Abstract
An unresolved issue in the field of diet and health is if and how changes in meal frequency affect energy metabolism in humans. We therefore evaluated the influence of reduced meal frequency without a reduction in energy intake on glucose metabolism in normal weight healthy male and female subjects. The study was a randomized cross-over design, with 2 eight-week treatment periods (with an intervening 11 week off-diet period) in which subjects consumed all of their calories for weight maintenance distributed in either 3 meals or 1 meal per day (consumed between 17:00 and 21:00). Energy metabolism was evaluated at designated time points throughout the study by performing morning oral glucose tolerance tests (OGTT) and measuring levels of glucose, insulin, glucagon, leptin, ghrelin, adiponectin, resistin and brain-derived neurotrophic factor (BDNF). Subjects consuming 1 meal/d exhibited higher morning fasting plasma glucose levels, greater and more sustained elevations of plasma glucose concentrations and a delayed insulin response in the OGTT compared to subjects consuming 3 meal/d. Levels of ghrelin were elevated in response to the 1 meal/d regimen. Fasting levels of insulin, leptin, ghrelin, adiponectin, resistin and BDNF were not significantly affected by meal frequency. Subjects consuming a single large daily meal exhibit elevated fasting glucose levels, and impaired morning glucose tolerance associated with a delayed insulin response, during a 2 month diet period compared to those consuming 3 meals/day. The impaired glucose tolerance was reversible and was not associated with alterations in the levels of adipokines or BDNF.
Keywords: adiponectin, BDNF, ghrelin, insulin, leptin, meal frequency, resistin
1. Introduction
Glucose intolerance and insulin resistance are prominent features of type 2 diabetes [1], and a more subtle impairment of glucose tolerance may increase the risk of diabetes, cardiovascular disease and stroke [2, 3]. Such “pre-diabetic” states in otherwise healthy individuals are characterized by modest elevations of fasting plasma glucose and insulin levels, and altered temporal profiles of plasma glucose and insulin levels in the oral glucose tolerance test (OGTT) with greater and more sustained elevations of glucose levels and a delayed insulin response [4–6]. Reduced insulin sensitivity of skeletal muscle cells, and decreased responsiveness of pancreatic beta-cells contribute to impaired glucose tolerance [7].
Adipokines are hormones produced by fat cells in response to feeding or fasting, that may play important roles in the development of obesity and diabetes [1]. For example, levels of circulating leptin are increased in obese and diabetic individuals, and leptin resistance in hypothalamic cells that normally suppress food intake likely contributes to overeating in these conditions [8, 9]. Levels of circulating adiponectin are low, and levels of resistin elevated, in obese and insulin resistant individuals [10]. However, the roles of alterations in adipokines in impaired glucose metabolism is unclear. In addition to insulin and adipokines, brain-derived neurotrophic factor (BDNF) has recently been suggested to play a role in glucose metabolism. Studies of BDNF heterozygous knockout mice [11], obese and diabetic animals administered BDNF [12, 13] and humans with type 2 diabetes [14] suggest that BDNF signaling enhances insulin sensitivity. An anti-diabetic action of BDNF in humans is suggested by a recent study that demonstrated an inverse association between fasting plasma BDNF levels and glucose levels, but not insulin levels [14]. However, the effects of variations in dietary energy intake on BDNF levels in humans are unknown.
Intermittent fasts over periods of days have been shown to improve glucose tolerance in obese subjects [15]. Similarly, intermittent feeding and fasting reduces diabetes incidence in rats [16]. Alternate day fasting (a 24 hour fast every other day) improves glucose regulation and indicators of cardiovascular health in mice and rats [17–19]. On the other hand, several epidemiological studies, and short-term (days) intervention experiments, have suggested an association between meal skipping (particularly breakfast) and poor health [20–22]. There is therefore a need for controlled studies that directly compare the effects of different meal frequencies on human health [23], a gap in knowledge identified by the 2005 Dietary Guidelines Advisory Committee Report as a future research direction [24]. Intermittent fasting usually results in an overall reduction in calorie intake in animals [25] and humans [26], raising the question of whether the effects of such diets are the result of caloric restriction rather than fasting. In addition, most studies of dietary energy restriction have been performed on overweight and/or diabetic human or animal subjects. In recent studies, non-obese subjects had an overall reduction in energy intake and lost weight when maintained on an alternate day calorie restriction regimen [27, 28]. We therefore performed a study to determine the effects of reduced meal frequency (1 meal/day) without caloric restriction on health indicators in normal weight, middle-aged male and female subjects.
2. Subjects and Methods
Subjects and Study Design
Details of the subject characteristics and study design have been reported previously [29]. Briefly, the subjects were healthy 40–50 year-old men and women with body mass indexes between 18 and 25 kg/m2 with a usual eating pattern of three-meals-per-day. Study entry was approved by a physician based on medical history, screening blood and urine test results, and a physical examination. The protocol was approved by the Johns Hopkins University Committee on Human Research and the MedStar Research Institute Institutional Review Board. All subjects gave their informed consent and were compensated for their participation in the study. Each subject underwent two 8-week controlled diet periods during which they consumed all of their calories for weight maintenance in either 3 meals/day (breakfast, lunch and dinner) or 1 meal/day (during a 4 hour time period in the early evening; 16:00 – 20:00 hours) in a randomized cross-over design with an 11 week off-diet period between the two controlled diet periods. In the experimental diet, breakfast and lunch food items were substituted for traditional evening meal items; the composition of the diets was reported previously (28). Energy intake was adjusted as necessary to maintain constant body weight during the study.
Glucose tolerance test and measurements of hormone levels
These methods have been described previously [30]. Briefly, all subjects fasted overnight (no food or caloric beverages after 20:00) prior to the oral glucose tolerance test (OGTT), and an initial blood sample was obtained for measurements of fasting glucose, insulin, leptin, ghrelin, adiponectin, resistin and BDNF. The subjects then drank 75 g of glucose in a 300 ml solution (SunDex; Fisherbrand, Pittsburgh, PA) and additional blood samples were obtained at 5, 10, 15, 20, 40, 60, 80, 100 and 120 min for plasma glucose and insulin measurements. Plasma glucose concentrations were measured using a glucose analyzer (Beckman Instruments, Brea, CA). Plasma insulin and resistin concentrations were measured using enzyme-linked immunosorbent assays (ELISA) (Alpco Diagnostics, Salem, NH) with intra-assay variations of 4.8–9.0% and 2.8–3.4% and inter-assay variations of 2.6–3.6% and 5.1–6.9%, respectively. Plasma leptin levels were measured using ELISA (LINCO Research, St. Charles, MO) with intra-assay variations of 1.09–4.98% and inter-assay variations of 3.89–5.33%. Plasma adiponectin levels were measured by radioimmunoassay (RIA) (LINCO) having an intra-assay and an inter-assay variation of 1.78–6.21% and 6.9–9.25%, respectively. Plasma ghrelin levels were measured by RIA (Phoenix Pharmaceuticals, Belmont, CA) with calculated intra-assay and inter-assay variations of 6.7% and 7.8%, respectively. Plasma BDNF levels were measured by ELISA (Promega Cooperation, Medison, WI) with the range of sensitivity from 7.8 to 500 pg/ml and inter-assay assay variation measured at 8.8% (low concentration), 2.9% (medium concentration), and 2.2% (high concentration).
Calculation of insulin sensitivity
We quantified insulin sensitivity by calculating the homeostatic model assessment of insulin resistance (HOMAIR) using fasting plasma glucose and insulin levels [31]. We also calculated the insulin sensitivity index (ISI), metabolic clearance rates (MCR), β-cell function during first phase secretion (β-cell function, first phase) and second phase secretion β-cell function, second phase) (as well as oral glucose- insulin sensitivity (OGIS: 0, 90 [mean of the 80 and 100 min value] and 120) [32–35].
Statistical analysis
A repeated measures analysis of variance (ANOVA) appropriate for a 2 period 2 treatment crossover study, where period was considered a repeated measure, was used to evaluate the effects of meal frequency, when observations were measured prior to the start of each treatment period (baseline) and at the end of the treatment period (the MIXED procedure in SAS, SAS Institute, Cary NC, version 9). Sequence, treatment, and period were included in the model as fixed effects. Subject within sequence was included as a random effect. Period specific baseline values were included as a covariate. When multiple measurements were made during a treatment period (i.e. glucose at 0, 20, 40, 60, 80, 100, 120 min), a similar statistical model was used to evaluate the effects of meal frequency, including time as an additional repeated measures variable. Observation time and the interaction between time and treatment were included as fixed effects in the model. Where the interaction between time and treatment were statistically significant (p < 0.05), within time treatment effects were evaluated. Data are presented as least squares means and SEMs.
3. Results
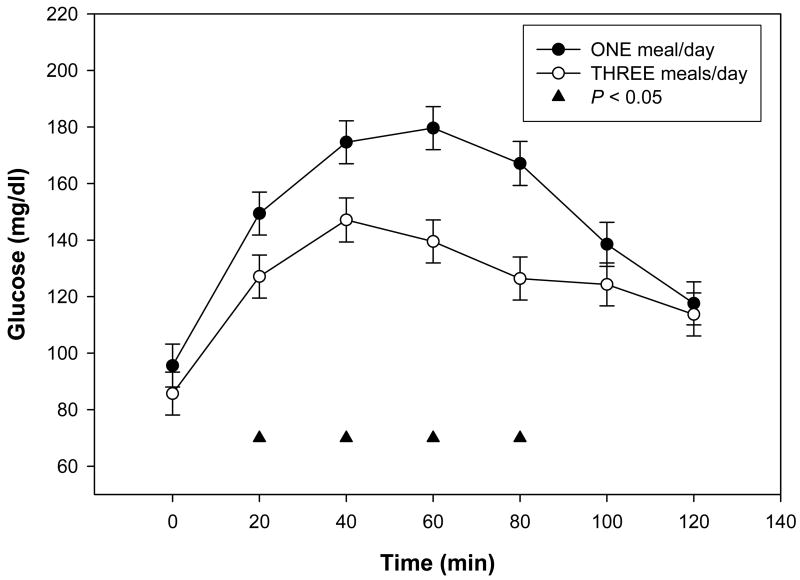
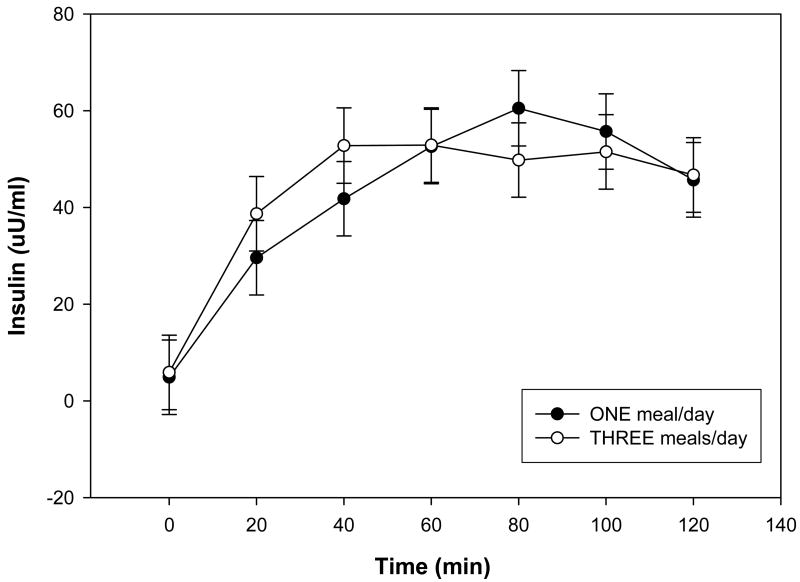
Morning plasma glucose concentrations were significantly greater in subjects when they were consuming 1 meal/d compared to when they were consuming 3 meals/d (Table 1). When consuming 1 meal/d the subjects exhibited poorer glucose tolerance as indicated by a significantly greater and more prolonged elevation of plasma glucose concentrations compared to subjects consuming 3 meals/d diet (Fig. 1). Fasting plasma insulin concentrations were not significantly affected by meal frequency (Table 1), and there were no significant effects of diet on insulin responses to glucose during the OGTT, although there was a trend towards a delayed insulin response when subjects consumed 1 meal/d (Fig. 2).
Table 1.
Biomarkers in subjects when consuming either one meal per day or three meals per day ¹
| One Meal | Three Meals | P2 | |
|---|---|---|---|
| Glucose mg/dl | 95.9 ± 1.7 | 85.4 ± 1.7 | 0.0002 |
| Insulin μU/ml | 5.0 ± 0.7 | 5.8 ± 0.7 | 0.4329 |
| Glucagon pg/ml | 66.5 ± 7.7 | 62.1 ± 7.4 | 0.6878 |
| HOMA-IR | 1.2 ± 0.2 | 1.3 ± 0.2 | 0.8718 |
| OGIS | 403.4 ± 14.0 | 458.8 ± 13.9 | 0.0114 |
| ISI | 0.1 ± 0.004 | 0.1 ± 0.004 | 0.6552 |
| MCR | 8.8 ± 0.3 | 9.2 ± 0.3 | 0.4011 |
| β-cell function 1st phase | 782.1 ± 66.0 | 1013.85 ± 66.1 | 0.0209 |
| β-cell function 2nd phase | 239.0 ± 19.0 | 253.7 ± 19.0 | 0.5894 |
| Adiponectin pg/ml | 13.5 ± 1.3 | 13.5 ± 1.3 | 0.9919 |
| Resistin ng/ml | 3.1 ± 0.3 | 2.8 ± 0.2 | 0.4147 |
| Leptin ng/ml | 20.2 ± 2.2 | 16.1 ± 2.1 | 0.18 |
| Ghrelin pg/ml | 163.2 ± 12.8 | 158.4 ± 12.8 | 0.7942 |
| BDNF ng/ml | 141.7 ± 26.7 | 148.1 ± 26.6 | 0.8175 |
Data are presented as least squares means ± SEM, n = 15 (10 women, 5 men).
P value for the comparison of one meal versus three meals.
Figure 1.
Plasma glucose concentrations during the oral glucose tolerance tests during each study period. Data are presented as least squares means ± SEM, n = 15 (10 women, 5 men), from a repeated measures ANOVA. There was a significant treatment effect between the 1 meal/d (●) and the 3 meals/d (○) for the OGTT at 20 min, 40 min, 60 min and 80 min, ▲ P < 0.05.
Figure 2.
Plasma insulin concentrations during the oral glucose tolerance tests during each study period. Data are presented as least squares means ± SEM, n = 15 (10 women, 5 men), from a repeated measures ANOVA. There was no significant treatment effect between the 1 meal/d (●) and the 3 meals/d (○) for plasma insulin concentrations during the oral glucose tolerance test.
There were no significant effects of meal frequency on HOMA-IR, ISI or MCR (Table 1). However, the OGIS values were significantly lower in subjects when on 1 meal/day compared to 3 meals/day (Table 1). In addition, when on 1 meal/d the values for the 1st phase of β-cell function were significantly lower than the value when on 3 meals/d (Table 1). Values for the 2nd phase of β-cell function were not significantly affected by diet.
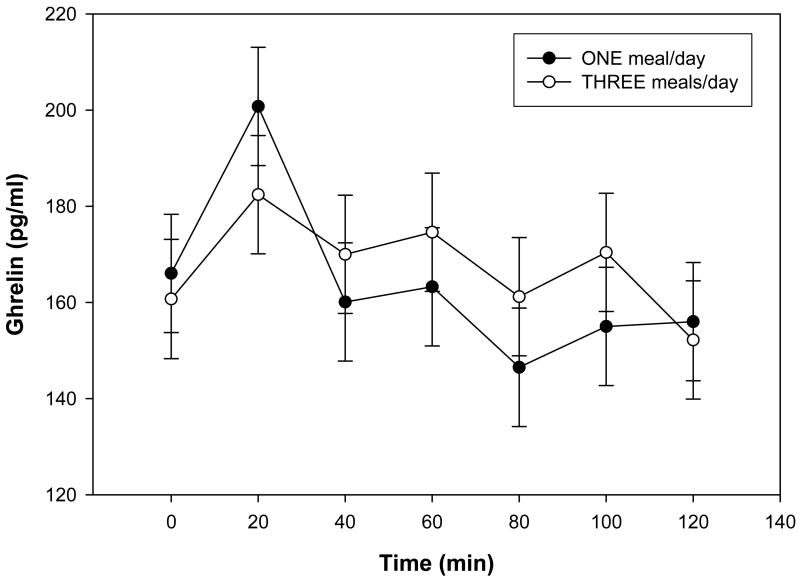
In order to further elucidate the effects of meal frequency without caloric restriction on energy metabolism, we measured fasting levels of several adipokines that are known to play important roles in regulating energy balance. The fasting plasma ghrelin concentration similar in subjects when on 1 meal/d or 3 meals/d (Table 1). There were no significant effects of diet on plasma ghrelin concentrations during the OGTT, although levels tended to be lower in subjects when on 1 meal/d for time points between 40 and 100 min after the glucose ingestion (Fig. 3). Diet had no significant effects on morning plasma concentrations of glucagon, leptin, adiponectin, resistin and BDNF (Table 1).
Figure 3.
Plasma ghrelin concentrations during the oral glucose tolerance tests during each study period. Data are presented as least squares means ± SEM, n = 15 (10 women, 5 men), from a repeated measures ANOVA. There was no significant treatment effect between the 1 meal/d (●) and the 3 meals/d (○) for plasma ghrelin concentrations during the oral glucose tolerance test.
4. Discussion
This controlled randomized dietary intervention study is among the first to evaluate the effects of meal frequency on glucose regulation in normal weight, middle-aged men and women. Each subject consumed the same amount of calories each day regardless of whether they ate one or three meals, and all subjects maintained their body weight within 2 kg of their initial weight throughout the 6 month period [29]. Most physiological variables measured, including heart rate, body temperature and blood chemicals, were unaffected by meal frequency; however, when on 1 meal/d, subjects exhibited: a significant reduction of fat mass, and significant increases in levels of total and LDL and HDL cholesterol [29]. . In the present study morning glucose tolerance was impaired when subjects were consuming 1 meal/day compared to 3 meals/day. Fasting (morning) plasma glucose levels were significantly elevated in subjects when they were consuming 1 meal/d compared to 3 meals/d. The latter difference in fasting glucose levels could be explained, in part, by continuing absorption of the greater amount of food consumed in the evening in the subjects on the 1 meal/d diet. Other studies have suggested an adverse effect of meal-skipping diets on insulin sensitivity [20–22]; however, these studies were either epidemiological (with inherent confounds) or involved very short-term (days) changes in diet. Whether the effect of the 1 meal/d diet on glucose tolerance would persist, exacerbate or resolve over time beyond the 2 month experimental diet period of our study is an important question relevant to long-term effects of the diet. However, we did find that the effect of the 1 meal/d diet on glucose tolerance was rapidly reversed upon return to the 3 meal/d diet, indicating that the diet had no long-lasting effect on glucose metabolism.
The cause of the impaired morning glucose tolerance in subjects consuming 1 meal/d compared to 3 meals/d is unclear. Fasting insulin, leptin and glucagon concentrations have been reported to be elevated in subjects with impaired glucose tolerance [36, 37]. However, there were no significant effects of diet on concentrations of the latter hormones in the present study. Similarly, there were no significant effects of meal frequency on plasma levels of ghrelin, adiponectin, resistin or BDNF. Thus, although fasting insulin and adipokine levels were not different between the two diet groups, insulin sensitivity was apparently decreased in subjects when consuming 1 meal/d. Indeed, the values for OGIS and 1st phase β-cell function were significantly lower in the subjects when they were consuming 1 meal/d compared to baseline, 3 meals/day and off-diet values. The latter results suggest a relative impairment of insulin sensitivity and pancreatic β-cell insulin responses in subjects on 1 meal/d compared to 3 meals/day.
The OGTTs were performed in the morning. Therefore, when on the 1 meal/d diet the subjects had consumed a much greater amount of food in proximity to the OGTT compared to subjects on 3 meals/d, which could have influenced morning insulin sensitivity. Moreover, circadian variations in glucose tolerance have been documented with tolerance being best in the morning [38]. When not accustomed to a morning meal, and then subjected to a morning OGTT, the subjects eating 1 meal/d may therefore exhibit poorer glucose tolerance compared to those adapted to eating breakfast. However, the current manner of eating by westernized society of consuming the largest meal in the evening would appear to be a maladaptive life-style.
Our findings show that consumption of one unusually large meal per day worsens morning glucose tolerance compared to an isocaloric diet spread across three meals. However, when on 1 meal/d the subjects would have eaten less than those on 3 meals/day if we had not asked them to consume the same amount of food that they normally eat on a 3 meal/d schedule. When rodents are subjected to an alternate day fasting regimen, their overall calorie intake is decreased by 10–30% and they maintain a lower body weight than animals on an ad libitum control diet, and exhibit increased insulin sensitivity and decreased blood pressure [11, 25]. Similarly, when maintained on an alternate day calorie restriction diet over a 2 month period, human subjects lost weight and exhibited improved cardiovascular disease and diabetes risk profiles [28]. In the latter study the subjects ate only 400–500 calories on CR days, which resulted in a reduction in plasma leptin levels and an elevation of β-hydroxybutyrate levels only on the CR days, but sustained decreases in plasma insulin levels suggesting improved insulin sensitivity. Collectively, the available data therefore suggest that meal skipping or intermittent CR diets can result in health benefits including improved glucose regulation, but only if there is an overall reduction in energy intake.
Acknowledgments
This research was supported by the Intramural Research Program of the National Institute on Aging. We thank K. Spears for technical assistance.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- 1.Arner P. Insulin resistance in type 2 diabetes – role of the adipokines. Curr Mol Med. 2005;5:333–339. doi: 10.2174/1566524053766022. [DOI] [PubMed] [Google Scholar]
- 2.Aygencel G, Karamercan A, Akinci E, Demircan A, Akeles A. Metabolic syndrome and its association with ischemic cerebrovascular disease. Adv Ther. 2006;23:495–501. doi: 10.1007/BF02850171. [DOI] [PubMed] [Google Scholar]
- 3.Schwartz EA, Reaven PD. Molecular and signaling mechanisms of atherosclerosis in insulin resistance. Endocrinol Metab Clin North Am. 2006;35:525–549. doi: 10.1016/j.ecl.2006.06.005. [DOI] [PubMed] [Google Scholar]
- 4.Efendic S, Grill V, Luft R, Wajngot A. Low insulin response: a marker of prediabetes. Adv Exp Med Biol. 1988;246:167–174. doi: 10.1007/978-1-4684-5616-5_20. [DOI] [PubMed] [Google Scholar]
- 5.Kanauchi M, Kawano T, Kanauchi K, Saito Y. New “pre-diabetes” category and the metabolic syndrome in Japanese. Horm Metab Res. 2005;37:622–626. doi: 10.1055/s-2005-870537. [DOI] [PubMed] [Google Scholar]
- 6.Osei K, Rhinesmith S, Gaillard T, Schuster D. Impaired insulin sensitivity, insulin secretion, and glucose effectiveness predict future development of impaired glucose tolerance and type 2 diabetes in pre-diabetic African Americans: implications for primary diabetes prevention. Diabetes Care. 2004;27:1439–1446. doi: 10.2337/diacare.27.6.1439. [DOI] [PubMed] [Google Scholar]
- 7.Haring HU, Mehnert H. Pathogenesis of type 2 (non-insulin-dependent) diabetes mellitus: candidates for a signal transmitter defect causing insulin resistance of the skeletal muscle. Diabetologia. 1993;36:176–182. doi: 10.1007/BF00399946. [DOI] [PubMed] [Google Scholar]
- 8.Hamann A, Matthaei S. Regulation of energy balance by leptin. Exp Clin Endocrinol Diabetes. 1996;104:293–300. doi: 10.1055/s-0029-1211457. [DOI] [PubMed] [Google Scholar]
- 9.Munzberg H, Myers MG., Jr Molecular and anatomical determinants of central leptin resistance. Nat Neurosci. 2005;8:566–570. doi: 10.1038/nn1454. [DOI] [PubMed] [Google Scholar]
- 10.Koerner A, Kratzsch J, Kiess W. Adipocytokines: leptin--the classical, resistin--the controversical, adiponectin--the promising, and more to come. Best Pract Res Clin Endocrinol Metab. 2005;19:525–546. doi: 10.1016/j.beem.2005.07.008. [DOI] [PubMed] [Google Scholar]
- 11.Duan W, Guo Z, Jiang H, Ware M, Mattson MP. Reversal of behavioral and metabolic abnormalities, and insulin resistance syndrome, by dietary restriction in mice deficient in brain-derived neurotrophic factor. Endocrinology. 2003;144:2446–2453. doi: 10.1210/en.2002-0113. [DOI] [PubMed] [Google Scholar]
- 12.Nakagawa T, Ogawa Y, Ebihara K, Yamanaka M, Tsuchida A, Taiji M, Noguchi H, Nakao K. Anti-obesity and anti-diabetic effects of brain-derived neurotrophic factor in rodent models of leptin resistance. Int J Obes Relat Metab Disord. 2003;27:557–565. doi: 10.1038/sj.ijo.0802265. [DOI] [PubMed] [Google Scholar]
- 13.Ono M, Itakura Y, Nonomura T, Nakagawa T, Nakayama C, Taiji M, Noguchi H. Intermittent administration of brain-derived neurotrophic factor ameliorates glucose metabolism in obese diabetic mice. Metabolism. 2000;49:129–133. doi: 10.1016/s0026-0495(00)90988-0. [DOI] [PubMed] [Google Scholar]
- 14.Krabbe KS, Nielsen AR, Krogh-Madsen R, Plomgaard P, Rasmussen P, Erikstrup C, Fischer CP, Lindegaard B, Petersen AM, Taudorf S, Secher NH, Pilegaard H, Bruunsgaard H, Pedersen BK. Brain-derived neurotrophic factor (BDNF) and type 2 diabetes. Diabetologia. 2007;50:431–438. doi: 10.1007/s00125-006-0537-4. [DOI] [PubMed] [Google Scholar]
- 15.Schless GL, Duncan GG. The beneficial effect of intermittent total fasts on the glucose tolerance in obese diabetic patients. Metabolism. 1966;15:98–102. doi: 10.1016/0026-0495(66)90029-1. [DOI] [PubMed] [Google Scholar]
- 16.Pedersen CR, Hagemann I, Bock T, Buschard K. Intermittent feeding and fasting reduces diabetes incidence in BB rats. Autoimmunity. 1999;30:243–250. doi: 10.3109/08916939908993805. [DOI] [PubMed] [Google Scholar]
- 17.Anson RM, Guo Z, de Cabo R, Iyun T, Rios M, Hagepanos A, Ingram DK, Lane MA, Mattson MP. Intermittent fasting dissociates beneficial effects of dietary restriction on glucose metabolism and neuronal resistance to injury from calorie intake. Proc Natl Acad Sci USA. 2003;100:6216–6220. doi: 10.1073/pnas.1035720100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Mattson MP, Wan R. Beneficial effects of intermittent fasting and caloric restriction on the cardiovascular and cerebrovascular systems. J Nutr Biochem. 2005;16:129–137. doi: 10.1016/j.jnutbio.2004.12.007. [DOI] [PubMed] [Google Scholar]
- 19.Mager DE, Wan R, Brown M, Cheng A, Wareski P, Abernethy DR, Mattson MP. Caloric restriction and intermittent fasting alter spectral measures of heart rate and blood pressure variability in rats. FASEB J. 2006;20:631–637. doi: 10.1096/fj.05-5263com. [DOI] [PubMed] [Google Scholar]
- 20.Jenkins DJ, Wolever TM, Vuksan V, et al. Nibbling versus gorging: metabolic advantages of increased meal frequency. N Engl J Med. 1989;321:929–934. doi: 10.1056/NEJM198910053211403. [DOI] [PubMed] [Google Scholar]
- 21.Keski-Rahkonen A, Kaprio J, Rissanen A, Virkkunen M, Rose RJ. Breakfast skipping and health-compromising behaviors in adolescents and adults. Eur J Clin Nutr. 2003;57:842–853. doi: 10.1038/sj.ejcn.1601618. [DOI] [PubMed] [Google Scholar]
- 22.Farshchi HR, Taylor MA, Macdonald IA. Deleterious effects of omitting breakfast on insulin sensitivity and fasting lipid profiles in healthy lean women. Am J Clin Nutr. 2005;81:388–396. doi: 10.1093/ajcn.81.2.388. [DOI] [PubMed] [Google Scholar]
- 23.Mattson MP. The need for controlled studies of the effects of meal frequency on health. Lancet. 2005;365:1978–1980. doi: 10.1016/S0140-6736(05)66667-6. [DOI] [PubMed] [Google Scholar]
- 24.Nutrition and Your Health:Dietary Guidelines for Americans, 2005 Dietary Guidelines Advisory Committee Report, Research Recommendations. 2005 http://www.health.gov/DietaryGuidelines/dga2005/report/HTML/F_ResearchRec.htm.
- 25.Wan R, Camandola S, Mattson MP. Intermittent food deprivation improves cardiovascular and neuroendocrine responses to stress in rats. J Nutr. 2003;133:1921–1929. doi: 10.1093/jn/133.6.1921. [DOI] [PubMed] [Google Scholar]
- 26.Martin A, Normand S, Sothier M, Peyrat J, Louche-Pelissier C, Laville M. Is advice for breakfast consumption justified? Results from a short-term dietary and metabolic experiment in young healthy men. Br J Nutr. 2000;84:337–344. doi: 10.1017/s0007114500001616. [DOI] [PubMed] [Google Scholar]
- 27.Heilbronn LK, Smith SR, Martin CK, Anton SD, Ravussin E. Alternate-day fasting in nonobese subjects: effects on body weight, body composition, and energy metabolism. Am J Clin Nutr. 2005;81:69–73. doi: 10.1093/ajcn/81.1.69. [DOI] [PubMed] [Google Scholar]
- 28.Johnson JB, Summer W, Cutler RG, Martin B, Hyun DH, Dixit VD, Pearson M, Nasar M, Tellijohan R, Maudsley S, Carlson O, John S, Laub DR, Mattson MP. Alternate day calorie restriction improves clinical findings and reduces markers of oxidative stress and inflammation in overweight adults with moderate asthma. Free Rad Biol Med. 2007;42:665–674. doi: 10.1016/j.freeradbiomed.2006.12.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Stote KS, Baer DJ, Spears K, Paul DR, Harris GK, Rumpler WV, Strycular P, Najjar SS, Ferrucci L, Ingram DK, Longo DL, Mattson MP. A controlled trial of reduced meal frequency without caloric restriction in healthy, normal weight middle-aged men and women. Am J Clin Nutr. 2007;85:981–988. doi: 10.1093/ajcn/85.4.981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Kim BJ, Carlson OD, Jang HJ, Elahi D, Berry C, Egan JM. Peptide YY is secreted after oral glucose administration in a gender-specific manner. J Clin Endocrinol Metab. 2005;90:6665–6671. doi: 10.1210/jc.2005-0409. [DOI] [PubMed] [Google Scholar]
- 31.Matthews DR, Hosker JP, Rudenski AS, Naylor BA, Treacher DF, Turner RC. Homeostasis model assessment: insulin resistance and beta-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia. 1985;28:412–419. doi: 10.1007/BF00280883. [DOI] [PubMed] [Google Scholar]
- 32.Kanauchi M, Yamano S, Kanauchi K, Saito Y. Homeostasis model assessment of insulin resistance, quantitative insulin sensitivity check index, and oral glucose insulin sensitivity index in nonobese, nondiabetic subjects with high-normal blood pressure. J Clin Endocrinol Metab. 2003;88:3444–3446. doi: 10.1210/jc.2002-021641. [DOI] [PubMed] [Google Scholar]
- 33.Theodorakis MJ, Muller DC, Carlson O, Egan JM. Assessment of insulin sensitivity and secretion indices from oral glucose tolerance testing in subjects with fasting euglycemia but impaired 2-hour plasma glucose. Metabolism. 2003;52:1523–1524. doi: 10.1016/j.metabol.2003.08.001. [DOI] [PubMed] [Google Scholar]
- 34.Stumvoll M, Mitrakou A, Pimenta W, et al. Use of the oral glucose tolerance test to assess insulin release and insulin sensitivity. Diabetes Care. 2000;23:295–301. doi: 10.2337/diacare.23.3.295. [DOI] [PubMed] [Google Scholar]
- 35.Stumvoll M, Van Haeften T, Fritsche A, Gerich J. Oral glucose tolerance test indexes for insulin sensitivity and secretion based on various availabilities of sampling times. Diabetes Care. 2001;24:796–797. doi: 10.2337/diacare.24.4.796. [DOI] [PubMed] [Google Scholar]
- 36.Segal KR, Landt M, Klein S. Relationship between insulin sensitivity and plasma leptin concentration in lean and obese men. Diabetes. 1996;45:988–991. doi: 10.2337/diab.45.7.988. [DOI] [PubMed] [Google Scholar]
- 37.Henkel E, Menschikowski M, Koehler C, Leonhardt W, Hanefeld M. Impact of glucagon response on postprandial hyperglycemia in men with impaired glucose tolerance and type 2 diabetes mellitus. Metabolism. 2005;54:1168–1173. doi: 10.1016/j.metabol.2005.03.024. [DOI] [PubMed] [Google Scholar]
- 38.Morgan L, Hampton S, Gibbs M, Arendt J. Circadian aspects of postprandial metabolism. Chronobiol Int. 2003;20:795–808. doi: 10.1081/cbi-120024218. [DOI] [PubMed] [Google Scholar]