Abstract
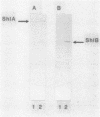
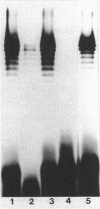
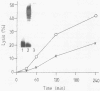
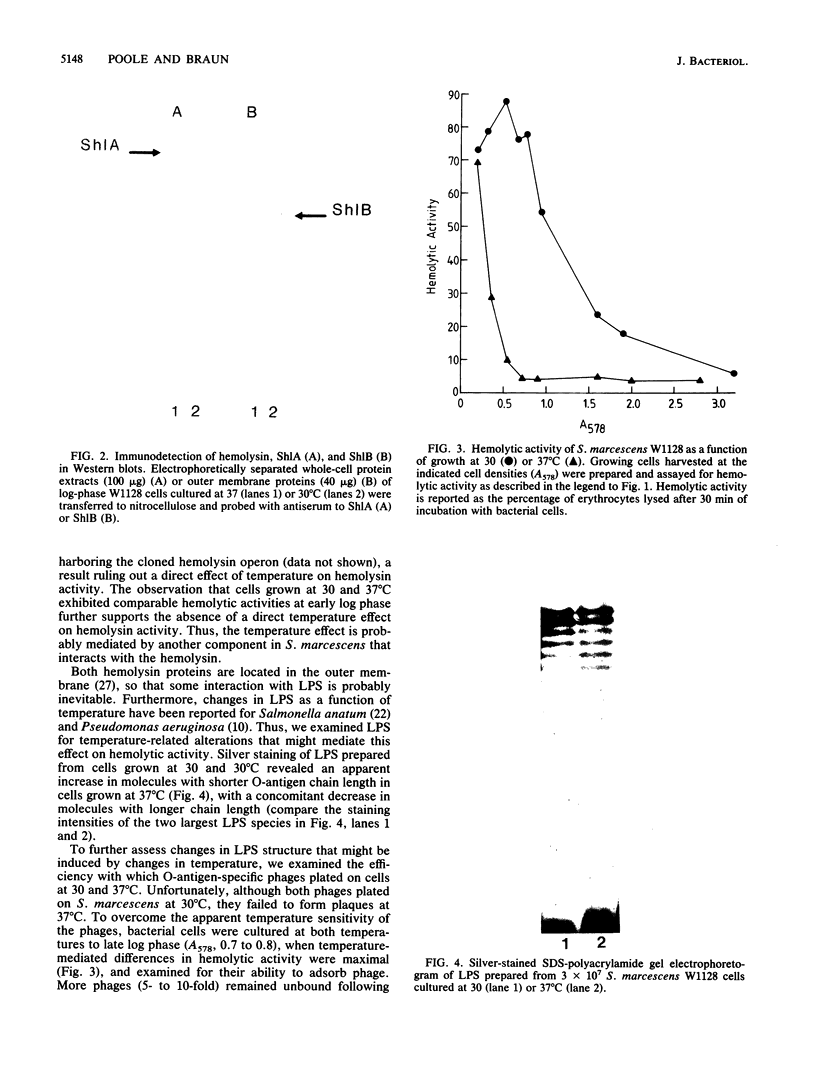
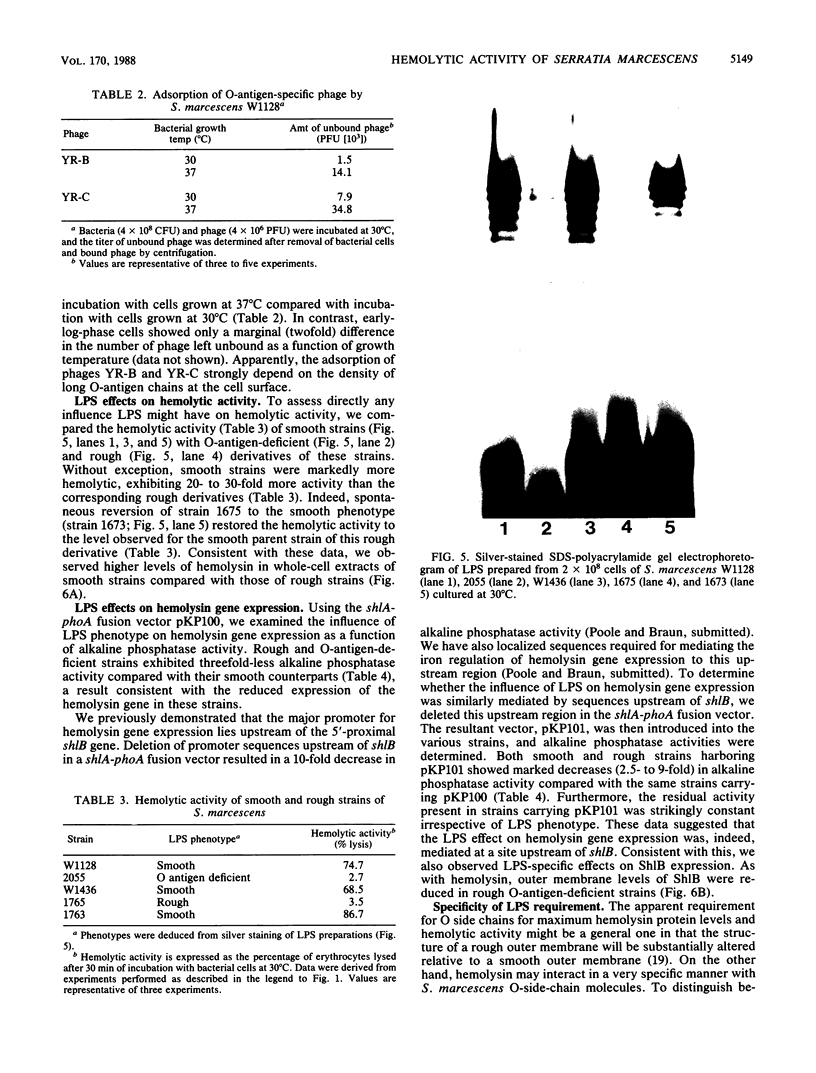
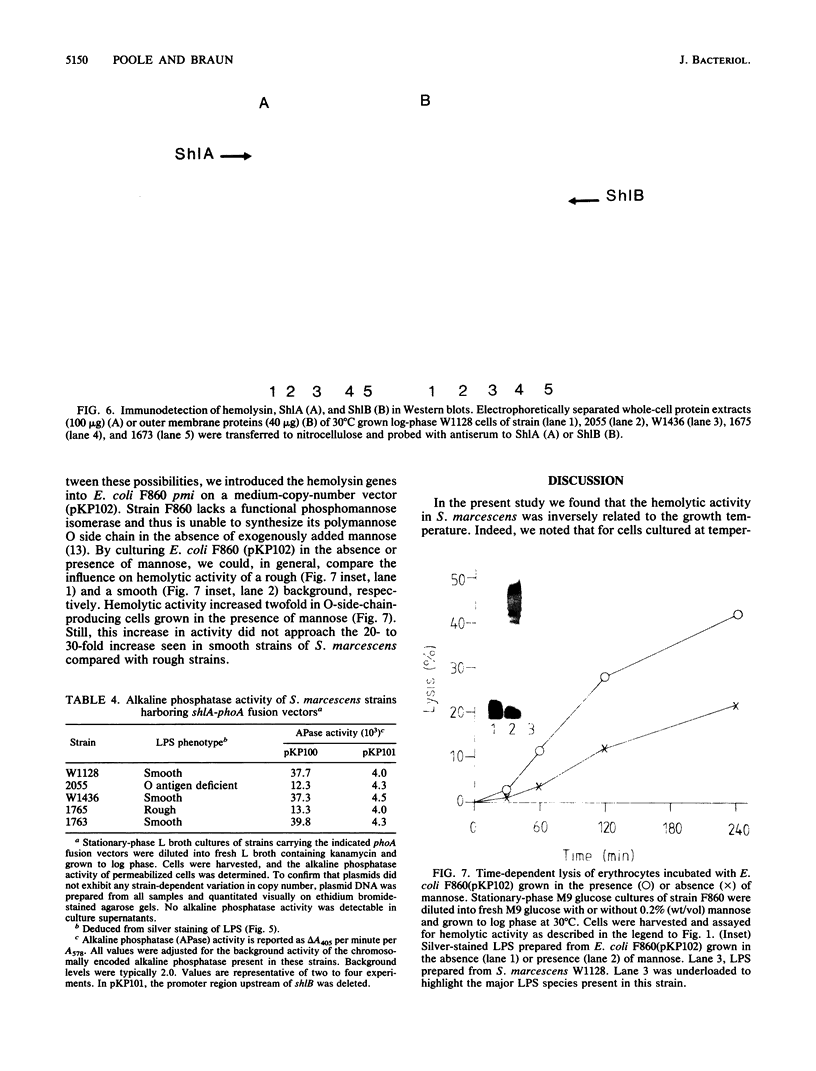
Log-phase cells of Serratia marcescens cultured at 30 degrees C were approximately 10-fold more hemolytic than those grown at 37 degrees C. By using a cloned gene fusion of the promoter-proximal part of the hemolysin gene (shlA) to the Escherichia coli alkaline phosphatase gene (phoA), hemolysin gene expression as a function of alkaline phosphatase activity was measured at 30 and 37 degrees C. No difference in alkaline phosphatase activity was observed as a function of growth temperature, although more hemolysin was detectable immunologically in whole-cell extracts of cells grown at 30 degrees C. The influence of temperature was, however, growth phase dependent, because the hemolytic activities of cells cultured to early log phase at 30 and 37 degrees C were comparable. Given the outer membrane location of the hemolysin, lipopolysaccharide (LPS) was examined as a candidate for mediating the temperature effect on hemolytic activity. Silver staining of LPS in polyacrylamide gels revealed a shift towards shorter O-antigen molecules at 37 degrees C relative to 30 degrees C. Moreover, there was less binding of O-antigen-specific bacteriophage to S. marcescens with increasing growth temperature, a finding consistent with temperature-mediated changes in LPS structure. Smooth strains of S. marcescens were 20- to 30-fold more hemolytic than rough derivatives, a result confirming that changes in LPS structure can influence hemolytic activity. The alkaline phosphatase activity of rough strains harboring the shlA-phoA fusion was threefold lower than that of smooth strains harboring the fusion plasmids, a result consistent with a decrease in hemolysin gene expression in rough strains. The absence of a similar effect of temperature on gene expression may be related to less-marked changes in LPS structure as a function of temperature compared with a smooth-to-rough mutational change.
Full text
PDF


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Allan B. J., Stevenson R. M. Extracellular virulence factors of Aeromonas hydrophila in fish infections. Can J Microbiol. 1981 Oct;27(10):1114–1122. doi: 10.1139/m81-174. [DOI] [PubMed] [Google Scholar]
- Ball T. K., Saurugger P. N., Benedik M. J. The extracellular nuclease gene of Serratia marcescens and its secretion from Escherichia coli. Gene. 1987;57(2-3):183–192. doi: 10.1016/0378-1119(87)90121-1. [DOI] [PubMed] [Google Scholar]
- Beher M., Pugsley A., Schnaitman C. Correlation between the expression of an Escherichia coli cell surface protein and the ability of the protein to bind to lipopolysaccharide. J Bacteriol. 1980 Jul;143(1):403–410. doi: 10.1128/jb.143.1.403-410.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bhakdi S., Mackman N., Nicaud J. M., Holland I. B. Escherichia coli hemolysin may damage target cell membranes by generating transmembrane pores. Infect Immun. 1986 Apr;52(1):63–69. doi: 10.1128/iai.52.1.63-69.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Birnboim H. C. A rapid alkaline extraction method for the isolation of plasmid DNA. Methods Enzymol. 1983;100:243–255. doi: 10.1016/0076-6879(83)00059-2. [DOI] [PubMed] [Google Scholar]
- Bohach G. A., Snyder I. S. Composition of affinity-purified alpha-hemolysin of Escherichia coli. Infect Immun. 1986 Aug;53(2):435–437. doi: 10.1128/iai.53.2.435-437.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Braun V., Günther H., Neuss B., Tautz C. Hemolytic activity of Serratia marcescens. Arch Microbiol. 1985 May;141(4):371–376. doi: 10.1007/BF00428852. [DOI] [PubMed] [Google Scholar]
- Braun V., Neuss B., Ruan Y., Schiebel E., Schöffler H., Jander G. Identification of the Serratia marcescens hemolysin determinant by cloning into Escherichia coli. J Bacteriol. 1987 May;169(5):2113–2120. doi: 10.1128/jb.169.5.2113-2120.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bullen J. J., Rogers H. J., Griffiths E. Role of iron in bacterial infection. Curr Top Microbiol Immunol. 1978;80:1–35. doi: 10.1007/978-3-642-66956-9_1. [DOI] [PubMed] [Google Scholar]
- Cadieux J. E., Kuzio J., Milazzo F. H., Kropinski A. M. Spontaneous release of lipopolysaccharide by Pseudomonas aeruginosa. J Bacteriol. 1983 Aug;155(2):817–825. doi: 10.1128/jb.155.2.817-825.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eaves G. N., Jeffries C. D. ISOLATION AND PROPERTIES OF AN EXOCELLULAR NUCLEASE OF SERRATIA MARCESCENS. J Bacteriol. 1963 Feb;85(2):273–278. doi: 10.1128/jb.85.2.273-278.1963. [DOI] [PMC free article] [PubMed] [Google Scholar]
- FUKUI G. M., LAWTON W. D., HAM D. A., JANSSEN W. A., SURGALLA M. J. The effect of temperature on the synthesis of virulence factors by Pasteurella pestis. Ann N Y Acad Sci. 1960 Nov 21;88:1146–1151. doi: 10.1111/j.1749-6632.1960.tb20104.x. [DOI] [PubMed] [Google Scholar]
- Heller K., Braun V. Polymannose O-antigens of Escherichia coli, the binding sites for the reversible adsorption of bacteriophage T5+ via the L-shaped tail fibers. J Virol. 1982 Jan;41(1):222–227. doi: 10.1128/jvi.41.1.222-227.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hitchcock P. J., Brown T. M. Morphological heterogeneity among Salmonella lipopolysaccharide chemotypes in silver-stained polyacrylamide gels. J Bacteriol. 1983 Apr;154(1):269–277. doi: 10.1128/jb.154.1.269-277.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Howard S. P., Buckley J. T. Membrane glycoprotein receptor and hole-forming properties of a cytolytic protein toxin. Biochemistry. 1982 Mar 30;21(7):1662–1667. doi: 10.1021/bi00536a029. [DOI] [PubMed] [Google Scholar]
- Jepsen P. K., Riise E., Biedermann K., Kristensen P. C., Emborg C. Two-level factorial screening for influence of temperature, pH, and aeration on production of Serratia marcescens nuclease. Appl Environ Microbiol. 1987 Oct;53(10):2593–2596. doi: 10.1128/aem.53.10.2593-2596.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- König W., Faltin Y., Scheffer J., Schöffler H., Braun V. Role of cell-bound hemolysin as a pathogenicity factor for Serratia infections. Infect Immun. 1987 Nov;55(11):2554–2561. doi: 10.1128/iai.55.11.2554-2561.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lugtenberg B., Meijers J., Peters R., van der Hoek P., van Alphen L. Electrophoretic resolution of the "major outer membrane protein" of Escherichia coli K12 into four bands. FEBS Lett. 1975 Oct 15;58(1):254–258. doi: 10.1016/0014-5793(75)80272-9. [DOI] [PubMed] [Google Scholar]
- Lugtenberg B., Van Alphen L. Molecular architecture and functioning of the outer membrane of Escherichia coli and other gram-negative bacteria. Biochim Biophys Acta. 1983 Mar 21;737(1):51–115. doi: 10.1016/0304-4157(83)90014-x. [DOI] [PubMed] [Google Scholar]
- Maurelli A. T., Blackmon B., Curtiss R., 3rd Temperature-dependent expression of virulence genes in Shigella species. Infect Immun. 1984 Jan;43(1):195–201. doi: 10.1128/iai.43.1.195-201.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McConnell M., Wright A. Variation in the structure and bacteriophage-inactivating capacity of Salmonella anatum lipopolysaccharide as a function of growth temperature. J Bacteriol. 1979 Feb;137(2):746–751. doi: 10.1128/jb.137.2.746-751.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Menestrina G., Mackman N., Holland I. B., Bhakdi S. Escherichia coli haemolysin forms voltage-dependent ion channels in lipid membranes. Biochim Biophys Acta. 1987 Nov 27;905(1):109–117. doi: 10.1016/0005-2736(87)90014-9. [DOI] [PubMed] [Google Scholar]
- Michaelis S., Inouye H., Oliver D., Beckwith J. Mutations that alter the signal sequence of alkaline phosphatase in Escherichia coli. J Bacteriol. 1983 Apr;154(1):366–374. doi: 10.1128/jb.154.1.366-374.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nicas T. I., Hancock R. E. Outer membrane protein H1 of Pseudomonas aeruginosa: involvement in adaptive and mutational resistance to ethylenediaminetetraacetate, polymyxin B, and gentamicin. J Bacteriol. 1980 Aug;143(2):872–878. doi: 10.1128/jb.143.2.872-878.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Parrisius J., Bhakdi S., Roth M., Tranum-Jensen J., Goebel W., Seeliger H. P. Production of listeriolysin by beta-hemolytic strains of Listeria monocytogenes. Infect Immun. 1986 Jan;51(1):314–319. doi: 10.1128/iai.51.1.314-319.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Poole K., Schiebel E., Braun V. Molecular characterization of the hemolysin determinant of Serratia marcescens. J Bacteriol. 1988 Jul;170(7):3177–3188. doi: 10.1128/jb.170.7.3177-3188.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reid J. D., Stoufer S. D., Ogrydziak D. M. Efficient transformation of Serratia marcescens with pBR322 plasmid DNA. Gene. 1982 Jan;17(1):107–112. doi: 10.1016/0378-1119(82)90106-8. [DOI] [PubMed] [Google Scholar]
- Rennie R. P., Freer J. H., Arbuthnott J. P. The kinetics of erythrocyte lysis by Escherichia coli haemolysin. J Med Microbiol. 1974 May;7(2):189–195. doi: 10.1099/00222615-7-2-189. [DOI] [PubMed] [Google Scholar]
- Springer W., Goebel W. Synthesis and secretion of hemolysin by Escherichia coli. J Bacteriol. 1980 Oct;144(1):53–59. doi: 10.1128/jb.144.1.53-59.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stinson M. W., Hayden C. Secretion of phospholipase C by Pseudomonas aeruginosa. Infect Immun. 1979 Aug;25(2):558–564. doi: 10.1128/iai.25.2.558-564.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stoker N. G., Fairweather N. F., Spratt B. G. Versatile low-copy-number plasmid vectors for cloning in Escherichia coli. Gene. 1982 Jun;18(3):335–341. doi: 10.1016/0378-1119(82)90172-x. [DOI] [PubMed] [Google Scholar]
- Timmis K., Winkler U. Isolation of covalently closed circular deoxyribonucleic acid from bacteria which produce exocellular nuclease. J Bacteriol. 1973 Jan;113(1):508–509. doi: 10.1128/jb.113.1.508-509.1973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Towbin H., Staehelin T., Gordon J. Electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose sheets: procedure and some applications. Proc Natl Acad Sci U S A. 1979 Sep;76(9):4350–4354. doi: 10.1073/pnas.76.9.4350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tsai C. M., Frasch C. E. A sensitive silver stain for detecting lipopolysaccharides in polyacrylamide gels. Anal Biochem. 1982 Jan 1;119(1):115–119. doi: 10.1016/0003-2697(82)90673-x. [DOI] [PubMed] [Google Scholar]
- Weinstein D. L., Holmes R. K., O'Brien A. D. Effects of iron and temperature on Shiga-like toxin I production by Escherichia coli. Infect Immun. 1988 Jan;56(1):106–111. doi: 10.1128/iai.56.1.106-111.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Welch R. A. Identification of two different hemolysin determinants in uropathogenic Proteus isolates. Infect Immun. 1987 Sep;55(9):2183–2190. doi: 10.1128/iai.55.9.2183-2190.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Williams R. P., Gott C. L., Qadri S. M., Scott R. H. Influence of temperature of incubation and type of growth medium on pigmentation in Serratia marcescens. J Bacteriol. 1971 May;106(2):438–443. doi: 10.1128/jb.106.2.438-443.1971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Winkler U., Heller K. B., Folle B. Pleiotropic consequences of mutations towards antibiotic-hypersensitivity in Serratia marcescens. Arch Microbiol. 1978 Mar;116(3):259–268. doi: 10.1007/BF00417849. [DOI] [PubMed] [Google Scholar]