Abstract
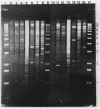
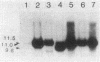
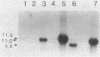
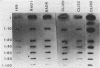
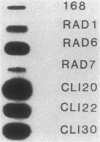
Endogenous chromosomal DNA amplifications with associated tetracycline resistance (Tcr) in Bacillus subtilis were first described by C. R. Wilson and A. E. Morgan (J. Bacteriol. 163:445-453, 1985). We have confirmed and extended their results, and we show that fusion of protoplasts from Tcs B. subtilis 168 trpC2 with polyethylene glycol and regeneration on medium containing 20 micrograms of tetracycline per ml induces Tcr regenerants that contain amplified DNA. This phenomenon appeared to be recE dependent and requires the addition of polyethylene glycol. Along with three regenerants kindly provided by Wilson and Morgan (RAD1, RAD6, and RAD7), we characterized three strains (CLI20, CLI22, CLI30) isolated in this laboratory. All six contain an amplified region of DNA which was independently cloned on plasmid pCIS7. Integration of pCIS7 into the wild-type (Tcs) B. subtilis chromosome and amplification of the plasmid sequences generated a Tcr phenotype, even though the DNA on pCIS7 was cloned from Tcs B. subtilis KS162 (Ives and Bott, J. Bacteriol. 171:1801-1810, 1989). The amplified DNA also showed homology (through hybridization analysis) with pAM alpha 1 delta 1, a gram-positive Tcr plasmid, indicating that B. subtilis normally contains a silent integrated copy of the gene whose amplification confers Tcr. The amplifications were determined to lie between purA and gyrB on the B. subtilis chromosome, and the endpoints were mapped. RAD6 and CLI30 may share the same left-hand endpoint, but the other endpoints are different in each isolate. The amplified DNAs of RAD1, RAD6, CLI20, and CLI30 end near known DNA membrane binding sites. The number of amplified units of DNA was determined through dot blot analysis to do approximately 80 to 100 copies per cell, with corresponding increases in transcription of RAD1, RAD6, CLI20, CLI22, and CLI30.
Full text
PDF








Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Albertini A. M., Galizzi A. Amplification of a chromosomal region in Bacillus subtilis. J Bacteriol. 1985 Jun;162(3):1203–1211. doi: 10.1128/jb.162.3.1203-1211.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Anderson P., Roth J. Spontaneous tandem genetic duplications in Salmonella typhimurium arise by unequal recombination between rRNA (rrn) cistrons. Proc Natl Acad Sci U S A. 1981 May;78(5):3113–3117. doi: 10.1073/pnas.78.5.3113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Anderson R. P., Roth J. R. Tandem genetic duplications in phage and bacteria. Annu Rev Microbiol. 1977;31:473–505. doi: 10.1146/annurev.mi.31.100177.002353. [DOI] [PubMed] [Google Scholar]
- Chai T. J., Foulds J. Isolation and partial characterization of protein E, a major protein found in certain Escherichia coli K-12 mutant strains: relationship to other outer membrane proteins. J Bacteriol. 1979 Aug;139(2):418–423. doi: 10.1128/jb.139.2.418-423.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chang S., Cohen S. N. High frequency transformation of Bacillus subtilis protoplasts by plasmid DNA. Mol Gen Genet. 1979 Jan 5;168(1):111–115. doi: 10.1007/BF00267940. [DOI] [PubMed] [Google Scholar]
- Clewell D. B., Yagi Y., Bauer B. Plasmid-determined tetracycline resistance in Streptococcus faecalis: evidence for gene amplification during growth in presence of tetracycline. Proc Natl Acad Sci U S A. 1975 May;72(5):1720–1724. doi: 10.1073/pnas.72.5.1720. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Deretic V., Tomasek P., Darzins A., Chakrabarty A. M. Gene amplification induces mucoid phenotype in rec-2 Pseudomonas aeruginosa exposed to kanamycin. J Bacteriol. 1986 Feb;165(2):510–516. doi: 10.1128/jb.165.2.510-516.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eccles S., Docherty A., Chopra I., Shales S., Ball P. Tetracycline resistance genes from Bacillus plasmid pAB124 confer decreased accumulation of the antibiotic in Bacillus subtilis but not in Escherichia coli. J Bacteriol. 1981 Mar;145(3):1417–1420. doi: 10.1128/jb.145.3.1417-1420.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Edlund T., Normark S. Recombination between short DNA homologies causes tandem duplication. Nature. 1981 Jul 16;292(5820):269–271. doi: 10.1038/292269a0. [DOI] [PubMed] [Google Scholar]
- Feinberg A. P., Vogelstein B. A technique for radiolabeling DNA restriction endonuclease fragments to high specific activity. Anal Biochem. 1983 Jul 1;132(1):6–13. doi: 10.1016/0003-2697(83)90418-9. [DOI] [PubMed] [Google Scholar]
- Hasegawa M., Hintermann G., Simonet J. M., Crameri R., Piret J., Hütter R. Certain chromosomal regions in Streptomyces glaucescens tend to carry amplifications and deletions. Mol Gen Genet. 1985;200(3):375–384. doi: 10.1007/BF00425720. [DOI] [PubMed] [Google Scholar]
- Hashiguchi K., Tanimoto A., Nomura S., Yamane K., Yoda K., Harada S., Mori M., Furusato T., Takatsuki A., Yamasaki M. Amplification of the amyE-tmrB region on the chromosome in tunicamycin-resistant cells of Bacillus subtilis. Mol Gen Genet. 1986 Jul;204(1):36–43. doi: 10.1007/BF00330184. [DOI] [PubMed] [Google Scholar]
- Ives C. L., Bott K. F. Cloned Bacillus subtilis chromosomal DNA mediates tetracycline resistance when present in multiple copies. J Bacteriol. 1989 Apr;171(4):1801–1810. doi: 10.1128/jb.171.4.1801-1810.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jannière L., Niaudet B., Pierre E., Ehrlich S. D. Stable gene amplification in the chromosome of Bacillus subtilis. Gene. 1985;40(1):47–55. doi: 10.1016/0378-1119(85)90023-x. [DOI] [PubMed] [Google Scholar]
- Long E. O., Dawid I. B. Repeated genes in eukaryotes. Annu Rev Biochem. 1980;49:727–764. doi: 10.1146/annurev.bi.49.070180.003455. [DOI] [PubMed] [Google Scholar]
- Mori M., Tanimoto A., Yoda K., Harada S., Koyama N., Hashiguchi K., Obinata M., Yamasaki M., Tamura G. Essential structure in the cloned transforming DNA that induces gene amplification of the Bacillus subtilis amyE-tmrB region. J Bacteriol. 1986 Jun;166(3):787–794. doi: 10.1128/jb.166.3.787-794.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Perkins J. B., Youngman P. Streptococcus plasmid pAM alpha 1 is a composite of two separable replicons, one of which is closely related to Bacillus plasmid pBC16. J Bacteriol. 1983 Aug;155(2):607–615. doi: 10.1128/jb.155.2.607-615.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Peterson B. C., Rownd R. H. Drug resistance gene amplification of plasmid NR1 derivatives with various amounts of resistance determinant DNA. J Bacteriol. 1985 Mar;161(3):1042–1048. doi: 10.1128/jb.161.3.1042-1048.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Piggot P. J., Curtis C. A. Analysis of the regulation of gene expression during Bacillus subtilis sporulation by manipulation of the copy number of spo-lacZ fusions. J Bacteriol. 1987 Mar;169(3):1260–1266. doi: 10.1128/jb.169.3.1260-1266.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- SAITO H., MIURA K. I. PREPARATION OF TRANSFORMING DEOXYRIBONUCLEIC ACID BY PHENOL TREATMENT. Biochim Biophys Acta. 1963 Aug 20;72:619–629. [PubMed] [Google Scholar]
- Sanchez-Rivas C., Lévi-Meyrueis C., Lazard-Monier F., Schaeffer P. Diploid state of phenotypically recombinant progeny arising after protoplast fusion in Bacillus subtilis. Mol Gen Genet. 1982;188(2):272–278. doi: 10.1007/BF00332687. [DOI] [PubMed] [Google Scholar]
- Sandman K., Losick R., Youngman P. Genetic analysis of Bacillus subtilis spo mutations generated by Tn917-mediated insertional mutagenesis. Genetics. 1987 Dec;117(4):603–617. doi: 10.1093/genetics/117.4.603. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sargent M. G., Bennett M. F. Amplification of a major membrane-bound DNA sequence of Bacillus subtilis. J Bacteriol. 1985 Feb;161(2):589–595. doi: 10.1128/jb.161.2.589-595.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sargent M. G., Bennett M. F., Burdett I. D. Identification of specific restriction fragments associated with a membrane subparticle from Bacillus subtilis. J Bacteriol. 1983 Jun;154(3):1389–1396. doi: 10.1128/jb.154.3.1389-1396.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schaeffer P., Cami B., Hotchkiss R. D. Fusion of bacterial protoplasts. Proc Natl Acad Sci U S A. 1976 Jun;73(6):2151–2155. doi: 10.1073/pnas.73.6.2151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schimke R. T., Alt F. W., Kellems R. E., Kaufman R. J., Bertino J. R. Amplification of dihydrofolate reductase genes in methotrexate-resistant cultured mouse cells. Cold Spring Harb Symp Quant Biol. 1978;42(Pt 2):649–657. doi: 10.1101/sqb.1978.042.01.067. [DOI] [PubMed] [Google Scholar]
- Schwartz D. C., Cantor C. R. Separation of yeast chromosome-sized DNAs by pulsed field gradient gel electrophoresis. Cell. 1984 May;37(1):67–75. doi: 10.1016/0092-8674(84)90301-5. [DOI] [PubMed] [Google Scholar]
- Smith G. E., Summers M. D. The bidirectional transfer of DNA and RNA to nitrocellulose or diazobenzyloxymethyl-paper. Anal Biochem. 1980 Nov 15;109(1):123–129. doi: 10.1016/0003-2697(80)90019-6. [DOI] [PubMed] [Google Scholar]
- Speer B. S., Salyers A. A. Characterization of a novel tetracycline resistance that functions only in aerobically grown Escherichia coli. J Bacteriol. 1988 Apr;170(4):1423–1429. doi: 10.1128/jb.170.4.1423-1429.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Spradling A. C. The organization and amplification of two chromosomal domains containing Drosophila chorion genes. Cell. 1981 Nov;27(1 Pt 2):193–201. doi: 10.1016/0092-8674(81)90373-1. [DOI] [PubMed] [Google Scholar]
- Williams G., Smith I. Chromosomal mutations causing resistance to tetracycline in Bacillus subtilis. Mol Gen Genet. 1979;177(1):23–29. doi: 10.1007/BF00267249. [DOI] [PubMed] [Google Scholar]
- Wilson C. R., Morgan A. E. Chromosomal-DNA amplification in Bacillus subtilis. J Bacteriol. 1985 Aug;163(2):445–453. doi: 10.1128/jb.163.2.445-453.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Winston S., Sueoka N. DNA-membrane association is necessary for initiation of chromosomal and plasmid replication in Bacillus subtilis. Proc Natl Acad Sci U S A. 1980 May;77(5):2834–2838. doi: 10.1073/pnas.77.5.2834. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yagi Y., Clewell D. B. Identification and characterization of a small sequence located at two sites on the amplifiable tetracycline resistance plasmid pAMalpha1 in Streptococcus faecalis. J Bacteriol. 1977 Jan;129(1):400–406. doi: 10.1128/jb.129.1.400-406.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Young M. Gene amplification in Bacillus subtilis. J Gen Microbiol. 1984 Jul;130(7):1613–1621. doi: 10.1099/00221287-130-7-1613. [DOI] [PubMed] [Google Scholar]
- Youngman P., Perkins J. B., Losick R. A novel method for the rapid cloning in Escherichia coli of Bacillus subtilis chromosomal DNA adjacent to Tn917 insertions. Mol Gen Genet. 1984;195(3):424–433. doi: 10.1007/BF00341443. [DOI] [PubMed] [Google Scholar]