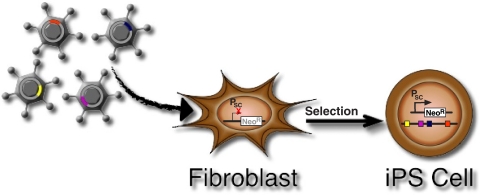
Last year a paper appeared in Cell that caused a great degree of excitement in the scientific community. Takahashi and Yamanaka (2006) described a system where murine fibroblasts, virally transduced with as few as four stem cell factors (Oct3/4, Sox2, c-Myc, and Klf4), reverted to an embryonic stem (ES) cell-like phenotype when placed in selective culture conditions. Last month, the same team extended this work, this time showing transduced cells selected for reactivation of the homeoprotein Nanog were even more similar to ES cells than before, and capable of germline transmission (Okita et al., 2007). Two other teams, those of Konrad Hochedlinger (Maherali et al., 2007) and Rudolf Jaenisch (Wernig et al., 2007), concurrently demonstrated the remarkable capacity of this new approach. Figure 1 illustrates the general experimental design used by all of these reports.
Fig. 1.
Reprogramming the differentiated state by viral transduction. The approach initially outlined by Takahashi and Yamananaka (2006) involves the transduction of fibroblast populations (both embryonic and adult) with four pluripotent stem cell factors Klf4, Oct3/4, Sox2, and c-Myc. Transduced cells are then placed under neomycin selection for reactivation of a stem cell-specific promoter (PSC), such as Fbx15, Oct3/4, or Nanog. Resultant colonies were subsequently analyzed functionally, genetically, and epigenetically to assess to what degree they behave like bona fide ES cells.
Taken together, the data presented by these four papers show the transduced cells display characteristics strikingly similar to ES cells in every assay to which they were subjected. Table 1 highlights the analyses used in each study. These cells reactivate pluripotency genes and present gene expression profiles strikingly similar to bona fide ES cells. Furthermore, epigenetic analysis showed the cells to have DNA methylation and histone modification patterns virtually indistinguishable from ES cells, both globally and at loci associated with pluripotency. Importantly, this also includes analysis at imprinted loci, suggesting that the cells being reprogrammed are not contaminating germ cells. Perhaps most significantly all three groups have subjected their cells to a battery of functional assays for developmental potential. These include formation of the three germ layers in embryoid bodies and teratomas, high chimeric contribution, and, crucially, germ line transmission, including live, fertile mice obtained by Yamanaka's group. Two additional experiments bolster this evidence of pluripotency: first, Hochedlinger's group has shown cells have the capacity to reprogram somatic cells by cell fusion, and second, Jaenisch's group presented evidence of mid-gestation tetraploid complementation embryos derived from these cells, and hence were named ‘induced Pluripotent Stem (iPS) cells’.
Table 1.
Summary of published iPS papers
| Paper (Group) | Takahashi and Yamanaka (2006) (Yamanaka) | Okita et al. (2007) (Yamanaka) | Wernig et al. (2007) (Jaenisch) | Maherali et al. (2007) (Hochedlinger) |
|---|---|---|---|---|
| Selection | Fbx15 | Nanog | Oct3/4, Nanog | Oct3/4, Nanog |
| Cell type(s) | MEF, TTF | MEF | MEF, TTF | MEF, TTF |
| Frequency (iPS/fibroblasts) | 0.01%–0.5% | 0.001%–0.03% | Oct3/4: 0.08% Nanog: 0.005% | ND |
| Reprograming assays | ||||
| Functional | ||||
| Chimera contribution | +/−1 | + | + | + |
| Teratoma formation | + | + | + | + |
| Germline contribution | From MEF | From MEF | From TTF | |
| Cell fusion | + | |||
| Genetic/epigenetic | ||||
| Gene expression analysis | + | + | + | + |
| Gene-specific DNA methylation | + | + | + | + |
| Genome-wide DNA methylation | + | + | ||
| Imprinting | + | + | + | |
| Xi reactivation | + | |||
Experimental details of the four iPS papers are shown, in addition to the functional and other assays used to illustrate nuclear reprogramming.
Chimeric contribution limited to midgestation embryos in this study.
MEF, murine embryonic fibroblasts; TTF, tail-tip fibroblasts; ND, not disclosed; iPS, induced pluripotent stem cell.
While the result presented by Hochedlinger's group that iPS cells generated from adult cells can contribute to a chimera's germline is compelling in contrast with the germline-competent reprogramming of embryonic fibroblasts shown by other groups, these adult tail-tip fibroblasts are nonetheless a very heterogeneous population and it remains to be demonstrated whether terminally differentiated cells can be reprogrammed in this fashion, as has been demonstrated with other reprogramming technologies (Tada et al., 2001; Hochedlinger and Jaenisch, 2002; Eggan et al., 2004; Inoue et al., 2005). Furthermore, the continued presence of transgenes encoding oncogenic factors and delivered by oncogenic retroviruses (Hochedlinger et al., 2005; Yamanaka, 2007) raises concerns about the long-term capacity of these cells to support normal development. Notably, of the only live F1 mice from germline iPS chimeras reported in these studies, approximately one-fifth developed tumors, likely due to reactivation of the c-Myc retrovirus (Okita et al., 2007). Perhaps this problem could be addressed by removal of the transgenes after reprogramming or by an inducible system for all four factors, as was carried out by Hochedlinger with Oct3/4 alone.
This second concern is particularly crucial to surmount if this system is to be of use clinically. While the recapitulation of this technology in human cells could address two important challenges currently faced by the stem cell field, the need to avoid immune rejection by creating pluripotent cells genetically identical to a patient and objections of some to the use of surplus preimplantation human embryos, this demonstrated connection to oncogenesis presents for now a significant barrier to such application. Likewise, identifying the appropriate factors and selection regime to produce human iPS cells may require an extensive amount of new work and insight. New methodologies to either deliver or activate these genes will also need to be developed, as suggested by both Yamanaka and Jaenisch, in order to avoid the use of potentially cancer-causing retroviruses.
In addition to describing a new system for generating pluripotent cells, which may be clinically relevant, this work provides major insights into how nuclear reprogramming itself proceeds and as such may be able to inform us about fundamental mechanisms of cellular identity. How amenable is cell identity to being altered in this fashion? Can this approach be generalized to reprogram a variety of different cell types? And, further, can it be used to identify various factors or regiments to regress differentiated states in a stepwise fashion, hence generating progenitors and precursors as well as fully pluripotent cells?
Certainly the advances reported in these studies create a myriad of opportunities for new directions in controlling and manipulating differentiation. It will be interesting to see how this field progresses.
References
- Eggan K. Mice cloned from olfactory sensory neurons. Nature. 2004;428:44–49. doi: 10.1038/nature02375. [DOI] [PubMed] [Google Scholar]
- Hochedlinger K. Monoclonal mice generated by nuclear transfer from mature B and T donor cells. Nature. 2002;415:1035–1038. doi: 10.1038/nature718. [DOI] [PubMed] [Google Scholar]
- Hochedlinger K. Ectopic expression of Oct-4 blocks progenitor-cell differentiation and causes dysplasia in epithelial tissues. Cell. 2005;121:465–477. doi: 10.1016/j.cell.2005.02.018. [DOI] [PubMed] [Google Scholar]
- Inoue K. Generation of cloned mice by direct nuclear transfer from natural killer T cells. Curr Biol. 2005;15:1114–1118. doi: 10.1016/j.cub.2005.05.021. [DOI] [PubMed] [Google Scholar]
- Maherali N. Directly reprogrammed fibroblasts show global epigenetic remodeling and widespread tissue contribution. Cell Stem Cell. 2007;1:55–70. doi: 10.1016/j.stem.2007.05.014. [DOI] [PubMed] [Google Scholar]
- Okita K. Generation of germline-competent induced pluripotent stem cells. Nature. 2007 doi: 10.1038/nature05934. Published online June 6, 2007 10.1038/nature05934. [DOI] [PubMed] [Google Scholar]
- Tada M. Nuclear reprogramming of somatic cells by in vitro hybridization with ES cells. Curr Biol. 2001;11:1553–1558. doi: 10.1016/s0960-9822(01)00459-6. [DOI] [PubMed] [Google Scholar]
- Takahashi K. Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell. 2006;126:663–676. doi: 10.1016/j.cell.2006.07.024. [DOI] [PubMed] [Google Scholar]
- Wernig M. In vitro reprogramming of fibroblasts into a pluripotent ES-cell-like state. Nature. 2007 doi: 10.1038/nature05944. Published online June 6, 2007 10.1038/nature05944. [DOI] [PubMed] [Google Scholar]
- Yamanaka S. Strategies and new developments in the generation of patient-specific pluripotent stem cells. Cell Stem Cell. 2007;1:39–49. doi: 10.1016/j.stem.2007.05.012. [DOI] [PubMed] [Google Scholar]