Abstract
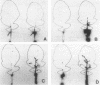
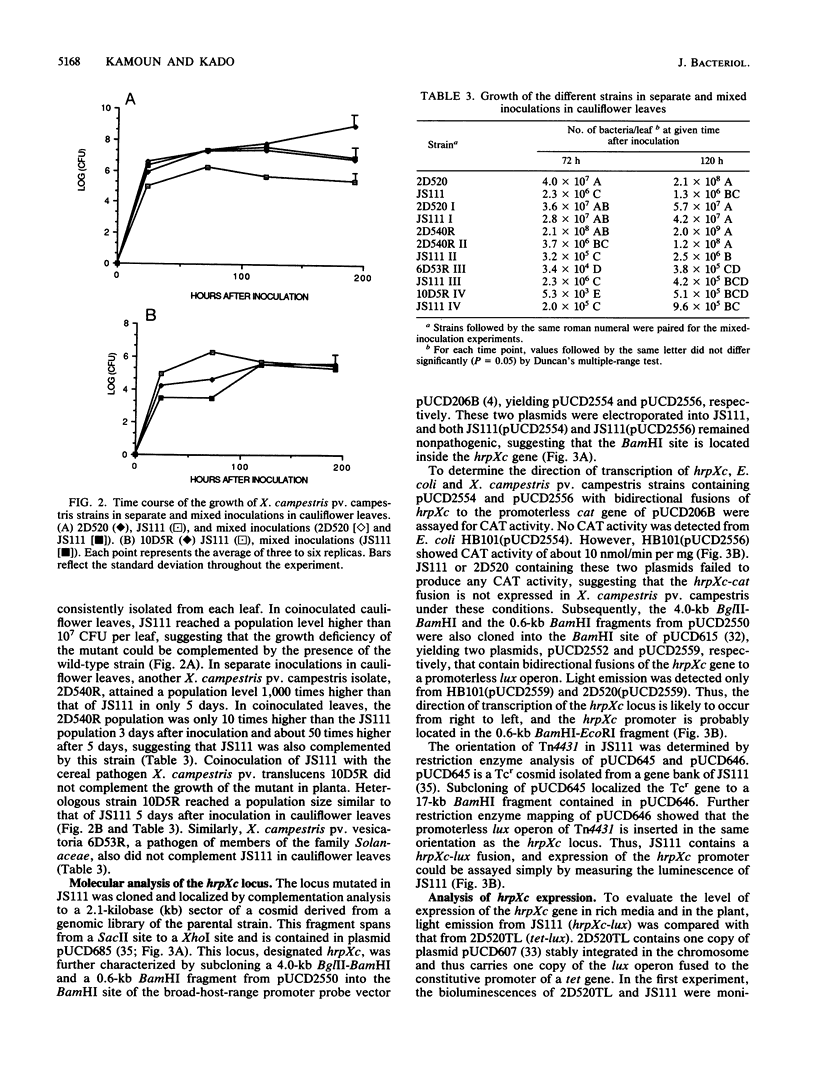
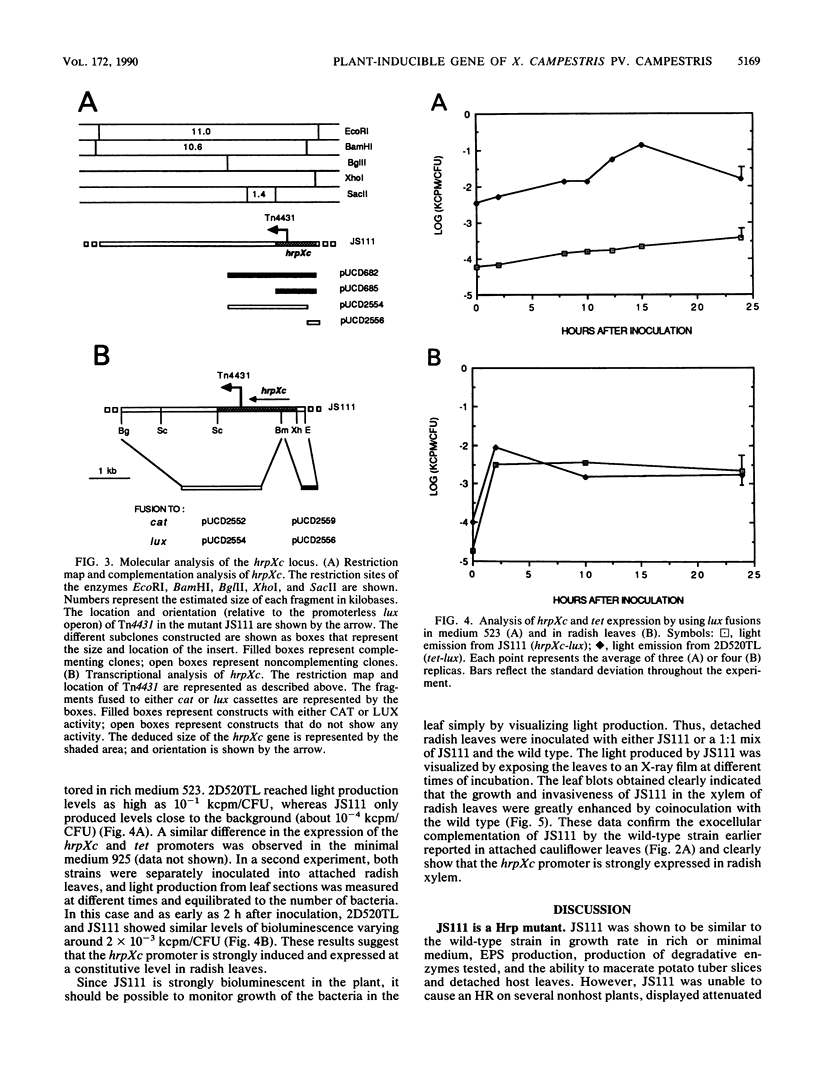
Using Tn4431, a transposon that allows transcriptional fusions to a promoterless luciferase (lux) operon, we have isolated a nonpathogenic mutant of Xanthomonas campestris pv. campestris, i.e., JS111, that does not incite any of the black rot symptoms on all tested cruciferous host plants (J. J. Shaw, L. G. Settles, and C. I. Kado, Mol. Plant Microbe Interact. 1:39-45, 1988). In the study reported here, we determined that in contrast to the wild-type strain, JS111 is unable to induce a hypersensitive necrotic response on nonhost plants such as datura, tomato, and cucumber, suggesting that JS111 is a nonpathogenic, nonhypersensitive Hrp mutant. JS111 displayed culture growth rates, exopolysaccharide production, and protease, pectate lysase, cellulase, amylase, and phosphatase activities comparable to those of the wild-type strain. However, the growth of JS111 in host leaves was markedly attenuated. Coinoculation of JS111 with the wild-type strain in cauliflower or radish leaves rescued the growth deficiency of the mutant to normal levels. The locus mutated in JS111 was cloned and named hrpXc, and transcriptional and genetic complementation analyses of the hrpXc locus were conducted. The regulation of hrpXc expression was also investigated in vitro and in planta, using fusions to a lux or chloramphenicol acetyltransferase reporter gene. The hrpXc gene was found to be strongly induced in radish leaves. This is the first report and analysis of a hrp locus from a Xanthomonas species.
Full text
PDF





Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Boucher C. A., Van Gijsegem F., Barberis P. A., Arlat M., Zischek C. Pseudomonas solanacearum genes controlling both pathogenicity on tomato and hypersensitivity on tobacco are clustered. J Bacteriol. 1987 Dec;169(12):5626–5632. doi: 10.1128/jb.169.12.5626-5632.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Close T. J., Tait R. C., Kado C. I. Regulation of Ti plasmid virulence genes by a chromosomal locus of Agrobacterium tumefaciens. J Bacteriol. 1985 Nov;164(2):774–781. doi: 10.1128/jb.164.2.774-781.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Daniels M. J., Barber C. E., Turner P. C., Sawczyc M. K., Byrde R. J., Fielding A. H. Cloning of genes involved in pathogenicity of Xanthomonas campestris pv. campestris using the broad host range cosmid pLAFR1. EMBO J. 1984 Dec 20;3(13):3323–3328. doi: 10.1002/j.1460-2075.1984.tb02298.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dower W. J., Miller J. F., Ragsdale C. W. High efficiency transformation of E. coli by high voltage electroporation. Nucleic Acids Res. 1988 Jul 11;16(13):6127–6145. doi: 10.1093/nar/16.13.6127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grimm C., Panopoulos N. J. The predicted protein product of a pathogenicity locus from Pseudomonas syringae pv. phaseolicola is homologous to a highly conserved domain of several procaryotic regulatory proteins. J Bacteriol. 1989 Sep;171(9):5031–5038. doi: 10.1128/jb.171.9.5031-5038.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hirooka T., Kado C. I. Location of the right boundary of the virulence region on Agrobacterium tumefaciens plasmid pTiC58 and a host-specifying gene next to the boundary. J Bacteriol. 1986 Oct;168(1):237–243. doi: 10.1128/jb.168.1.237-243.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hirooka T., Rogowsky P. M., Kado C. I. Characterization of the virE locus of Agrobacterium tumefaciens plasmid pTiC58. J Bacteriol. 1987 Apr;169(4):1529–1536. doi: 10.1128/jb.169.4.1529-1536.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kamoun S., Cooley M. B., Rogowsky P. M., Kado C. I. Two chromosomal loci involved in production of exopolysaccharide in Agrobacterium tumefaciens. J Bacteriol. 1989 Mar;171(3):1755–1759. doi: 10.1128/jb.171.3.1755-1759.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Keen N. T., Dahlbeck D., Staskawicz B., Belser W. Molecular cloning of pectate lyase genes from Erwinia chrysanthemi and their expression in Escherichia coli. J Bacteriol. 1984 Sep;159(3):825–831. doi: 10.1128/jb.159.3.825-831.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lindgren P. B., Peet R. C., Panopoulos N. J. Gene cluster of Pseudomonas syringae pv. "phaseolicola" controls pathogenicity of bean plants and hypersensitivity of nonhost plants. J Bacteriol. 1986 Nov;168(2):512–522. doi: 10.1128/jb.168.2.512-522.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Loper J. E., Kado C. I. Host range conferred by the virulence-specifying plasmid of Agrobacterium tumefaciens. J Bacteriol. 1979 Aug;139(2):591–596. doi: 10.1128/jb.139.2.591-596.1979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Manoil C., Beckwith J. TnphoA: a transposon probe for protein export signals. Proc Natl Acad Sci U S A. 1985 Dec;82(23):8129–8133. doi: 10.1073/pnas.82.23.8129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mellano V. J., Cooksey D. A. Development of Host Range Mutants of Xanthomonas campestris pv. translucens. Appl Environ Microbiol. 1988 Apr;54(4):884–889. doi: 10.1128/aem.54.4.884-889.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Niepold F., Anderson D., Mills D. Cloning determinants of pathogenesis from Pseudomonas syringae pathovar syringae. Proc Natl Acad Sci U S A. 1985 Jan;82(2):406–410. doi: 10.1073/pnas.82.2.406. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rogowsky P. M., Close T. J., Chimera J. A., Shaw J. J., Kado C. I. Regulation of the vir genes of Agrobacterium tumefaciens plasmid pTiC58. J Bacteriol. 1987 Nov;169(11):5101–5112. doi: 10.1128/jb.169.11.5101-5112.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Staskawicz B. J., Dahlbeck D., Keen N. T. Cloned avirulence gene of Pseudomonas syringae pv. glycinea determines race-specific incompatibility on Glycine max (L.) Merr. Proc Natl Acad Sci U S A. 1984 Oct;81(19):6024–6028. doi: 10.1073/pnas.81.19.6024. [DOI] [PMC free article] [PubMed] [Google Scholar]