Abstract
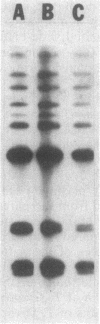
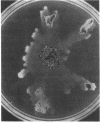
Two solid medium formulations, designated 100:10 and 10:10, were developed for the growth of Thiobacillus ferrooxidans. The new media contain a mixture of both ferrous iron and thiosulfate as available energy sources, permitting the detection of colony morphology variants that arise spontaneously in a wild-type population. Several morphological and physiological characteristics of a class of T. ferrooxidans variants, termed LSC for large spreading colony, are described. LSC variants lack the ability to oxidize iron but retain the capacity to utilize thiosulfate or tetrathionate as energy sources. An LSC colony spreads on the surface of solid 100:10 medium as a monolayer of cells in a fashion resembling that of certain swarming or gliding bacteria. The LSC variant reverts to a parental wild type at frequencies that vary in different independently arising isolates. The identity of the LSC variant as a derivative of the parental wild-type T. ferrooxidans was established by Southern blot hybridization.
Full text
PDF







Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Brock T. D., Gustafson J. Ferric iron reduction by sulfur- and iron-oxidizing bacteria. Appl Environ Microbiol. 1976 Oct;32(4):567–571. doi: 10.1128/aem.32.4.567-571.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Claassen P. A., Kortstee G. J., Oosterveld-van Vliet W. M., van Neerven A. R. Colonial heterogeneity of Thiobacillus versutus. J Bacteriol. 1986 Nov;168(2):791–794. doi: 10.1128/jb.168.2.791-794.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dworkin M., Keller K. H., Weisberg D. Experimental observations consistent with a surface tension model of gliding motility of Myxococcus xanthus. J Bacteriol. 1983 Sep;155(3):1367–1371. doi: 10.1128/jb.155.3.1367-1371.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fuscoe F. J. The role of extracellular slime secretion in the swarming of Proteus. Med Lab Technol. 1973 Oct;30(4):373–382. [PubMed] [Google Scholar]
- Gaffney T. D., Lessie T. G. Insertion-sequence-dependent rearrangements of Pseudomonas cepacia plasmid pTGL1. J Bacteriol. 1987 Jan;169(1):224–230. doi: 10.1128/jb.169.1.224-230.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hagblom P., Segal E., Billyard E., So M. Intragenic recombination leads to pilus antigenic variation in Neisseria gonorrhoeae. Nature. 1985 May 9;315(6015):156–158. doi: 10.1038/315156a0. [DOI] [PubMed] [Google Scholar]
- Henrichsen J. Bacterial surface translocation: a survey and a classification. Bacteriol Rev. 1972 Dec;36(4):478–503. doi: 10.1128/br.36.4.478-503.1972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- KELLOGG D. S., Jr, PEACOCK W. L., Jr, DEACON W. E., BROWN L., PIRKLE D. I. NEISSERIA GONORRHOEAE. I. VIRULENCE GENETICALLY LINKED TO CLONAL VARIATION. J Bacteriol. 1963 Jun;85:1274–1279. doi: 10.1128/jb.85.6.1274-1279.1963. [DOI] [PMC free article] [PubMed] [Google Scholar]
- PARKER C. D., PRISK J. The oxidation of inorganic compounds of sulphur by various sulphur bacteria. J Gen Microbiol. 1953 Jun;8(3):344–364. doi: 10.1099/00221287-8-3-344. [DOI] [PubMed] [Google Scholar]
- Paruchuri D. K., Harshey R. M. Flagellar variation in Serratia marcescens is associated with color variation. J Bacteriol. 1987 Jan;169(1):61–65. doi: 10.1128/jb.169.1.61-65.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Robinson M., Lewis E., Napier E. Occurrence of reiterated DNA sequences in strains of Streptomyces produced by an interspecific protoplast fusion. Mol Gen Genet. 1981;182(2):336–340. doi: 10.1007/BF00269680. [DOI] [PubMed] [Google Scholar]
- SILVERMAN M. P., LUNDGREN D. G. Studies on the chemoautotrophic iron bacterium Ferrobacillus ferrooxidans. I. An improved medium and a harvesting procedure for securing high cell yields. J Bacteriol. 1959 May;77(5):642–647. doi: 10.1128/jb.77.5.642-647.1959. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sapienza C., Doolittle W. F. Unusual physical organization of the Halobacterium genome. Nature. 1982 Feb 4;295(5848):384–389. doi: 10.1038/295384a0. [DOI] [PubMed] [Google Scholar]
- Schnaitman C. A., Korczynski M. S., Lundgren D. G. Kinetic studies of iron oxidation by whole cells of Ferrobacillus ferrooxidans. J Bacteriol. 1969 Aug;99(2):552–557. doi: 10.1128/jb.99.2.552-557.1969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Slutsky B., Buffo J., Soll D. R. High-frequency switching of colony morphology in Candida albicans. Science. 1985 Nov 8;230(4726):666–669. doi: 10.1126/science.3901258. [DOI] [PubMed] [Google Scholar]
- Slutsky B., Staebell M., Anderson J., Risen L., Pfaller M., Soll D. R. "White-opaque transition": a second high-frequency switching system in Candida albicans. J Bacteriol. 1987 Jan;169(1):189–197. doi: 10.1128/jb.169.1.189-197.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Soll D. R., Langtimm C. J., McDowell J., Hicks J., Galask R. High-frequency switching in Candida strains isolated from vaginitis patients. J Clin Microbiol. 1987 Sep;25(9):1611–1622. doi: 10.1128/jcm.25.9.1611-1622.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Southern E. M. Detection of specific sequences among DNA fragments separated by gel electrophoresis. J Mol Biol. 1975 Nov 5;98(3):503–517. doi: 10.1016/s0022-2836(75)80083-0. [DOI] [PubMed] [Google Scholar]
- Sugio T., Domatsu C., Munakata O., Tano T., Imai K. Role of a Ferric Ion-Reducing System in Sulfur Oxidation of Thiobacillus ferrooxidans. Appl Environ Microbiol. 1985 Jun;49(6):1401–1406. doi: 10.1128/aem.49.6.1401-1406.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sugio T., Katagiri T., Moriyama M., Zhèn Y. L., Inagaki K., Tano T. Existence of a new type of sulfite oxidase which utilizes ferric ions as an electron acceptor in Thiobacillus ferrooxidans. Appl Environ Microbiol. 1988 Jan;54(1):153–157. doi: 10.1128/aem.54.1.153-157.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Swanson J., Kraus S. J., Gotschlich E. C. Studies on gonococcus infection. I. Pili and zones of adhesion: their relation to gonococcal growth patterns. J Exp Med. 1971 Oct 1;134(4):886–906. doi: 10.1084/jem.134.4.886. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tuovinen O. H., Kelly D. P. Studies on the growth of Thiobacillus ferrooxidans. I. Use of membrane filters and ferrous iron agar to determine viable numbers, and comparison with 14 CO 2 -fixation and iron oxidation as measures of growth. Arch Mikrobiol. 1973;88(4):285–298. [PubMed] [Google Scholar]
- Tuovinen O. H., Kelly D. P. Studies on the growth of Thiobacillus ferrooxidans. V. Factors affecting growth in liquid culture and development of colonies on solid media containing inorganic sulphur compounds. Arch Microbiol. 1974 Jul 22;98(4):351–364. doi: 10.1007/BF00425295. [DOI] [PubMed] [Google Scholar]
- Tuovinen O. H., Niemelä S. I., Gyllenberg H. G. Tolerance of Thiobacillus ferrooxidans to some metals. Antonie Van Leeuwenhoek. 1971;37(4):489–496. doi: 10.1007/BF02218519. [DOI] [PubMed] [Google Scholar]
- Wood N. B., Rake A. V., Shapiro L. Structure of Caulobacter deoxyribonucleic acid. J Bacteriol. 1976 Jun;126(3):1305–1315. doi: 10.1128/jb.126.3.1305-1315.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yates J. R., Holmes D. S. Two families of repeated DNA sequences in Thiobacillus ferrooxidans. J Bacteriol. 1987 May;169(5):1861–1870. doi: 10.1128/jb.169.5.1861-1870.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]