Abstract
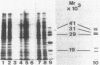
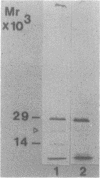
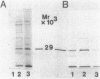
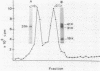
Membrane-associated gene products of shock-sensitive bacterial transport operons are often difficult to detect. A 4.5-kilobase DNA fragment, known to completely encode the Salmonella typhimurium tctI operon, was cloned in both orientations behind the T7 phage promoter phi 10 and expressed by using the T7 polymerase-promoter system of Tabor and Richardson (S. Tabor and C. C. Richardson, Proc. Natl. Acad. Sci. USA 82:1074-1078, 1985). Under these conditions, five proteins were clearly demonstrated. One DNA strand was shown to encode the periplasmic (29,000-Mr) C protein (as a 31,000-Mr precursor), a 19,000-Mr protein, and a 40,000- to 45,000-Mr protein which ran as a diffuse band on sodium dodecyl sulfate-polyacrylamide gel electrophoresis. The opposite strand carried the information for two additional proteins of 29,000 and 14,000 Mr. By Tn5 mutagenesis, subcloning of Tn5 insertions, and subcloning of various deletion mutants it was shown that the tctI system is divergently transcribed. The periplasmic binding protein (C protein) is the first product of one operon, followed by the 19,000-Mr and 45,000-Mr integral inner membrane proteins. On the opposite strand only the 29,000-Mr protein was essential for tctI function, and it was found to be weakly attached to the inner membrane. Thus tctI encodes four proteins, one periplasmic, two integral, and one peripheral to the cytoplasmic membrane, with the genes arranged as tctA tctB tctC tctD.
Full text
PDF




Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Ames G. F. Bacterial periplasmic transport systems: structure, mechanism, and evolution. Annu Rev Biochem. 1986;55:397–425. doi: 10.1146/annurev.bi.55.070186.002145. [DOI] [PubMed] [Google Scholar]
- Ames G. F. Resolution of bacterial proteins by polyacrylamide gel electrophoresis on slabs. Membrane, soluble, and periplasmic fractions. J Biol Chem. 1974 Jan 25;249(2):634–644. [PubMed] [Google Scholar]
- Bochner B. R., Savageau M. A. Generalized indicator plate for genetic, metabolic, and taxonomic studies with microorganisms. Appl Environ Microbiol. 1977 Feb;33(2):434–444. doi: 10.1128/aem.33.2.434-444.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boeke J. D., Model P. A prokaryotic membrane anchor sequence: carboxyl terminus of bacteriophage f1 gene III protein retains it in the membrane. Proc Natl Acad Sci U S A. 1982 Sep;79(17):5200–5204. doi: 10.1073/pnas.79.17.5200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dale R. M., McClure B. A., Houchins J. P. A rapid single-stranded cloning strategy for producing a sequential series of overlapping clones for use in DNA sequencing: application to sequencing the corn mitochondrial 18 S rDNA. Plasmid. 1985 Jan;13(1):31–40. doi: 10.1016/0147-619x(85)90053-8. [DOI] [PubMed] [Google Scholar]
- Horazdovsky B. F., Hogg R. W. High-affinity L-arabinose transport operon. Gene product expression and mRNAs. J Mol Biol. 1987 Sep 5;197(1):27–35. doi: 10.1016/0022-2836(87)90606-1. [DOI] [PubMed] [Google Scholar]
- McAllister W. T., Morris C., Rosenberg A. H., Studier F. W. Utilization of bacteriophage T7 late promoters in recombinant plasmids during infection. J Mol Biol. 1981 Dec 15;153(3):527–544. doi: 10.1016/0022-2836(81)90406-x. [DOI] [PubMed] [Google Scholar]
- Russel M., Model P. Filamentous phage pre-coat is an integral membrane protein: analysis by a new method of membrane preparation. Cell. 1982 Jan;28(1):177–184. doi: 10.1016/0092-8674(82)90387-7. [DOI] [PubMed] [Google Scholar]
- Somers J. M., Kay W. W. Genetic fine structure of the tricarboxylate transport (tct) locus of Salmonella typhimurium. Mol Gen Genet. 1983;190(1):20–26. doi: 10.1007/BF00330319. [DOI] [PubMed] [Google Scholar]
- Sweet G. D., Kay C. M., Kay W. W. Tricarboxylate-binding proteins of Salmonella typhimurium. Purification, crystallization, and physical properties. J Biol Chem. 1984 Feb 10;259(3):1586–1592. [PubMed] [Google Scholar]
- Tabor S., Richardson C. C. A bacteriophage T7 RNA polymerase/promoter system for controlled exclusive expression of specific genes. Proc Natl Acad Sci U S A. 1985 Feb;82(4):1074–1078. doi: 10.1073/pnas.82.4.1074. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Towbin H., Staehelin T., Gordon J. Electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose sheets: procedure and some applications. Proc Natl Acad Sci U S A. 1979 Sep;76(9):4350–4354. doi: 10.1073/pnas.76.9.4350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Widenhorn K. A., Boos W., Somers J. M., Kay W. W. Cloning and properties of the Salmonella typhimurium tricarboxylate transport operon in Escherichia coli. J Bacteriol. 1988 Feb;170(2):883–888. doi: 10.1128/jb.170.2.883-888.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- de Bruijn F. J., Lupski J. R. The use of transposon Tn5 mutagenesis in the rapid generation of correlated physical and genetic maps of DNA segments cloned into multicopy plasmids--a review. Gene. 1984 Feb;27(2):131–149. doi: 10.1016/0378-1119(84)90135-5. [DOI] [PubMed] [Google Scholar]