivo. Lactobacillus may contribute to induce the development of ‘regulated states’ of immune response like conventional condition because constant stimulation of the immune cells from germ-free mice did not induce strongly antigen-specific cytokine production. On the other hand, we expected Bacteroides induce the development of ‘excessive states’ of immune response because Bacteroides induced effective expression of cell surface molecules on APCs and more strongly antigen-specific cytokine production than Lactobacillus, although Bacteorides is considered to have significant roles in the development of immune systems since Bacteroides species have the largest proportion of intestinal microflora and a report (Mazmanian et al. 2005) has shown that B. fragilis can enhance the proportion of CD4+ cells and then, improve the immune response of germ-free animals into conventional type.
In conclusion we suggest here that the gram-positive bacteria Lactobacillus and the gram-negative bacteria Bacteroides can induce different patterns of OVA-specific cytokine production via APCs. This suggests that the different genera of intestinal bacteria can modulate different patterns of antigen-specific immune responses as seen in the adaptive immune system, indicating that colonization by an appropriate balance of intestinal bacteria in the gut might be important for the development or regulation of a healthy immune system but not for excessive immune responses like allergy.
Acknowledgments
We thank Dr. Sonoko Habu and Dr. Takehito Sato (Tokai University, School of Medicine) for generously providing the TCR-transgenic mice.
Abbreviations
- APCs
Antigen-presenting cells
- BA
Bacteroides acidofaciens
- DCs
Dendritic cells
- LA
Lactobacillus johnsonii
- LPS
Lipopolysaccharide
- LTA
Lipotechoic acid
- Mϕs
Macrophages
- MLN(s)
Mesenteric lymph node(s)
- OVA
Ovalbumin
- OVA-Tg
OVA-specific T cell receptor transgenic
- PAMPs
Pathogen associated molecular patterns
- PGN
Peptidegrycan
- PRRs
Pattern recognition receptors
- TLRs
Toll-like receptors
References
- Ahrne S, Nobaek S, Jeppsson B, Adlerberth I, Wold AE, Molin G (1998) The normal Lactobacillus flora of healthy human rectal and oral mucosa. J Appl Microbiol 85:88–94 [DOI] [PubMed]
- Bjorksten B, Naaber P, Sepp E, Mikelsaar M (1999) The intestinal microflora in allergic Estonian and Swedish 2-year-old children. Clin Exp Allergy 29:342–346 [DOI] [PubMed]
- Chen Y, Song K, Eck SL (2000) An intra-Peyer’s patch gene transfer model for studying mucosal tolerance: distinct roles of B7 and IL-12 in mucosal T cell tolerance. J Immunol 165:3145–3153 [DOI] [PubMed]
- Christensen HR, Frokiaer H, Pestka JJ (2002) Lactobacilli differentially modulate expression of cytokines and maturation surface markers in murine dendritic cells. J Immunol 168:171–178 [DOI] [PubMed]
- Drakes M, Blanchard T, Czinn S (2004) Bacterial probiotic modulation of dendritic cells. Infect Immun 72:3299–3309 [DOI] [PMC free article] [PubMed]
- Fujioka M, Hachimura S, Hosono A, Nakamura R, Hirayama K, Itoh K, Kaminogawa S (2004) Establishment and analysis of germfree T cell receptor transgenic mice. Anim Cell Technol: Basic & Appl Aspects 13:243–247
- Hessle C, Andersson B, Wold AE (2000) Gram-positive bacteria are potent inducers of monocytic interleukin-12 (IL-12) while gram-negative bacteria preferentially stimulate IL-10 production. Infect Immun 68:3581–3586 [DOI] [PMC free article] [PubMed]
- Itoh K, Mitsuoka T (1985) Characteriazation of clostridia isolated from faeces of limited flora mice and their effect on caecal size when associated with germ-free mice. Lab Anim 19:111–118 [DOI] [PubMed]
- Itoh K, Mitsuoka T, Sudo K, Suzuki K (1983) Comparison of fecal lactobacilli in mice of different strains under different housing conditions. Z Versuchstierkd 25:193–200 [PubMed]
- Kabelitz D (2007) Expression and function of Toll-like receptors in T lymphocytes. Curr Opin Immunol 19:39–45 [DOI] [PubMed]
- Kaisho T, Takeuchi O, Kawai T, Hoshino K, Akira S (2001) Endotoxin-induced maturation of MyD88-deficient dendritic cells. J Immunol 166:5688–5694 [DOI] [PubMed]
- Kaisho T, Hoshino K, Iwabe T, Takeuchi O, Yasui T, Akira S (2002) Endotoxin can induce MyD88-deficient dendritic cells to support T(h)2 cell differentiation. Int Immunol 14:695–700 [DOI] [PubMed]
- Kalinski P, Hilkens CM, Wierenga EA, Kapsenberg ML (1999) T-cell priming by type-1 and type-2 polarized dendritic cells: the concept of a third signal. Immunol Today 20:561–567 [DOI] [PubMed]
- Kalliomaki M, Kirjavainen P, Eerola E, Kero P, Salminen S, Isolauri E (2001) Distinct patterns of neonatal gut microflora in infants in whom atopy was and was not developing. J Allergy Clin Immunol 107:129–134 [DOI] [PubMed]
- Kononen E, Jousimies-Somer H, Asikainen S (1992) Relationship between oral gram-negative anaerobic bacteria in saliva of the mother and the colonization of her edentulous infant. Oral Microbiol Immunol 7:273–276 [DOI] [PubMed]
- Mazmanian SK, Liu CH, Tzianabos AO, Kasper DL (2005) An immunomodulatory molecule of symbiotic bacteria directs maturation of the host immune system. Cell 122:107–118 [DOI] [PubMed]
- Nakajima-Adachi H, Ebihara A, Kikuchi A, Ishida T, Sasaki K, Hirano K, Watanabe H, Asai K, Takahashi Y, Kanamori Y, Shimojo N, Matsuda H, Kohno Y, Hachimura S, Kaminogawa S (2006) Food antigen causes TH2-dependent enteropathy followed by tissue repair in T-cell receptor transgenic mice. J Allergy Clin Immunol 117:1125–1132 [DOI] [PubMed]
- Park SH, Itoh K (2005) Species-specific oligonucleotide probes for the detection and identification of Lactobacillus isolated from mouse faeces. J Appl Microbiol 99:51–57 [DOI] [PubMed]
- Pasare C, Medzhitov R (2004) Toll-dependent control mechanisms of CD4 T cell activation. Immunity 21:733–741 [DOI] [PubMed]
- Raymond CR, Wilkie BN (2005) Toll-like receptor, MHC II, B7 and cytokine expression by porcine monocytes and monocyte-derived dendritic cells in response to microbial pathogen-associated molecular patterns. Vet Immunol Immunopathol 107:235–247 [DOI] [PubMed]
- Sato S, Takeuchi O, Fujita T, Tomizawa H, Takeda K, Akira S (2002) A variety of microbial components induce tolerance to lipopolysaccharide by differentially affecting MyD88-dependent and -independent pathways. Int Immunol 14:783–791 [DOI] [PubMed]
- Shida K, Hachimura S, Ametani A, Ishimori M, Ling M, Hashiguchi M, Ueda Y, Sato T, Kumagai Y, Takamizawa K, Habu S, Kaminogawa S (2000) Serum IgE response to orally ingested antigen: a novel IgE response model with allergen-specific T-cell receptor transgenic mice. J Allergy Clin Immunol 105:788–795 [DOI] [PubMed]
- Snel J, Bakker MH, Heidt PJ (1997) Quantification of antigen-specific immunoglobulin A after oral booster immunization with ovalbumin in mice mono-associated with segmented filamentous bacteria or Clostridium innocuum. Immunol Lett 58:25–28 [DOI] [PubMed]
- Umesaki Y, Setoyama H, Matsumoto S, Imaoka A, Itoh K (1999) Differential roles of segmented filamentous bacteria and clostridia in development of the intestinal immune system. Infect Immun 67:3504–3511 [DOI] [PMC free article] [PubMed]
- Wang Q, McLoughlin RM, Cobb BA, Charrel-Dennis M, Zaleski KJ, Golenbock D, Tzianabos AO, Kasper DL (2006) A bacterial carbohydrate links innate and adaptive responses through Toll-like receptor 2. J Exp Med 203:2853–2863 [DOI] [PMC free article] [PubMed]
- Werling D, Hope JC, Howard CJ, Jungi TW (2004) Differential production of cytokines, reactive oxygen and nitrogen by bovine macrophages and dendritic cells stimulated with Toll-like receptor agonists. Immunology 111:41–52 [DOI] [PMC free article] [PubMed]


