Abstract
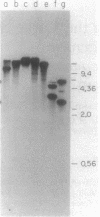
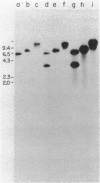
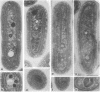
Transformation of the high-CO2-requiring mutants (hcr) O221 and E1 derived from the cyanobacterium Synechococcus sp. strain PCC 7942 by a wild-type DNA library restored their ability to grow at the level of CO2 in air. A plasmid (pE12) containing a 10-kilobase DNA insert was rescued from a O221 heterogenote and proved to transform both O221 and E1 to the wild-type phenotype. The capacity of the pE12 subclones to confer the wild-type phenotype to O221 transformants enabled the mapping of the mutation in O221 (designated hcrO221) within a 232-base-pair PstI-BstXI DNA restriction fragment. Sequence analysis revealed two open reading frames (ORFs) at positions -1745 to -1262 (ORFI) and -1218 to -393 (ORFII) upstream of the rbcL gene. A 3-kilobase PstI fragment of O221 was cloned, and hcrO221 was found to be a point mutation within the PstI-BstXI region -1309 nucleotides upstream of the rbcL gene. The significance of this flanking region for adaptation to air levels of CO2 was further demonstrated by the generation of new hcr mutants following insertional inactivation of wild-type DNA in the BstXI site. Electron microscopy revealed aberrant carboxysome structures in growing cells of the hcr mutants, a defect that was possibly related to the mutation, since transformation with pE12 derivatives restored the carboxysome structure to normal.
Full text
PDF







Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Badger M. R., Kaplan A., Berry J. A. Internal Inorganic Carbon Pool of Chlamydomonas reinhardtii: EVIDENCE FOR A CARBON DIOXIDE-CONCENTRATING MECHANISM. Plant Physiol. 1980 Sep;66(3):407–413. doi: 10.1104/pp.66.3.407. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Badger M. R. Kinetic properties of ribulose 1,5-bisphosphate carboxylase/oxygenase from Anabaena variabilis. Arch Biochem Biophys. 1980 Apr 15;201(1):247–254. doi: 10.1016/0003-9861(80)90509-3. [DOI] [PubMed] [Google Scholar]
- Friedberg D., Seijffers J. Controlled gene expression utilising lambda phage regulatory signals in a cyanobacterium host. Mol Gen Genet. 1986 Jun;203(3):505–510. doi: 10.1007/BF00422077. [DOI] [PubMed] [Google Scholar]
- Friedberg D. Use of reporter genes in cyanobacteria. Methods Enzymol. 1988;167:736–747. doi: 10.1016/0076-6879(88)67085-6. [DOI] [PubMed] [Google Scholar]
- Golden S. S., Nalty M. S., Cho D. S. Genetic relationship of two highly studied Synechococcus strains designated Anacystis nidulans. J Bacteriol. 1989 Jan;171(1):24–29. doi: 10.1128/jb.171.1.24-29.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marcus Y., Schwarz R., Friedberg D., Kaplan A. High CO(2) Requiring Mutant of Anacystis nidulans R(2). Plant Physiol. 1986 Oct;82(2):610–612. doi: 10.1104/pp.82.2.610. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marcus Y., Zenvirth D., Harel E., Kaplan A. Induction of HCO(3) Transporting Capability and High Photosynthetic Affinity to Inorganic Carbon by Low Concentration of CO(2) in Anabaena variabilis. Plant Physiol. 1982 May;69(5):1008–1012. doi: 10.1104/pp.69.5.1008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miller A. G., Colman B. Active transport and accumulation of bicarbonate by a unicellular cyanobacterium. J Bacteriol. 1980 Sep;143(3):1253–1259. doi: 10.1128/jb.143.3.1253-1259.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ogawa T., Kaneda T., Omata T. A Mutant of Synechococcus PCC7942 Incapable of Adapting to Low CO(2) Concentration. Plant Physiol. 1987 Jul;84(3):711–715. doi: 10.1104/pp.84.3.711. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oka A., Sugisaki H., Takanami M. Nucleotide sequence of the kanamycin resistance transposon Tn903. J Mol Biol. 1981 Apr 5;147(2):217–226. doi: 10.1016/0022-2836(81)90438-1. [DOI] [PubMed] [Google Scholar]
- Omata T., Ogawa T. Biosynthesis of a 42-kD Polypeptide in the Cytoplasmic Membrane of the Cyanobacterium Anacystis nidulans Strain R2 during Adaptation to Low CO(2) Concentration. Plant Physiol. 1986 Feb;80(2):525–530. doi: 10.1104/pp.80.2.525. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Omata T., Ogawa T., Marcus Y., Friedberg D., Kaplan A. Adaptation to Low CO(2) Level in a Mutant of Anacystis nidulans R(2) which Requires High CO(2) for Growth. Plant Physiol. 1987 Apr;83(4):892–894. doi: 10.1104/pp.83.4.892. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sanger F., Nicklen S., Coulson A. R. DNA sequencing with chain-terminating inhibitors. Proc Natl Acad Sci U S A. 1977 Dec;74(12):5463–5467. doi: 10.1073/pnas.74.12.5463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schwarz R., Friedberg D., Kaplan A. Is there a role for the 42 kilodalton polypeptide in inorganic carbon uptake by cyanobacteria? Plant Physiol. 1988 Oct;88(2):284–288. doi: 10.1104/pp.88.2.284. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shinozaki K., Yamada C., Takahata N., Sugiura M. Molecular cloning and sequence analysis of the cyanobacterial gene for the large subunit of ribulose-1,5-bisphosphate carboxylase/oxygenase. Proc Natl Acad Sci U S A. 1983 Jul;80(13):4050–4054. doi: 10.1073/pnas.80.13.4050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Spurr A. R. A low-viscosity epoxy resin embedding medium for electron microscopy. J Ultrastruct Res. 1969 Jan;26(1):31–43. doi: 10.1016/s0022-5320(69)90033-1. [DOI] [PubMed] [Google Scholar]
- Tabita F. R., Small C. L. Expression and assembly of active cyanobacterial ribulose-1,5-bisphosphate carboxylase/oxygenase in Escherichia coli containing stoichiometric amounts of large and small subunits. Proc Natl Acad Sci U S A. 1985 Sep;82(18):6100–6103. doi: 10.1073/pnas.82.18.6100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vieira J., Messing J. Production of single-stranded plasmid DNA. Methods Enzymol. 1987;153:3–11. doi: 10.1016/0076-6879(87)53044-0. [DOI] [PubMed] [Google Scholar]
- Williams J. G., Szalay A. A. Stable integration of foreign DNA into the chromosome of the cyanobacterium Synechococcus R2. Gene. 1983 Sep;24(1):37–51. doi: 10.1016/0378-1119(83)90129-4. [DOI] [PubMed] [Google Scholar]