Abstract
Anaplasma phagocytophilum is the agent of human anaplasmosis, the second most common tick-borne illness in the United States. This pathogen, which is closely related to obligate intracellular organisms in the genera Rickettsia, Ehrlichia, and Anaplasma, persists in ticks and mammalian hosts; however, the mechanisms for survival in the arthropod are not known. We now show that A. phagocytophilum induces expression of the Ixodes scapularis salp16 gene in the arthropod salivary glands during vector engorgement. RNA interference–mediated silencing of salp16 gene expression interfered with the survival of A. phagocytophilum that entered ticks fed on A. phagocytophilum–infected mice. A. phagocytophilum migrated normally from A. phagocytophilum–infected mice to the gut of engorging salp16-deficient ticks, but up to 90% of the bacteria that entered the ticks were not able to successfully infect I. scapularis salivary glands. These data demonstrate the specific requirement of a pathogen for a tick salivary protein to persist within the arthropod and provide a paradigm for understanding how Rickettsia-like pathogens are maintained within vectors.
Arthropod-borne infectious diseases are a major threat to human life. Human anaplasmosis, caused by Anaplasma phagocytophilum, is an emerging tick-borne illness that has been reported in the United States, Europe, and Asia (1–4). Ixodes scapularis is the most common vector in North America. A. phagocytophilum is an obligate intracellular pathogen closely related to organisms in the genera Rickettsia and Ehrlichia that primarily resides within the neutrophils of its mammalian hosts (5). Infection may be self-limiting, or it may result in severe illness or death, particularly in older or immunocompromised individuals.
A. phagocytophilum persists within the secretory salivary acini of tick salivary glands (6). Tick feeding stimulates the replication and migration of the bacteria from the salivary glands to the mammalian host (7). Studies suggest that transmission of A. phagocytophilum occurs between 24 (8) and 48 h (9, 10) after tick attachment. Once in the host, A. phagocytophilum invades granulocytes and disseminates (7). It has been shown that the acquisition of A. phagocytophilum by uninfected I. scapularis larva begins within 2 d of tick attachment on A. phagocytophilum–infected mice (9). Once in the tick, A. phagocytophilum moves through the gut and infect the salivary glands. Our studies have detected A. phagocytophilum within tick salivary glands as early as 24 h after engorgement (unpublished data).
As vector-borne microbes have evolved with arthropods, they have developed an intimate relationship to facilitate survival and transmission. In one strategy, the pathogen attempts to use arthropod or mammalian factors to help complete the microbial life cycle within the vector or reservoir host. As examples, Borrelia burgdorferi, the Lyme disease agent, uses a tick salivary protein to help infect the mammalian host (11), and Leishmania species use galectins within the sandfly to adhere to the midgut and possibly bind to galectins within the mammalian host (12). Plasmodium species may also require carbohydrate-modified ligands to persist in the mosquito gut and salivary glands (13, 14). The obligate intracellular nature of Anaplasma in both the arthropod and mammalian host provides an excellent opportunity to study the factors that are essential for the survival of A. phagocytophilum and possibly other Rickettsia-like organisms within vectors. In this study, we show that an I. scapularis salivary protein, Salp16, is required for the survival of A. phagocytophilum within the arthropod salivary glands.
RESULTS
A. phagocytophilum selectively alters the expression of tick salivary gland genes
To study whether A. phagocytophilum selectively modulates tick salivary gland gene transcription, the expression of 14 salp (salivary protein) genes that encode antigens recognized by the tick-exposed host sera was analyzed in the salivary glands of A. phagocytophilum–infected I. scapularis nymphs. Salivary glands were dissected at 24 and 72 h of engorgement. Because A. phagocytophilum is transstadially transmitted, A. phagocytophilum–infected nymphs were produced by feeding larva on A. phagocytophilum–infected C3H mice and subsequently allowing them to molt into nymphs. The nymphal infection rate was 82 ± 13%. To generate uninfected nymphs to be used as controls, larvae were fed to repletion on uninfected mice and were permitted to molt.
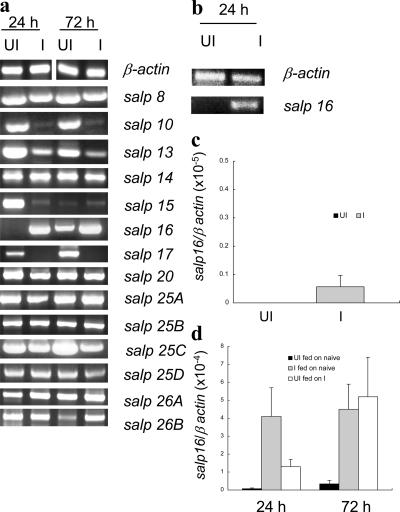
For the experiments, 25 infected or 25 uninfected nymphs were fed upon three naive C3H mice. Groups of 10 ticks were collected from each mouse at 24 and 72 h after attachment. Tick salivary glands were isolated, RNA was extracted, and PCR was performed to specifically amplify the 14 salp genes. Differential expression of several salp genes in A. phagocytophilum–infected tick salivary glands compared with uninfected glands was noted at both 24 and 72 h after tick attachment (Fig. 1 a). Notably, the expression of salp16 was most strongly up-regulated in the salivary glands of A. phagocytophilum–infected ticks. In contrast, the expression of salp10, 13, and 17 were down-regulated. Expression of the other salp genes, including salp 8, 14, 15, 20, 25A, 25B, 25C, 25D, 26A, and 26B, was not markedly influenced by A. phagocytophilum. The relative change in selected salp gene expression upon A. phagocytophilum infection was confirmed by quantitative RT-PCR (Table I). The up-regulation of salp16 was further verified by examining individual salivary glands from a batch of A. phagocytophilum–infected ticks rather than pooled samples (Fig. 1 b), and one of many representative examples is presented.
Figure 1.
Anaplasma phagocytophilum selectively alters salp gene expression. (a) The expression levels of 14 salp genes in the salivary glands of A. phagocytophilum–infected (I) Ixodes scapularis nymphs were compared with uninfected (UI) nymphs at 24 and 72 h after feeding using RT-PCR. (b) Verification of the up-regulation of salp16 expression in A. phagocytophilum–infected single tick salivary glands at 24 h after infection by RT-PCR. (c) Expression of salp16 in the salivary glands of uninfected and infected unfed ticks by quantitative RT-PCR. (d) Expression of salp16 at 24 and 72 h in the salivary glands of uninfected and infected ticks fed on naive mice or uninfected ticks fed on infected mice by quantitative RT-PCR. Results are expressed as the means ± SEM (error bars) from three independent experiments. P < 0.05 was considered statistically significant (Student's t test).
Table I.
A. phagocytophilum selectively alters salp gene expression
| Gene
|
Fold change
|
|
|---|---|---|
| 24 h | 72 h | |
| salp8 | 0.9 ± 0.1 | 0.98 ± 0.03 |
| salp10 | 0.04 ± 0.01 | 0.1 ± 0.1 |
| salp13 | 0.6 ± 0.14 | 0.3 ± 0.1 |
| salp14 | 0.98 ± 0.1 | 1.02 ± 0.01 |
| salp15 | 0.04 ± 0.001 | 1.2 ± 0.2 |
| salp16 | 54 ± 4.5 | 11.8 ± 1.2 |
| salp17 | 0.06 ± 0.01 | 0.03 ± 0.01 |
| salp20 | 0.9 ± 0.01 | 0.97 ± 0.1 |
| salp 25A | 0.99 ± 0.23 | 1.1 ± 0.3 |
| salp 25B | 1.04 ± 0.05 | 1.01 ± 0.1 |
| salp 25C | 0.9 ± 0.002 | 0.3 ± 0.04 |
| salp 25D | 1.1 ± 0.01 | 1.0 ± 0.02 |
| salp 26A | 1.2 ± 0.01 | 1.2 ± 0.1 |
| salp 26B | 1.01 ± 0.8 | 3.2 ± 1.2 |
Fold change of the expression levels of 14 salp genes in the salivary glands of A. phagocytophilum–infected I. scapularis nymphs compared with uninfected nymphs at 24 and 72 h after feeding. Tick β-actin gene expression was used to normalize the amount of cDNA. Results shown are expressed as the ratio of expression levels of each gene (nanograms of cDNA) in infected versus uninfected ticks, which was determined by quantitative RT-PCR analysis.
Because the expression of salp16 was highly up-regulated in A. phagocytophilum–infected tick salivary glands, we characterized the temporal relationship of salp16 induction with tick engorgement and A. phagocytophilum infection. For the experiments, five sample groups, each having three pooled salivary glands, were examined at each time point. The samples were taken at precisely the same time point during the feeding cycle. Tick salivary glands were isolated, RNA was extracted, and PCR was performed. First, in the salivary glands of unfed and uninfected ticks, salp16 was not expressed (Fig. 1 c). When uninfected ticks were fed on naive mice, salp16 expression was barely detectable at 24 h and showed a modest up-regulation at 72 h (fivefold increase from 24 to 72 h; P < 0.05), indicating that salp16 expression is partially induced by feeding, (Fig. 1 d). In the infected unfed ticks, there was a very low level of expression of salp16 (Fig. 1 c). salp16 was strongly up-regulated in infected fed ticks (55-fold increase in comparison with uninfected fed ticks; P < 0.01) at an early time point (24 h) during engorgement (Fig. 1 d). Moreover, when uninfected ticks were fed on A. phagocytophilum–infected mice, salp16 was up-regulated 18-fold (P < 0.05) at 24 h and 15-fold (P < 0.05) at 72 h in comparison with that of uninfected ticks that fed on naive mice for identical intervals (Fig. 1 d). It is interesting to note that the salp16 up-regulation at 72 h, when uninfected ticks engorged on infected mice, was comparable with that observed when infected ticks were fed on naive mice (Fig. 1 d). These data suggest that the presence of A. phagocytophilum in the salivary gland during feeding up-regulates salp16 irrespective of whether A. phagocytophilum was already present in the salivary gland or had newly entered the tick during engorgement on an infected mouse (Fig. 1 d). salp16 mRNA was not detected in the gut of uninfected ticks (15) but was observed upon A. phagocytophilum infection (unpublished data).
Silencing of the salp16 gene using RNAi reduces acquisition of A. phagocytophilum by the tick
To examine whether salp16 plays a role in the survival of A. phagocytophilum within ticks, salp16-deficient ticks were generated using RNAi. For the construction of salp16 double-stranded RNA (dsRNA), salp16 was amplified and cloned from the cDNA of engorged tick salivary glands. Injection of the RNAi construct through the idiosoma of the ticks and the feeding and recovery of I. scapularis were performed as described in Materials and methods. There was no visible phenotypic change or impairment in feeding in salp16-injected ticks when compared with control buffer-injected ticks.
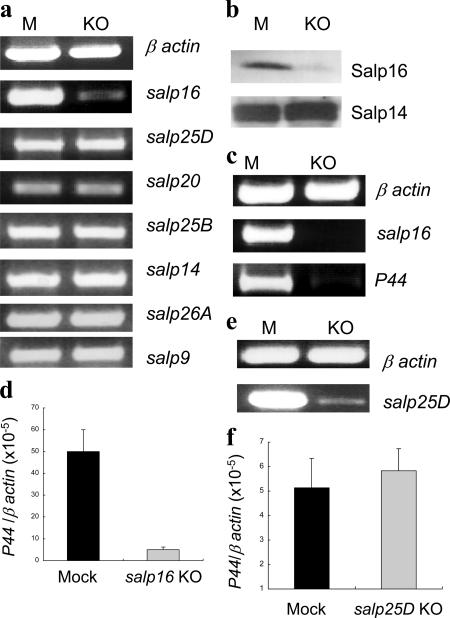
To assess the efficacy of the RNAi treatment, total RNA isolated from the salivary glands of mock (buffer alone) and salp16 dsRNA–injected ticks was used as a template to amplify salp16. salp16 mRNA was substantially decreased in the salp16 dsRNA–injected group (Fig. 2 a), and the percentage of inhibition was 93 ± 3% as quantified by ImageJ software (National Institutes of Health [NIH]). The expression levels of other salp genes, including salp25D, salp14, salp20, salp25B, salp26A, and salp9, were not affected by the silencing of salp16 (Fig. 2 a). The nucleotide sequence of salp16 is not related to any of the other salp genes investigated in this study. The protein extract of mock and salp16 dsRNA–injected tick salivary glands was analyzed by immunoblotting using recombinant Salp16 antisera to confirm gene silencing at the protein level. Salp16 in salivary glands of ticks injected with salp16 dsRNA was reduced by 81 ± 6% (Fig. 2 b). The specificity of gene silencing was determined by probing a duplicate immunoblot with antisera raised against another salivary gland protein, Salp14, and equivalent levels of Salp14 were evident in the control and experimental groups (Fig. 2 b).
Figure 2.
Silencing of the salp16 gene using RNAi reduces the acquisition of Anaplasma phagocytophilum by the tick. The efficiency and specificity of RNAi-dependent knockdown of the salp16 gene was assessed using RT-PCR (a) and immunoblotting (b). (a) The expression of salp25D, salp14, salp20, salp25B, salp26A, and salp9 in salp16-deficient tick salivary glands. As shown in b, the specificity of knockdown was confirmed using a duplicate immunoblot probed with Salp14 antisera. (c and d) RT-PCR– (c) and quantitative RT-PCR (d)–based demonstrations of the reduced ability of salp16-deficient ticks to acquire A. phagocytophilum from infected mice. RNAi-injected ticks were fed on infected mice for 72 h, and the level of A. phagocytophilum was assessed in the salivary gland using RT-PCR with the P44 gene as the marker. (e) RT-PCR showing the ablation of salp25D after the injection of ticks with a salp25D RNAi construct. (f) The acquisition of A. phagocytophilum by salp25D-depleted ticks. Results are expressed as the mean ± SEM (error bars) from three independent experiments. P < 0.05 was considered statistically significant (Student's t test). M, mock; KO, knockout.
We then explored whether the silencing of salp16 gene expression affects the ability of ticks to acquire A. phagocytophilum from the infected mammalian host. Control ticks and ticks injected with salp16 dsRNA were allowed to feed on each of three A. phagocytophilum–infected C3H mice (20 ticks per mice) for 72 h and were then collected. To assess the levels of salp16 and A. phagocytophilum in the salivary glands, RT-PCR was performed using total RNA from mock and salp16 dsRNA–injected tick salivary glands (from groups of three pairs of glands). The level of the gene encoding a major outer membrane antigen, P44 of A. phagocytophilum, was used to assess the number of viable bacteria. As expected, salp16 expression was diminished significantly in salp16 dsRNA–injected ticks (Fig. 2 c). Surprisingly, the level of A. phagocytophilum was markedly lower (reduced 10-fold; P < 0.01; Fig. 2 d) in salp16-deficient ticks. To rule out any bias on A. phagocytophilum acquisition by the tick as a result of mouse-to-mouse variations in the bacterial infection, we repeated the acquisition experiments by placing both mock and salp16-deficient ticks on the same A. phagocytophilum–infected mice. The results were identical to those observed when the acquisition experiment was performed using separate mice for mock and salp16-deficient ticks (unpublished data). To further demonstrate that the reduced acquisition of A. phagocytophilum by salp16-deficient ticks was specific, the acquisition experiment was repeated by silencing another salp gene, salp25D (Fig. 2 e). A. phagocytophilum levels were the same in the mock and salp25D-deficient groups (Fig. 2 f).
salp16 silencing does not affect the acquisition of B. burgdorferi
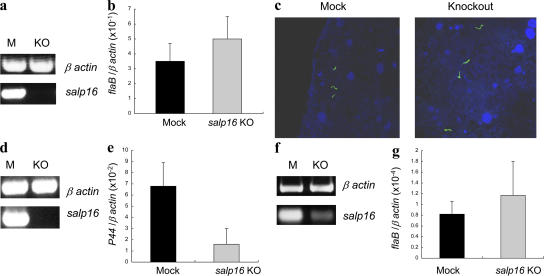
We determined whether salp16 was required for A. phagocytophilum acquisition by ticks or were used by another tick-borne pathogen by examining the levels of B. burgdorferi in salp16 dsRNA–injected nymphs after feeding on B. burgdorferi–infected C3H mice. B. burgdorferi infects the tick gut and moves to the salivary gland immediately before transmission. The guts of engorged ticks were collected at 72 h and examined. As expected, salp16 expression was markedly reduced in the guts of ticks administered salp16 dsRNA (Fig. 3 a). Quantitative RT-PCR demonstrated that there was no difference in the level of B. burgdorferi in salp16-deficient or control ticks (Fig. 3 b). Imaging of the tick gut for B. burgdorferi by confocal microscopy yielded similar results (Fig. 3 c).
Figure 3.
Silencing of salp16 expression does not affect the acquisition of B. burgdorferi. salp16 RNAi–injected ticks were allowed to feed on B. burgdorferi–infected mice, and the guts were analyzed at 72 h after infection. (a) RT-PCR–based confirmation of the silencing of salp16 in the knockout tick guts. (b) Analysis of the levels of B. burgdorferi in the gut of knockout ticks (KO) in comparison with mock-injected ticks (M) by quantitative RT-PCR analysis. flaB gene expression was used to measure the levels of B. burgdorferi. Results are means ± SEM (error bars) from one representative experiment. (c) Confirmation of the levels of B. burgdorferi in the gut of mock and knockout ticks by confocal microscopy. Gut samples from mock and knockout ticks were probed with FITC-labeled anti–B. burgdorferi antibody (green), and nuclei were stained with TO-PRO. (d) Knockout of salp16 in the salivary glands and (f) gut of ticks fed on coinfected mice for 72 h assessed by RT-PCR. The levels of A. phagocytophilum (e) and B. burgdorferi (g) in ticks fed on coinfected mice was performed by measuring the levels of A. phagocytophilum P44 and B. burgdorferi flaB. Results are the means ± SEM from three quantitative PCR experiments. P < 0.05 was considered statistically significant (Student's t test).
B. burgdorferi and A. phagocytophilum are both transmitted by I. scapularis and can sometimes simultaneously infect a host. Therefore, we examined the role of salp16 in the acquisition of B. burgdorferi and A. phagocytophilum from coinfected mice. The acquisition experiment involved feeding 20 salp16-deficient ticks on each of the three coinfected mice. Ticks were collected at 72 h after feeding. Groups of three ticks from each mouse were used to check the levels of A. phagocytophilum in the salivary glands and B. burgdorferi in the gut. As expected, salp16 was repressed in the tick salivary glands (Fig. 3 d) and gut (Fig. 3 f). The level of A. phagocytophilum was significantly lower in the salp16-deficient group in comparison with the mock group (P < 0.05; Fig. 3 e), and there was no change in the level of B. burgdorferi (P > 0.05; Fig. 3 g). Overall, these data show that salp16 disruption affects the acquisition of A. phagocytophilum but not B. burgdorferi.
Transmission of A. phagocytophilum is not affected in salp16-deficient ticks
To examine the role of salp16 in the transmission of A. phagocytophilum from ticks, 10 infected salp16-deficient or control nymphs were fed on each of three naive C3H mice. The mice were bled on days 5 and 8 to check the levels of A. phagocytophilum in the peripheral blood, time points at which infection is known to be detectable in the bloodstream. Quantitative PCR analysis showed that the bacterial load in the blood of mice fed upon by either control or salp16-deficient ticks was similar (copies of P44 gene per 1011 copies of mouse β-actin gene were 2.5 ± 0.8 and 2.3 ± 0.6 on day 5 and 5.9 ± 1.4 and 7.6 ± 1.5 on day 8, respectively), thereby demonstrating that A. phagocytophilum does not require salp16 for infection of the mammalian host.
A. phagocytophilum does not use salp16 in the mammalian host just before acquisition by the arthropod vector
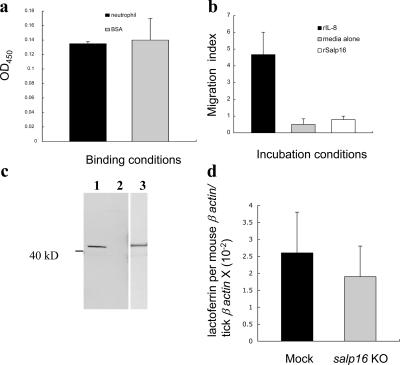
A. phagocytophilum in mice may be passively recruited to the tick bite site by the chemotaxis of granulocytes (7). It is possible that tick-derived molecules could attract neutrophils and facilitate the entry of A. phagocytophilum into the arthropod. Therefore, we examined whether Salp16 interacts with neutrophils, potentially leading to the enhanced entry of A. phagocytophilum into ticks. Extracts of human neutrophils did not bind rSalp16 (Fig. 4 a), and rSalp16 did not induce neutrophil chemotaxis (Fig. 4 b). Tick immune sera recognized rSalp16 (Fig. 4 c), and rSalp16 antisera bound native Salp16 (Fig. 2 b), demonstrating that the recombinant and native protein are similar enough to elicit cross-reactive antibodies. The binding assay and the chemotaxis assay were repeated with rSalp16 expressed in Drosophila cells, which also showed similar results (unpublished data). rSalp16 expressed in Drosophila cells has posttranslational modifications that may not be present in protein expressed in Escherichia coli; however, it still may not be identical to native Salp16. We then investigated whether the presence of Salp16 enhances the acquisition levels of neutrophils by ticks. Neutrophil-specific marker-based PCR analysis of salp16-deficient and wild-type ticks after feeding showed equal numbers of neutrophils, demonstrating that neutrophil entry into ticks is independent of Salp16 (Fig. 4 d). These experiments support the hypothesis that the functional requirement of A. phagocytophilum for Salp16 occurs within the vector alone.
Figure 4.
A. phagocytophilum does not use Salp16 in the mammalian host before acquisition by the arthropod. (a) Binding of Salp16 with human neutrophils. Extracts of human neutrophils or BSA (control) were incubated with recombinant Salp16, and a binding study was performed as described in Materials and methods. Bars represent the mean ± SEM (error bars) from three studies. (b) A neutrophil chemotaxis assay using Salp16. Freshly isolated human neutrophils were used as chemoattractants in 96-well Neuro Probe TX chambers together with recombinant IL-8 as a positive control and medium as a negative control as described in Materials and methods. The results are expressed as the migration index after subtraction of background values (medium alone). The means ± SEM of three independent studies are shown. (c) Immunoblot showing that recombinant Salp16 (expressed in bacteria) is recognized by tick immune sera. Lane 1 shows recombinant Salp16 (GST fusion) probed with rabbit tick immune sera; lane 2 shows GST protein probed with tick immune sera; and lane 3 shows recombinant Salp16 probed with Salp16 antisera. (d) Comparison of the number of neutrophils entering the mock and salp16 knockout (KO) ticks. After the feeding of mock and salp16 knockout ticks on infected mice, the number of neutrophils within the whole ticks was assessed by measuring the lactoferrin (neutrophil-specific gene) levels (normalized to mouse β-actin gene and tick β-actin gene) by quantitative RT-PCR analysis. The results are expressed as means ± SEM from three independent experiments.
A. phagocytophilum requires Salp16 for initial infection of the tick salivary gland
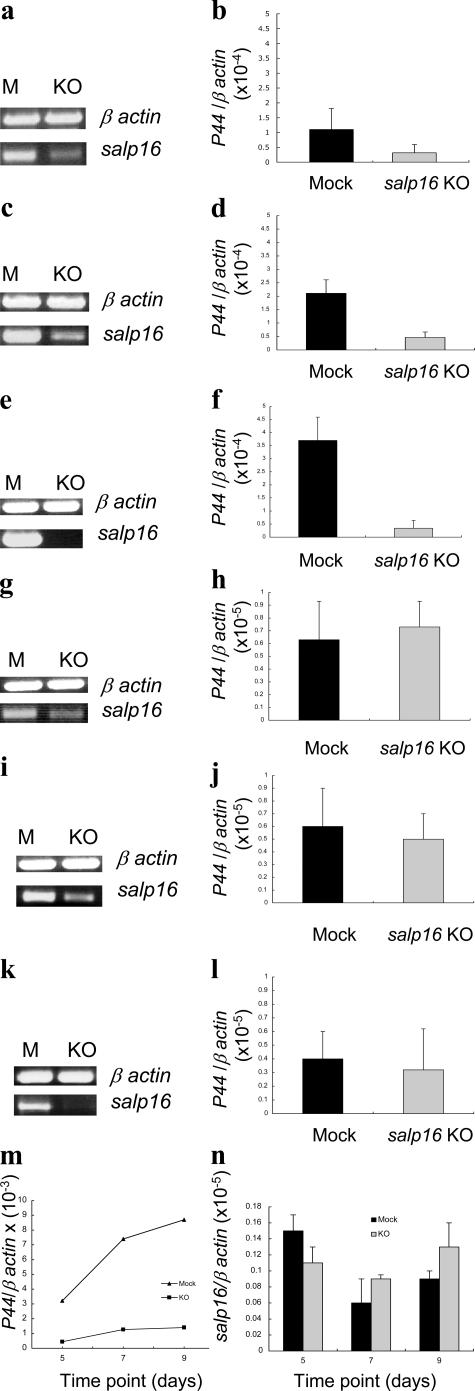
We then determined whether Salp16 is involved in the entry of A. phagocytophilum to the tick gut or salivary gland. A time course assessment of the level of A. phagocytophilum in the gut and salivary glands of feeding ticks assessed whether Salp16 is involved in the ingress of the bacteria into these tissues. Mock or salp16-deficient ticks were placed on each of three A. phagocytophilum–infected C3H mice (20 ticks on each mice), and, at 24, 48, and 72 h after feeding, three ticks from each group were collected. Salivary gland and gut samples were analyzed for the level of salp16 and A. phagocytophilum. Fig. 5 (a, c, and e) shows the repression of salp16 in the salivary glands at 24, 48, and 72 h. Similarly, Fig. 5 (g, i, and k) represents the repression of salp16 in the gut at 24, 48, and 72 h. There was a significant reduction in the levels of A. phagocytophilum in the salivary glands of salp16-deficient ticks at 24 (P < 0.05; Fig. 5 b), 48 (P < 0.05; Fig. 5 d), and 72 h (P < 0.01; Fig. 5 f). The relative reduction of A. phagocytophilum in salp16-deficient glands compared with bacteria in mock glands gradually increased from 73 to 90% as the feeding time increased from 24 to 72 h. In contrast, the levels of A. phagocytophilum in the gut did not show any change at any time points when mock ticks were compared with salp16 dsRNA–injected ticks (P > 0.05; Fig. 5, h, j, and l). The presence of similar numbers of A. phagocytophilum in the gut at all intervals indicates that salp16 is not required by the bacteria in the gut. Fig. 5 m shows that at later postfeeding time points (days 5, 7, and 9), the relative reduction of A. phagocytophilum in salp16-deficient salivary glands compared with mock glands were 86.3, 82.8, and 83.9% (P < 0.05), respectively. However, as indicated in Fig. 5 n, the expression level of salp16 in both mock and salp16-deficient ticks were barely detectable at these time points and were comparable with its levels in the salivary glands of unfed infected ticks. The level of A. phagocytophilum was substantially lower in the salivary glands of salp16-silenced ticks in comparison with mock salivary glands at all time points examined, suggesting that A. phagocytophilum specifically requires salp16 to infect salivary glands.
Figure 5.
A. phagocytophilum requires Salp16 for the initial infection of the tick salivary gland. A time course assessment of the levels of A. phagocytophilum within the salivary glands and guts of salp16-deficient (KO) and mock (M) ticks fed on infected mice for 24, 48, and 72 h is shown. Repression of salp16 in the salivary glands of ticks fed for 24 (a), 48 (c), and 72 h (e) by RT-PCR. The levels of A. phagocytophilum P44 within the salivary glands of ticks fed for 24 (b), 48 (d), and 72 h (f) by quantitative RT-PCR. Repression of salp16 within the corresponding gut samples of ticks fed for 24 (g), 48 (i), and 72 h (k) by RT-PCR. The levels of A. phagocytophilum P44 in the gut samples of ticks fed for 24 (h), 48 (j), and 72 h (l). (m) Levels of A. phagocytophilum P44 in the salivary glands of ticks on days 5, 7, and 9 after feeding, and (n) levels of salp16 in the salivary glands at the respective time points. The results are expressed as means ± SEM (error bars) from a representative experiment. P < 0.05 was considered statistically significant (Student's t test).
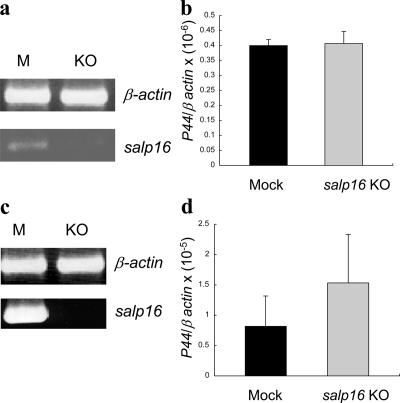
We then studied the role of salp16 in the survival of A. phagocytophilum within a previously infected salivary gland. We monitored the levels of A. phagocytophilum in the salivary glands of infected nymphal ticks (infected as larvae and molted to nymph) in which salp16 was subsequently repressed. The A. phagocytophilum–infected salp16-deficient ticks were examined either in an unfed state (Fig. 6 a) or after 72 h (Fig. 6 c) of feeding. There was no significant difference in the levels of A. phagocytophilum when we compared mock with salp16 dsRNA–treated groups (P > 0.05; Fig. 6, b and d). These results show that salp16 is not required for the maintenance of A. phagocytophilum in the salivary glands of an infected tick.
Figure 6.
salp16 is not required for the maintenance of A. phagocytophilum within infected tick salivary glands. salp16 RNAi was injected into A. phagocytophilum–infected ticks and were analyzed either in an unfed state or in a 72-h fed state. (a) Knockdown of salp16 expression in A. phagocytophilum–infected unfed ticks by RT-PCR, and (b) quantification of A. phagocytophilum P44 levels by quantitative RT-PCR analysis. (c) Knockdown of salp16 expression in 72-h fed ticks by RT-PCR, and (d) quantitative RT-PCR data showing A. phagocytophilum P44 levels. Results are means ± SEM (error bars) from three independent experiments. P < 0.05 was considered statistically significant (Student's t test). M, mock; KO, knockout.
DISCUSSION
Ixodes ticks transmit many important human and veterinary pathogens, including B. burgdorferi (16), A. phagocytophilum (6), Babesia microti (17), Bartonella species (18), and tick-borne encephalitis virus (19). A tick receptor that enables B. burgdorferi to attach to the tick gut and, thereby, infect I. scapularis has been recently identified (20). However, the arthropod ligands participating in pathogen survival in ticks and tick salivary glands, particularly of the related group of obligate intracellular microbes that include Anaplasma, Rickettsia, Ehrlichia, and Neorickettsia, are unknown (21). In this study, we demonstrate that A. phagocytophilum requires an I. scapularis protein, Salp16, to infect tick salivary glands.
The striking temporal relationship between A. phagocytophilum infection, tick feeding, and the strong up-regulation of salp16 first raised the possibility that A. phagocytophilum may need this protein within the vector. The lack of a notable induction of salp16 in an unfed A. phagocytophilum–infected tick compared with an unfed uninfected tick (in contrast with the robust stimulation of salp16 during I. scapularis engorgement) suggested that A. phagocytophilum does not require salp16 to persist once infection of the tick has been established. Indeed, when salp16 expression was knocked down in a tick previously infected with A. phagocytophilum (infected as a larvae and molted to nymph), there was no difference in the level of A. phagocytophilum within salivary glands compared with that of control ticks. In contrast, the acquisition of A. phagocytophilum from the infected mammalian host was severely inhibited (up to a 10-fold reduction from controls) in salp16-deficient ticks. Collectively, these data show a specific dependence of A. phagocytophilum on Salp16 for the microbe's successful acquisition by I. scapularis.
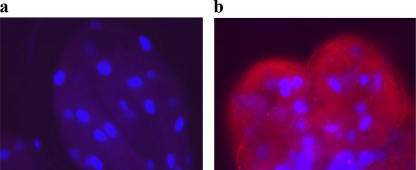
We then determined when in the A. phagocytophilum life cycle Salp16 was necessary. Ticks may secrete Salp16 into mice during the process of engorgement because Salp16 protein has been found in tick saliva by antigen-capture ELISA (15, 22). Moreover, Salp16 antibodies have been identified in animals exposed to repeated tick bite (15, 22), and Salp16 has a putative secretory signal sequence, as assessed by the SignalP 3.0 program (www.cbs.dtu.dk/services/SignalP/), with the cleavage of the signal peptide predicted to occur between amino acid residues 18 and 19. We also showed that tick immune sera, which is raised against secreted tick proteins, recognizes rSalp16 (Fig. 4 c). The immunostaining of Salp16 in the engorged tick salivary glands using anti-rSalp16 antisera shows that Salp16 is distributed throughout the salivary gland (Fig. 7), similar to the pattern of two other secreted salivary gland proteins, Salp14 and Salp25D (23). We performed studies to examine whether A. phagocytophilum–Salp16 interactions could be important in the host environment. These data (Fig. 4) suggested that A. phagocytophilum does not use Salp16 in the host just before acquisition by the vector. Therefore, we focused on delineating the stage of A. phagocytophilum life cycle within I. scapularis that is influenced by Salp16. Temporal acquisition studies (Fig. 5, c, e, i, and k) in salp16-depleted ticks showed a marked reduction (up to 90%) in the levels of A. phagocytophilum within salp16-deficient I. scapularis salivary glands compared with controls. In contrast, the levels of A. phagocytophilum within the gut of salp16-deficient and control ticks remained the same at all time periods, demonstrating that Salp16 is not required by A. phagocytophilum in the gut. A postfeeding time course analysis showed that the reduction in the level of A. phagocytophilum within the salivary glands of salp16-deficient ticks was consistently maintained throughout the 9-d examination period (Fig. 5 m). Collectively, these results show that Salp16 is required by A. phagocytophilum for the infection of I. scapularis salivary glands.
Figure 7.
Confocal microscopy showing the distribution of Salp16 within engorged tick salivary glands. Ticks were fed for 72 h, and salivary glands were isolated and probed with anti-GST antisera (negative control; a) and anti-Salp16 polyclonal antisera (b). Binding was visualized using tetramethylrhodamine isothiocyanate–conjugated secondary antibody, and the samples were counterstained with nuclear stain DAPI.
I. scapularis ticks also vector B. burgdorferi, the causative agent of Lyme disease and the most common tick-transmitted bacterium in North America (16). Coinfections and cotransmissions of B. burgdorferi and A. phagocytophilum are known to occur (24). Thus, there was an interest to examine whether Salp16 was also critical for the acquisition of B. burgdorferi. B. burgdorferi infection did not alter the expression level of salp16 in ticks (unpublished data). Furthermore, RNAi knockdown experiments (Fig. 3 b) demonstrated that Salp16 was not essential for B. burgdorferi acquisition. The observation that the abrogation of salp16 impairs the acquisition of A. phagocytophilum and not B. burgdorferi from a coinfected mouse host (Fig. 3 g) underscores the functional importance of Salp16 in A. phagocytophilum infection. I. scapularis ticks can also harbor and transmit B. microti (17). Like A. phagocytophilum, B. microti, a protozoan pathogen, infects the salivary gland of ticks. However, unlike A. phagocytophilum, B. microti infection did not alter the expression of salp16 (unpublished data). Further testing with various tick-borne pathogens will determine whether salp16 is used by any other pathogens.
The mechanism by which Salp16 enables A. phagocytophilum to infect I. scapularis salivary glands remains to be elucidated. The reduction in the level of A. phagocytophilum in the salivary glands of salp16-deficient ticks compared with control ticks as feeding progresses suggests that Salp16 may be required by the bacterium during an early stage of infection of the salivary glands, perhaps at entry or immediately after entry. In addition, the postfeeding (>72 h) multiplication rates of A. phagocytophilum within the salivary glands of mock and salp16-deficient ticks were similar (Fig. 5 m), further suggesting that the pathogen requires Salp16 early during infection. Finally, the integrity of the vacuolar membrane surrounding the A. phagocytophilum morulae within neutrophils is critical for its survival (25), and it is logical to expect that the Anaplasma-containing vacuole within the arthropod is also vital for bacterial growth. This vacuole could certainly contain both tick and bacterial components. Salp16 could be directly or indirectly involved in this process in the salivary glands; however, Anaplasma-containing vacuoles are likely to also be important in the gut, where Salp16 does not appear to be essential. It is conceivable that during tick feeding and pathogen acquisition, the bacterium may be predominantly contained in morulae within the host neutrophils entering the gut along with the blood meal. As A. phagocytophilum leaves these morulae, poised to infect tick salivary glands, the role of Salp16 may be vital. To date, little is known of how A. phagocytophilum transitions from the host neutrophil to the tick salivary glands.
Obligate intracellular pathogens in the genera closely related to Anaplasma are among the most poorly understood pathogens despite their historical and current importance. As examples, Rickettsia prowazekii, the agent of epidemic typhus, has been a leading cause of death during war and famine throughout history (26). Rocky mountain spotted fever, caused by Rickettsia rickettsia, has recently been described in new locations in the United States (27), and A. phagocytophilum is now the second most common tick-borne disease in North America. These data now identify a vector molecule that is essential for the survival of A. phagocytophilum within I. scapularis and serves as a paradigm for understanding how obligate intracellular pathogens may survive within arthropods. It is likely that related agents may also require specific vector antigens to successfully complete their life cycle. The molecular aspects of arthropod–pathogen interactions are only beginning to be explored (11, 12, 28). The identification of these fundamental interactions will enable us to develop new methods to interrupt the pathogen–vector–host cycle and may lead to novel methods to prevent or contain disease.
MATERIALS AND METHODS
Mice.
4–6-wk-old female C3H/HeN and C3H/SCID mice were purchased from the NIH and The Jackson Laboratory, respectively. The maintenance of mice and the animal experiments were performed according to the protocols approved by the Institutional Animal Care and Use Committee at the Yale University School of Medicine.
Infection of C3H/HeN mice with A. phagocytophilum and/or B. burgdorferi.
The A. phagocytophilum isolate NCH-1 (6) used in these studies was maintained through serial passage of infected blood in C3H/SCID mice as described previously (29). Quantitative PCR was performed on an aliquot of the pooled blood collected from the SCID mice to quantitate and standardize the infectious dose of A. phagocytophilum.
For the acquisition experiments, C3H/HeN mice were injected intraperitoneally with 100 μl of pooled A. phagocytophilum–infected or uninfected (controls) anticoagulated blood. The bacterial burden in the peripheral mouse blood was assessed on day 6. Total genomic DNA was isolated from the peripheral blood using the DNeasy tissue kit (QIAGEN) according to the manufacturer's protocol, and the levels of P44 were measured using quantitative PCR. Ticks were then allowed to engorge upon the infected mice on day 7, the interval of maximal infection of A. phagocytophilum in the peripheral blood.
B. burgdorferi isolate N40 was used to infect C3H/HeN mice (104 bacteria per mouse). 2 wk after infection, the mice were tested by taking ear punches to amplify flaB DNA in order to confirm the presence of spirochetes. DNA extraction and PCR analysis were performed as described previously (20).
To generate coinfected mice, the C3H/HeN mice were first challenged with 104 B. burgdorferi followed by challenge with A. phagocytophilum on day 7. On day 14, after the infection with B. burgdorferi and 7 d after infection with A. phagocytophilum, the mice were tested for the presence of both B. burgdorferi and A. phagocytophilum as described above.
Ticks.
I. scapularis nymphs and larvae and B. burgdorferi–infected nymphs (70% infected) were obtained from continuously maintained tick colonies. To generate A. phagocytophilum–infected nymphs, the larvae were fed to repletion on A. phagocytophilum–infected C3H/HeN mice and molted into nymphs. To produce uninfected nymphs (controls), larvae engorged on uninfected mice and were then allowed to molt. Tick rearing was conducted in an incubator at 23°C with 85% relative humidity and a 14/10 h light/dark photo period regimen. 10% of the molted nymphs from each infection group were individually tested by PCR to confirm infection and to determine prevalence. The infection rate was 82 ± 13% for A. phagocytophilum–infected nymphs.
RNAi.
Salivary glands isolated from engorged nymphs were used to isolate cDNA. salp16 was amplified using the primers S16RNAiF (5′-GAAGATCTGTACTCGCTGGTTTATGTTTC-3′) and S16RNAiR (5′-GGGGTACCATTTGTCTTTGTTACTGTTGC-3′) containing BglII and KpnI restriction enzyme sites. The amplified salp16 fragment was cloned into the L4440 double T7 Script II vector, and dsRNA was synthesized using the Megascript RNAi kit (Ambion). 5 nl (5 × 1012 molecules/μl) salp16 dsRNA was injected into uninfected A. phagocytophilum– or B. burgdorferi–infected nymphal ticks. The dsRNA was inoculated through the idiosoma of the ticks into the hoemocoel. The feeding and recovery of ticks were performed as previously described (23). The control ticks (mock) received 0.5 nl of the injection buffer (10 mM Tris-HCl, pH 7.5, and 1 mM EDTA). Ticks fed for the respective time points were collected, cDNA was made from the salivary glands, and the expression of salp16 was assessed using primers 16F-RNIC (5′-CTGAAGTTCTTTATTCTCTTC-3′) and 16R-RNIC (5′-GCAGGGTCCTTCTTCGGG-3′). The construction of salp25D RNAi was performed in a similar fashion using primers salp25DRNAiF (5′-AGATCTCCACGAATGGCTCGGC-3′) and salp25DRNAiR (5′-GGTACCGGAACAGCTTGAGAATC-3′). The salp25D RNAi injection, feeding of ticks, and cDNA synthesis from the salivary glands were performed as described for salp16. The expression of salp25D after the RNAi experiment was assessed using primers salp25DRT-F (5′-CCTTTCCCCAACTTCACC-3′) and salp25RT-R (5′-GTCCATGGTTGTTCGGAG-3′).
Immunoblots.
Salivary glands (10 pooled samples) isolated from the mock and salp16-deficient ticks were resuspended in sterile PBS with protease inhibitors (protease inhibitor cocktail; Sigma-Aldrich) and homogenized. Equal amounts of salivary gland protein (10 μg) from mock or salp16 dsRNA–injected ticks were electrophoresed on an SDS/12% polyacrylamide gel and processed for immunoblotting. The immunoblots were probed with recombinant Salp16 antisera. A duplicate immunoblot was probed with recombinant Salp14 antisera. For the immunoblot showing tick immune sera recognizing recombinant Salp16 (glutathione S-transferase [GST] fusion), 1 μg of bacterial recombinant Salp16 and 1 μg GST protein were electrophoresed on an SDS/12% polyacrylamide gel and probed with rabbit tick immune sera. As a positive control, rSalp16 was probed with anti-Salp16 antisera. The bound antibodies were detected by using horseradish peroxidase–conjugated rabbit anti–mouse and goat anti–rabbit secondary antibodies (Sigma-Aldrich), and the blots were developed using the enhanced chemiluminescence Western Blotting Detection System (GE Healthcare).
Acquisition experiment.
For the acquisition experiments, control ticks and ticks injected with salp16 dsRNA were allowed to feed on A. phagocytophilum–infected, B. burgdorferi–infected, or coinfected C3H/HeN mice for 72 h and were collected. We used three mice in each group (experimental and control/mock), and 20 ticks were placed on each mouse. cDNA was made from pooled salivary glands or guts of three fed ticks. After the confirmation of the knockdown of salp16, the levels of the P44 gene of A. phagocytophilum and the flaB gene of B. burgdorferi were measured by quantitative RT-PCR to assess the levels of viable bacteria. For the time point acquisition experiments, mock and salp16 dsRNA–injected ticks were fed for 24, 48, and 72 h on A. phagocytophilum–infected C3H/HeN mice, and salivary glands (pooled in groups of three pairs) from each group were collected at each time point and analyzed as described for the 72-h acquisition experiments. To determine the levels of A. phagocytophilum in the salivary glands of mock and salp16 dsRNA–injected ticks at later time points (days 5, 7, and 9), the engorged ticks were maintained in an incubator at 23°C with 85% relative humidity, and the salivary glands (three pairs pooled) were dissected on the indicated days.
To rule out the possibility of any bias on A. phagocytophilum acquisition by the tick as a result of variation in the infection levels in various mice, we also performed an acquisition experiment with single mice. In these experiments, the mock group of nymphs was injected with buffer containing 1 nM Ponceau S (Sigma-Aldrich). 20 ticks (salp16 dsRNA injected or mock injected) were placed on a single mouse, allowed to feed for 72 h, and collected. The Ponceau S dye stained the injection site pink and enabled us to distinguish mock-injected and dsRNA-injected ticks.
Transmission experiment.
A. phagocytophilum–infected and salp16-deficient or control nymphs (10 ticks each) were allowed to feed on each of three naive C3H/HeN mice. Once the ticks fed to repletion, the mice were bled on days 5 and 8. Total genomic DNA was isolated from the peripheral blood using the DNeasy tissue kit (QIAGEN), and the level of P44 was measured using quantitative PCR.
RNA extraction, cDNA synthesis, RT-PCR, quantitative RT-PCR, and genomic DNA isolation.
Total RNA was extracted from the salivary glands and gut samples using TRIzol reagent (Invitrogen). The RNA was treated with TURBO DNase (Ambion) to remove the contaminating DNA. The purified RNA was used for RT-PCR and quantitative PCR analysis.
cDNA was made from the tick salivary glands and gut samples using the iScript cDNA synthesis kit (Bio-Rad Laboratories). Quantitative PCR was performed using an iCycler Real-Time Detection System (Bio-Rad Laboratories). Real-time PCR was performed according to the iQ SYBR Green Supermix (Bio-Rad Laboratories) protocol. The relative gene expression levels of each transcript were determined by comparison with a standard curve. The salivary gland or gut cDNA levels were normalized to the I. scapularis β-actin gene, and the levels of P44 and flaB genes were then quantified (iCycler IQ software version 3.1; Bio-Rad Laboratories). In the mouse infection studies, the mouse β-actin gene was used to normalize the levels of DNA in the samples, and P44 was used to assess the levels of A. phagocytophilum in the mouse blood samples. The sequence-specific primers for the P44 gene were HF (5′-CTACTAGCTAAGGAGTTAGC-3′) and HB (5′-CACAGAAGTAGAAGAAACCG-3′; reference 30) and for the flab gene were FlabF (5′-TTCAATCAGGTAACGGCACA-3′) and FlabR (5′-GACGCT TGAGACCCTGAAAG-3′). The primers used for RT-PCR, quantitative RT-PCR to analyze the expression of 14 salp genes, and their GenBank accession numbers are as described previously (15, 22).
To quantify neutrophil entry into mock and salp16-deficient ticks during acquisition feeding, total genomic DNA was isolated from whole ticks that had fed for 72 h using the DNeasy tissue kit (QIAGEN). The expression level of a neutrophil-specific gene, lactoferrin, was used as a direct readout of the number of neutrophils entering the ticks during feeding. Equal amounts of total tick genomic DNA (0.5 μg) from either mock or salp16-deficient ticks were used as a template to amplify the mouse neutrophil lactoferrin gene using the primers LACTMF (5′-CGGACTCACTACTATGC-3′) and LACTMR (5′-TCCTCAAGGGATGCAGGT-3′), and the levels of DNA were normalized to the mouse β-actin gene and to the tick β-actin gene.
Protein expression and preparation of polyclonal antibody.
salp16 was amplified using primers 5′-CCGCTCGAGGATACAAGTCCCAGTGAGACAGGA-3′ and 5′-ATAAGAATGCGGCCGCATGCAGCTTGGCAAGGG-3′ and was cloned into pGEX6P-1 vector (GE Healthcare) for bacterial expression or into pIZ/V5/His vector (Invitrogen) for Drosophila expression. In BL21 bacterial cells, the salp16 gene was induced with 1 mM isopropyl-β-d-thiogalactoside for 3 h at 37°C, and the culture pellet was lysed using a French press. After centrifugation at 7,500 g for 30 min, the supernatant was allowed to pass through a glutathione–Sepharose 4B column (GE Healthcare), washed extensively, and eluted. The glutathione transferase fusion protein tag was cleaved using PreScission Protease (GE Healthcare). Salp16 was expressed in a Drosophila cell-based system and purified using Talon affinity chromatography (CLONTECH Laboratories, Inc.). The antibody against bacterially expressed Salp16 was raised in both mice and rabbit using standard protocols.
In vitro binding assay.
Human neutrophils were isolated as described previously (31, 32). The binding assays of rSalp16 to neutrophil extracts were performed as previously described (33). In brief, 0.5 μg of neutrophil extract was coated on ELISA plates overnight at 4°C. After blocking the nonspecific sites, the wells were incubated with rSalp16 (1 μg/well) for 1 h followed by incubation with rSalp16 antisera. Binding was detected using anti–mouse IgG–horseradish peroxidase conjugate as secondary reagent, and TMB Microwell peroxidase substrate was used for color development. The OD was measured at 450 nm.
Neutrophil chemotaxis assay.
2 × 106 freshly isolated human neutrophils suspended in 50 μl of Hank's balanced salt solution (Invitrogen) were added to the upper compartment of a 96-well ChemoTx System Chemotaxis Chamber (Neuro Probe, Inc). 1 μg/well rSalp16 was added to the lower compartment. 0.1 μg/well of human recombinant IL-8 was used as the positive control and the medium alone as the negative control. After washing off the cells attached under the filter after 30 min of incubation at 37°C with 5% CO2, the migrated cells were determined by reading the absorbency at 490 nm after the addition of CellTiter 96 AQueous assay reagent (Promega). The OD values of the samples were expressed relative to the standard curve values, yielding an index of cells that migrated across the membrane.
Confocal microscopy.
The levels of B. burgdorferi within the mock and salp16-deficient tick gut samples were analyzed by confocal microscopy as described previously (34). In brief, the acetone-fixed gut samples were incubated with FITC-conjugated anti–B. burgdorferi antisera (Kirkegaard and Perry Laboratories, Inc.) followed by the nuclear stain TO-PRO (Invitrogen). Staining of Salp16 within tick salivary glands was performed as previously described (23). Acetone-fixed salivary gland samples were incubated with control GST antisera and anti-Salp16 antisera, respectively. The binding was visualized using tetramethylrhodamine isothiocyanate–conjugated secondary antibody. The samples were counterstained with the nuclear stain DAPI and viewed with a scanning laser confocal microscope (LSM 510; Carl Zeiss MicroImaging, Inc.).
Statistical analysis.
Results are expressed as the means ± SE. The significance of the differences between the mean values of two groups of mice or time periods was evaluated by Student's t test. Calculated P values of <0.05 were considered significant.
Acknowledgments
We thank Michele Papero for maintaining and providing I. scapularis larvae and A. phagocytophilum–infected nymphs, Deborah Beck for help with the mice, and Utpal Pal and Ruth Montgomery for assistance with microscopy.
This work was supported by grants from the NIH.
The authors have no conflicting financial interests.
Abbreviations used: dsRNA, double-stranded RNA; GST, glutathione S-transferase.
References
- 1.Cao, W.C., Q.M. Zhao, P.H. Zhang, J.S. Dumler, X.T. Zhang, L.Q. Fang, and H. Yang. 2000. Granulocytic Ehrlichiae in Ixodes persulcatus ticks from an area in China where Lyme disease is endemic. J. Clin. Microbiol. 38:4208–4210. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Chen, S.M., J.S. Dumler, J.S. Bakken, and D.H. Walker. 1994. Identification of a granulocytotropic Ehrlichia species as the etiologic agent of human disease. J. Clin. Microbiol. 32:589–595. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Dumler, J.S., and J.S. Bakken. 1998. Human ehrlichioses: newly recognized infections transmitted by ticks. Annu. Rev. Med. 49:201–213. [DOI] [PubMed] [Google Scholar]
- 4.Park, J.H., E.J. Heo, K.S. Choi, J.S. Dumler, and J.S. Chae. 2003. Detection of antibodies to Anaplasma phagocytophilum and Ehrlichia chaffeensis antigens in sera of Korean patients by western immunoblotting and indirect immunofluorescence assays. Clin. Diagn. Lab. Immunol. 10:1059–1064. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Bakken, J.S., J.S. Dumler, S.M. Chen, M.R. Eckman, L.L. Van Etta, and D.H. Walker. 1994. Human granulocytic ehrlichiosis in the upper Midwest United States. A new species emerging? JAMA. 272:212–218. [PubMed] [Google Scholar]
- 6.Telford, S.R., J.E. Dawson, P. Katavolos, C.K. Warner, C.P. Kolbert, and D.H. Persing. 1996. Perpetuation of the agent of human granulocytic ehrlichiosis in a deer tick-rodent cycle. Proc. Natl. Acad. Sci. USA. 93:6209–6214. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Hodzic, E., D.L. Borjesson, S. Feng, and S.W. Barthold. 2001. Acquisition dynamics of Borrelia burgdorferi and the agent of human granulocytic ehrlichiosis at the host-vector interface. Vector Borne Zoonotic Dis. 1:149–158. [DOI] [PubMed] [Google Scholar]
- 8.des Vignes, F., J. Piesman, R. Heffernan, T.L. Schulze, K.C. Stafford 3rd, and D. Fish. 2001. Effect of tick removal on transmission of Borrelia burgdorferi and Ehrlichia phagocytophila by Ixodes scapularis nymphs. J. Infect. Dis. 183:773–778. [DOI] [PubMed] [Google Scholar]
- 9.Hodzic, E., D. Fish, C.M. Maretzki, A.M. De Silva, S. Feng, and S.W. Barthold. 1998. Acquisition and transmission of the agent of human granulocytic ehrlichiosis by Ixodes scapularis ticks. J. Clin. Microbiol. 36:3574–3578. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Katavolos, P., P.M. Armstrong, J.E. Dawson, and S.R. Telford. 1998. Duration of tick attachment required for transmission of granulocytic ehrlichiosis. J. Infect. Dis. 177:1422–1425. [DOI] [PubMed] [Google Scholar]
- 11.Ramamoorthi, N., S. Narasimhan, U. Pal, F. Bao, X.F. Yang, D. Fish, J. Anguita, M.V. Norgard, F.S. Kantor, J.F. Anderson, et al. 2005. The Lyme disease agent exploits a tick protein to infect the mammalian host. Nature. 436:573–577. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Kamhawi, S., M. Ramalho-Ortigao, V.M. Pham, S. Kumar, P.G. Lawyer, S.J. Turco, C. Barillas-Mury, D.L. Sacks, and J.G. Valenzuela. 2004. A role for insect galectins in parasite survival. Cell. 119:329–341. [DOI] [PubMed] [Google Scholar]
- 13.Zieler, H., J.P. Nawrocki, and M. Shahabuddin. 1999. Plasmodium gallinaceum ookinetes adhere specifically to the midgut epithelium of Aedes aegypti by interaction with a carbohydrate ligand. J. Exp. Biol. 202:485–495. [DOI] [PubMed] [Google Scholar]
- 14.Matuschewski, K., A.C. Nunes, V. Nussenzweig, and R. Menard. 2002. Plasmodium sporozoite invasion into insect and mammalian cells is directed by the same dual binding system. EMBO J. 21:1597–1606. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Das, S., N. Marcantonio, K. Deponte, S.R. Telford, J.F. Anderson, F.S. Kantor, and E. Fikrig. 2000. SALP16, a gene induced in Ixodes scapularis salivary glands during tick feeding. Am. J. Trop. Med. Hyg. 62:99–105. [DOI] [PubMed] [Google Scholar]
- 16.Barbour, A.G., and D. Fish. 1993. The biological and social phenomenon of Lyme disease. Science. 260:1610–1616. [DOI] [PubMed] [Google Scholar]
- 17.Mather, T.N., S.R. Telford, S.I. Moore, and A. Spielman. 1990. Borrelia burgdorferi and Babesia microti: efficiency of transmission from reservoirs to vector ticks (Ixodes dammini). Exp. Parasitol. 70:55–61. [DOI] [PubMed] [Google Scholar]
- 18.Schouls, L.M., I. Van De Pol, S.G. Rijpkema, and C.S. Schot. 1999. Detection and identification of Ehrlichia, Borrelia burgdorferi sensu lato, and Bartonella species in Dutch Ixodes ricinus ticks. J. Clin. Microbiol. 37:2215–2222. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Telford, S.R., P.M. Armstrong, P. Katavolos, I. Foppa, A.S. Garcia, M.L. Wilson, and A. Spielman. 1997. A new tick-borne encephalitis-like virus infecting New England deer ticks, Ixodes dammini. Emerg. Infect. Dis. 3:165–170. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Pal, U., X. Li, T. Wang, R.R. Montgomery, N. Ramamoorthi, A.M. Desilva, F. Bao, X. Yang, M. Pypaert, D. Pradhan, et al. 2004. TROSPA, an Ixodes scapularis receptor for Borrelia burgdorferi. Cell. 119:457–468. [DOI] [PubMed] [Google Scholar]
- 21.Dumler, J.S., A.F. Barbet, C.P. Bekker, G.A. Dasch, G.H. Palmer, S.C. Ray, Y. Rikihisa, and F.R. Rurangirwa. 2001. Reorganization of genera in the families Rickettsiaceae and Anaplasmataceae in the order Rickettsiales: unification of some species of Ehrlichia with Anaplasma, Cowdria with Ehrlichia and Ehrlichia with Neorickettsia, descriptions of six new species combinations and designation of Ehrlichia equi and ‘HGE agent’ as subjective synonyms of Ehrlichia phagocytophila. Int. J. Syst. Evol. Microbiol. 51:2145–2165. [DOI] [PubMed] [Google Scholar]
- 22.Das, S., G. Banerjee, K. DePonte, N. Marcantonio, F.S. Kantor, and E. Fikrig. 2001. Salp25D, an Ixodes scapularis antioxidant, is 1 of 14 immunodominant antigens in engorged tick salivary glands. J. Infect. Dis. 184:1056–1064. [DOI] [PubMed] [Google Scholar]
- 23.Narasimhan, S., R.R. Montgomery, K. DePonte, C. Tschudi, N. Marcantonio, J.F. Anderson, J.R. Sauer, M. Cappello, F.S. Kantor, and E. Fikrig. 2004. Disruption of Ixodes scapularis anticoagulation by using RNA interference. Proc. Natl. Acad. Sci. USA. 101:1141–1146. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Levin, M.L., and D. Fish. 2000. Acquisition of coinfection and simultaneous transmission of Borrelia burgdorferi and Ehrlichia phagocytophila by Ixodes scapularis ticks. Infect. Immun. 68:2183–2186. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Carlyon, J.A., and E. Fikrig. 2003. Invasion and survival strategies of Anaplasma phagocytophilum. Cell. Microbiol. 5:743–754. [DOI] [PubMed] [Google Scholar]
- 26.Azad, A.F., and S. Radulovic. 2003. Pathogenic rickettsiae as bioterrorism agents. Ann. NY Acad. Sci. 990:734–738. [DOI] [PubMed] [Google Scholar]
- 27.Demma, L.J., M.S. Traeger, W.L. Nicholson, C.D. Paddock, D.M. Blau, M.E. Eremeeva, G.A. Dasch, M.L. Levin, J. Singleton Jr., S.R. Zaki, et al. 2005. Rocky Mountain spotted fever from an unexpected tick vector in Arizona. N. Engl. J. Med. 353:587–594. [DOI] [PubMed] [Google Scholar]
- 28.Rogers, M.E., T. Ilg, A.V. Nikolaev, M.A. Ferguson, and P.A. Bates. 2004. Transmission of cutaneous leishmaniasis by sand flies is enhanced by regurgitation of fPPG. Nature. 430:463–467. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Hodzic, E., J.W. Ijdo, S. Feng, P. Katavolos, W. Sun, C.H. Maretzki, D. Fish, E. Fikrig, S.R. Telford, and S.W. Barthold. 1998. Granulocytic ehrlichiosis in the laboratory mouse. J. Infect. Dis. 177:737–745. [DOI] [PubMed] [Google Scholar]
- 30.IJdo, J.W., C. Wu, S.R. Telford, and E. Fikrig. 2002. Differential expression of the p44 gene family in the agent of human granulocytic ehrlichiosis. Infect. Immun. 70:5295–5298. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Carlyon, J.A., D. Abdel-Latif, M. Pypaert, P. Lacy, and E. Fikrig. 2004. Anaplasma phagocytophilum utilizes multiple host evasion mechanisms to thwart NADPH oxidase-mediated killing during neutrophil infection. Infect. Immun. 72:4772–4783. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Webster, P., J.W. IJdo, L.M. Chicoine, and E. Fikrig. 1998. The agent of Human Granulocytic Ehrlichiosis resides in an endosomal compartment. J. Clin. Invest. 101:1932–1941. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Pal, U., A.M. de Silva, R.R. Montgomery, D. Fish, J. Anguita, J.F. Anderson, Y. Lobet, and E. Fikrig. 2000. Attachment of Borrelia burgdorferi within Ixodes scapularis mediated by outer surface protein A. J. Clin. Invest. 106:561–569. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Pal, U., R.R. Montgomery, D. Lusitani, P. Voet, V. Weynants, S.E. Malawista, Y. Lobet, and E. Fikrig. 2001. Inhibition of Borrelia burgdorferi-tick interactions in vivo by outer surface protein A antibody. J. Immunol. 166:7398–7403. [DOI] [PubMed] [Google Scholar]