Abstract
Background
Subjective visual vertical (SVV) perception can be perturbed after stroke, but its effect on balance recovery is not yet known.
Aim
To evaluate the influence of SVV perturbations on balance recovery after stroke.
Methods
28 patients (14 with a right hemisphere lesion (RHL) and 14 with a left hemisphere lesion (LHL)) were included, 5 were lost to follow‐up. SVV perception was initially tested within 3 months after stroke, then at 6 months, using a luminous line, which the patients adjusted to the vertical position in a dark room. Mean deviation (V) and uncertainty (U), defined as the standard deviation of the SVV, were calculated for eight trials. Balance was initially assessed by the Postural Assessment Scale for Stroke (PASS), and at 6 months by the PASS (PASS6), a force platform (lateral and sagittal stability limits (LSL6 and SSL6)), the Rivermead Mobility Index (RMI6) and gait velocity (v6). Functional outcome was also assessed by the Functional Independence Measure at 6 months (FIM6).
Results
The scores for balance and for FIM6 were related to the initial V value: PASS6 (p = 0.01, τ = −0.38); RMI6 (p = 0.002, τ = −0.48), LSL6 (p = 0.06, τ = −0.29), SSL6 (p = 0.004, τ = −0.43), v6 (p = 0.01, τ = −0.36) and FIM6 (p = 0.001, τ = −0.49), as well as to the initial U value: PASS6 (p = 0.03, τ = −0.32), RMI6 (p = 0.02, τ = −0.35), SSL6 (p = 0.005, τ = −0.43) and FIM6 (p = 0.01, τ = −0.38).
Conclusions
Initial misperception of verticality was related to a poor score for balance after stroke. This relationship seems to be independent of motricity and neglect. Rehabilitation programmes should take into account verticality misperceptions, which could be an important factors influencing balance recovery after stroke.
Balance recovery is crucial for the acquisition of autonomy in patients with hemiplegia after stroke. In addition to weakness, increase in stiffness or sensory disorders leading to lower limb instability, recent articles have emphasised the role of space exploration disturbance, such as hemineglect in imbalance after stroke. Balance difficulties are more frequent and recovery takes a longer time after a right than after a left stroke, especially in patients with visuospatial neglect.1,2 An additional argument supporting the crucial role of space exploration disturbance in patients with balance disorder after stroke is that sensory stimulations such as vestibular caloric stimulation1 or neck transcutaneous electric nerve stimulation3 reduce simultaneously space disturbance and postural imbalance. Further, space exploration and balance seem to be closely linked, as the ability to perform space exploration depends on trunk orientation and vice versa in normal people as well as in patients with stroke.4,5,6,7,8,9 The assumption for this predominance of imbalance in patients with hemineglect and more generally in patients with right stroke is that spatial cognition is processed in the right hemisphere.1,10
Nethertheless, disorders of spatial cognition after stroke are not limited to space exploration. Spatial abilities such as the sense of straight ahead,11 central space representation12 or perception of verticality can be perturbed after stroke.13,14,15 Such abilities can be assumed to have a major role in maintaining balance. To our knowledge, no systematic study has been conducted to assess the role of these abilities on balance recovery. Regarding the perception of visual verticality, which is tested in a dark room asking patients to adjust a luminous line to a vertical position, two kinds of perturbations have been described after hemispheric stroke: a mean tilt, most of the time contralateral to the cerebral lesion,13,14,15 and an excessive range of uncertainty between the different adjustments.14,15 In this study, we aimed to determine whether initial SVV misperceptions (abnormal tilt or uncertainty) influence balance recovery after stroke. The results would give new insight into postural disturbance after stroke and may change clinical practice. Rehabilitation programmes of postural control for patients with hemiplegia after stroke should take into account the possible impairment of verticality perception.
Patients and methods
Patients admitted to the Physical and Medicine Rehabilitation Department, Groupe Hospitalier Lariboisiere‐F Widal APHP Paris, Paris, France, between 1 August 2004 and 30 June 2005 were included if they had experienced their first and only cerebral hemispheric stroke within 3 months, resulting in at least an initial motor and balance impairment. Patients were excluded if they had a history of neurological disturbances, vertigo or vestibular dysfunction, amblyopia or diplopia, or severe aphasia. Only patients who were able to understand the instructions for SVV adjustments and gave informed written consent to participate in the study before testing were included. The study was approved by the Clinical Investigation Unit of the Lariboisiere University Hospital. All patients underwent a traditional programme of rehabilitation including physiotherapy, occupational therapy and speech therapy if needed. This programme was not focused on verticality perception recovery.
At baseline, we recorded sex, age at stroke, type of stroke, location and side of the lesion according to computed tomography scan or magnetic resonance imaging performed at diagnosis, and time since stroke. We performed a complete neurological examination in which the following were evaluated: motricity using the Motricity Index (0–200);16 visual field, assessed at the bedside, possibly completed by a campylometry assessment when abnormal; and visuospatial neglect, using the Bell's cancellation test,17 line bisection18 and scene copy tests19: visuospatial neglect was considered to be present when the difference between the bells omitted on the left and right sides in the Bells Cancellation Test was >3, when the bias in line bisection was >0.6 cm and when at least one element was omitted in the scene copy test; the joint position sense for the big toe, ankle, knee and hip was evaluated. Patients were then classified into three groups for sensitivity: normal for position sense (2), hyposensitivity (1) and anaesthesia (0).
Procedure
Assessment of SVV perception
SVV perception was tested in a dark room to eliminate any visual reference cues, with patients sitting in a wheelchair or normal chair. They were tested using specific software for the measurement of SVV perception. A white luminous line was projected with a video projector on a dark background on the wall in front of the patient at a distance of 2 m (fig 1). The observer rotated the line around its centre until the patient indicated that he or she perceived it as exactly vertical; there was no time limit. The head was maintained in a fixed position by a supporting chinrest. Eight trials were conducted in random order: for four of them, the starting orientation of the line was tilted 40° to the left of the vertical objective, and the patient had to adjust it with a clockwise movement; for the other four, the starting orientation of the line was tilted 40° to the right of the vertical objective for a counterclockwise adjustment. The deviation was evaluated in degrees. The following parameters were analysed: mean absolute SVV deviation (V) and the uncertainty (U), defined as the standard deviation (SD).
Figure 1 Subjective visual vertical perception was tested in a dark room to eliminate any visual reference cues, with patients sitting in a wheelchair or normal chair. Using a video projector, a white luminous line was projected on to a dark background on the wall in front of the patient at a distance of 2 m. The observer rotated the line around its centre until the subject indicated that he or she perceived it as exactly vertical; there was no time limit. The head was maintained in a fixed position by a supporting chinrest.
Postural assessment
The only test for balance initially used was a specific validated ordinal scale for recent stroke, ranging from 0 to 36, called the Postural Assessment Scale for Stroke (PASS): PASS0.20 At 6 months, in addition to the PASS (PASS6), which was the primary outcome variable, balance was assessed by the Rivermead Mobility Index (RMI), a simple questionnaire with answers ranging from 0 to 10, evaluating locomotion in real life (RMI6),21 the measurement of lateral stability limits (LSL6) and sagittal stability limits (SSL6) and gait velocity (v6). Limits of LSL6 and SSL6 were measured while the patient was standing barefoot on a force platform and asked to lean as far as he or she could to the left, to the right, forward and then backward. The force platform (Satel, Blanac, France) was an upper stable support (480×480 mm2) fixed on three strain‐gauge force transducers that recorded the course of the patient's centre of pressure. This platform was connected to a personal computer with a serial connection. The course of the patient's centre of pressure was recorded for 52 s and the limits of stability were calculated as the maximum LSL6 and then SSL6 displacement of the centre of pressure. Two trials were performed and the highest LSL and SSL scores were chosen. Gait velocity (v6) was assessed while patients walked at their most comfortable pace for 10 m using their usual assistive device and orthoses. Functional independence was measured initially (FIM0) and at 6 months (FIM6) by the Functional Independence Measure, score ranging from 0 to 126,22 and used as a global indicator of stability for the activities of daily living.
Identification of the cerebral lesion
Lesion locations were accurately determined using the Talairach and Turnoud atlas23 with 0 signifying no lesion and 1 signifying a lesion of a given area. Nine sites were considered: thalamus, internal capsule, striatum, corona radiata, frontal cortex, Rolando's cortex, parietal cortex, temporal cortex and temporoparietal junction (TPJ). Lesion size was estimated from the number of areas injured.
Statistical analysis
Data were summarised by medians and interquartile distance (IQD) because the variables were not gaussian. SVV values (ie, V and U) were compared with those of normal controls with the Mann–Whitney U test. The relationship between variables describing SVV (ie, V and U) and, the characteristics of the patients (age and motricity), and, balance (PASS0, FIM0, PASS6, RMI6, LSL6, SSL6, v6 and FIM6) was tested using the Kendall's tau test. Variables describing SVV were compared, using the Mann–Whitney U test, between the visual neglect and non‐visual neglect groups, between the right (RHL) and the left hemispheric lesion (LHL) groups, and between each of these groups and values obtained from healthy controls. For the relationship between SVV and sensitivity, the Krukall–Wallis test was used. To estimate the role of motricity in the relationship between SVV variables and balance, partial coefficients of Kendall were calculated. For all tests, the significance level was fixed at 0.05, using StatView software (Cary, North Carolina, USA). The sensitivity and specificity of the initial SVV misperceptions (tilt and uncertainty) to detect severe imbalance at 6 months (PASS<31, RMI<10, LSL<110, SSL<75 and FIM<110) were tested.
Results
Patient characteristics
Table 1 gives the clinical and demographic data of the 28 patients included in the study.
Table 1 Clinical and demographic data of the 28 patients.
| Patients | Side | Age (years) | Sex | Lesion localisation | I/H | Days since lesion | Neglect | FIM | Motricity | Sensory | V0 | U0 | V6 | U6 |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| R1 | R | 76 | M | Ic/C/Fc/Rc/ P/T/TPJ | I | 16 | 1 | 76 | 28 | 0 | 8.2 | 5.5 | 4.9 | 5.6 |
| R2 | R | 30 | F | Ic/S/C/Fc/ Rc/T/TPJ | I | 31 | 0 | 90 | 67 | 2 | 0.6 | 2.4 | 0.3 | 1.7 |
| R3 | R | 46 | M | Ic/S/C/Fc/ T/TPJ | I | 18 | 0 | 54 | 0 | 1 | 4.5 | 13.7 | 8.9 | 3.5 |
| R4 | R | 39 | F | Ic/S/C/Fc/ Rc/P/T/TPJ | I | 75 | 1 | 49 | 0 | 0 | 11 | 13.6 | 3.3 | 2.9 |
| R5 | R | 71 | M | T/IC/C | H | 17 | 1 | 36 | 0 | 0 | 7.9 | 3.9 | 1.8 | 3.2 |
| R6 | R | 45 | F | Fc/Rc/P/ T/TPJ | I | 25 | 0 | 67 | 0 | 1 | 2.2 | 17.4 | 2.1 | 2.7 |
| R7 | R | 44 | F | Ic/S/CR/Fc/ Rc/P/T/TPJ | I | 51 | 1 | 48 | 0 | 0 | 3.1 | 3.2 | 1.7 | 5.4 |
| R8 | R | 60 | M | Ic/C | I | 17 | 0 | 102 | 121 | 1 | 1 | 4.9 | 2.7 | 9.8 |
| R9 | R | 82 | M | Ic/S/C/Fc/ Rc/P/T/TPJ | I | 20 | 1 | 54 | 92 | 0 | 3.1 | 9.3 | 4 | 3.9 |
| R10 | R | 49 | M | Ic/S/C/Fc/ Rc/P/T/TPJ | I | 58 | 1 | 36 | 0 | 0 | 7.8 | 7.7 | 3.1 | 2.9 |
| R11 | R | 84 | M | Fc/Rc | I | 18 | 0 | 79 | 136 | 2 | 2.2 | 1.2 | 2.6 | 2.4 |
| R12 | R | 65 | F | C/R | H | 22 | 1 | 70 | 139 | 2 | 2.7 | 2.5 | 3.2 | 1.2 |
| R13 | R | 60 | F | S/C/Rc/Fc/ P/T/TPJ | I | 66 | 1 | 73 | 86 | 1 | 2.8 | 7.5 | ||
| R14 | R | 57 | F | Ic/S/C/Rc/ Fc/P/T/TPJ | I | 55 | 0 | 77 | 122 | 1 | 4.1 | 1.4 | 1.9 | 1.4 |
| L1 | L | 72 | F | Ic/S/C/T | I | 23 | 0 | 83 | 158 | 1 | 0.7 | 1.2 | 1.6 | 1.0 |
| L2 | L | 32 | M | T/IC/S/Tc | H | 46 | 0 | 82 | 119 | 1 | 11 | 1.6 | 2.9 | 1.0 |
| L3 | L | 60 | M | T/IC | H | 14 | 0 | 111 | 158 | 1 | 0.8 | 0.8 | 2.3 | 0.8 |
| L4 | L | 56 | M | Ic/S/C/Rc/ Fc/P/Tc/TPJ | I | 50 | 0 | 84 | 75 | 1 | 4.9 | 2.3 | 0.3 | 1.2 |
| L5 | L | 45 | M | Ic/S/C/Rc/ Fc/P/Tc/TPJ | I | 40 | 0 | 63 | 62 | 1 | 6.7 | 0.8 | 0.7 | 1.5 |
| L6 | L | 46 | F | Ic/S/C/TPJ | H | 66 | 0 | 40 | 0 | 0 | 3.9 | 1.2 | 7.5 | 1.5 |
| L7 | L | 60 | F | S/C/Fc/TPJ | I | 12 | 0 | 76 | 93 | 2 | 1.6 | 3.4 | 0.9 | 1.6 |
| L8 | L | 35 | F | Ic/S/C | I | 9 | 0 | 95 | 103 | 2 | 0.7 | 1.0 | 1.0 | 2.0 |
| L9 | L | 33 | M | Ic/S/C/Fc/TPJ | I | 90 | 0 | 115 | 98 | 2 | 0.9 | 0.9 | 0.4 | 0.8 |
| L10 | L | 78 | M | Ic/C | I | 20 | 0 | 103 | 174 | 2 | 0.2 | 2.9 | ||
| L11 | L | 58 | M | C/F/Rc/TPJ | I | 69 | 0 | 103 | 158 | 2 | 0.6 | 1.6 | ||
| L12 | L | 63 | F | Ic/S/C | I | 19 | 0 | 71 | 158 | 2 | 2.9 | 2.7 | ||
| L13 | L | 69 | F | I/S/C/Fc/Rc/ P/Tc/TPJ | I | 21 | 0 | 27 | 51 | 0 | 11.8 | 3.8 | ||
| L14 | L | 49 | F | P | I | 22 | 0 | 120 | 48 | 1 | 1.5 | 1.2 | 0.5 | 1.9 |
| Median | 57.5 | 22.5 | 76 | 89 | 2.8 | 2.6 | 2.1 | 1.9 | ||||||
| IQD | 22 | 32.5 | 38.5 | 123 | 4.8 | 3.8 | 2.2 | 1.9 |
C, corona radiate; F, frontal cortex; FIM, Functional Independent Measure; H, haemorrhagic; I, ischaemic; Ic, internal capsule; IQD, interquartile distance; L, left hemispheric lesion; P, parietal cortex; R, right hemispheric lesion; Rc, Rolando's cortex; S, striatum; T, thalamus; Tc, temporal cortex, TPJ, temporoparietal junction; U0 and U6, range of uncertainty of the subjective visual vertical perception (initial and at 6 months); V0 and V6, subjective visual vertical tilt (initially and at 6 months). Values in bold are considered abnormal.
In all, 28 patients (14 with LHL and 14 with RHL) were included and gave informed written consent to participate in the study. Median (IQD) age was 57.5 (22) years, time since stroke was 22.5 (33) days. The stroke was haemorrhagic in five patients and ischaemic in 23 patients. Visual field defect was observed in three patients. Visuospatial neglect was noted in eight patients (all RHL). Nine patients had normal sensitivity, 11 hyposensitivity, and 8 had anaesthesia. Five patients (4 with LHL and 1 with RHL) were lost to follow‐up: two died, one underwent a myocardial infarct and two did not attend the 6‐month appointment.
SVV values
Table 2 gives the initial and 6‐month SVV values in patients with and without visual neglect.
Table 2 Subjective visual vertical perception values and scores for balance in controls and patients with and without visual neglect, initially and at 6 months.
| V0 (°) | U0 (°) | V6 (°) | U6 (°) | PASS6 | |
|---|---|---|---|---|---|
| Controls (n = 20) | 0.8 (1.4) | 1.2 (0.7) | |||
| All patients (n = 28) | 2.8 (4.8)* | 2.6 (3.8)* | 2.1 (2.2)* | 1.9 (1.9)* | 30 (9.7) |
| Patients with non‐visual neglect (n = 20) | 1.9 (3.5)*† | 1.6 (1.8)† | 1.7 (2.0)† | 1.5 (1.0)† | 32 (7)† |
| Patients with visual neglect (n = 8) | 5.4 (5.0)* | 6.5 (4.9)* | 3.2 (1.7)* | 3.2 (2.0)* | 22 (9) |
PASS, Postural Assessment Scale for Stroke; V and U, tilt and range of uncertainty, respectively, in the perception of verticality (initial, V0 and U0, at 6 months, V6 and U6).
Values are median (IQD). Values in parentheses are in degrees.
*Significantly different from controls (p⩽0.01).
†Significantly different between patients with and without visual neglect (p<0.05).
Normal data on controls were supplied by 20 healthy people, who were age matched with the patients (median (IQD) age 60 (19) years) and underwent the same SVV perception tests. For the controls, the median (IQD) V was 0.8° (IQD 1.4°) and the maximum individual V was 2.8°. Consequently, the results for each patient were considered different from control values when V>3°, which was clearly larger than the deviation for the controls, and was similar to the normal range previously reported.24 The median (IQD) U was 1.2° (0.7°) and the maximum individual difference was 2.5°. Therefore for U, 3° was considered markedly abnormal.
The initial SVV perception values (V0 and U0) of all patients were significantly different from those of the controls (p = 0.001 and p = 0.006). Of the 28 patients, 13 (46%) had an abnormal SVV tilt (8 with RHL and 5 with LHL) and 12 (43%) an abnormal range of uncertainty (10 with RHL and 2 with LHL). Among the five patients who were lost to follow‐up, two had SVV misperception, the first (with RHL) had an uncertainty and the second (with LHL) had both misperceptions. Six (4 with LHL and 2 with RHL) of the 12 patients with initial SVV tilt, followed up to 6 months, recovered, as did 4 (all with LHL) of the 10 patients with initial SVV uncertainty. At 6 months, of the 23 patients, 7 (30%) had an abnormal SVV tilt (6 RHL and 1 LHL) and 6 (26%) an abnormal range of uncertainty (all RHL).
Relationship between SVV perception values and initial clinical status
V0 and U0 were significantly related to initial motricity, balance (PASS0) and independence (FIM0), and were significantly different in the three groups for joint position sense. V0 and U0 were higher in patients with RHL than with LHL: V0 = 3.1 (IQD 11.2) v 1.5 (IQD 4.2) and U0 = 5.2 (IQD 6.7) v 1.5 (IQD 1.5), but the difference reached significance only for U0. SVV perception values were significantly higher in the RHL group than those from controls (for V0, p<0.001; V6, p = 0.001; U0, p<0.001; and U6, p<0.003).
V and U were significantly higher in the neglect than in non‐neglect group, initially (p = 0.02 and 0.005, respectively), and at 6 months (p = 0.04 and 0.01, respectively). The V value was initially higher in the non‐neglect group than in controls (p = 0.03), whereas V and U values were higher initially and at 6 months in the neglect group than in the controls (for V, p<0.001 initially and 0.001 at 6 months, and for U, p<0.001 and p = 0.001, respectively). Five patients without neglect had SVV misperception (L2, L4–7, L10–11, L13).
V0 but not U0 was linked to the size of the lesion. A higher V value was found in the groups with parietal and temporal lesions than in the groups without such lesions (p = 0.006 and 0.02). A higher value of uncertainty was found in the groups with temporal or TPJ lesions than in the groups without such lesions (p = 0.02 and 0.04).
Relationship between SVV perception values and balance
Table 3 and figs 2 and 3 describe the relationship between SVV perception values and balance.
Table 3 Relationship (p value) between initial subjective visual vertical perception and postural values and characteristics of the 28 patients.
| Motricity | Sensory | Neglect | Side | Size of lesion | PASS0 | FIM0 | FIM6 | |
|---|---|---|---|---|---|---|---|---|
| V0 | ⩽0.01 | ⩽0.001 | 0.02 | 0.10 | ⩽0.01 | ⩽0.001 | ⩽0.001 | ⩽0.001 |
| U0 | ⩽0.01 | 0.07 | ⩽0.01 | ⩽0.001 | 0.10 | ⩽0.01 | ⩽0.01 | ⩽0.01 |
| PASS0 | ⩽0.001 | ⩽0.01 | ⩽0.01 | 0.05 | 0.10 | |||
| FIM0 | ⩽0.001 | ⩽0.001 | ⩽0.01 | 0.03 | ⩽0.01 | |||
| PASS6 | 0.02 | ⩽0.01 | ⩽0.01 | 0.02 | 0.70 | |||
| RMI6 | ⩽0.01 | ⩽0.01 | ⩽0.01 | 0.03 | 0.60 | |||
| v6 | 0.06 | ⩽0.01 | ⩽0.01 | 0.10 | 0.20 | |||
| LSL | ⩽0.01 | ⩽0.01 | ⩽0.01 | 0.07 | 0.20 | |||
| SSL | ⩽0.001 | ⩽0.01 | ⩽0.01 | 0.03 | ⩽0.01 | |||
| FIM6 | ⩽0.001 | ⩽0.001 | ⩽0.01 | 0.10 | 0.05 |
FIM, Functional Independent Measure (FIM0, initial; and FIM6, at 6 months); LSL, lateral stability limit; PASS Postural Assessment Scale for Stroke (PASS0, initial and PASS6, at 6 months); RMI Rivermead Mobility Index; SSL, Sagittal stability limits; SVV, subjective visual vertical; V0 and U0, initial tilt and range of uncertainty, respectively, in the perception of verticality.
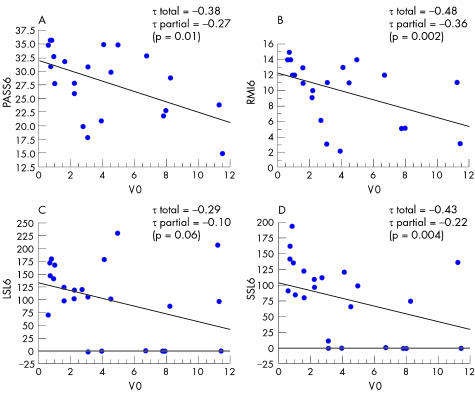
Figure 2 Relationship between balance at 6 months and the initial subjective visual vertical (SVV) perception values (V0). τ Values are the Kendall coefficient correlations and τ partial is the partial Kendall correlations coefficient excluding the role of motricity in the relationship between initial SVV perception values and balance.
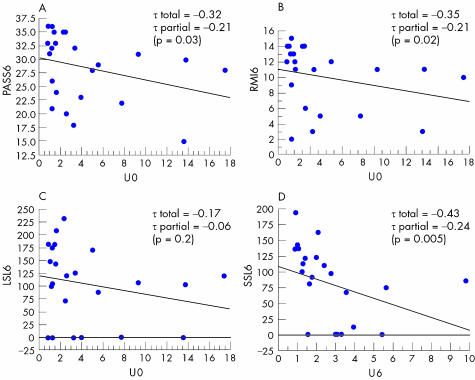
Figure 3 Relationship between balance at 6 months and the initial subjective visual vertical (SVV) perception values (U0). τ Values are the Kendall coefficient correlations and τ partial is the partial Kendall correlations coefficient excluding the role of motricity in the relationship between initial SVV perception values and balance.
Initial SVV perception values (V0 and U0) were significantly related to initial scores for balance and to scores for balance and independence at 6 months (PASS6: p = 0.01, τ = −0.38; RMI6: p = 0.002, τ = −0.48; LSL6: p = 0.06, τ = −0.29; SSL6: p = 0.004, τ = −0.43; v6: p = 0.01, τ = −0.36; FIM6: p0.001, τ = −0.49), as well as the initial U value (PASS6: p = 0.03, τ = −0.32; RMI6: p = 0.02, τ = −0.35; SSL6: p = 0.005, τ = −0.43; and FIM6: p = 0.01, τ = −0.38).
The value of the partial Kendall coefficients decreased only from about one third compared with the total coefficient values. The sensitivity values of tilt perturbation to detect imbalance was good: 0.72 for PASS, 0.85 for RMI, 0.75 for LSL, 1 for SSL and 0.9 for FIM. The specificity values were relatively modest: 0.58, 0.56, 0.63, 0.71 and 0.75, respectively. The sensitivity of uncertainty perturbation to detect imbalance was 0.72 for PASS, 0.71 for RMI, 0.58 for LSL, 0.77 for SSL and 0.72 for FIM. The specificity values were 0.75, 0.50, 0.63, 0.71 and 0.75, respectively.
In patients with non‐visual neglect, V0 was significantly related to initial scores for balance (p = 0.004 for PASS0, 0.002 for functional independence measure (FIM)0) and to scores for balance and independence at 6 months (p = 0.003 for RMI6, 0.03 for V6, 0.07 for SSL6 and 0.006 for FIM6); U0 was related to SSL (p = 0.05) and FIM6 (p = 0.05).
Relationship between balance and initial clinical status
Initial and 6‐month scores for balance were all significantly related to motricity, higher in the neglect group than in the non‐visual neglect group and in the RHL than in the LHL group, different in the three groups for joint position sense but not related to the size of lesion (table 3).
Discussion
We aimed to determine the influence of initial misperception of visual verticality on balance recovery after stroke. We focused on a population of 28 patients with recent stroke and followed them up for 6 months. Five were lost to follow‐up.
The relevance of SVV perception tasks in patients from the neglect group could be questionable because of their left visual exploratory deficit. Our opinion is, as emphasised by Kerkhoff, that the results are not affected by visuospatial neglect as firstly, the time is unlimited, secondly, the visual angle used in the task is only 15° maximum to the left, usually easily explored in cases of visuospatial neglect, and third, no major starting effect of the rod was found.14
SVV tilt was abnormal in about half the patients (46%) and was almost equally present in patients with acute RHL and LHL. The number of patients with initial tilted SVV perception was similar to that reported in our previous study of 40 similar patients after stroke (46% v 40%).15 Recovery of tilt was complete during the first 6 months for 50% of the patients with an initially tilted SVV perception, with a better recovery in those with LHL than in those with RHL. A tendency towards recovery of normal SVV perception has been suggested by Brandt et al,13 who followed the recovery of a few of their series of 71 patients with stroke. Such recovery of SVV perception, rapid and predominant in patients with LHL, explains why the prevalence of abnormal SVV perception varies in different studies, depending on the time elapsing since stroke. Tilt after hemispheric stroke was shown to be a consequence of lesions involving the central vestibular pathways (from the thalamus to the vestibular cortex, which is a polysensory area) on either side.13,24 We found here that tilt was higher in patients with temporal as well as parietal lesions. Such a relationship between tilt and lesions in regions concerned with visuospatial analysis has been suggested by Kerkhoff, as in his series, patients with parietal hemineglect had a tilt whereas those without neglect were not different from normal people.25
The uncertainty in the perception of verticality is sometimes spectacular, as an adjustment made to the left can be immediately followed by an adjustment to the right. The uncertainty, defined as an abnormal variability in SVV adjustments, could be a reflection of the difficulty some patients experience in assessing the visual vertical, as if they have lost the sense of verticality. This has long been noted in visual14,15,26 and in haptic modality27 and related to visuospatial neglect. Uncertainty was found in 43% of our patients. Contrarily to tilt, uncertainty was predominant, even recently after stroke, in patients with RHL (71% in RHL v 14% in LHL). The range of uncertainty was higher in patients with lesions in the TPJ and temporal cortex. The TPJ, which resembles that of the parietoinsular vestibular cortex in the monkey, is of great interest, firstly because it is of crucial importance to balance after stroke,10 and secondly, because it is a polymodal sensory area, as it responds to visual, somatosensory, tactile or vestibular input.1,3 The close relationship between uncertainty and such a polymodal sensory area, with the predominance of uncertainty in patients with right cerebral lesions and visual neglect which is a multisensory disorder, suggests that uncertainty stems from difficulty in multisensory integration because of a lesion located in multisensory areas of the right hemisphere.
Therefore, two components of SVV misperceptions exist after stroke. These are tilt, which is initially equally present in those with RHL or LHL, and uncertainty, which obviously predominates during the whole period in patients with RHL. Uncertainty and tilt were often dissociated, some patients percieved a vertical tilt without any uncertainty, whereas others assessed it with a wide range of uncertainty without mean tilt. The dissociation between tilt and uncertainty in some patients, together with the predominance of uncertainty in patients with RHL and patients in the neglect group argue for different underlying mechanisms for tilt and uncertainty. Visual vertical perception, tested in a dark room without visual afference, needs vestibular inputs13,28 and somatosensory information.29,30 Each element of information takes its sensory pathway and all are finally processed together in polymodal sensory areas.13,31 In patients after stroke, correct vestibular and somatosensory information is produced by peripheral receptors; the possible erroneous perception of gravity might then be due to a dysfunction situated on the vestibular or somatosensory pathways, between the receptors and the polysensory cortex. Tilt, contrary to uncertainty, is seen in patients with otolithic or vestibular pathway impairment. Tilt after hemispheric stroke might be related to a lesion on the vestibular or somatosensory pathways, providing right or left asymmetrical information to the sensory integration centre. As described earlier, uncertainty could be related to a central inability to analyse spatial information in the right hemisphere.
For a long time, visual verticality misperception has been intuitively assumed to be responsible for postural disturbance after stroke.26,32 However, SVV perception was tested, as usual, in a dark room to eliminate any visual reference, exploring the perception of the verticality elaborated in the internal graviceptors of the patient (otolith and somatosensory receptors). Contrary to what happens in the experimental task where any visual reference is absent, in real life, external visual cues such as trees, buildings and people are available and give pertinent information of verticality. Such visual cues of verticality are used in normal and vestibular patients to resolve conflict between sensory information.33 However, in patients after stroke, the use of such external visual cues in the perception of visual verticality is not known. Thus, the effect of SVV misperception on balance is not evident and has not yet been systematically investigated. In patients with a specific and unusual disturbance of balance after stroke, called Pusher syndrome, Karnath et al34 found a misperception in the verticality of one's own body, known as the subjective postural vertical, but recorded no bias in SVV perception. Such result contrasted with those of Saj et al,35 who found an ipsilateral SVV tilt in the patients with Pusher syndrome. In another study, poor ambulation of patients after stroke was related to SVV misperception, but ambulation was scored on a rough three‐step scale (patients were classified as fully mobile, walking up to 50 m or requiring a wheelchair).25
In our study, SVV perturbations were found to be related to initial balance disturbances and to poor balance recovery 6 months after stroke. The relationship between SVV perturbations and balance was highly significant although correlation coefficients were relatively not impressive because of the particular distribution of the tau statistics used here. The sensitivity of SVV perturbations to detect imbalance at 6 months was found to be good. So, the probability for patients with SVV perturbations to have a poor balance recovery is high. However, the specificity of such perturbations to detect imbalance was relatively modest, which is not surprising because of the other components influencing balance. SVV misperceptions are also related to motor and sensory impairments and visual neglect. The question arises whether the initial SVV perception values are related to balance because they are strongly related to other factors of imbalance, such as motor impairment, sensory impairment or visuospatial neglect, or whether they are related because the misperception of verticality itself underlies imbalance. Unfortunately, in our study, the number of patients was too small to allow separate analyses for patients with or without sensory defect, or with LHL compared with RHL. Regarding the role of motricity, the results of partial Kendall correlation coefficients suggest that a relevant part of the relationship between balance and SVV perception variables was independent from the score for motricity, as partial Kendall correlation coefficients are diminished from only about one third of the value of the total Kendall coefficients. Regarding the role of visuospatial neglect, the relationships between SVV tilt and scores for balance are appreciable even in the non–visual neglect group. Moreover, here as in other studies, neglect and SVV misperception were often dissociated.15,25 Thus, some patients with severe neglect had no tilt in SVV perception and other patients with non‐visual neglect expressed an SVV tilt or an abnormal range of uncertainty. Kerkhoff and Zoelch14 suggest that this close but not systematic relation between neglect and SVV misperception might be because neglect and verticality perception result from lesions in anatomically very close or overlapping regions or to the fact that a related or partially contingent network of corticovestibular areas is involved in the perception of verticality and spatial exploration.
SVV misperceptions are related to poor balance recovery after stroke. The results of this pilot study suggest that SVV misperceptions influence balance recovery independently from other factors of imbalance, and could be considered as an inner factor of imbalance after stroke. Therefore, verticality misperception seems to be one of the main factors influencing balance recovery after stroke. However, a multivariate analysis, with a larger sample of patients has to be performed to confirm these results and to evaluate the predictive effect of SVV perturbations on balance recovery. Misperception of verticality could be the reflection of central impairment of space construction. It would be interesting systematically to measure verticality perception after stroke and to take into account its possible misperception for establishing rehabilitation programmes and include new exercises designed to improve the sense of verticality.
Abbreviations
FIM - Functional Independent Measure
IQD - interquartile distance
LHL - left hemispheric lesion
LSL - lateral stability limit
PASS - Postural Assessment Scale for Stroke
RHL - right hemispheric lesion
RMI - Rivermead Mobility Index
SSL - sagittal stability limit
SVV - subjective visual vertical
TPJ - temporoparietal junction
Footnotes
Competing interests: None.
Ethical approval: This study was approved by the local ethics committee of the Clinical Investigation Unit, Groupe Hospitalier Lariboisiere‐F.Widal APHP Paris, University Paris, Paris, France.
References
- 1.Rode G, Tiliket C, Charlopain P.et al Postural asymmetry reduction by vestibular caloric stimulation in left hemiparetic patients. Scand J Rehabil Med 1998309–14. [DOI] [PubMed] [Google Scholar]
- 2.Perennou D, Benaim C, Rouget E.et al Postural balance following stroke: towards a disadvantage of right brain‐damaged hemisphere. Rev Neuro 1999155281–290. [PubMed] [Google Scholar]
- 3.Perennou D A, Leblond C, Amblard B.et al Transcutaneous electric nerve stimulation reduces neglect‐related postural instability after stroke. Arch Phys Med Rehabil 200182440–448. [DOI] [PubMed] [Google Scholar]
- 4.Hasselbach‐Heitzeg M M, Reuter‐Lorenz P A. Egocentric body‐centered coordinates modulate visuomotor performance. Neurospychologia 2002401822–1833. [DOI] [PubMed] [Google Scholar]
- 5.Chokron S, Imbert M. Variations of the egocentric reference among normal subjects and patient with unilateral neglect. Neuropsychologia 199533703–711. [DOI] [PubMed] [Google Scholar]
- 6.Mennemeier M, Chatterjee A, Heilman K M. A comparison of the influences of body and environment centred reference frames on neglect. Brain 19941171013–1021. [DOI] [PubMed] [Google Scholar]
- 7.Karnath H O, Schenkel P, Fischer B. Trunk orientation as the determining factor of the contralateral deficit in the neglect syndrome and as the physical anchor of the internal representation of body orientation in space. Brain 19911141997–2014. [DOI] [PubMed] [Google Scholar]
- 8.Pizzamiglio L, Valar G, Dorrichi F. Gravity and hemineglect. Neuroreport 19957370–371. [PubMed] [Google Scholar]
- 9.Schindler I, Kerkhoff G. Head and trunk orientation modulate visual neglect. Neuroreport 199782681–2685. [DOI] [PubMed] [Google Scholar]
- 10.Perennou D A, Leblond C, Amblard B.et al The polymodal sensory cortex is crucial for controlling lateral postural stability: Evidence from stroke patients. Brain Res 200053359–365. [DOI] [PubMed] [Google Scholar]
- 11.Vallar G, Lobel E, Galati G.et al A fronto‐parietal system for computing the egocentric spatial frame of reference in humans. Exp Brain Res 1999124281–286. [DOI] [PubMed] [Google Scholar]
- 12.Girardi M, McIntosh R D, Michel C.et al Sensorimotor effects on central space representation: prism adaptation influences haptic and visual representations in normal subjects. Neuropsychologia 2004421477–1487. [DOI] [PubMed] [Google Scholar]
- 13.Brandt Th, Dieterich M, Danek A. Vestibular cortex lesions affect the perception of verticality. Ann Neurol 199435403–412. [DOI] [PubMed] [Google Scholar]
- 14.Kerkhoff G, Zoelch Ch. Disorders of visuospatial orientation in the frontal plane in patients with visual neglect following right or parietal lesions. Exp Brain Res 1998122108–120. [DOI] [PubMed] [Google Scholar]
- 15.Yelnik A P, Lebreton F O, Bonan I V.et al Perception of verticality after recent cerebral hemispheric stroke. Stroke 2002332247–2253. [DOI] [PubMed] [Google Scholar]
- 16.Collin C, Wade D T. Assessing motor impairment after stroke: a pilot reliability study. J Neurol Neurosurg Psychiatry 199053576–579. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Gauthier L, Dehaut F, Joanette Y. The Bells Test for visual neglect: a quantitative and qualitative test for visual neglect. Int J Neuropsychol 19891149–54. [Google Scholar]
- 18.Shenkenberg T, Bradford D C, Ajax E T. Line bisection and unilateral neglect in patients with neurologic impairment. Neurol 198025695–699. [DOI] [PubMed] [Google Scholar]
- 19.Ogden Ja. Anterio‐posterior interhemispheric differences in the loci of lesions producing visual hemineglect. Brain Cogn 1985459–75. [DOI] [PubMed] [Google Scholar]
- 20.Benaim Ch, Perennou D A, Villy J.et al Validation of a standardized assessment of postural control in stroke patients. Stroke 1999301862–1868. [DOI] [PubMed] [Google Scholar]
- 21.Hsieh C L, Hsueh I P, Mao H F. Validity and responsiveness of the Rivermead Mobility Index in stroke patients. Scand J Rehab Med 200032140–142. [DOI] [PubMed] [Google Scholar]
- 22.Granger C V, Hamilton B B. The uniform data system for medical rehabilitation report of first admissions for 1990. Am J Phys Med Rehabil 19927133–38. [DOI] [PubMed] [Google Scholar]
- 23.Talairach J, Tournoux P.Co‐planar stereotaxic atlas of the human brain: 3‐dimensional proportional system: an approach to cerebral imaging. Stuttgart: Thieme, 1988
- 24.Dieterich M, Brandt T. Ocular torsion and tilt of subjective visual vertical are sensitive brainstem signs. Ann Neurol 199333292–299. [DOI] [PubMed] [Google Scholar]
- 25.Kerkhoff G. Multimodal spatial orientation deficits in left‐sided visual neglect. Neuropsychologia 1999371387–1405. [DOI] [PubMed] [Google Scholar]
- 26.Bruell J H, Peszczy M, Albee G W. Disturbance of perception of verticality in patients with hemiplegia: a preliminary report. Arch Phys Med 1956677–681. [PubMed]
- 27.Gentaz E, Badan M, Luyat M.et al The manual haptic perception of orientations and the oblique effect in patients with left visuo‐spatial neglect. Neuroreport 200213327–331. [DOI] [PubMed] [Google Scholar]
- 28.Dieterich M, Brandt T. Thalamic infarctions: differential effects on vestibular function in the roll plane. Neurology 1993431732–1740. [DOI] [PubMed] [Google Scholar]
- 29.Anastasopoulos D, Bronstein A, Haslwanter T.et al The role of somatosensory input for the perception of verticality. Ann N Y Acad Sci 1999871379–383. [DOI] [PubMed] [Google Scholar]
- 30.Yardley L. Contribution of somatosensory information to perception of the visual vertical with body tilt and rotating visual field. Percept Psychophys 199048131–134. [DOI] [PubMed] [Google Scholar]
- 31.Lopez C, Lacour M, Ballester M.et al Brain activation during subjective visual vertical judgement: a functional magnetic resonance imaging study. Gait Posture 200521S49–S50. [Google Scholar]
- 32.Levensen C. Rehabilitation of the stroke hemiplegia patient. In: Krusen FH, Kottke FJ, Elwood PM, eds. Handbook of physical medicine and rehabilitation. Kidlington: WB Saunders, 1965
- 33.Howard I P, Austis T, Lucia H C. The relative lability of mobile and stationary components in a visual‐motor adaptation task. Q J Exp Psychol 197426293–300. [DOI] [PubMed] [Google Scholar]
- 34.Karnath H O, Ferber S, Dichgans J. The origin of contraversive pushing. Evidence for a second graviceptive system in humans. Neurology 2000551298–1304. [DOI] [PubMed] [Google Scholar]
- 35.Saj A, Honoré J, Coello Y.et al The visual vertical in the Pusher syndrome. Influence of hemispace and body position. J Neurol 2005252885–891. [DOI] [PubMed] [Google Scholar]