Abstract
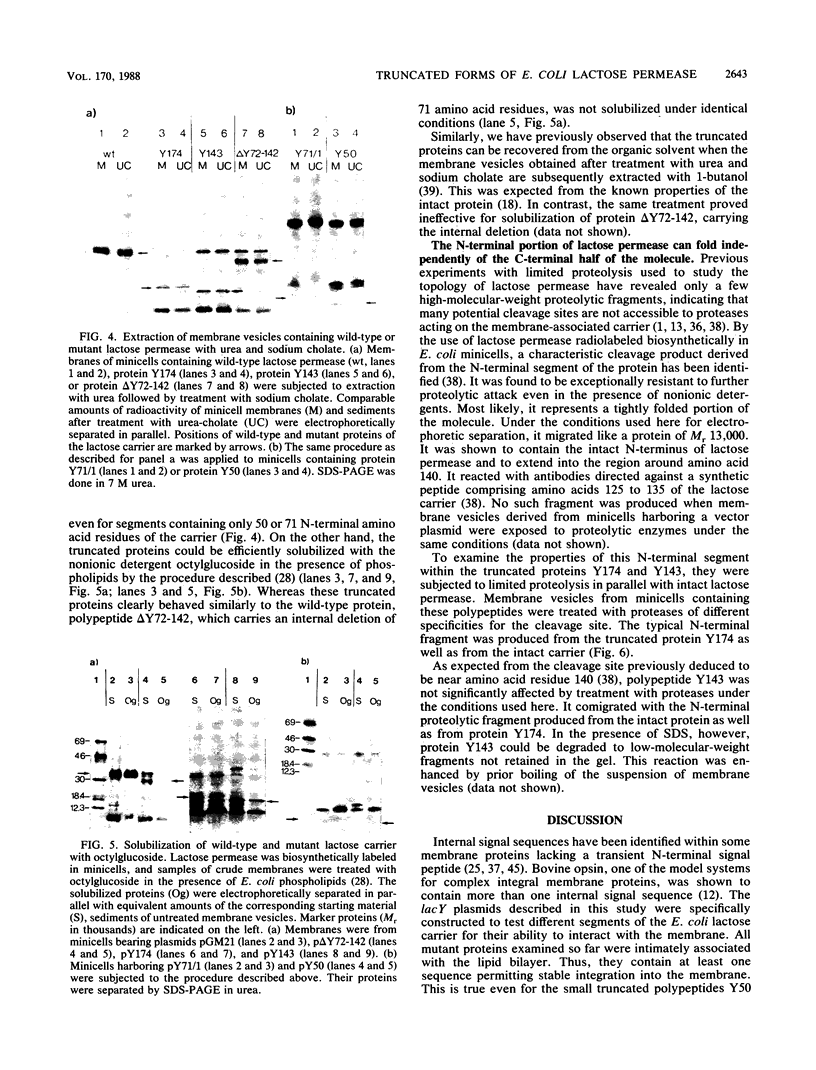
Using in vitro DNA manipulations, we constructed different lacY alleles encoding mutant proteins of the Escherichia coli lactose carrier. With respect to structural models developed for lactose permease, the truncated polypeptides represent model systems containing approximately one, two, four, and five of the N-terminal membrane-spanning alpha-helices. In addition, a protein carrying a deletion of predicted helices 3 and 4 was obtained. The different proteins were radiolabeled in plasmid-bearing E. coli minicells and were found to be stably integrated into the lipid bilayer. The truncated polypeptides of 50, 71, 143, and 174 N-terminal amino acid residues resembled the wild-type protein in their solubilization characteristics, whereas the mutant protein carrying an internal deletion of amino acid residues 72 to 142 of the lactose carrier behaved differently. Minicell membrane vesicles containing truncated proteins comprising amino acid residues 1 to 143 or 1 to 174 were subjected to limited proteolysis. Upon digestion with proteases of different specificities, the same characteristic fragment that was also produced from the membrane-associated wild-type protein was found to accumulate under these conditions. It has previously been shown to contain the intact N terminus of lactose permease. This supports the idea of an independent folding and membrane insertion of this segment even in the absence of the C-terminal part of the molecule. The results suggest that the N-terminal region of the lactose permease represents a well-defined structural domain.
Full text
PDF





Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bieseler B., Prinz H., Beyreuther K. Topological studies of lactose permease of Escherichia coli by protein sequence analysis. Ann N Y Acad Sci. 1985;456:309–325. doi: 10.1111/j.1749-6632.1985.tb14882.x. [DOI] [PubMed] [Google Scholar]
- Brooker R. J., Wilson T. H. Isolation and nucleotide sequencing of lactose carrier mutants that transport maltose. Proc Natl Acad Sci U S A. 1985 Jun;82(12):3959–3963. doi: 10.1073/pnas.82.12.3959. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brooker R. J., Wilson T. H. Site-specific alteration of cysteine 176 and cysteine 234 in the lactose carrier of Escherichia coli. J Biol Chem. 1986 Sep 5;261(25):11765–11769. [PubMed] [Google Scholar]
- Burr F. A., Burr B. Slab gel system for the resolution of oligopeptides below molecular weight of 10,000. Methods Enzymol. 1983;96:239–244. doi: 10.1016/s0076-6879(83)96022-6. [DOI] [PubMed] [Google Scholar]
- Büchel D. E., Gronenborn B., Müller-Hill B. Sequence of the lactose permease gene. Nature. 1980 Feb 7;283(5747):541–545. doi: 10.1038/283541a0. [DOI] [PubMed] [Google Scholar]
- Carrasco N., Antes L. M., Poonian M. S., Kaback H. R. lac permease of Escherichia coli: histidine-322 and glutamic acid-325 may be components of a charge-relay system. Biochemistry. 1986 Aug 12;25(16):4486–4488. doi: 10.1021/bi00364a004. [DOI] [PubMed] [Google Scholar]
- Chang A. C., Cohen S. N. Construction and characterization of amplifiable multicopy DNA cloning vehicles derived from the P15A cryptic miniplasmid. J Bacteriol. 1978 Jun;134(3):1141–1156. doi: 10.1128/jb.134.3.1141-1156.1978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ehring R., Beyreuther K., Wright J. K., Overath P. In vitro and in vivo products of E. coli lactose permease gene are identical. Nature. 1980 Feb 7;283(5747):537–540. doi: 10.1038/283537a0. [DOI] [PubMed] [Google Scholar]
- Engelman D. M., Steitz T. A. The spontaneous insertion of proteins into and across membranes: the helical hairpin hypothesis. Cell. 1981 Feb;23(2):411–422. doi: 10.1016/0092-8674(81)90136-7. [DOI] [PubMed] [Google Scholar]
- Foster D. L., Boublik M., Kaback H. R. Structure of the lac carrier protein of Escherichia coli. J Biol Chem. 1983 Jan 10;258(1):31–34. [PubMed] [Google Scholar]
- Friedlander M., Blobel G. Bovine opsin has more than one signal sequence. 1985 Nov 28-Dec 4Nature. 318(6044):338–343. doi: 10.1038/318338a0. [DOI] [PubMed] [Google Scholar]
- Goldkorn T., Rimon G., Kaback H. R. Topology of the lac carrier protein in the membrane of Escherichia coli. Proc Natl Acad Sci U S A. 1983 Jun;80(11):3322–3326. doi: 10.1073/pnas.80.11.3322. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Griesser H. W., Müller-Hill B., Overath P. Characterization of beta-galactosidase--lactose-permease chimaeras of Escherichia coli. Eur J Biochem. 1983 Dec 15;137(3):567–572. doi: 10.1111/j.1432-1033.1983.tb07863.x. [DOI] [PubMed] [Google Scholar]
- Hanahan D. Studies on transformation of Escherichia coli with plasmids. J Mol Biol. 1983 Jun 5;166(4):557–580. doi: 10.1016/s0022-2836(83)80284-8. [DOI] [PubMed] [Google Scholar]
- Heffron F., So M., McCarthy B. J. In vitro mutagenesis of a circular DNA molecule by using synthetic restriction sites. Proc Natl Acad Sci U S A. 1978 Dec;75(12):6012–6016. doi: 10.1073/pnas.75.12.6012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kaback H. R. The lac carrier protein in Escherichia coli. J Membr Biol. 1983;76(2):95–112. doi: 10.1007/BF02000610. [DOI] [PubMed] [Google Scholar]
- Kuhnke G., Krause A., Heibach C., Gieske U., Fritz H. J., Ehring R. The upstream operator of the Escherichia coli galactose operon is sufficient for repression of transcription initiated at the cyclic AMP-stimulated promoter. EMBO J. 1986 Jan;5(1):167–173. doi: 10.1002/j.1460-2075.1986.tb04192.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- König B., Sandermann H., Jr Beta-D-galactoside transport in Escherichia coli. Solubilization in organic solvent and reconstitution of binding. Eur J Biochem. 1984 Dec 3;145(2):397–402. doi: 10.1111/j.1432-1033.1984.tb08567.x. [DOI] [PubMed] [Google Scholar]
- Lugtenberg B., Meijers J., Peters R., van der Hoek P., van Alphen L. Electrophoretic resolution of the "major outer membrane protein" of Escherichia coli K12 into four bands. FEBS Lett. 1975 Oct 15;58(1):254–258. doi: 10.1016/0014-5793(75)80272-9. [DOI] [PubMed] [Google Scholar]
- Markgraf M., Bocklage H., Müller-Hill B. A change of threonine 266 to isoleucine in the lac permease of Escherichia coli diminishes the transport of lactose and increases the transport of maltose. Mol Gen Genet. 1985;198(3):473–475. doi: 10.1007/BF00332941. [DOI] [PubMed] [Google Scholar]
- Maxam A. M., Gilbert W. Sequencing end-labeled DNA with base-specific chemical cleavages. Methods Enzymol. 1980;65(1):499–560. doi: 10.1016/s0076-6879(80)65059-9. [DOI] [PubMed] [Google Scholar]
- Mieschendahl M., Büchel D., Bocklage H., Müller-Hill B. Mutations in the lacY gene of Escherichia coli define functional organization of lactose permease. Proc Natl Acad Sci U S A. 1981 Dec;78(12):7652–7656. doi: 10.1073/pnas.78.12.7652. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mueckler M., Lodish H. F. The human glucose transporter can insert posttranslationally into microsomes. Cell. 1986 Feb 28;44(4):629–637. doi: 10.1016/0092-8674(86)90272-2. [DOI] [PubMed] [Google Scholar]
- Nelson S. O., Wright J. K., Postma P. W. The mechanism of inducer exclusion. Direct interaction between purified III of the phosphoenolpyruvate:sugar phosphotransferase system and the lactose carrier of Escherichia coli. EMBO J. 1983;2(5):715–720. doi: 10.1002/j.1460-2075.1983.tb01490.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Neuhaus J. M., Soppa J., Wright J. K., Riede I., Blöcker H., Frank R., Overath P. Properties of a mutant lactose carrier of Escherichia coli with a Cys148----Ser148 substitution. FEBS Lett. 1985 Jun 3;185(1):83–88. doi: 10.1016/0014-5793(85)80745-6. [DOI] [PubMed] [Google Scholar]
- Newman M. J., Foster D. L., Wilson T. H., Kaback H. R. Purification and reconstitution of functional lactose carrier from Escherichia coli. J Biol Chem. 1981 Nov 25;256(22):11804–11808. [PubMed] [Google Scholar]
- Newman M. J., Wilson T. H. Solubilization and reconstitution of the lactose transport system from Escherichia coli. J Biol Chem. 1980 Nov 25;255(22):10583–10586. [PubMed] [Google Scholar]
- Oliver D. Protein secretion in Escherichia coli. Annu Rev Microbiol. 1985;39:615–648. doi: 10.1146/annurev.mi.39.100185.003151. [DOI] [PubMed] [Google Scholar]
- Overath P., Weigel U., Neuhaus J. M., Soppa J., Seckler R., Riede I., Bocklage H., Müller-Hill B., Aichele G., Wright J. K. Lactose permease of Escherichia coli: properties of mutants defective in substrate translocation. Proc Natl Acad Sci U S A. 1987 Aug;84(16):5535–5539. doi: 10.1073/pnas.84.16.5535. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Püttner I. B., Sarkar H. K., Poonian M. S., Kaback H. R. lac permease of Escherichia coli: histidine-205 and histidine-322 play different roles in lactose/H+ symport. Biochemistry. 1986 Aug 12;25(16):4483–4485. doi: 10.1021/bi00364a003. [DOI] [PubMed] [Google Scholar]
- Reeve J. N. Bacteriophage infection of minicells: a general method for identification of "in vivo" bacteriophage directed polypeptide biosynthesis. Mol Gen Genet. 1977 Dec 14;158(1):73–79. doi: 10.1007/BF00455121. [DOI] [PubMed] [Google Scholar]
- Sarkar H. K., Viitanen P. V., Poonian M. S., Kaback H. R. Substitution of glutamine-60 with glutamic acid causes the lac permease of Escherichia coli to become temperature sensitive. Biochemistry. 1986 May 20;25(10):2778–2781. doi: 10.1021/bi00358a006. [DOI] [PubMed] [Google Scholar]
- Seckler R., Möröy T., Wright J. K., Overath P. Anti-peptide antibodies and proteases as structural probes for the lactose/H+ transporter of Escherichia coli: a loop around amino acid residue 130 faces the cytoplasmic side of the membrane. Biochemistry. 1986 May 6;25(9):2403–2409. doi: 10.1021/bi00357a016. [DOI] [PubMed] [Google Scholar]
- Seckler R., Wright J. K., Overath P. Peptide-specific antibody locates the COOH terminus of the lactose carrier of Escherichia coli on the cytoplasmic side of the plasma membrane. J Biol Chem. 1983 Sep 25;258(18):10817–10820. [PubMed] [Google Scholar]
- Spiess M., Lodish H. F. An internal signal sequence: the asialoglycoprotein receptor membrane anchor. Cell. 1986 Jan 17;44(1):177–185. doi: 10.1016/0092-8674(86)90496-4. [DOI] [PubMed] [Google Scholar]
- Stochaj U., Bieseler B., Ehring R. Limited proteolysis of lactose permease from Escherichia coli. Eur J Biochem. 1986 Jul 15;158(2):423–428. doi: 10.1111/j.1432-1033.1986.tb09770.x. [DOI] [PubMed] [Google Scholar]
- Stochaj U., Ehring R. The N-terminal region of Escherichia coli lactose permease mediates membrane contact of the nascent polypeptide chain. Eur J Biochem. 1987 Mar 16;163(3):653–658. doi: 10.1111/j.1432-1033.1987.tb10914.x. [DOI] [PubMed] [Google Scholar]
- Sutcliffe J. G. Complete nucleotide sequence of the Escherichia coli plasmid pBR322. Cold Spring Harb Symp Quant Biol. 1979;43(Pt 1):77–90. doi: 10.1101/sqb.1979.043.01.013. [DOI] [PubMed] [Google Scholar]
- Teather R. M., Bramhall J., Riede I., Wright J. K., Fürst M., Aichele G., Wilhelm U., Overath P. Lactose carrier protein of Escherichia coli. Structure and expression of plasmids carrying the Y gene of the lac operon. Eur J Biochem. 1980;108(1):223–231. doi: 10.1111/j.1432-1033.1980.tb04715.x. [DOI] [PubMed] [Google Scholar]
- Viitanen P. V., Menick D. R., Sarkar H. K., Trumble W. R., Kaback H. R. Site-directed mutagenesis of cysteine-148 in the lac permease of Escherichia coli: effect on transport, binding, and sulfhydryl inactivation. Biochemistry. 1985 Dec 17;24(26):7628–7635. doi: 10.1021/bi00347a020. [DOI] [PubMed] [Google Scholar]
- Vogel H., Wright J. K., Jähnig F. The structure of the lactose permease derived from Raman spectroscopy and prediction methods. EMBO J. 1985 Dec 16;4(13A):3625–3631. doi: 10.1002/j.1460-2075.1985.tb04126.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wickner W. T., Lodish H. F. Multiple mechanisms of protein insertion into and across membranes. Science. 1985 Oct 25;230(4724):400–407. doi: 10.1126/science.4048938. [DOI] [PubMed] [Google Scholar]
- Wright J. K., Seckler R., Overath P. Molecular aspects of sugar:ion cotransport. Annu Rev Biochem. 1986;55:225–248. doi: 10.1146/annurev.bi.55.070186.001301. [DOI] [PubMed] [Google Scholar]
- von Heijne G., Blomberg C. Trans-membrane translocation of proteins. The direct transfer model. Eur J Biochem. 1979 Jun;97(1):175–181. doi: 10.1111/j.1432-1033.1979.tb13100.x. [DOI] [PubMed] [Google Scholar]