Abstract
Major histocompatibility complex (MHC) class II molecules are transported to intracellular MHC class II compartments via a transient association with the invariant chain (Ii). After removal of the invariant chain, peptides can be loaded onto class II molecules, a process catalyzed by human leukocyte antigen-DM (HLA-DM) molecules. Here we show that MHC class II compartments consist of two physically and functionally distinct organelles. Newly synthesized MHC class II/Ii complexes were targeted to endocytic organelles lacking HLA-DM molecules, where Ii degradation occurred. From these organelles, class II molecules were transported to a distinct organelle containing HLA-DM, in which peptides were loaded onto class II molecules. This latter organelle was not directly accessible via fluid phase endocytosis, suggesting that it is not part of the endosomal pathway. Uptake via antigen-specific membrane immunoglobulin resulted however in small amounts of antigen in the HLA-DM positive organelles. From this peptide-loading compartment, class II–peptide complexes were transported to the plasma membrane, in part after transit through endocytic organelles. The existence of two separate compartments, one involved in Ii removal and the other functioning in HLA-DM–dependent peptide loading of class II molecules, may contribute to the efficiency of antigen presentation by the selective recruitment of peptide-receptive MHC class II molecules and HLA-DM to the same subcellular location.
Major histocompatibility complex (MHC)1 class II molecules present peptides on the cell surface of antigen presenting cells to T helper cells (Germain, 1994). Such peptides are usually derived from antigens internalized into the endocytic/lysosomal pathway while newly synthesized class II molecules are targeted to endocytic compartments via their association with the invariant chain (Ii) (Cresswell, 1994a ; Pieters, 1997a ).
Association of class II molecules with Ii occurs directly after translation in the endoplasmic reticulum (Kvist et al., 1982), and serves at least three functions (Wolf and Ploegh, 1995; Pieters, 1997a). First, Ii guides the folding and egress of the class II complex from the endoplasmic reticulum (Anderson and Miller, 1992). Second, Ii association with class II molecules prevents them from binding antigenic peptides at early stages during biosynthesis (Teyton et al., 1990; Roche and Cresswell, 1991). Third, Ii provides targeting signals for transport of class II molecules to the endosomal/lysosomal pathway (Bakke and Dobberstein, 1990; Lotteau et al., 1990; Pieters et al., 1993).
The targeting of newly synthesized class II/Ii complexes to the endocytic/lysosomal pathway occurs via dileucine based signals that reside within the Ii cytoplasmic tail (Bakke and Dobberstein, 1990; Lotteau et al., 1990; Pieters et al., 1993). Upon arrival in endosomal organelles, the class II– associated Ii is stepwise degraded. Degradation of Ii occurs from the COOH-terminal, lumenal side and results in the formation of NH2-terminal, cytoplasmic Ii fragments that remain associated to class II molecules (Blum and Cresswell, 1988; Pieters et al., 1991). Besides containing the endosomal targeting sequences, the class II–associated Ii fragments also retain the class II–associated invariant chain peptide (CLIP). CLIP associates with class II molecules in a manner quite similar to antigenic peptides (Ghosh et al., 1995), and therefore CLIP or CLIP-containing fragments need to be removed before antigenic peptide loading (Roche, 1996; Pieters, 1997a ).
Removal of Ii fragments from the class II complex is mediated by human leukocyte antigen-DM (HLA-DM) molecules, class II like molecules that consist of an αβ heterodimer (Cho et al., 1991; Kelly et al., 1991; Denzin and Cresswell, 1995; Stebbins et al., 1996). Mutant cell lines lacking these molecules are defective in presenting peptides derived from intact antigens (Mellins et al., 1990) and class II molecules expressed by such mutants are predominantly loaded with CLIP (Riberdy et al., 1992). This implied that DM may facilitate the exchange of Ii fragments for antigenic peptides (Cresswell, 1994b ; Roche, 1995) and, indeed, several groups have confirmed a peptide-exchange function for HLA-DM (Denzin and Cresswell, 1995; Sherman et al., 1995; Sloan et al., 1995). More recent work revealed that HLA-DM can also edit peptide loading of class II in the absence of Ii fragments (Katz et al., 1996; Kropshofer et al., 1996; Roche, 1996; van Ham et al., 1996; Weber et al., 1996). HLA-DM is located in class II positive organelles, to which it is targeted via a tyrosine-based signal that is present in the cytoplasmic tail of the HLA-DM β chain (Karlsson et al., 1994; Sanderson et al., 1994; Marks et al., 1995; Copier et al., 1996; Pierre et al., 1996).
How the processes involved in the generation of class II–peptide complexes, i.e., invariant chain degradation and HLA-DM–dependent peptide loading are controlled in time and in space within an antigen-presenting cell is not known. Recent work from several laboratories has revealed the existence of specialized organelles, MHC class II compartments, in a variety of antigen-presenting cells (Peters et al., 1991; Pieters et al., 1991; Amigorena et al., 1994; Qiu et al., 1994; Tulp et al., 1994; West et al., 1994). Within these class II compartments, the class II–associated invariant chain is degraded and peptides are loaded onto class II molecules. From these organelles, peptide-loaded class II dimers are transported to the cell surface (Amigorena et al., 1994; Tulp et al., 1994; West et al., 1994).
MHC class II compartments, as revealed by immunocytochemistry, appear as multivesicular or multilaminar organelles (Peters et al., 1991, 1995; Pieters et al., 1991). In addition to being morphologically heterogeneous, these compartments differ in their content of Ii as well as endocytic markers (Pieters et al., 1991; Peters et al., 1995). As defined by the degradation of Ii and class II peptide loading, class II compartments can biochemically be distinguished from early and late endosomes as well as from lysosomes in several cell types (Amigorena et al., 1994; Tulp et al., 1994; West et al., 1994; Pieters, 1997b ). In some cases, however, class II–containing organelles were found to colocalize predominantly with lysosomal compartments (Qiu et al., 1994) or with both endosomes and lysosomes (Castellino and Germain, 1995). Together, the morphological and biochemical evidence suggest that class II molecules may be distributed among various compartments linked to the endosomal/lysosomal pathway. Whether or not distinct functions in class II peptide loading can be assigned to different organelles is not known.
Here, we demonstrate that the MHC class II compartments, as originally defined to function in Ii degradation and antigenic peptide loading, consist of two physically and functionally distinct organelles. MHC class II–Ii complexes were first transported to an endocytic compartment where the associated Ii was degraded. From there, class II complexes were transported to a distinct, Ii negative organelle population that contained HLA-DM. The latter compartment was not part of the endocytic pathway, and functioned in antigenic peptide loading. In contrast to fluid phase endocytosed protein, antigen internalized via specific immunoglobulin receptor molecules did reach this peptide loading compartment. The existence of a compartment involved in peptide loading, that is distinct from the one where Ii is degraded and not accessible to the bulk of endocytosed material, may contribute to the efficiency of antigen presentation to CD4+ T cells.
Materials and Methods
Cell Lines and Antibodies
The human melanoma cell line Mel JuSo (Johnson et al., 1981) was grown in RPMI-1640 supplemented with 5% fetal calf of serum (RPMI-FCS) as described (Pieters et al., 1991). Mel JuSo cells that were transfected with the heavy and light chain of immunoglobulin specific for tetanus toxin have been described (Knight et al., 1997). Tetanus toxin C fragment was labeled using iodobeads (Pierce Chem. Co., Rockford, IL) with Na125I to a specific activity of ∼2.7 × 106 cpm/μg (Markwell, 1982). The following antibodies were used: anti-Ii rabbit antiserum MDDQ, raised against the NH2-terminal fragment of Ii; VIC-Y1 (anti-IiN mAb), a kind gift from Dr. W. Knapp (University of Vienna, Vienna, Austria); mAb L243, a kind gift from Dr. T. Johnson (University of Munich, Munich, Germany); mAb Tu36 (PharMingen, San Diego, CA); anti-MHC class II rabbit antiserum, a kind gift of Dr. H.L. Ploegh (Harvard University, Cambridge, MA); anti–HLA-DM antiserum, a kind gift of Dr. E. Mellins (Stanford University, Stanford, CA); mAb 5C1 (anti–HLA-DM) (Sanderson et al., 1996), a kind gift of Dr. J. Trowsdale (University of Cambridge, Cambridge,UK); and DM.K8/2b, a kind gift of Dr. G. Hammerling (German Cancer Research Center, Heidelberg, Germany). Texas-red and FITC-coupled goat anti–rabbit and goat anti–mouse antibodies were purchased from Southern Biotechnology Associates (Birmingham, AL) or Zymed Laboratories Inc. (Seattle, WA).
Subcellular Fractionation
Monolayers of Mel JuSo cells were grown to subconfluency in tissue culture dishes. After scraping the cells in homogenization buffer (10 mM triethanolamine, 10 mM acetic acid, 1 mM EDTA, 0.25 M sucrose, pH 7.4), a membrane fraction was prepared as described (Tulp et al., 1994). The homogenate was adjusted to 1.5 mg protein/500 μl in homogenization buffer and layered on top of a 0.8–1.2 M sucrose gradient prepared in homogenization buffer using the Gradient Master (Nycomed Pharma, Oslo, Norway) in a Beckman SW 28 tube. After centrifugation (12 h at 27,000 rpm in a Beckman SW28 rotor at 4°C), 0.4–1-ml fractions were collected from the top. The variation of the fraction volume size in the different experiments is reflected by different fraction numbers shown in the Figures. Organelle electrophoresis was carried out essentially as described (Tulp et al., 1994; Engering et al., 1997). In brief, membranes in the different fractions after sucrose density centrifugation were collected and sedimented after dilution to 0.25 M sucrose by centrifugation in a SW28 rotor (Beckman) at 27,000 rpm for 1 h at 4°C. The membranes were resuspended in homogenization buffer containing 6% Ficoll-70 (Pharmacia) and electrophoresed for 55 min at 10.4 mA. 0.5-ml fractions were collected and assayed for the different markers (see below). For concentration of membranes, pooled fractions were sedimented in an Optima TLX Ultracentrifuge (Beckman Instrs., Carlsbad, CA) (45 min at 55,000 rpm in a TLA 120 rotor at 4°C).
Analysis of Organelle Markers
For endosome labeling, cells were incubated with horse radish peroxidase (Sigma type VIa) at 2 mg/ml in internalization medium (DMEM supplemented with 10 mM glucose and 10 mM Hepes pH 7.4 [Gruenberg et al., 1989]). After 5 min at 37°C, cells were cooled on ice, washed three times with ice cold Dulbecco's PBS containing 10% BSA, once with prewarmed RPMI-FCS, and then incubated for the times indicated at 37°C in the same medium. The activity of the enzyme β-hexosaminidase was assayed as described (Koldovsky and Palmieri, 1971). Total protein was determined according to Bradford (Bradford, 1976), using bovine gamma globulin (Bio-Rad Labs., Hercules, CA) as a standard.
Metabolic Labeling and Pulse Chase
Before labeling, the cells were grown for 30 min in methionine/cysteine-free medium containing 5% dialyzed FCS. Labeling was performed by placing the cells in methionine/cysteine-free medium containing 5% dialyzed FCS and 0.15–0.3 mCi/ml [35S]methionine/cysteine mix (Promix; Amersham Corp., Arlington Heights, IL) for 20 min. After the pulse, cells were washed twice in RPMI-FCS containing 5 mM cold methionine and 5 mM cold cysteine. Chase was performed at 37°C in RPMI-FCS supplemented with 5 mM of methionine and cysteine each.
Immunoprecipitation and Immunoblotting
For immunoprecipitation, subcellular fractions were adjusted to 10 mM Tris-HCl, pH 7.4, 100 mM NaCl, 5 mM MgCl2, and 1% Triton X-100 and a mixture of protease inhibitors (chymostatin [10 μg/ml], leupeptin [10 μg/ml], aprotinin [10 μg/ml], antipain [10 μg/ml], pepstatin [10 μg/ml], and PMSF [1 mM]) was added to the supernatant. After incubation with the antibodies indicated, the samples were centrifuged at 20,000 g for 15 min, after which 50 μl of a protein A–Sepharose (1:1; Pharmacia) slurry was added. After a further 2-h incubation at 4°C, the protein A beads were washed and eluted as described (Pieters et al., 1991). SDS-PAGE was performed according to Laemmli (Laemmli, 1970). For detection of SDS-stable class II complexes, immunoprecipitated material was eluted from the protein A–Sepharose beads at room temperature for 20 min before SDS-PAGE and fluorography. For quantitation, the radioactivity associated with proteins fixed in the dried gels was determined by Phosphor Image analysis (Molecular Dynamics, Sunnyvale, CA). Alternatively, autoradiographs were scanned using a Molecular Dynamics scanner, and relative intensities were determined using the software supplied. For detection of polypeptides following immunoblotting, membrane fractions were either sedimented in an Optima TLX Ultracentrifuge (Beckman) (45 min at 55,000 rpm in a TLA 120.1 rotor at 4°C) or proteins were concentrated by precipitation in 10% trichloric acetic acid on ice, followed by centrifugation at 20,000 g for 30 min. Membrane or protein pellets were resuspended in Laemmli sample buffer, proteins were separated by SDS-PAGE and transferred to nitrocellulose using a Trans-Blot device (Bio-Rad Labs.). Alternatively, 20–100-μl aliquots from the fractions were applied directly to nitrocellulose. After incubation of the membrane in 5% lowfat milk in PBS containing 0.2% Tween-20, the nitrocellulose was incubated with specific antibodies, washed and incubated with peroxidase-coupled secondary antibodies and developed by chemoluminescence (ECL; Amersham). For quantitation, resulting films were scanned after which the relative intensity was determined using the NIH Image Software.
Immunofluorescence Microscopy
Cells were grown on glass coverslips and fixed in 3% paraformaldehyde followed by permeabilization in 0.1% Triton X-100 or fixed in methanol as described (Pieters et al., 1993). Coverslips were incubated with primary antibodies for 40 min at room temperature, washed in PBS containing 5% fetal calf serum and then incubated with FITC and Texas red coupled goat anti-rabbit and goat anti-mouse antibodies. Coverslips were mounted in Mowiol (Hoechst) containing 100 mg/ml DABCO (Sigma) or in FluoroGuard Antifade Mounting Reagent (Bio-Rad Labs.), and analyzed using a confocal laser scanning microscope system (MRC-1024, equipped with 522/32-nm and 605/32-nm band pass filter for FITC and Texas red, respectively; Bio-Rad Labs.) attached to an Axiovert 100 microscope (Carl Zeiss, Inc., Thornwood, NY). Images were collected using the 488 nm (FITC) and 568 nm (Texas red) lines of an argon krypton laser, with pinhole settings at 2.0 and 2.5 for FITC and Texas red, respectively.
Results
Isolation of Distinct MHC Class II Containing Organelles
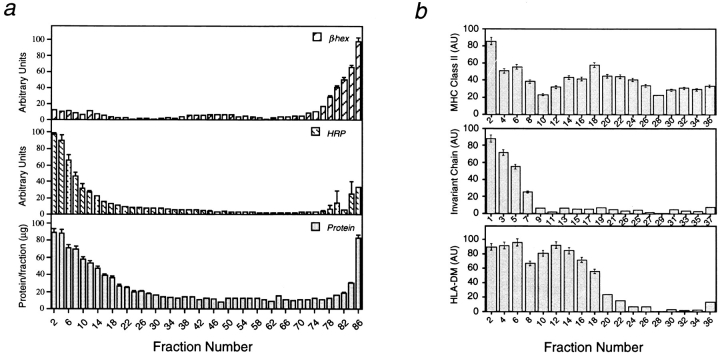
The human HLA-DR3 positive cell line Mel JuSo is a useful model cell line for studying the cell biology of antigen processing and presentation in that it efficiently processes and presents antigens to CD4+ T cells (Chen and Hersey, 1991; Tulp et al., 1994; Knight et al., 1997). Besides expressing HLA-DR, it is positive for the class II–associated Ii as well as for HLA-DM molecules (Pieters et al., 1991; Sanderson et al., 1994; Tulp et al., 1994) and contains large numbers of MHC class II positive organelles (Pieters et al., 1991; Tulp et al., 1994). When such organelles are isolated from other subcellular organelles on the basis of their charge characteristics, they can be distinguished from early and late endosomes but comigrate in such separation procedures with lysosomes (Tulp et al., 1994). To separate class II positive organelles from lysosomes, conditions were established using isopycnic centrifugation in sucrose (De Duve, 1965) that enriched class II–containing organelles while depleting lysosomes. Mel JuSo cells were allowed to internalize the fluid phase endocytic tracer horse radish peroxidase (HRP) for 5 min, followed by a 15-min chase to allow transport to class II–containing organelles (Tulp et al., 1994). Membranes were prepared as described in Materials and Methods, and separated using various gradients ranging from 0.6–1.6 M sucrose (not shown). We found that a 0.8–1.2 M sucrose gradient resulted in a recovery of HRP in membranes migrating in the top fractions of the gradient while lysosomes, identified by the presence of β-hexosaminidase, sedimented at the bottom of the gradient (Fig. 1 a, top and middle). Small amounts of β-hexosaminidase activity were also present in the light (fraction 1–25) and intermediate (25–60) density fractions (Fig. 1 a, top). The bulk of subcellular organelles, including the endoplasmic reticulum and Golgi compartments migrated in the top fractions of the gradient, as judged by the analysis of the total amount of protein in the different fractions (Fig. 1 a, bottom). Thus, the here established 0.8–1.2 M sucrose gradient resulted in the depletion of lysosomes from most other subcellular organelles.
Figure 1.
(a) Sedimentation profiles of endosomal and lysosomal organelles after isopycnic centrifugation is sucrose. Mel JuSo cells were allowed to internalize HRP for 5 min followed by a 15-min chase. Cells were homogenized and a post-nuclear supernatant was loaded on a sucrose gradient (0.8–1.2 M sucrose in homogenization buffer). After centrifugation and fractionation of the gradients (left top, right bottom), each fraction was analyzed for the activity of β-hexosaminidase (β-hex, top) and HRP (middle). (Bottom) Total protein content in the different fractions as analyzed by Bradford. Shown are mean (± SD) from three separate experiments. (b) Steady state distribution of MHC class II, Ii and HLA-DM molecules in subcellular organelles. Proteins from each fraction after sucrose density gradient centrifugation (left top, right bottom) were allowed to bind to nitrocellulose after which the amount of MHC class II (top), Ii (middle) or HLA-DM (bottom) was analyzed by immunostaining, using specific antibodies. Shown are mean (± SD) obtained from three separate gradients.
To analyze the distribution of MHC class II–containing organelles across the 0.8—1.2 M sucrose gradient, an aliquot of the different fractions was subjected to slot blot analysis followed by immunostaining with the anti–MHC class II mAb L243. As shown in Fig. 1 b, top, two major populations of MHC class II molecules migrated in the top and the middle part of the gradient, respectively. A minor population of MHC class II molecules comigrated with the lysosomal marker β-hexosaminidase. The distribution of the Ii, was similarly analyzed using an antibody raised against the NH2-terminal cytoplasmic tail of Ii. Ii containing organelles were almost exclusively present in the light part of the gradient (Fig. 1 b, middle). HLA-DM molecules were distributed in the light as well as in the intermediate density fractions, with only small amounts present in the dense fractions (Fig. 1 b, bottom), in agreement with its distribution in B cells (Kropshofer et al., 1997). The slightly different distribution of class II and HLA-DM molecules most probably resulted from plasma membrane fragments containing large amounts of class II molecules. Indeed, the plasma membrane was recovered from the light, intermediate and dense parts of the gradient, as analyzed by the presence of metabolically labeled and chased MHC class I molecules (data not shown).
Thus, following isopycnic centrifugation in sucrose, two class II–containing organelle populations displaying different buoyant density could be distinguished; only within the lighter of these two populations the class II–associated invariant was detected, while both populations contained HLA-DM.
Localization of Invariant Chain and HLA-DM in Two Distinct MHC Class II Positive Organelles
As noted above, while sucrose density gradient centrifugation results in separation of organelles having a different buoyant density, several types of membranes comigrate during such a procedure. To achieve a better separation of subcellular organelles, density centrifugation was combined with organelle electrophoresis. During such electrophoresis, organelles can be separated that display differences in their charge characteristics (Marsh et al., 1987; Tulp et al., 1993; Engering et al., 1997).
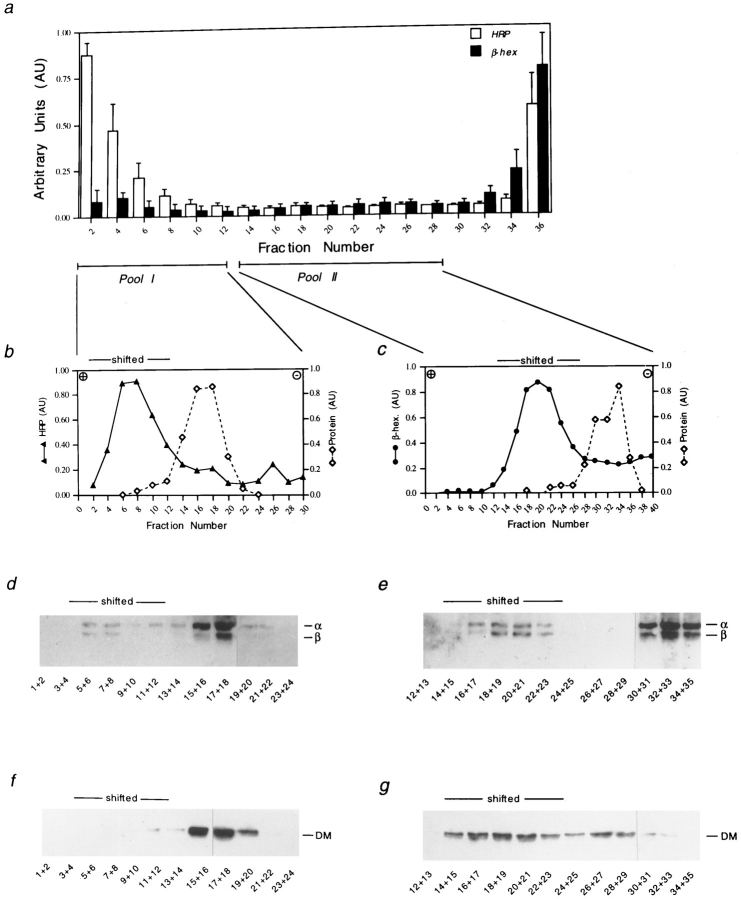
To purify MHC class II–containing organelles from the two class II positive pools resulting after isopycnic centrifugation in sucrose, a two-step fractionation procedure was established. Cells were allowed to internalize HRP for 5 min followed by a 15-min chase after which a homogenate was prepared. In a first step, organelles were separated by isopycnic centrifugation in sucrose to deplete lysosomal organelles from other subcellular organelles. Fractions 1-12 (Pool I) and fractions 13-28 (Pool II) as indicated in Fig. 2 a, were pooled separately. Membranes from each pool were sedimented at 100,000 g, resuspended, and then subjected to organelle electrophoresis as described in Materials and Methods; acidic organelles, including endosomes and lysosomes shift towards the anode during such separation (Harms et al., 1980; Engering et al., 1997), and are recovered in the first fractions (Fig. 2, b and c, shifted). Most other subcellular organelles do not migrate in the electric field and are recovered at the cathodic side. The resulting fractions were analyzed for the presence of HRP and β-hexosaminidase activity as well as for the amount of total protein to analyze the migration of other subcellular organelles. As shown in Fig. 2 b and c, each pool could be separated in a anodally “shifted” fraction and a fraction containing the bulk of the protein, including ER/Golgi as well as plasma membrane components (unshifted fractions) (Tulp et al., 1994). The anodally shifted membranes from pool I contained little β-hexosaminidase activity but contained the endocytic marker HRP (Fig. 2 b). In contrast, anodally shifted membranes from pool II contained no measurable HRP activity, but were positive for β-hexosaminidase (Fig. 2 c).
Figure 2.
Isolation of distinct MHC class II–containing organelles. (a) Mel JuSo cells were allowed to internalize HRP for 5 min, following by a 15-min chase. Cells were homogenized and a postnuclear supernatant was applied to a 0.8–1.2 M sucrose gradient (left top, right bottom). After centrifugation, fractions were analyzed for HRP activity (open bars) and β-hexosaminidase (filled bars). Fractions 1-12 (Pool I) and 13-28 (Pool II) were sedimented at 100,000 g and each pool was applied to organelle electrophoresis. (b and c) Distribution of HRP (b, filled triangles) and β-hexosaminidase activity (c, filled circles) and total amount of protein (b, and c, open diamonds) after electrophoresis of pool I and pool II, respectively. Anode, left, cathode, right. Fractions after electrophoresis were sedimented at 100,000 g and proteins were subjected to 12% SDS-PAGE and transferred to nitrocellulose. The presence of MHC class II molecules (d and e) as well as HLA-DM (f and g) was detected by immunostaining of the membrane.
To analyze the distribution of MHC class II and HLA-DM, membranes in the different fractions were pooled as indicated in Fig. 2, d–g and sedimented at 100,000 g. After SDS-PAGE and transfer to nitrocellulose, the presence of MHC class II molecules and HLA-DM was detected by immunostaining. As expected, anodally shifted membranes from both pool I and pool II contained MHC class II molecules (Fig. 2, d and e). In contrast, only the anodally shifted membranes from pool II contained HLA-DM, whereas after electrophoresis of pool I, HLA-DM was exclusively detected in unshifted fractions containing ER/Golgi compartments (Fig. 2, f and g).
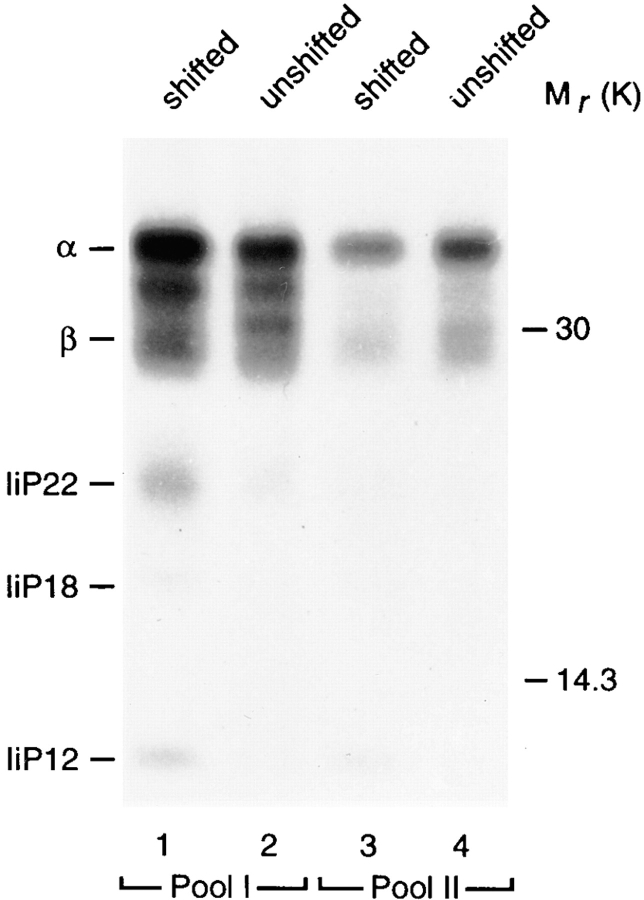
MHC class II–associated invariant chain degradation fragments are being generated in Mel JuSo cells 2–4 h after biosynthesis (Pieters et al., 1991; Tulp et al., 1994). To reveal the presence of class II–associated Ii degradation products in the two class II positive organelle populations, the above described two-step fractionation procedure was applied to membranes prepared from cells that were metabolically labeled for 20 min followed by a 3-h chase. From both the shifted and unshifted membranes obtained after electrophoresis of pool I and pool II, class II molecules were immunoprecipitated and analyzed by SDS-PAGE. As shown in Fig. 3, class II molecules in the shifted membranes from the light pool (pool I) were assembled with Ii degradation fragments IiP22, IiP18, and IiP12 (Fig. 3, lane 1). Interestingly, small amounts of IiP12 were present in the shifted fractions from the denser pool (pool II; Fig. 3, lane 3). IiP12 most probably includes the so-called CLIP fragment (Pieters et al., 1991, Amigorena et al., 1995), and like CLIP, this fragment can be removed from MHC class II molecules by HLA-DM in exchange for peptides (Denzin and Cresswell, 1995; Stebbins et al., 1996).
Figure 3.
Detection of Ii fragments in class II–containing organelles. Cells were metabolically labeled for 20 min with [35S]-methionine/ cysteine followed by a 3-h chase. Membranes were prepared and fractionated by sucrose density centrifugation followed by electrophoresis as described in Fig. 2. After electrophoresis, shifted (lanes 1 and 3) and unshifted (lanes 2 and 4) membranes from pool I (lanes 1 and 2) and pool II (lanes 3 and 4) were pooled and class II molecules immunoprecipitated followed by SDS-PAGE analysis and fluorography.
Taken together, these data show that two physically distinct MHC class II containing organelle populations can be defined based on density centrifugation; in the lighter organelles, the Ii associated with MHC class II molecules is being degraded, whereas the denser vesicle population contains HLA-DM.
Localization of Ii and HLA-DM by Immunofluorescence Microscopy
The finding that Ii degradation products and HLA-DM reside in two distinct class II–containing organelles led us to analyze the steady state distribution of Ii and HLA-DM by confocal immunofluorescence microscopy. Cells were grown on coverslips, fixed and permeabilized, and then stained for Ii using anti-Ii NH2-terminal antibodies as well as for HLA-DM molecules. Ii was localized in the Golgi area as well as in a punctated pattern throughout the cytoplasm (Fig. 4 a, a and d). HLA-DM was distributed in a punctated staining pattern, as analyzed using both a purified polyclonal antiserum (Fig. 4 a, b) and an mAb (Fig. 4 a, e). Some colocalization is apparent in the Golgi area, but most HLA-DM positive structures did not label for Ii, as is shown in Fig. 4 a, c and f, which depicts merging of the two images. Both Ii and HLA-DM colocalized to a large degree with MHC class II molecules, as expected (Fig. 4 b), although the class II staining did not completely overlap with Ii (Fig. 4 b, c) or HLA-DM (Fig. 4 b, f). These results further corroborate the existence of two distinct MHC class II organelle populations; one containing Ii immunoreactivity, the other positive for HLA-DM.
Figure 4.
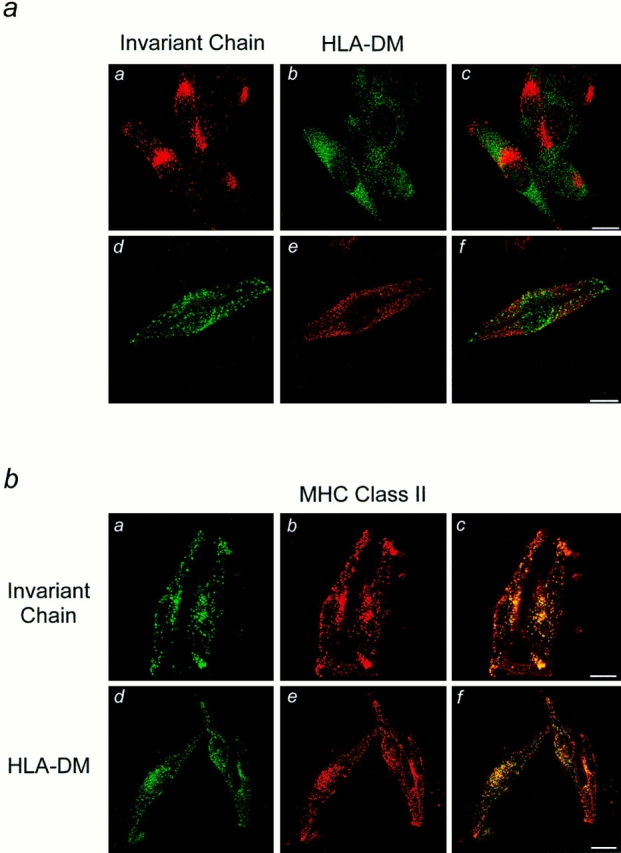
Localization of Ii and HLA-DM by immunofluorescence microscopy. (a) Cells grown on coverslips were fixed, permeabilized and labeled for Ii using anti-Ii NH2-terminal monoclonal (a) or polyclonal (d) antibodies, and HLA-DM using polyclonal (b) or monoclonal antibodies (e) followed by FITC (b and d) or Texas red (a and e) conjugated secondary antibodies. The superposition of the images is shown in c and f. In b, cells were labeled for MHC class II molecules using monoclonal (b) or polyclonal (e) antibodies and double labeled for Ii using polyclonal anti-Ii NH2-terminal antibodies (a) or HLA-DM using mAbs (d), followed by Texas red (b and e) or FITC (a and d) conjugated secondary antibodies. c and f show the merged images. Bars: 20 μm.
Transport of MHC Class II Complexes through Distinct Class II Positive Organelles
To analyze the kinetics of transport of newly synthesized class II molecules through the Ii positive and HLA-DM positive organelle population, pulse-chase analysis was combined with the two-step subcellular fractionation. Cells were metabolically labeled with [35S]methionine/cysteine for 20 min and chased for the times indicated in Fig. 5. At each time point, membranes were prepared and subjected to isopycnic centrifugation in sucrose, followed by organelle electrophoresis of the separate pools. After electrophoresis, the shifted and unshifted membranes were pooled, and the presence of newly synthesized class II complexes analyzed by immunoprecipitation and SDS-PAGE. As shown in Fig. 5, after 2 h of chase, small amounts of class II molecules had accessed the lighter, Ii positive class II–containing organelles, whereas very little was present in the denser, HLA-DM positive class II organelles. Most class II molecules, however, comigrated with ER/Golgi markers in the unshifted pools. With increasing chase times (Fig. 5, T = 3 hours and T = 4 hours), class II molecules migrated from the Ii positive class II vesicle population (Fig. 5, pool I, shifted) to the HLA-DM positive class II organelles (pool II, shifted). Surprisingly, during 5 and 7 h of chase, class II molecules became equally distributed between both class II–containing organelles. Finally, class II molecules are expected to be transported to the plasma membrane: indeed, 12 and 48 h after biosynthesis all class II molecules had migrated out of both class II containing vesicle populations and comigrated with plasma membrane in the unshifted pools (Fig. 5, T = 12 hours and T = 48 hours).
Figure 5.
Pulse-chase analysis of class II transport through distinct subcellular organelles. Cells were labeled for 20 min with [35S-]methionine/cysteine, followed by a chase for the times indicated in the absence of radiolabel. At each time point, cells were homogenized and membranes fractionated by sucrose density centrifugation followed by organelle electrophoresis as described in Fig. 2. Shifted (lanes 1 and 3) and unshifted (lanes 2 and 4) membranes were pooled, class II molecules immunoprecipitated and analyzed by SDS-PAGE and fluorography.
Quantitation of the resulting autoradiographs revealed that 3 h after biosynthesis, ∼50% of total newly synthesized MHC class II molecules comigrated with the Ii positive class II–containing organelles, while only ∼5% was present in the HLA-DM positive organelles. After 4 h of chase, ∼30% of total class II molecules was present in the HLA-DM positive class II containing organelle population whereas ∼5% remained in the Ii positive organelles. After 5 and 7 h of chase, ∼15% of the class II molecules were present in both the Ii and HLA-DM positive shifted fractions; at later chase times (T = 12 hours and T = 48 hours), <1% of class II molecules remained in the class II– containing shifted vesicle populations.
In general, MHC class II molecules are not subject to degradation and indeed no loss of class II signal was observed up to 72 h after biosynthesis (results not shown). Therefore, the time course of class II transport through subcellular compartments suggests a sequential transit of newly synthesized class II complexes from the trans-Golgi network through organelles where the associated Ii is degraded, to the vesicle population containing HLA-DM. From these latter, HLA-DM positive organelles, class II complexes were exported to the plasma membrane, possibly via endocytic organelles (see below).
Peptide Loading in MHC Class II–containing Organelles
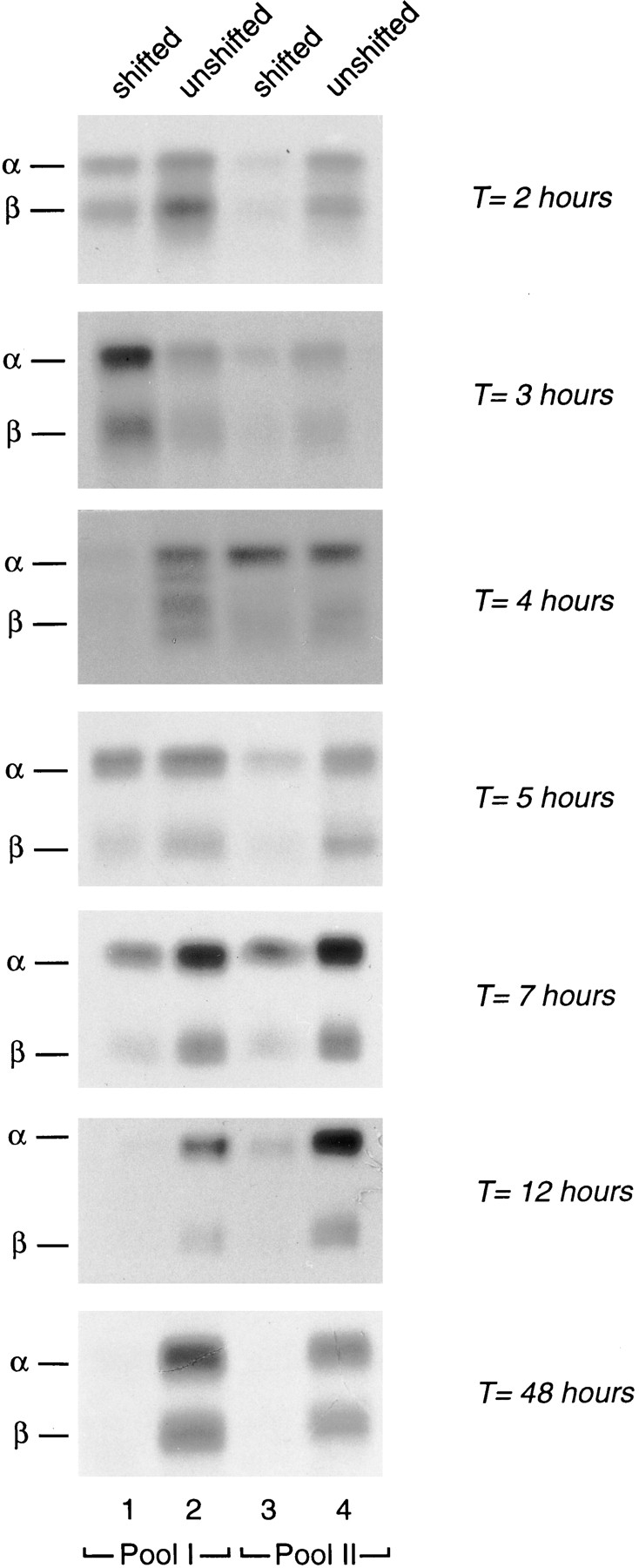
The presence of HLA-DM in the denser of the MHC class II positive vesicle population together with the arrival of newly synthesized class II molecules in these organelles after Ii degradation suggests that peptide loading would occur in these organelles preferentially. To address the formation of class II–peptide complexes, a series of pulse-chases combined with the two-step subcellular fractionation was performed. After immunoprecipitation, MHC-class II complexes were resolved by SDS-PAGE analysis under mildly denaturing conditions. As peptide loading renders class II dimers SDS resistant, peptide-loaded class II molecules appear as αβ dimers migrating at ∼56 kD under these conditions (Germain and Hendrix, 1991; Tulp et al., 1994). In Mel JuSo cells, SDS-stable class II complexes start to be formed 2 h after biosynthesis (Tulp et al., 1994 and data not shown). Cells were biosynthetically labeled for 20 min with [35S]methionine/cysteine followed by chases in the absence of radiolabeled amino acids for the times indicated in Fig. 6. To evaluate the percentage of class II complexes that had acquired SDS resistance in the two organelle populations, the fluorographs shown represent an equivalent class II signal in pool I and II per time point. 2 h after biosynthesis, which is the time class II molecules start to be transported into MHC class II–containing compartments (Amigorena et al., 1994; Tulp et al., 1994), low amounts of SDS-stable αβ dimers comigrated with the HLA-DM positive organelles (Fig. 6, T = 2 hours, Pool II; ∼10% of SDS-stable dimers present), but not with the Ii positive vesicle population (Fig. 6, T = 2 hours Pool I; note that the fluorograph representing pool II has been exposed five times longer than the one representing pool I). Interestingly, a portion of SDS-stable class II dimers were present in nonshifted fractions as well, probably reflecting a rapid transport of peptide loaded class II molecules to the plasma membrane. 4 h after biosynthesis, SDS-stable αβ dimers almost exclusively comigrated with the HLA-DM containing organelles (Fig. 6, T = 4 hours Pool II; ∼20% SDS-stable dimers present), while little class II molecules remained in the shifted fractions of pool I (Fig. 6, T = 4 hours). At this time point, unshifted membranes from both pools contained SDS-stable dimers, most probably reflecting peptide-loaded class II dimers that have reached the plasma membrane. After 7 h of chase, SDS-stable αβ dimers were present in shifted membranes from both pool I and pool II, indicating that peptide-loaded class II dimers had again gained access to Ii positive membranes. At late times of chase (Fig. 6, T = 12 hours) SDS-stable class II dimers were predominantly found to comigrate with plasma membrane fractions.
Figure 6.
Formation of SDS-stable αβ dimers in class II–containing subcellular organelles. Cells were labeled for 20 min with [35S]methionine/cysteine, followed by a chase for the times indicated in the absence of radiolabel. At each time point, cells were homogenized and membranes fractionated by sucrose density centrifugation and organelle electrophoresis as described in Fig. 2. Fractions after organelle electrophoresis were pooled as indicated, and class II molecules immunoprecipitated followed by SDS-PAGE under mildly denaturing conditions and fluorography. The protein peak migrated at fraction 26 (Pool I) and fraction 30 (Pool II), respectively. Note that the fluorographs represent equal amount of the class II signal in pool I and II (see text).
In conclusion, the formation of peptide-loaded MHC class II dimers in the HLA-DM positive vesicle population defines this site as the peptide-loading compartment.
Accessibility of the MHC Class II Positive Compartments to Internalized Antigen
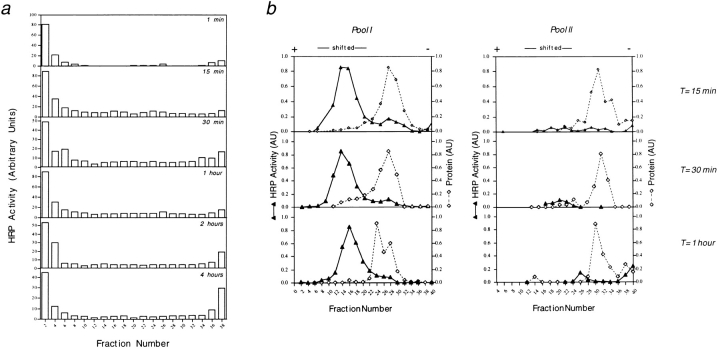
Epitopes presented by MHC class II molecules at the plasma membrane to T cells are usually derived from antigens that are internalized in the endocytic pathway of the presenting cell. The finding that the fluid phase marker HRP, internalized for 15 min, is not found in membranes comigrating with the peptide-loading compartment after density centrifugation (see Figs. 1 and 2), suggested that these organelles are not accessible via the endocytic pathway. To analyze whether HRP accessed the loading compartment at different times of chase, cells were pulsed for 5 min with HRP at 37°C, and chased in the absence of HRP from 1 min up to 4 h. After homogenization and density centrifugation of membranes, the HRP activity was determined in the different fractions (Fig. 7 a). The bulk of HRP activity at any chase time point was present in fractions containing the organelle population where Ii was degraded, with little HRP reactivity appearing in pool II at the 15-min to 1-h chase times. To directly analyze the presence of HRP in both class II positive organelles at these time points, membranes present in pool I and II from the density gradient were subjected to electrophoresis. As shown in Fig. 7 b, virtually all HRP reactivity was recovered in shifted membranes from pool I, consistent with HRP being distributed in endosomal organelles at these time points (Griffiths et al., 1988; Parton et al., 1989; Zachgo et al., 1992). Only trace amounts were detected in shifted membranes from pool II, although some HRP reactivity was recovered at the loading phase (corresponding to fraction 50) probably representing HRP aggregates (not shown).
Figure 7.
Kinetics of transport of HRP through intracellular compartments. Cells were allowed to internalize HRP for 5 min, followed by a chase for the times indicated in the different panels. Membranes were prepared and fractionated by sucrose density centrifugation (a) and organelle electrophoresis (b) for the times indicated.
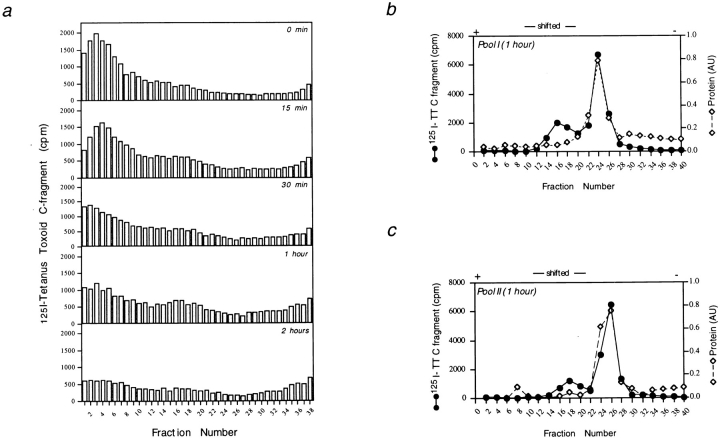
While fluid phase endocytosis is important for nonspecific uptake of antigens, e.g., in dendritic cells (Steinman and Swanson, 1995; Lanzavecchia, 1996), antigen-specific B cells internalize antigens via membrane bound immunoglobulin receptors (Lanzavecchia, 1987). The possibility was investigated whether receptor-mediated antigen uptake allowed transport of antigen into the HLA-DM positive peptide-loading compartment. When Mel JuSo cells are transfected with mIgG specific for tetanus toxin, they can present this antigen at 1,000-fold lower concentrations to T cells as compared to nontransfected Mel JuSo cells (Knight et al., 1997). To analyze whether antigen uptake via receptor-mediated internalization accessed the HLA-DM positive class II compartment, mIgG transfected Mel JuSo cells were allowed to internalize 125I-labeled tetanus toxin C fragment for 5 min at 37°C, after which the cells were chased in the absence of antigen for the times indicated in Fig. 8 a. At each time point, cells were homogenized, membranes fractionated by density centrifugation in sucrose, and the radioactivity recovered in each fraction was determined. As shown in Fig. 8 a, after 5 min internalization at 37°C (0-min chase), most of the radiolabel was present in pool I, comigrating with the Ii positive class II compartment. A small amount of radioactivity was also detected in pool II. With increasing chase times, the amount of radiolabel recovered from pool I gradually decreased, while the counts recovered from pool II containing the peptide-loading compartment remained approximately the same. To establish whether the activity recovered in pool II was associated with the plasma membrane or had accessed the class II positive compartments, fractions obtained after density gradient centrifugation were pooled and subjected to organelle electrophoresis. As shown in Fig. 8, b and c, after 1 h of chase, while most of the radiolabel was in fact associated with unshifted membranes (presumably plasma membrane and early endosomes [Tulp et al., 1993]), ∼30% of the label was associated with shifted membranes from pool I, whereas ∼20% was found in shifted membranes from pool II. Together these data indicate that after uptake via specific antigen receptors, small but significant amounts of antigen were transported to the peptide-loading compartment, in contrast to material internalized via fluid phase endocytosis.
Figure 8.
Kinetics of transport of 125I-tetanus toxin C fragment through intracellular compartments in Mel JuSo cells transfected with mIgG. Cells were allowed to internalize 125I-tetanus toxin C fragment for 5 min, followed by a chase for the times indicated in the different panels. Membranes were prepared and fractionated by sucrose density centrifugation (a) and organelle electrophoresis (b and c) for the times indicated.
Discussion
Two chaperones function at different stages in the MHC class II peptide-loading pathway: the Ii and the HLA-DM molecules. Whereas Ii serves to target class II molecules to endocytic compartments, the HLA-DM dimer functions in catalyzing peptide loading onto class II molecules that these encounter intracellularly (Wolf and Ploegh, 1995; Pieters, 1997a,b). In this paper, we characterized the subcellular compartments where Ii is being degraded and where HLA-DM–dependent peptide loading occurs. Surprisingly, these two events were found to occur in two physically distinct organelles; the class II–associated Ii was degraded in a light, endosomal compartment, whereas HLA-DM was located in a distinct compartment of intermediate density in which peptides were loaded onto class II molecules. This DM positive compartment could not be accessed via fluid phase endocytosis; it was, however, accessible to antigen after receptor mediated internalization. Thus, the recently characterized MHC class II compartment that was shown to mediate both Ii degradation and peptide loading, in fact consists of two distinct organelles, each carrying out a specific function.
A class II positive nonlysosomal compartment with similar characteristics to the organelles described here has been isolated from human and murine B cells. In these organelles, Ii is being degraded and peptides loaded onto class II complexes before their transport to the cell surface (Amigorena et al., 1994, 1995; West et al., 1994). Interestingly, in these B cells the class II compartments are only partially accessible to endocytic tracers (Amigorena et al., 1994), and, antigen internalized via mIgG receptors is rapidly transported to class II compartments (West et al., 1994). This suggest that also in B cells the class II compartment comprises an endocytic organelle population, as well as a compartment that functions in peptide loading of class II molecules.
A Separate Peptide-loading Compartment
Degradation of the class II associated Ii results in the formation of various Ii NH2-terminal fragments associated with class II (IiP22, IiP18, and IiP12 [Blum and Cresswell, 1988; Pieters et al., 1991]). IiP12, that most probably includes the CLIP sequence, remains associated with class II molecules for prolonged times (>4 h) (Pieters et al., 1991; Amigorena et al., 1995), and, class II associated with Ii NH2-terminal fragments such as IiP12 can serve as a substrate for HLA-DM, in a manner similar to class II-CLIP complexes (Denzin and Cresswell, 1995; Stebbins et al., 1996). Indeed, 3 h after synthesis, which is the time class II–peptide complexes were beginning to be detected, αβ-IiP12 was found in the peptide loading compartment. There, the presence of HLA-DM would result in removal of IiP12 from class II in favor of antigenic peptides; it can, however, not be excluded that CLIP is formed from IiP12 in the peptide loading compartment before DM assisted antigenic peptide loading of class II molecules.
Newly synthesized class II molecules first accessed the peptide loading compartment 2–3 h after biosynthesis. Within 4 h after biosynthesis, most class II complexes were transported to the peptide loading compartment, where they acquired resistance to SDS, indicative of the acquisition of peptides. What causes transport of class II molecules into the peptide-loading compartment remains to be established. One possibility is that degradation of the lumenal part of Ii leads to differences in the oligomeric state of class II complexed with Ii fragments. Indeed, recent work suggests that the oligomeric state of Ii dictates its precise subcellular localization (Arneson and Miller, 1995). In that case, IiP12 may target the associated class II to the peptide loading compartment. Alternatively, degradation of the Ii lumenal part may unmask a targeting signal in the class II molecules themselves, possibly residing in the class II β-chain (Chervonsky et al., 1994).
Whether or not the here described compartments originate from each other through the process of maturation or whether cargo is transported between preexisting organelles through budding and fusion of transport vesicles, remains to be established (Griffiths and Gruenberg, 1991; Murphy, 1991; Mellman, 1996). Most subcellular organelles that are part of the biosynthetic and endocytic pathway are interconnected, receiving a continuous flow of both newly synthesized protein and lipid moieties as well as material internalized from the plasma membrane (Kornfeld and Mellman, 1989; Mellman, 1996). If the organelles described here originate from each other through the process of maturation, the different distributions of class II–associated Ii fragments and HLA-DM might reflect encounter of proteases that degrade Ii at a stage that precedes accumulation of HLA-DM in a denser organelle. Alternatively, the here described organelle populations represent distinct compartments, as suggested from the absence of HLA-DM from the lighter, Ii positive endocytic organelles and the fact that HRP (being protease resistant) was not found in the HLA-DM–containing organelle.
Interestingly, targeting of Ii and HLA-DM from the trans-Golgi network to class II–containing post-Golgi organelles is mediated via distinct types of signals: Ii is targeted via dileucine based signals that reside within the cytoplasmic tail (Bakke and Dobberstein, 1990; Lotteau et al., 1990; Pieters et al., 1993) whereas HLA-DM contains a tyrosine-based motif in its cytoplasmic tail (Copier et al. 1996; Lindstedt et al. 1995; Marks et al. 1995). Both dileucine and tyrosine based signals appear to interact with adaptor proteins associated with clathrin (Hunziker and Fumey, 1994; Ohno et al., 1995). However, Marks et al. (1996) recently showed that dileucine and tyrosine based targeting events are mediated by distinct saturable components. While these authors suggested that Ii and HLA-DM may travel via different routes to MHC class II compartments, our results suggests that di-leucine and tyrosine based signals may specify different locations.
Transport of Peptide-loaded Class II Complexes to the Plasma Membrane
How are peptide loaded class II molecules transported to the plasma membrane? The results presented here suggest that more then one possibility exists. First, at the earliest time points after biosynthesis that SDS stable αβ complexes could be detected (2 h), small amounts of stable complexes comigrated with plasma membrane fractions. This suggests a rapid and direct transport of peptide-loaded class II complexes from the peptide-loading compartment to the plasma membrane, possibly involving early endosomes (Tulp et al., 1994; Raposo et al., 1996; Pond and Watts, 1997). Alternatively, peptide-loaded class II molecules may follow retrograde transport from the peptide-loading compartment through the Ii-containing endocytic organelles to reach the plasma membrane.
An interesting possibility is that depending on the nature of the peptides acquired in the loading compartment, class II complexes may either directly be deposited at the plasma membrane, or recycle between endocytic organelles and the HLA-DM–containing loading compartment. Recycling may continue until a productive peptide–class II interaction has occurred that will result in stable cell surface expression. The observed transit of class II complexes through endocytic organelles even after prolonged times of chase, is compatible with such a model. During such recycling, peptides might also be exchanged on class II molecules in endocytic organelles in an HLA-DM–independent manner (Pinet et al., 1995; Warmerdam et al., 1996; Cella et al., 1997).
Class II Compartments and the Endosomal/Lysosomal Pathway
Remarkably, the peptide-loading compartment, in contrast to the compartment where Ii is being degraded, was not directly accessible from the endocytic pathway. Whereas the fluid phase endocytic tracer HRP did not access the peptide-loading compartment, significant amounts of antigen that were internalized via mIgG molecules, however, could be detected in the HLA-DM positive loading compartment. In the latter case, relatively low levels of radiolabeled antigen were detected, which may be due either to extensive proteolysis of the internalized antigen, or to the fact that a large portion of the antigen cycles between the plasma membrane and early endosomes before transport to antigen degradation or peptide-loading compartments (Watts and Davidson, 1988). Thus, antigen would not necessarily accumulate in this compartment. However, consistent with the efficient antigen presentation capacity of these cells (Knight et al., 1997), our data do suggest that in contrast to fluid phase internalized material, antigen internalized via mIgG could access the peptide-loading compartment. At present, it is not known where precisely antigens are being degraded. In fact, distinct mechanisms may be involved in antigen capture, processing, and transport to the peptide-loading compartment. In support of this, Mitchell et al. (1995) recently characterized a mutation in the transmembrane region of mIgM, that abolished antigen presentations, despite a normal uptake and degradation of the antigen (Mitchell et al., 1995).
In principle, MHC class II molecules are able to bind any peptide as well as large polypeptide fragments (Stern et al., 1994; Busch et al., 1996). Thus, if invariant chain degradation, antigen proteolysis, and MHC class II loading all occurred within the same compartment, it might be difficult to load enough class II molecules with appropriate peptides to ensure adequate expression on the cell surface (Germain and Rinker, 1993; Lanzavecchia, 1996). Segregation of the peptide loading event from the bulk endocytic pathway may thus prevent unproductive MHC class II–(poly) peptide complex formation and thereby contribute to the efficiency and specificity of antigen presentation through selective recruitment of peptide-receptive class II molecules and HLA-DM within the same subcellular compartment.
Acknowledgments
We thank Mark Dessing for help with the confocal microscopy and Dr. Mutilineos (University of Heidelberg, Heidelberg, Germany) for HLA typing of the Mel JuSo cell line; Drs. G. Hammerling, W. Knapp, E. Mellins, H.L. Ploegh, and J. Trowsdale for antibodies, and Drs. S. Gilfillan, A. Lanzavecchia, and F. Letourneur for critically reviewing the manuscript.
Abbreviations used in this paper
- CLIP
class II–associated invariant chain peptide
- HLA-DM
human leukocyte antigen-DM
- Ii
invariant chain
- MHC
major histocompatibility complex
Footnotes
The Basel Institute for Immunology was founded and is supported by F. Hoffmann-La Roche Ltd. (Basel, Switzerland).
Address all correspondence to Jean Pieters, Basel Institute for Immunology, Grenzacherstrasse 487, Postfach, CH 4005 Basel, Switzerland. Tel.: (41) 61 605 12 68. Fax: (41) 61 605 13 64. E-mail: pieters@bii.ch
References
- Amigorena S, Drake JR, Webster P, Mellman I. Transient accumulation of new MHC molecules in a novel endocytic compartment in B lymphocytes. Nature. 1994;369:113–120. doi: 10.1038/369113a0. [DOI] [PubMed] [Google Scholar]
- Amigorena S, Webster P, Drake J, Newcomb J, Cresswell P, Mellman I. Invariant chain cleavage and peptide loading in major histocompatibility complex class II vesicles. J Exp Med. 1995;181:1729–1741. doi: 10.1084/jem.181.5.1729. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Anderson MS, Miller J. Invariant chain can function as a chaperone protein for class II major histocompatibility complex molecules. Proc Natl Acad Sci USA. 1992;89:2282–2286. doi: 10.1073/pnas.89.6.2282. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Arneson LS, Miller J. Efficient endosomal localization of major histocompatibility complex class II-invariant chain complexes requires multimerization of the invariant chain targeting sequence. J Cell Biol. 1995;129:1217–1228. doi: 10.1083/jcb.129.5.1217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bakke O, Dobberstein B. MHC class II-associated invariant chain contains a sorting signal for endosomal compartments. Cell. 1990;63:707–716. doi: 10.1016/0092-8674(90)90137-4. [DOI] [PubMed] [Google Scholar]
- Blum JS, Cresswell P. Role for intracellular proteases in the processing and transport of class II HLA antigens. Proc Natl Acad Sci USA. 1988;85:3975–3979. doi: 10.1073/pnas.85.11.3975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bradford M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem. 1976;72:248–254. doi: 10.1006/abio.1976.9999. [DOI] [PubMed] [Google Scholar]
- Busch R, Cloutier I, Sekaly R-P, Hammerling GJ. Invariant chain protects class II histocompatibility antigens from binding intact polypeptides in the endoplasmic reticulum. EMBO (Eur Mol Biol Organ) J. 1996;15:418–428. [PMC free article] [PubMed] [Google Scholar]
- Castellino F, Germain RN. Extensive trafficking of MHC class II-invariant chain complexes in the endocytic pathway and appearance of peptide-loaded class II in multiple compartments. Immunity. 1995;2:73–88. doi: 10.1016/1074-7613(95)90080-2. [DOI] [PubMed] [Google Scholar]
- Cella M, Engering A, Pinet V, Pieters J, Lanzavecchia A. Inflammatory stimuli induce accumulation of MHC class II complexes on dendritic cells. Nature. 1997;388:782–786. doi: 10.1038/42030. [DOI] [PubMed] [Google Scholar]
- Chen Q, Hersey P. MHC-restricted responses of CD8+ and CD4+ T-cell clones from regional lymph nodes of melanoma patients. Int J Cancer. 1992;51:218–224. doi: 10.1002/ijc.2910510209. [DOI] [PubMed] [Google Scholar]
- Chervonsky AV, Gordon L, Sant A. A segment in the MHC class II β chain plays a critical role in targeting class II molecules to the endocytic pathway. Int Immunol. 1994;6:973–982. doi: 10.1093/intimm/6.7.973. [DOI] [PubMed] [Google Scholar]
- Cho S, Attaya M, Monaco JJ. New class II-like genes in the murine MHC. Nature. 1991;353:573–576. doi: 10.1038/353573a0. [DOI] [PubMed] [Google Scholar]
- Copier J, Kleijmeer MJ, Ponnambalam S, Oorschot V, Potter P, Trowsdale J, Kelly A. Targeting signal and subcellular compartments involved in the intracellular trafficking of HLA-DMB. J Immunol. 1996;157:1017–1027. [PubMed] [Google Scholar]
- Cresswell P. Assembly, transport, and function of MHC class II molecules. Annu Rev Immunol. 1994a;12:259–293. doi: 10.1146/annurev.iy.12.040194.001355. [DOI] [PubMed] [Google Scholar]
- Cresswell P. Antigen presentation. Getting peptides into MHC class II molecules. Curr Biol. 1994b;4:541–543. doi: 10.1016/s0960-9822(00)00119-6. [DOI] [PubMed] [Google Scholar]
- De Duve C. The separation and characterization of subcellular particles. Harvey Lect. 1965;59:49–87. [PubMed] [Google Scholar]
- Denzin, L.K., and P. Cresswell. HLA-DM induces CLIP dissociation from MHC class II alpha beta dimers and facilitates peptide loading. Cell. 82:155–165. [DOI] [PubMed]
- Engering, A., I. Lefkovits, and J. Pieters. 1997. Molecular analysis of organelles involved in MHC class II restricted antigen presentation. Electrophoresis. In press. [DOI] [PubMed]
- Germain RN. MHC-dependent antigen processing and peptide presentation: providing ligands for T lymphocyte activation. Cell. 1994;76:287–299. doi: 10.1016/0092-8674(94)90336-0. [DOI] [PubMed] [Google Scholar]
- Germain RN, Hendrix LR. MHC class II structure, occupancy and surface expression determined by post-endoplasmic reticulum antigen binding. Nature. 1991;353:134–139. doi: 10.1038/353134a0. [DOI] [PubMed] [Google Scholar]
- Germain RN, Rinker AG. Peptide binding inhibits protein aggregation of invariant-chain free class II dimers and promotes surface expression of occupied molecules. Nature. 1993;363:725–728. doi: 10.1038/363725a0. [DOI] [PubMed] [Google Scholar]
- Ghosh P, Amaya M, Mellins E, Wiley DC. The structure of an intermediate in class II MHC maturation: CLIP bound to HLA-DR3. Nature. 1995;378:457–462. doi: 10.1038/378457a0. [DOI] [PubMed] [Google Scholar]
- Griffiths G, Gruenberg J. The arguments for pre-existing early and late endosomes. Trends Cell Biol. 1991;1:5–9. doi: 10.1016/0962-8924(91)90047-d. [DOI] [PubMed] [Google Scholar]
- Griffiths G, Hoflack B, Simons K, Mellman I, Kornfeld S. The mannose 6-phosphate receptor and the biogenesis of lysosomes. Cell. 1988;52:329–341. doi: 10.1016/s0092-8674(88)80026-6. [DOI] [PubMed] [Google Scholar]
- Gruenberg J, Griffiths G, Howell KE. Characterization of the early endosomes and putative endocytic carrier vesicles in vivo and with an assay of vesicle fusion in vitro. J Cell Biol. 1989;108:1301–1316. doi: 10.1083/jcb.108.4.1301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harms E, Kern H, Schneider JA. Human lysosomes can be purified from diploid skin fibroblasts by free-flow electrophoresis. Proc Natl Acad Sci USA. 1980;77:6139–6143. doi: 10.1073/pnas.77.10.6139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hunziker W, Fumey C. A di-leucine motif mediates endocytosis and basolateral sorting of macrophage IgG Fc receptors in MDCK cells. EMBO (Eur Mol Biol Organ) J. 1994;13:2963–2967. doi: 10.1002/j.1460-2075.1994.tb06594.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Johnson JP, Demmer-Dieckmann M, Meo T, Hadam MR, Rietmuller G. Surface antigens of human melanoma cells defined by monoclonal antibodies. I. Biochemical characterization of two antigens found on cell lines and fresh tumors of diverse tissue origin. Eur J Immunol. 1981;11:825–831. doi: 10.1002/eji.1830111015. [DOI] [PubMed] [Google Scholar]
- Karlsson L, Peleraux A, Lindstedt R, Liljedahl M, Peterson PA. Reconstitution of an operational MHC class II compartment in nonantigen-presenting cells. Science. 1994;266:1569–1573. doi: 10.1126/science.7985028. [DOI] [PubMed] [Google Scholar]
- Katz JF, Stebbins C, Appella E, Sant AJ. Invariant chain and DM edit self-peptide presentation by major histocompatibility complex (MHC) class II molecules. J Exp Med. 1996;184:1747–1753. doi: 10.1084/jem.184.5.1747. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kelly AP, Monaco JJ, Cho S, Trowsdale J. A new human HLA class II-related locus, DM. Nature. 1991;353:517–573. doi: 10.1038/353571a0. [DOI] [PubMed] [Google Scholar]
- Knight AM, Luccocq JM, Prescott AR, Ponnambalam S, Watts C. Antigen endocytosis and presentation mediated by human membrane IgG1 in the absence of the Igα/Igβ dimer. EMBO (Eur Mol Biol Organ) J. 1997;16:3842–3850. doi: 10.1093/emboj/16.13.3842. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Koldovsky O, Palmieri M. Cortisone-evoked decrease of acid β-galactosidase, β-glucuronidase, N-acetyl-β-glucosaminidase and acrylsulphatase in the ileum of sucling rats. Biochem J. 1971;125:697–701. doi: 10.1042/bj1250697. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kornfeld S, Mellman I. The biogenesis of lysosomes. Annu Rev Cell Biol. 1989;5:483–525. doi: 10.1146/annurev.cb.05.110189.002411. [DOI] [PubMed] [Google Scholar]
- Kropshofer H, Vogt AB, Moldenhauer G, Hammer J, Blum JS, Hammerling GJ. Editing of the HLA-DR-peptide repertoire by HLA-DM. EMBO (Eur Mol Biol Organ) J. 1996;15:6144–6154. [PMC free article] [PubMed] [Google Scholar]
- Kropshofer H, Arndt SO, Moldenhauer G, Hammerling CJ, Vogt AB. HLA-DM acts as a molecular chaperone and rescues empty HLA-DR molecules at lysosomal pH. Immunity. 1997;6:293–302. doi: 10.1016/s1074-7613(00)80332-5. [DOI] [PubMed] [Google Scholar]
- Kvist S, Wiman K, Claesson L, Peterson PA, Dobberstein B. Membrane insertion and oligomeric assembly of HLA-DR histocompatibility antigens. Cell. 1982;29:61–69. doi: 10.1016/0092-8674(82)90090-3. [DOI] [PubMed] [Google Scholar]
- Laemmli UK. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970;227:680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Lanzavecchia A. Antigen uptake and accumulation in antigen-specific B cells. Immunol Rev. 1987;99:39–51. doi: 10.1111/j.1600-065x.1987.tb01171.x. [DOI] [PubMed] [Google Scholar]
- Lanzavecchia A. Mechanisms of antigen uptake for presentation. Curr Opin Immunol. 1996;8:348–354. doi: 10.1016/s0952-7915(96)80124-5. [DOI] [PubMed] [Google Scholar]
- Lotteau V, Teyton L, Peleraux A, Nilsson T, Karlsson L, Schmid SL, Quaranta V, Peterson PA. Intracellular transport of class II MHC molecules directed by invariant chain. Nature. 1990;348:600–605. doi: 10.1038/348600a0. [DOI] [PubMed] [Google Scholar]
- Marks MS, Roche PA, van Donselaar E, Woodruff L, Peters PJ, Bonifacino JS. A lysosomal targeting signal in the cytoplasmic tail of the beta chain directs HLA-DM to MHC class II compartments. J Cell Biol. 1995;131:351–369. doi: 10.1083/jcb.131.2.351. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marks MS, Woodruff L, Ohno H, Bonifacino JS. Protein targeting by tyrosine- and di-leucine-based signals: Evidence for distinct saturable components. J Cell Biol. 1996;135:341–354. doi: 10.1083/jcb.135.2.341. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Markwell MA. A new solid-state reagent to iodinate proteins. I. Conditions for the efficient labeling of antiserum. Anal Biochem. 1982;125:427–432. doi: 10.1016/0003-2697(82)90025-2. [DOI] [PubMed] [Google Scholar]
- Marsh M, Schmid S, Kern H, Harms E, Male P, Mellman I, Helenius A. Rapid analytical and preparative isolation of functional endosomes by free flow electrophoresis. J Cell Biol. 1987;104:875–886. doi: 10.1083/jcb.104.4.875. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mellins E, Smith L, Arp B, Cotner T, Celis E, Pious D. Defective processing and presentation of exogenous antigens in mutants with normal HLA class II genes. Nature. 1990;343:71–74. doi: 10.1038/343071a0. [DOI] [PubMed] [Google Scholar]
- Mellman I. Endocytosis and molecular sorting. Ann Rev Cell Dev Biol. 1996;12:575–625. doi: 10.1146/annurev.cellbio.12.1.575. [DOI] [PubMed] [Google Scholar]
- Mitchell RN, Barnes KA, Grupp SA, Sanchez M, Misulovin Z, Nussenzweig MC, Abbas AK. Intracellular targeting of antigens internalized by membrane immunoglobulin in B lymphocytes. J Exp Med. 1995;181:1705–1714. doi: 10.1084/jem.181.5.1705. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Murphy RF. Maturation models for endosome and lysosome biogenesis. Trends Cell Biol. 1991;1:77–82. doi: 10.1016/0962-8924(91)90022-2. [DOI] [PubMed] [Google Scholar]
- Ohno H, Stewart J, Fournier MC, Bosshart H, Rhee I, Miyatake S, Saito T, Gallusser A, Kirchhausen T, Bonifacino JS. Interaction of tyrosine-based sorting signals with clathrin-associated proteins. Science. 1995;269:1872–1875. doi: 10.1126/science.7569928. [DOI] [PubMed] [Google Scholar]
- Parton RG, Prydz K, Bomsel M, Simons K, Griffiths G. Meeting of the apical and basolateral endocytic pathways of the Madin-Darby canine kidney cell in late endosomes. J Cell Biol. 1989;109:3259–3272. doi: 10.1083/jcb.109.6.3259. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Peters PJ, Neefjes JJ, Oorschot V, Ploegh HL, Geuze HJ. Segregation of MHC Class II molecules from MHC Class I molecules in the Golgi complex for transport to lysosomal compartments. Nature. 1991;349:669–676. doi: 10.1038/349669a0. [DOI] [PubMed] [Google Scholar]
- Peters PJ, Raposo G, Neefjes JJ, Oorschot V, Leijendekker RL, Geuze HJ, Ploegh HL. Major histocompatibility complex class II compartments in human B lymphoblastoid cells are distinct from early endosomes. J Exp Med. 1995;182:325–334. doi: 10.1084/jem.182.2.325. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pierre P, Denzin LK, Hammond C, Drake JR, Amigorena S, Cresswell P, Mellman I. HLA-DM is localized to conventional and unconventional MHC class II-containing endocytic compartments. Immunity. 1996;4:229–239. doi: 10.1016/s1074-7613(00)80431-8. [DOI] [PubMed] [Google Scholar]
- Pieters J. MHC class II restricted antigen presentation. Curr Opin Immunol. 1997a;9:89–96. doi: 10.1016/s0952-7915(97)80164-1. [DOI] [PubMed] [Google Scholar]
- Pieters A. MHC Class II compartments: specialized organelles of the endocytic pathway in antigen presenting cells. Biol Chem. 1997b;378:751–758. [PubMed] [Google Scholar]
- Pieters J, Horstmann H, Bakke O, Griffiths G, Lipp J. Intracellular transport and localization of Major Histocompatibility Complex class II molecules and associated invariant chain. J Cell Biol. 1991;115:1213–1223. doi: 10.1083/jcb.115.5.1213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pieters J, Bakke O, Dobberstein B. The MHC class II-associated Invariant chain contains two endosomal sorting signals within its cytoplasmic tail. J Cell Sci. 1993;106:831–846. doi: 10.1242/jcs.106.3.831. [DOI] [PubMed] [Google Scholar]
- Pinet, V., M. Vergelli, R. Martin, O. Bakke, and E.O. Long. Antigen presentation mediated by recycling of surface HLA-DR molecules. Nature. 375:603–606. [DOI] [PubMed]
- Pond L, Watts C. Characterization of transport of newly assembled, T cell-stimulatory MHC class II-peptide complexes from MHC class II compartments to the cell surface. J Immunol. 1997;159:543–553. [PubMed] [Google Scholar]
- Qiu Y, Xu X, Wandinger-Ness A, Dalke DP, Pierce SK. Separation of subcellular compartments containing distinct functional forms of MHC class II. J Cell Biol. 1994;125:595–605. doi: 10.1083/jcb.125.3.595. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Raposo G, Nijman HW, Stoorvogel W, Leijendekker R, Harding CV, Melief CJ, Geuze HJ. B lymphocytes secrete antigen-presenting vesicles. J Exp Med. 1996;183:1161–1172. doi: 10.1084/jem.183.3.1161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Riberdy JM, Newcomb JR, Surman MJ, Barbosa JA, Cresswell P. HLA-DR molecules from an antigen-processing mutant cell line are associated with invariant chain peptides. Nature. 1992;360:474–477. doi: 10.1038/360474a0. [DOI] [PubMed] [Google Scholar]
- Roche PA. HLA-DM: an in vivo facilitator of MHC class II peptide loading. Immunity. 1995;3:259–262. doi: 10.1016/1074-7613(95)90111-6. [DOI] [PubMed] [Google Scholar]
- Roche PA. Out, damned CLIP! Out, I say! . Science. 1996;274:526–527. doi: 10.1126/science.274.5287.526. [DOI] [PubMed] [Google Scholar]
- Roche PA, Cresswell P. Proteolysis of the class II-associated invariant chain generates a peptide binding site in intracellular HLA-DR molecules. Proc Natl Acad Sci USA. 1991;88:3150–3154. doi: 10.1073/pnas.88.8.3150. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sanderson F, Kleijmeer MJ, Kelly A, Verwoerd D, Tulp A, Neefjes JJ, Geuze HJ, Trowsdale J. Accumulation of HLA-DM, a regulator of antigen presentation, in MHC class II compartments. Science. 1994;266:1566–1569. doi: 10.1126/science.7985027. [DOI] [PubMed] [Google Scholar]
- Sanderson F, Thomas C, Neefjes J, Trowsdale J. Association between HLA-DM and HLA-DR in vivo. Immunity. 1996;4:87–96. doi: 10.1016/s1074-7613(00)80301-5. [DOI] [PubMed] [Google Scholar]
- Sherman MA, Weber DA, Jensen PE. DM enhances peptide binding to class II MHC by release of invariant chain-derived peptide. Immunity. 1995;3:197–205. doi: 10.1016/1074-7613(95)90089-6. [DOI] [PubMed] [Google Scholar]
- Sloan VS, Cameron P, Porter G, Gammon M, Amaya M, Mellins E, Zaller DM. Mediation by HLA-DM of dissociation of peptides from HLA-DR. Nature. 1995;375:802–806. doi: 10.1038/375802a0. [DOI] [PubMed] [Google Scholar]
- Stebbins CC, Peterson ME, Suh WM, Sant AJ. DM-mediated release of a naturally occurring invariant chain degradation intermediate from MHC class II molecules. J Immunol. 1996;157:4892–4898. [PubMed] [Google Scholar]
- Steinman RM, Swanson J. The endocytic activity of dendritic cells. J Exp Med. 1995;182:283–288. doi: 10.1084/jem.182.2.283. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stern LJ, Brown JH, Jardetzky TS, Gorga JC, Urban RG, Strominger JL, Wiley DC. Crystal structure of the human class II MHC protein HLA-DR1 complexed with an influenza virus peptide. Nature. 1994;368:215–221. doi: 10.1038/368215a0. [DOI] [PubMed] [Google Scholar]
- Teyton L, O'Sullivan D, Dickson PW, Lotteau V, Sette A, Fink P, Peterson PA. Invariant chain distinguishes between the exogenous and endogenous antigen presentation pathways. Nature. 1990;348:39–44. doi: 10.1038/348039a0. [DOI] [PubMed] [Google Scholar]
- Tulp A, Verwoerd D, Pieters J. Application of an improved density gradient electrophoresis apparatus to the separation of proteins, cells and subcellular organelles. Electrophoresis. 1993;14:1295–1301. doi: 10.1002/elps.11501401198. [DOI] [PubMed] [Google Scholar]
- Tulp A, Verwoerd D, Dobberstein B, Ploegh HL, Pieters J. Isolation and characterization of the intracellular MHC class II compartment. Nature. 1994;369:120–126. doi: 10.1038/369120a0. [DOI] [PubMed] [Google Scholar]
- van Ham SM, Gruneberg U, Malcherek G, Broker I, Melms A, Trowsdale J. Human histocompatibility leukocyte antigen (HLA)-DM edits peptides presented by HLA-DR according to their ligand binding motifs. J Exp Med. 1996;184:2019–2024. doi: 10.1084/jem.184.5.2019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Warmerdam PA, Long EO, Roche PA. Isoforms of the invariant chain regulate transport of MHC class II molecules to antigen processing compartments. J Cell Biol. 1996;133:281–291. doi: 10.1083/jcb.133.2.281. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Watts C, Davidson HW. Endocytosis and recycling of specific antigen by human B cell lines. EMBO (Eur Mol Biol Organ) J. 1988;7:1937–1945. doi: 10.1002/j.1460-2075.1988.tb03031.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weber, D.A., B.D. Evavold, and P.E. Jensen. Enhanced dissociation of HLA-DR-bound peptides in the presence of HLA-DM. Science 274:618–620. [DOI] [PubMed]
- West MA, Lucocq JM, Watts C. Antigen processing and class II MHC peptide-loading compartments in human B-lymphoblastoid cells. Nature. 1994;369:147–151. doi: 10.1038/369147a0. [DOI] [PubMed] [Google Scholar]
- Wolf PR, Ploegh HL. How MHC class II molecules acquire peptide cargo: Biosynthesis and trafficking through the endocytic pathway. Annu Rev Cell Dev Biol. 1995;11:267–306. doi: 10.1146/annurev.cb.11.110195.001411. [DOI] [PubMed] [Google Scholar]
- Zachgo S, Dobberstein B, Griffiths G. A block in degradation of MHC class II-associated invariant chain correlates with a reduction in transport from endosome carrier vesicles to the prelysosomal compartments. J Cell Sci. 1992;103:811–822. doi: 10.1242/jcs.103.3.811. [DOI] [PubMed] [Google Scholar]