Abstract
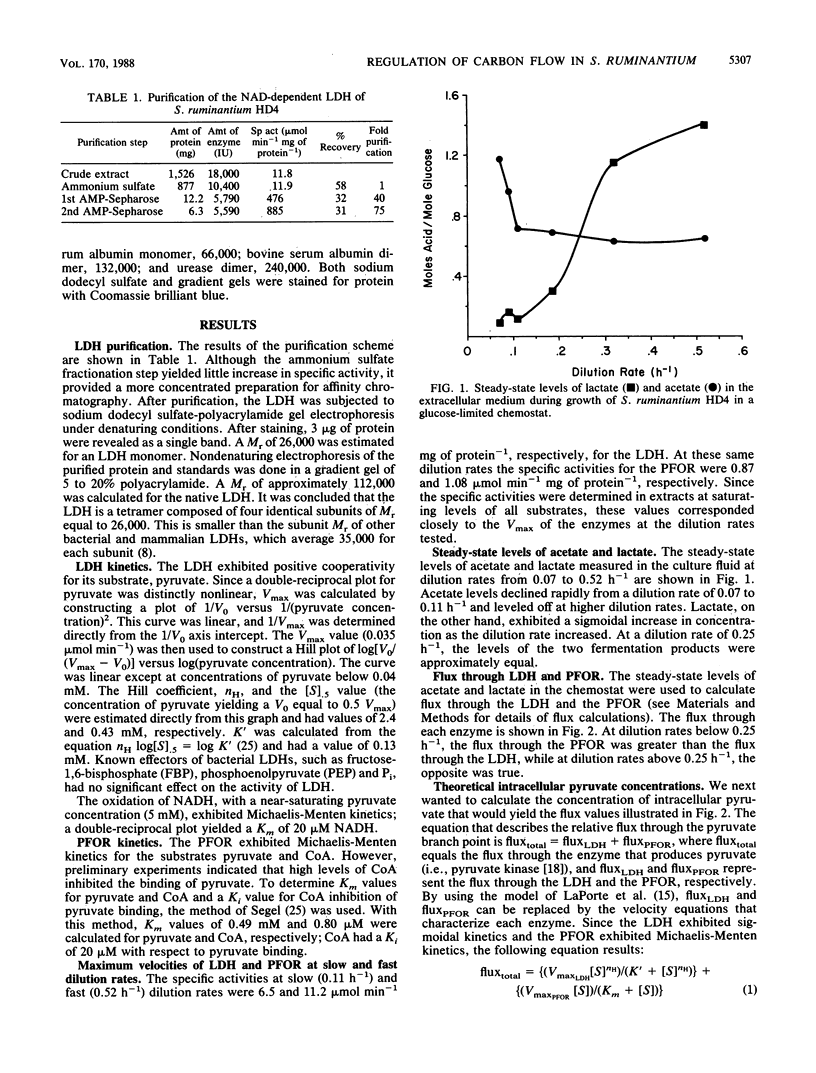
We have applied a model that permits the estimation of the sensitivity of flux through branch point enzymes (D. C. LaPorte, K. Walsh, and D. E. Koshland, J. Biol. Chem. 259:14068-14075, 1984) in order to analyze the control of flux through the lactate-acetate branch point of Selenomonas ruminantium grown in glucose-limited continuous culture. At this branch point, pyruvate is the substrate of both the NAD-dependent L-(+)-lactate dehydrogenase (LDH) and the pyruvate:ferredoxin oxidoreductase (PFOR). The LDH was purified, and it exhibited positive cooperativity for the binding of pyruvate. The LDH had an [S].5 for pyruvate of 0.43 mM, a Hill coefficient of 2.4, and a K' equal to 0.13 mM. The PFOR, assayed in cell extracts, exhibited Michaelis-Menten kinetics for pyruvate, with a Km of 0.49 mM. Carbon flux through the LDH and the PFOR increased 80-fold and 3-fold, respectively, as the dilution rate was increased from 0.07 to 0.52 h-1 in glucose-limited continuous culture. There was nearly a twofold increase, from 6.5 to 11.2 mumol min-1 mg of protein-1 in the specific activity (i.e., maximum velocity) of the LDH at dilution rates of 0.11 and 0.52 h-1, respectively. A flux equation was used to calculate the intracellular concentration of pyruvate; a fourfold increase in pyruvate, from 0.023 to 0.093 mM, was thereby predicted as the dilution rate was increased from 0.07 to 0.52 h-1. When these calculated values of intracellular pyruvate concentration were inserted into the flux equation, the predicted values of flux through the LDH and the PFOR were found to match closely the flux actually measured in the chemostat-grown cells. Thus, the 80-fold increase in flux through the LDH was due to a twofold increase in the maximum velocity of the LDH and a fourfold increase in the intracellular pyruvate concentration. In addition, the flux through the LDH exhibited ultrasensitivity to changes in both the maximum velocity of the LDH and the intracellular concentration of pyruvate. The flux through the PFOR exhibited ultrasensitivity to changes in the maximum velocity of the LDH and hyperbolic sensitivity to changes in the intracellular concentration of pyruvate.
Full text
PDF





Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- BRYANT M. P. The characteristics of strains of Selenomonas isolated from bovine rumen contents. J Bacteriol. 1956 Aug;72(2):162–167. doi: 10.1128/jb.72.2.162-167.1956. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bradford M. M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem. 1976 May 7;72:248–254. doi: 10.1016/0003-2697(76)90527-3. [DOI] [PubMed] [Google Scholar]
- Caldwell D. R., Bryant M. P. Medium without rumen fluid for nonselective enumeration and isolation of rumen bacteria. Appl Microbiol. 1966 Sep;14(5):794–801. doi: 10.1128/am.14.5.794-801.1966. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Collins L. B., Thomas T. D. Pyruvate kinase of Streptococcus lactis. J Bacteriol. 1974 Oct;120(1):52–58. doi: 10.1128/jb.120.1.52-58.1974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Garvie E. I. Bacterial lactate dehydrogenases. Microbiol Rev. 1980 Mar;44(1):106–139. doi: 10.1128/mr.44.1.106-139.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Henderson C. The influence of extracellular hydrogen on the metabolism of Bacteroides ruminicola, Anaerovibrio lipolytica and Selenomonas ruminantium. J Gen Microbiol. 1980 Aug;119(2):485–491. doi: 10.1099/00221287-119-2-485. [DOI] [PubMed] [Google Scholar]
- LaPorte D. C., Walsh K., Koshland D. E., Jr The branch point effect. Ultrasensitivity and subsensitivity to metabolic control. J Biol Chem. 1984 Nov 25;259(22):14068–14075. [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Mason P. W., Carbone D. P., Cushman R. A., Waggoner A. S. The importance of inorganic phosphate in regulation of energy metabolism of Streptococcus lactis. J Biol Chem. 1981 Feb 25;256(4):1861–1866. [PubMed] [Google Scholar]
- Melville S. B., Michel T. A., Macy J. M. Pathway and sites for energy conservation in the metabolism of glucose by Selenomonas ruminantium. J Bacteriol. 1988 Nov;170(11):5298–5304. doi: 10.1128/jb.170.11.5298-5304.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nilsson K., Mosbach K. p-Toluenesulfonyl chloride as an activating agent of agarose for the preparation of immobilized affinity ligands and proteins. Eur J Biochem. 1980 Nov;112(2):397–402. doi: 10.1111/j.1432-1033.1980.tb07218.x. [DOI] [PubMed] [Google Scholar]
- Relationship of lactate dehydrogenase specificity and growth rate to lactate metabolism by Selenomonas ruminantium. Appl Microbiol. 1975 Dec;30(6):916–921. doi: 10.1128/am.30.6.916-921.1975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roggenkamp R., Reipen G., Hollenberg C. P. Mediation, by Saccharomyces cerevisiae translocation signals, of beta-lactamase transport through the Escherichia coli inner membrane and sensitive method for detection of signal sequences. J Bacteriol. 1986 Oct;168(1):467–469. doi: 10.1128/jb.168.1.467-469.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Russell J. B., Baldwin R. L. Comparison of maintenance energy expenditures and growth yields among several rumen bacteria grown on continuous culture. Appl Environ Microbiol. 1979 Mar;37(3):537–543. doi: 10.1128/aem.37.3.537-543.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- SCHMIDT K., LIAAENJENSEN S., SCHLEGEL H. G. DIE CAROTINOIDE DER THIORHODACEAE. I. OKENON ALS HAUPTEAROTINOID VON CHROMATIUM OKENII PERTY. Arch Mikrobiol. 1963 Aug 1;46:117–126. [PubMed] [Google Scholar]
- Thauer R. K., Jungermann K., Decker K. Energy conservation in chemotrophic anaerobic bacteria. Bacteriol Rev. 1977 Mar;41(1):100–180. doi: 10.1128/br.41.1.100-180.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thomas T. D., Ellwood D. C., Longyear V. M. Change from homo- to heterolactic fermentation by Streptococcus lactis resulting from glucose limitation in anaerobic chemostat cultures. J Bacteriol. 1979 Apr;138(1):109–117. doi: 10.1128/jb.138.1.109-117.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thompson J., Torchia D. A. Use of 31P nuclear magnetic resonance spectroscopy and 14C fluorography in studies of glycolysis and regulation of pyruvate kinase in Streptococcus lactis. J Bacteriol. 1984 Jun;158(3):791–800. doi: 10.1128/jb.158.3.791-800.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wallace R. J. Control of lactate production by Selenomonas ruminantium: homotropic activation of lactate dehydrogenase by pyruvate. J Gen Microbiol. 1978 Jul;107(1):45–52. doi: 10.1099/00221287-107-1-45. [DOI] [PubMed] [Google Scholar]



