Abstract
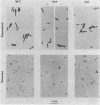
We examined several aspects of cell division regulation in Escherichia coli which have been thought to be controlled by cyclic AMP (cAMP) and its receptor protein (CAP). Mutants lacking adenyl cyclase (cya) or CAP (crp) were rod shaped, not spherical, during exponential growth in LB broth or glucose-Casamino Acids medium, and lateral wall elongation was normal; in broth, stationary-phase cells became ovoid. Cell mass was smaller for the mutants than for the wild type, but it remained appropriate for their slower growth rate and thus probably does not reflect early (uncontrolled) septation. The slow growth did not seem to reflect a gross metabolic disorder, since the mutants gave a normal yield on limiting glucose; surprisingly, however, the cya mutant (unlike crp) was unable to grow anaerobically on glucose, suggesting a role for cAMP (but not for CAP) in the expression of some fermentation enzyme. Both cya and crp mutants are known to be resistant to mecillinam, an antibiotic which inhibits penicillin-binding protein 2 (involved in lateral wall elongation) and also affects septation. This resistance does not reflect a lack of PBP2. Furthermore, it was not simply the result of slow growth and small cell mass, since small wild-type cells growing in acetate remained sensitive. The cAMP-CAP complex may regulate the synthesis of some link between PBP2 and the septation apparatus. The ftsZ gene, coding for a cell division protein, was expressed at a higher level in the absence of cAMP, as measured with an ftsZ::lacZ fusion, but the amount of protein per cell, shown by others to be invariable over a 10-fold range of cell mass, was independent of cAMP, suggesting that ftsZ expression is not regulated by the cAMP-CAP complex.
Full text
PDF




Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Aono R., Yamasaki M., Tamura G. High and selective resistance to mecillinam in adenylate cyclase-deficient or cyclic adenosine 3',5'-monophosphate receptor protein-deficient mutants of Escherichia coli. J Bacteriol. 1979 Feb;137(2):839–845. doi: 10.1128/jb.137.2.839-845.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Begg K. J., Spratt B. G., Donachie W. D. Interaction between membrane proteins PBP3 and rodA is required for normal cell shape and division in Escherichia coli. J Bacteriol. 1986 Sep;167(3):1004–1008. doi: 10.1128/jb.167.3.1004-1008.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Benner D., Müller N., Boos W. Temperature-sensitive catabolite activator protein in Escherichia coli BUG6. J Bacteriol. 1985 Jan;161(1):347–352. doi: 10.1128/jb.161.1.347-352.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Botsford J. L. Cyclic nucleotides in procaryotes. Microbiol Rev. 1981 Dec;45(4):620–642. doi: 10.1128/mr.45.4.620-642.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Braedt G., Gallant J. Role of the rel gene product in the control of cyclic adenosine 3',5'-monophosphate accumulation. J Bacteriol. 1977 Jan;129(1):564–566. doi: 10.1128/jb.129.1.564-566.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brickman E., Soll L., Beckwith J. Genetic characterization of mutations which affect catabolite-sensitive operons in Escherichia coli, including deletions of the gene for adenyl cyclase. J Bacteriol. 1973 Nov;116(2):582–587. doi: 10.1128/jb.116.2.582-587.1973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Canepari P., Botta G., Satta G. Inhibition of lateral wall elongation by mecillinam stimulates cell division in certain cell division conditional mutants of Escherichia coli. J Bacteriol. 1984 Jan;157(1):130–133. doi: 10.1128/jb.157.1.130-133.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dills S. E., Dobrogosz W. J. Cyclic adenosine 3',5'-monophosphate regulation of membrane energetics in Escherichia coli. J Bacteriol. 1977 Sep;131(3):854–865. doi: 10.1128/jb.131.3.854-865.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Freedberg W. B., Lin E. C. Three kinds of controls affecting the expression of the glp regulon in Escherichia coli. J Bacteriol. 1973 Sep;115(3):816–823. doi: 10.1128/jb.115.3.816-823.1973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Freundlich M. Cyclic AMP can replace the relA-dependent requirement for derepression of acetohydroxy acid synthase in E. coli K-12. Cell. 1977 Dec;12(4):1121–1126. doi: 10.1016/0092-8674(77)90174-x. [DOI] [PubMed] [Google Scholar]
- Holland I. B., Jones C. The role of the FtsZ protein (SfiB) in UV-induced division inhibition and in the normal Escherichia coli cell division cycle. Ann Inst Pasteur Microbiol. 1985 Jan-Feb;136A(1):165–171. doi: 10.1016/s0769-2609(85)80037-5. [DOI] [PubMed] [Google Scholar]
- Jaffé A., Chabbert Y. A., Derlot E. Selection and characterization of beta-lactam-resistant Escherichia coli K-12 mutants. Antimicrob Agents Chemother. 1983 Apr;23(4):622–625. doi: 10.1128/aac.23.4.622. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jaffé A., D'Ari R., Norris V. SOS-independent coupling between DNA replication and cell division in Escherichia coli. J Bacteriol. 1986 Jan;165(1):66–71. doi: 10.1128/jb.165.1.66-71.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kumar S., Prakash N., Agarwal K. N. Cyclic AMP control of the envelope growth in Escherichia coli: envelope morphology of the mutants in cya and crp genes. Indian J Exp Biol. 1979 Apr;17(4):325–327. [PubMed] [Google Scholar]
- Kumar S. Properties of adenyl cyclase and cyclic adenosine 3',5'-monophosphate receptor protein-deficient mutants of Escherichia coli. J Bacteriol. 1976 Feb;125(2):545–555. doi: 10.1128/jb.125.2.545-555.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lo T. C., Rayman M. K., Sanwal B. D. Transport of succinate in Escherichia coli. I. Biochemical and genetic studies of transport in whole cells. J Biol Chem. 1972 Oct 10;247(19):6323–6331. [PubMed] [Google Scholar]
- Matsuhashi S., Kamiryo T., Blumberg P. M., Linnett P., Willoughby E., Strominger J. L. Mechanism of action and development of resistance to a new amidino penicillin. J Bacteriol. 1974 Feb;117(2):578–587. doi: 10.1128/jb.117.2.578-587.1974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Park J. T., Burman L. FL-1060: a new penicillin with a unique mode of action. Biochem Biophys Res Commun. 1973 Apr 16;51(4):863–868. doi: 10.1016/0006-291x(73)90006-5. [DOI] [PubMed] [Google Scholar]
- Pastan I., Adhya S. Cyclic adenosine 5'-monophosphate in Escherichia coli. Bacteriol Rev. 1976 Sep;40(3):527–551. doi: 10.1128/br.40.3.527-551.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Patrick J. M., Dobrogosz W. J. The effect of cyclic AMP on anaerobic growth of Escherichia coli. Biochem Biophys Res Commun. 1973 Sep 18;54(2):555–561. doi: 10.1016/0006-291x(73)91458-7. [DOI] [PubMed] [Google Scholar]
- Postma P. W., Lengeler J. W. Phosphoenolpyruvate:carbohydrate phosphotransferase system of bacteria. Microbiol Rev. 1985 Sep;49(3):232–269. doi: 10.1128/mr.49.3.232-269.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sabourin D., Beckwith J. Deletion of the Escherichia coli crp gene. J Bacteriol. 1975 Apr;122(1):338–340. doi: 10.1128/jb.122.1.338-340.1975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smith M. W., Neidhardt F. C. 2-Oxoacid dehydrogenase complexes of Escherichia coli: cellular amounts and patterns of synthesis. J Bacteriol. 1983 Oct;156(1):81–88. doi: 10.1128/jb.156.1.81-88.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Spratt B. G., Boyd A., Stoker N. Defective and plaque-forming lambda transducing bacteriophage carrying penicillin-binding protein-cell shape genes: genetic and physical mapping and identification of gene products from the lip-dacA-rodA-pbpA-leuS region of the Escherichia coli chromosome. J Bacteriol. 1980 Aug;143(2):569–581. doi: 10.1128/jb.143.2.569-581.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Spratt B. G. Distinct penicillin binding proteins involved in the division, elongation, and shape of Escherichia coli K12. Proc Natl Acad Sci U S A. 1975 Aug;72(8):2999–3003. doi: 10.1073/pnas.72.8.2999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Spratt B. G., Pardee A. B. Penicillin-binding proteins and cell shape in E. coli. Nature. 1975 Apr 10;254(5500):516–517. doi: 10.1038/254516a0. [DOI] [PubMed] [Google Scholar]
- Spratt B. G. The mechanism of action of mecillinam. J Antimicrob Chemother. 1977 Jul;3 (Suppl B):13–19. doi: 10.1093/jac/3.suppl_b.13. [DOI] [PubMed] [Google Scholar]
- Tormo A., Ayala J. A., de Pedro M. A., Aldea M., Vicente M. Interaction of FtsA and PBP3 proteins in the Escherichia coli septum. J Bacteriol. 1986 Jun;166(3):985–992. doi: 10.1128/jb.166.3.985-992.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Unden G., Duchene A. On the role of cyclic AMP and the Fnr protein in Escherichia coli growing anaerobically. Arch Microbiol. 1987 Mar;147(2):195–200. doi: 10.1007/BF00415284. [DOI] [PubMed] [Google Scholar]
- Unden G., Guest J. R. Cyclic AMP and anaerobic gene expression in E. coli. FEBS Lett. 1984 May 21;170(2):321–325. doi: 10.1016/0014-5793(84)81336-8. [DOI] [PubMed] [Google Scholar]
- Utsumi R., Nakamoto Y., Kawamukai M., Himeno M., Komano T. Involvement of cyclic AMP and its receptor protein in filamentation of an Escherichia coli fic mutant. J Bacteriol. 1982 Aug;151(2):807–812. doi: 10.1128/jb.151.2.807-812.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ward J. E., Jr, Lutkenhaus J. Overproduction of FtsZ induces minicell formation in E. coli. Cell. 1985 Oct;42(3):941–949. doi: 10.1016/0092-8674(85)90290-9. [DOI] [PubMed] [Google Scholar]
- Westling-Häggström B., Normark S. Genetic and physiological analysis of an envB spherelike mutant of Escherichia coli K-12 and characterization of its transductants. J Bacteriol. 1975 Jul;123(1):75–82. doi: 10.1128/jb.123.1.75-82.1975. [DOI] [PMC free article] [PubMed] [Google Scholar]