Abstract
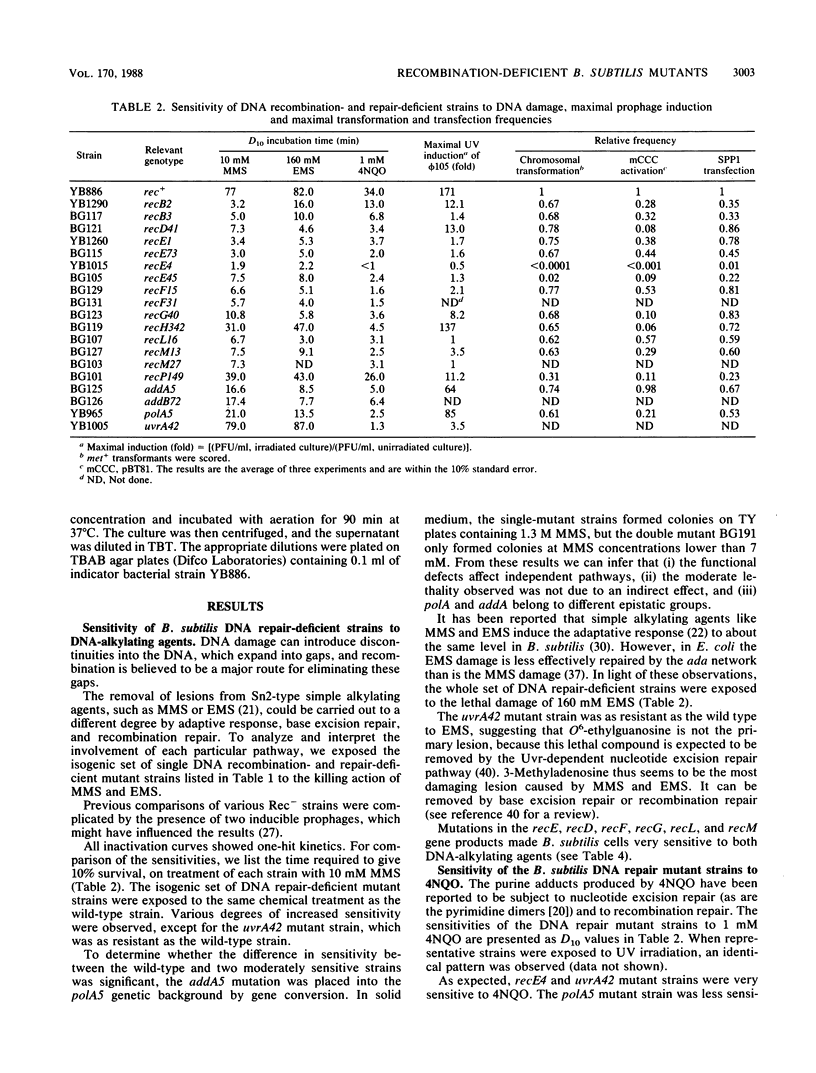
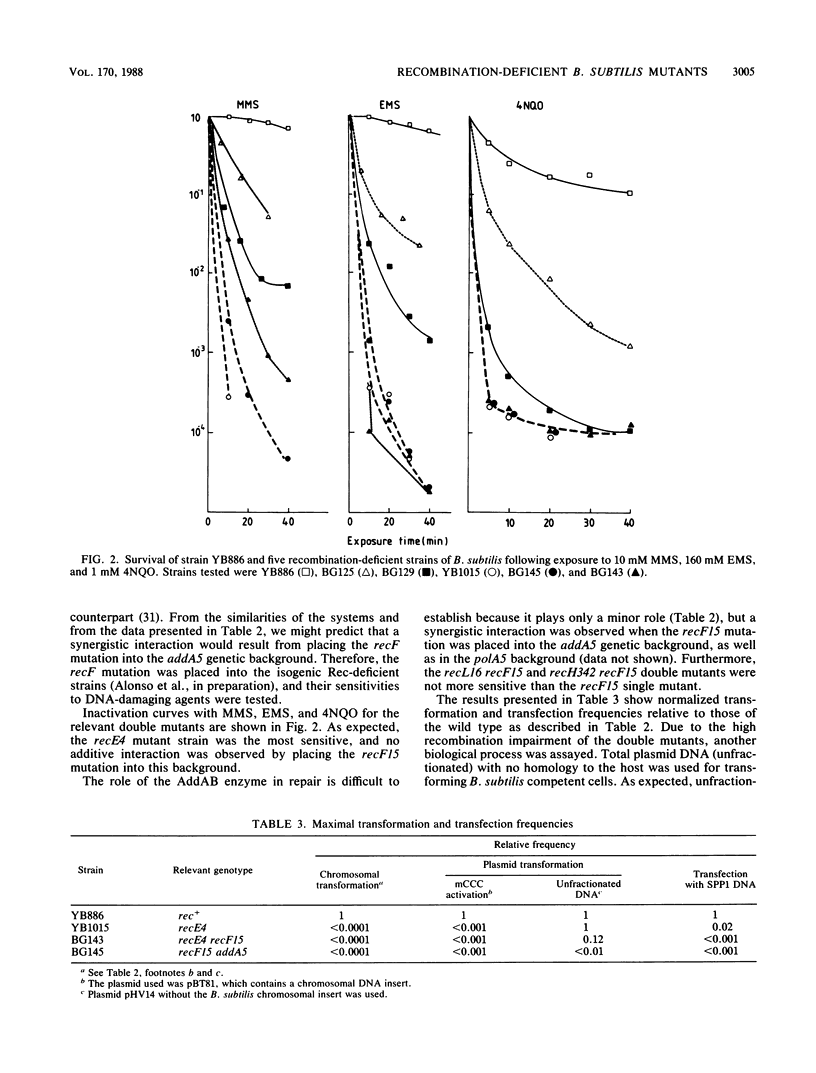
An isogenic set of "prophage-free," DNA repair-proficient and -deficient strains of Bacillus subtilis were characterized phenotypically. The mutant strains were provisionally classified into four categories on the basis of their sensitivity to DNA-damaging agents, their ability to release phage after lysogenization followed by damage to chromosomal DNA, and their impairment in genetic exchange. The properties of double Rec- mutants showed that recF and addA belong to different epistatic groups, whereas recF, recL, and recH fall into the same group. More than one pathway for genetic exchange might be operative in B. subtilis.
Full text
PDF




Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Alonso J. C., Trautner T. A. A gene controlling segregation of the Bacillus subtilis plasmid pC194. Mol Gen Genet. 1985;198(3):427–431. doi: 10.1007/BF00332934. [DOI] [PubMed] [Google Scholar]
- Alonso J. C., Viret J. F., Tailor R. H. Plasmid maintenance in Bacillus subtilis recombination-deficient mutants. Mol Gen Genet. 1987 Jun;208(1-2):349–352. doi: 10.1007/BF00330464. [DOI] [PubMed] [Google Scholar]
- Biswal N., Kleinschmidt A. K., Spatz H. C., Trautner T. A. Physical properties of the DNA of bacteriophage SP50. Mol Gen Genet. 1967;100(1):39–55. doi: 10.1007/BF00425774. [DOI] [PubMed] [Google Scholar]
- Canosi U., Iglesias A., Trautner T. A. Plasmid transformation in Bacillus subtilis: effects of insertion of Bacillus subtilis DNA into plasmid pC194. Mol Gen Genet. 1981;181(4):434–440. doi: 10.1007/BF00428732. [DOI] [PubMed] [Google Scholar]
- Canosi U., Morelli G., Trautner T. A. The relationship between molecular structure and transformation efficiency of some S. aureus plasmids isolated from B. subtilis. Mol Gen Genet. 1978 Nov 9;166(3):259–267. doi: 10.1007/BF00267617. [DOI] [PubMed] [Google Scholar]
- Chestukhin A. V., Shemyakin M. F., Kalinina N. A., Prozorov A. A. Some properties of ATP dependent deoxyribonucleases from normal and rec-mutant strains of Bacillus subtilis. FEBS Lett. 1972 Jul 15;24(1):121–125. doi: 10.1016/0014-5793(72)80841-x. [DOI] [PubMed] [Google Scholar]
- Clark A. J., Sandler S. J., Willis D. K., Chu C. C., Blanar M. A., Lovett S. T. Genes of the RecE and RecF pathways of conjugational recombination in Escherichia coli. Cold Spring Harb Symp Quant Biol. 1984;49:453–462. doi: 10.1101/sqb.1984.049.01.051. [DOI] [PubMed] [Google Scholar]
- Deichelbohrer I., Alonso J. C., Lüder G., Trautner T. A. Plasmid transduction by Bacillus subtilis bacteriophage SPP1: effects of DNA homology between plasmid and bacteriophage. J Bacteriol. 1985 Jun;162(3):1238–1243. doi: 10.1128/jb.162.3.1238-1243.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dodson L. A., Hadden C. T. Capacity for postreplication repair correlated with transducibility in Rec- mutants of Bacillus subtilis. J Bacteriol. 1980 Nov;144(2):608–615. doi: 10.1128/jb.144.2.608-615.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Doly J., Le Roscouët D., Anagnostopoulos C. Substrate specificity and adenosine triphosphatase activity of the ATP-dependent deoxyribonuclease of Bacillus subtilis. Eur J Biochem. 1981 Mar;114(3):493–499. doi: 10.1111/j.1432-1033.1981.tb05172.x. [DOI] [PubMed] [Google Scholar]
- Doly J., Sasarman E., Anagnostopoulos C. ATP-dependent deoxyribonuclease in Bacillus subtilis and a mutant deficient in this activity. Mutat Res. 1974 Jan;22(1):15–23. doi: 10.1016/0027-5107(74)90003-7. [DOI] [PubMed] [Google Scholar]
- Dubnau D., Davidoff-Abelson R., Scher B., Cirigliano C. Fate of transforming deoxyribonucleic acid after uptake by competent Bacillus subtilis: phenotypic characterization of radiation-sensitive recombination-deficient mutants. J Bacteriol. 1973 Apr;114(1):273–286. doi: 10.1128/jb.114.1.273-286.1973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Friedman B. M., Yasbin R. E. The genetics and specificity of the constitutive excision repair system of Bacillus subtilis. Mol Gen Genet. 1983;190(3):481–486. doi: 10.1007/BF00331080. [DOI] [PubMed] [Google Scholar]
- Goldmark P. J., Linn S. Purification and properties of the recBC DNase of Escherichia coli K-12. J Biol Chem. 1972 Mar 25;247(6):1849–1860. [PubMed] [Google Scholar]
- Hadden C. T. Transformability of DNA polymerase-deficient mutants of Bacillus subtilis. Biochem Biophys Res Commun. 1973 Apr 2;51(3):501–506. doi: 10.1016/0006-291x(73)91342-9. [DOI] [PubMed] [Google Scholar]
- Hoch J. A., Anagnostopoulos C. Chromosomal location and properties of radiation sensitivity mutations in Bacillus subtilis. J Bacteriol. 1970 Aug;103(2):295–301. doi: 10.1128/jb.103.2.295-301.1970. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kondo S., Ichikawa H., Iwo K., Kato T. Base-change mutagenesis and prophage induction in strains of Escherichia coli with different DNA repair capacities. Genetics. 1970 Oct;66(2):187–217. doi: 10.1093/genetics/66.2.187. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lawley P. D. Some chemical aspects of dose-response relationships in alkylation mutagenesis. Mutat Res. 1974 Jun;23(3):283–295. doi: 10.1016/0027-5107(74)90102-x. [DOI] [PubMed] [Google Scholar]
- Lindahl T. DNA repair enzymes. Annu Rev Biochem. 1982;51:61–87. doi: 10.1146/annurev.bi.51.070182.000425. [DOI] [PubMed] [Google Scholar]
- Love P. E., Yasbin R. E. Genetic characterization of the inducible SOS-like system of Bacillus subtilis. J Bacteriol. 1984 Dec;160(3):910–920. doi: 10.1128/jb.160.3.910-920.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Love P. E., Yasbin R. E. Induction of the Bacillus subtilis SOS-like response by Escherichia coli RecA protein. Proc Natl Acad Sci U S A. 1986 Jul;83(14):5204–5208. doi: 10.1073/pnas.83.14.5204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lovett C. M., Jr, Roberts J. W. Purification of a RecA protein analogue from Bacillus subtilis. J Biol Chem. 1985 Mar 25;260(6):3305–3313. [PubMed] [Google Scholar]
- Martinez S., Lopez P., Espinosa M., Lacks S. A. Complementation of Bacillus subtilis polA mutants by DNA polymerase I from Streptococcus pneumoniae. Mol Gen Genet. 1987 Dec;210(2):203–210. doi: 10.1007/BF00325685. [DOI] [PubMed] [Google Scholar]
- Mazza G., Fortunato A., Ferrari E., Canosi U., Falaschi A., Polsinelli M. Genetic and enzymic studies on the recombination process in Bacillus subtilis. Mol Gen Genet. 1975;136(1):9–30. doi: 10.1007/BF00275445. [DOI] [PubMed] [Google Scholar]
- McPartland A., Green L., Echols H. Control of recA gene RNA in E. coli: regulatory and signal genes. Cell. 1980 Jul;20(3):731–737. doi: 10.1016/0092-8674(80)90319-0. [DOI] [PubMed] [Google Scholar]
- Morohoshi F., Munakata N. Two classes of Bacillus subtilis mutants deficient in the adaptive response to simple alkylating agents. Mol Gen Genet. 1986 Feb;202(2):200–206. doi: 10.1007/BF00331637. [DOI] [PubMed] [Google Scholar]
- Ogasawara N., Moriya S., von Meyenburg K., Hansen F. G., Yoshikawa H. Conservation of genes and their organization in the chromosomal replication origin region of Bacillus subtilis and Escherichia coli. EMBO J. 1985 Dec 1;4(12):3345–3350. doi: 10.1002/j.1460-2075.1985.tb04087.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ohi S., Sueoka N. Adenosine triphosphate-dependent deoxyribonuclease in Bacillus subtilis. J Biol Chem. 1973 Nov 10;248(21):7336–7344. [PubMed] [Google Scholar]
- Riva S., Polsinelli M., Falaschi A. A new phage of Bacillus subtilis with infectious DNA having separable strands. J Mol Biol. 1968 Jul 28;35(2):347–356. doi: 10.1016/s0022-2836(68)80029-4. [DOI] [PubMed] [Google Scholar]
- Rottländer E., Trautner T. A. Genetic and transfection studies with B, subtilis phage SP 50. I. Phage mutants with restricted growth on B. subtilis strain 168. Mol Gen Genet. 1970;108(1):47–60. doi: 10.1007/BF00343184. [DOI] [PubMed] [Google Scholar]
- Rutberg L. Mapping of a temperate bacteriophage active on Bacillus subtilis. J Virol. 1969 Jan;3(1):38–44. doi: 10.1128/jvi.3.1.38-44.1969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sargent M. G., Davies S., Bennett M. F. Potentiation of a nucleolytic activity in Bacillus subtilis. J Gen Microbiol. 1985 Oct;131(10):2795–2804. doi: 10.1099/00221287-131-10-2795. [DOI] [PubMed] [Google Scholar]
- Sedgwick B., Lindahl T. A common mechanism for repair of O6-methylguanine and O6-ethylguanine in DNA. J Mol Biol. 1982 Jan 5;154(1):169–175. doi: 10.1016/0022-2836(82)90424-7. [DOI] [PubMed] [Google Scholar]
- Shemyakin M. F., Grepachevsky A. A., Chestukhin A. V. Properties of Bacillus subtilis ATP-dependent deoxyribonuclease. Eur J Biochem. 1979 Aug 1;98(2):417–423. doi: 10.1111/j.1432-1033.1979.tb13201.x. [DOI] [PubMed] [Google Scholar]
- Smith K. C., Wang T. V., Sharma R. C. recA-dependent DNA repair in UV-irradiated Escherichia coli. J Photochem Photobiol B. 1987 Sep;1(1):1–11. doi: 10.1016/1011-1344(87)80002-7. [DOI] [PubMed] [Google Scholar]
- Walker G. C. Inducible DNA repair systems. Annu Rev Biochem. 1985;54:425–457. doi: 10.1146/annurev.bi.54.070185.002233. [DOI] [PubMed] [Google Scholar]
- Yamamoto K., Higashikawa T., Ohta K., Oda Y. A loss of uvrA function decreases the induction of the SOS functions recA and umuC by mitomycin C in Escherichia coli. Mutat Res. 1985 May;149(3):297–302. doi: 10.1016/0027-5107(85)90144-7. [DOI] [PubMed] [Google Scholar]
- Yasbin R. E., Fields P. I., Andersen B. J. Properties of Bacillus subtilis 168 derivatives freed of their natural prophages. Gene. 1980 Dec;12(1-2):155–159. doi: 10.1016/0378-1119(80)90026-8. [DOI] [PubMed] [Google Scholar]


