Abstract
The β chemokine known as 6-C-kine, secondary lymphoid-tissue chemokine (SLC), TCA4, or Exodus-2 (herein referred to as 6CK/SLC) can trigger rapid integrin-dependent arrest of lymphocytes rolling under physiological shear and is highly expressed by high endothelial venules, specialized vessels involved in lymphocyte homing from the blood into lymph nodes and Peyer's patches. We show that 6CK/SLC is an agonist for the lymphocyte chemoattractant receptor, CCR7 (EBI-1, BLR-2), previously described as a receptor for the related β chemokine MIP-3β (ELC or Exodus-3). Moreover, 6CK/SLC and MIP-3β attract the same major populations of circulating lymphocytes, including naive and memory T cells > B cells (but not natural killer cells); desensitization to MIP-3β inhibits lymphocyte chemotaxis to 6CK/SLC but not to the α chemokine SDF-1 (stromal cell–derived factor); and 6CK/SLC competes for MIP-3β binding to resting mouse lymphocytes. The findings suggest that the majority of circulating lymphocytes respond to 6CK/SLC and MIP-3β in large part through their common receptor CCR7 and that these molecules may be important mediators of physiological lymphocyte recirculation in vivo.
Lymphocytes cycle throughout the body and are in constant flux, traveling long distances via the circulation, entering tissues where they may encounter antigens and eventually reentering the circulation. Secondary lymphoid organs (lymph nodes, Peyer's patches, and spleen) are major sites of lymphocyte traffic, as these organs are specialized for the support of antigen presentation and for facilitating lymphocyte interactions. Lymphocyte recruitment from the blood into lymph nodes and Peyer's patches and into sites of severe chronic inflammation is controlled by interactions with specialized high endothelial venules (HEV).1 These interactions are thought to be mediated by a multistep process, involving in many instances: (1a) tethering of blood borne cells through receptors presented on microvilli, followed by (1b) rolling along the vessel wall, (2) rapid activation of lymphocyte integrins, (3) integrin-dependent adhesion resistant to shear, and (4) diapedesis (2, 8, 36). The rapid adhesion triggering (step 2) and transendothelial migration (step 4) are thought to be mediated by Gi α-linked receptors of the chemoattractant subfamily. Chemoattractant cytokines (chemokines) act through this class of receptors (1, 30, 40), and it has been speculated that they mediate these lymphocyte trafficking events (7, 8, 31, 32).
In the context of physiological lymphocyte recirculation, recent studies have focused our attention on two chemokines in particular: the β chemokine 6-C-kine or secondary lymphoid-tissue chemokine (6CK/SLC; also known as TCA4 and Exodus-2) (15, 17, 24, 34), whose receptor has not been defined, and MIP-3β (ELC, Exodus-3) (27, 39), whose receptor is CCR7 (also known as EBI-1 and BLR-2). Although antibodies to these chemokines remain to be produced, Northern blot and cDNA library analyses suggest that 6CK/SLC and MIP-3β are most highly expressed in lymphoid organs (15, 17, 24, 27, 39). Of particular interest is the finding that 6CK/SLC message is abundant in HEV (14), the vessels involved in lymphocyte recruitment into lymph nodes and Peyer's patches in vivo, and that 6CK/SLC and MIP-3β can trigger rapid β2 integrin activation in a major proportion of resting human and mouse lymphocytes, inducing adhesion to ICAM-1 and promoting rapid lymphocyte arrest under physiologic shear (12). 6CK/SLC is also a highly selective chemoattractant for lymphocytes, attracting naive but also memory T cells and, to a lesser extent, B cells (14, 17). Although chemotactic activity of MIP-3β on resting lymphocytes has not been reported previously, several T cell lines are attracted efficiently to MIP-3β, and neither 6CK/SLC nor MIP-3β appear to act on monocytes or granulocytes (14, 17, 34, 39). This is an important issue because circulating monocytes and neutrophils share with lymphocytes the L-selectin and β2 integrin adhesion molecules required for rolling and arrest in lymph node HEVs, leading to the current hypothesis that the adhesion triggering (and potentially chemoattractant) agonists associated with these specialized venules must be responsible for the highly selective lymphocyte accumulation observed, and thus must preferentially or exclusively act on recirculating lymphocytes (18, 19, 36). In contrast to 6CK/SLC and MIP-3β, message for the other major chemoattractant for recirculating lymphocytes, stromal cell–derived factor 1α (SDF-1α), is widely expressed in diverse tissues (35), and SDF-1 attracts monocytes, natural killer (NK) cells, and other cell types as well (reference 5 and vide infra). Other chemokines with reported attractant activity for subsets of circulating lymphocytes, such as MCP-1 and IL-8, also lack lymphocyte selectivity, displaying prominent chemoattractant activities for myeloid or monocytic cells (1, 28, 30).
Here we report that the HEV-expressed chemokine 6CK/SLC signals through the MIP-3β receptor CCR7. Moreover, the same major lymphocyte subpopulations that chemotax to 6CK/SLC also migrate to MIP-3β, and desensitization of resting lymphocytes to MIP-3β inhibits their responses to 6CK/SLC, as well. We propose that, along with the specialized adhesion/homing receptors involved, 6CK/SLC, MIP-3β, and their common receptor CCR7 may play an important role in the physiological recirculation and homing of lymphocytes.
Materials and Methods
Preparation of Human and Mouse Lymphocytes
Human peripheral blood was collected in heparinized tubes from healthy donors. Granulocytes were removed as described (25). Monocytes were removed by two 30-min rounds of adherence to a culture flask (model T-175; Nunc, Roskilde, Denmark) at 37°C and 8% CO2 in RPMI 1642 medium supplemented with 10% calf serum. Mouse lymph nodes (cervical, axillary, inguinal, and mesenteric) were gently ground between the frosted ends of microscope slides and filtered through nylon mesh, and adherent cells were removed as above. BALB/c or C57BL/6 mice were used, as indicated.
Chemotaxis
All migration assays were performed in 24-well plate tissue culture inserts (Costar Corp., Cambridge, MA) with 5-μm pore polycarbonate filters. 5 × 105 cells were placed in the upper chamber in 100 μl, and 600 μl of a given chemokine dilution in the lower well (unless otherwise indicated, as for the checkerboard assays). Recombinant human and mouse 6CK/SLC were obtained from R&D Systems (Minneapolis, MN). Synthetic human MIP-3β and SDF-1α were synthesized by the authors (M.A. Siani and D. Thomas, Gryphon Sciences) by chemical ligation (13). Human MIP-3β and SDF-1α were used for both the human and mouse systems since the murine MIP-3β homologue has not yet been reported. The use of human MIP-3β on mouse lymphocytes is validated by its cross-reactivity with mouse CCR7 (see Figs. 1 and 2), although the K d may not be identical. Human and mouse SDF-1α differ by only two conservative amino acid substitutions (35), and their activities are reportedly similar. Chemotaxis medium consisted of RPMI-1642/10% calf serum for mouse lymphocytes and L1/2 cells, and RPMI-1642/0.5%BSA Fr. V (Sigma Chemical Co., St. Louis, MO) for human lymphocytes. Migration times were 30–60 min for mouse lymphocytes, 90 min for human lymphocytes, and 3 h for L1/2 cells. Migrated cells were counted as described (11, 12). All migration assays were carried out at 37°C and 8% CO2.
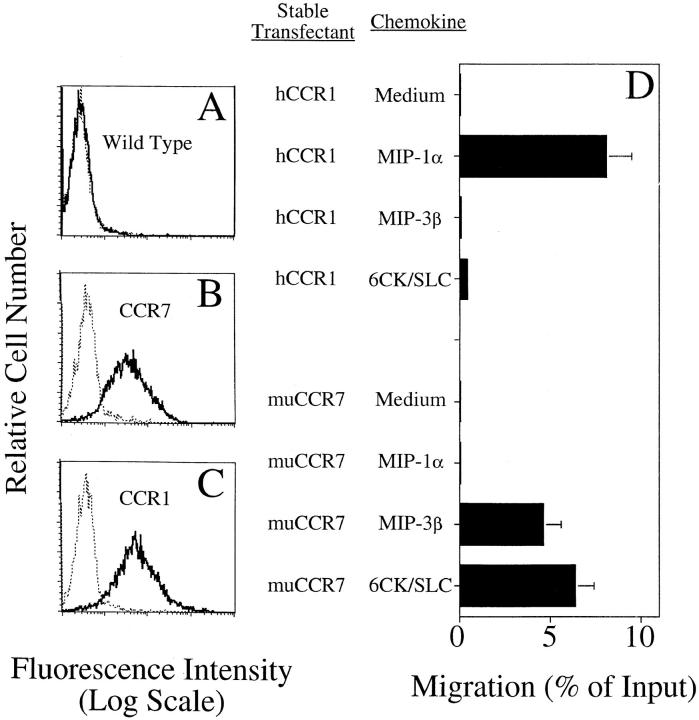
Figure 1.
6CK/SLC is a chemotactic agonist for the lymphocyte chemokine receptor CCR7 (stable L1/2 transfectants). (A–C) Anti-FLAG staining of Giα-linked receptor L1/2 transfectants. Solid line, M1 anti-FLAG mAb; dotted line, isotype control. Staining of (A) wild-type L1/2, (B) FLAGged CCR7 transfectants, and (C) FLAGged CCR1 transfectants. (D) Chemotaxis of L1/2 cells stably transfected with muCCR7 or hCCR1 to various chemokines or control medium. Stably transfected cells were selected to homogeneity by several rounds of sorting for the FLAG epitope tag. Mean and range of duplicate wells is shown. Representative of three individual experiments.
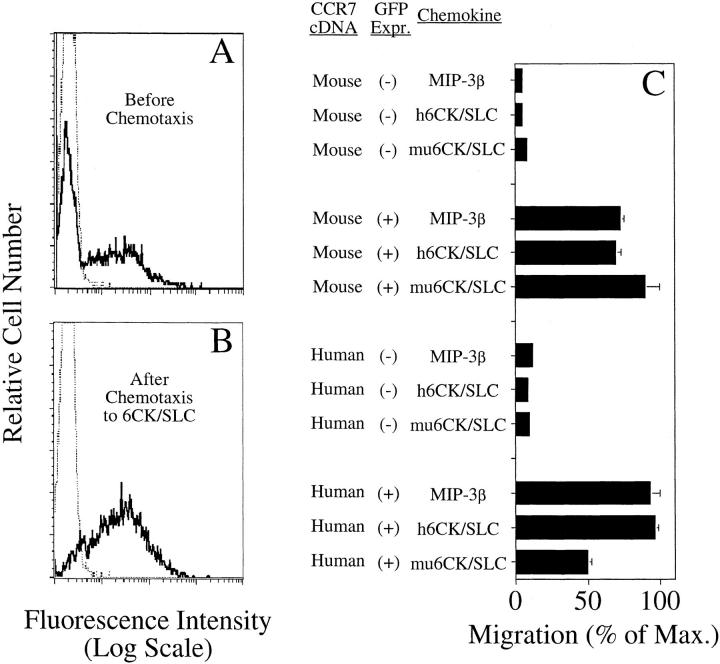
Figure 2.
6CK/SLC is a chemotactic agonist for the lymphocyte chemokine receptor CCR7 (transient CCR7 transfectants). (A and B) GFP expression of transient CCR7 transfectants. (A) hCCR7/GFP transient transfectants before chemotaxis (solid line) overlaid on mock transfected control (dotted line). (B) hCCR7/GFP transient transfectants after chemotaxis to h6CK/ SLC (solid line) overlaid on mock transfected control (dotted line). (C) Chemotaxis of transient CCR7 transfectants. Cells were transfected with either human or mouse CCR7 cDNA constructs along with a GFP vector. 24 h after transfection, when the chemotaxis was performed, the muCCR7 population was ∼25% GFP(+), and the hCCR7 population was ∼42% GFP(+). For each receptor/chemokine combination, chemotaxis was performed in six replicate wells, and the number of migrated cells enumerated as described previously (10, 11). Migrated cells in the replicate wells of each receptor/chemokine combination were then pooled and analyzed by flow cytometry for the frequency of GFP-expressing (transfected) cells. The number of GFP(+) and GFP(−) cells was determined for the starting population and for each migrated population, and the percent migration was determined from these numbers. Maximum migration was 4.8% for hCCR7 transfectants and 2.6% for muCCR7 transfectants. Mean and range of duplicate wells are shown. Representative of two individual experiments.
Optimal chemotactic doses for each chemokine on each cell type were determined by testing 10-fold dilutions between 1 μM and 1 nM. Twofold dilutions were then carried out within the most effective decade. The optimal chemotactic doses were: 100 nM for murine 6CK/SLC on mouse lymphocytes and L1/2 transfectants; 1 μM for human 6CK/SLC on human lymphocytes and 100 nM on L1/2 transfectants; 50 nM for human SDF-1α on mouse lymphocytes and 100 nM on human lymphocytes; and 25 nM human MIP-3β on mouse lymphocytes, 100 nM on L1/2 transfectants, and 1 μM on human lymphocytes.
Determination of Lymphocyte Subsets
The number of cells of each subtype was determined for the starting population of each chemotactic assay. The number of cells belonging to the same subtype was determined in the migrated population, and the percent migration was determined from these two numbers. Six to eight wells were chemotaxed for each chemokine, and the migrated cells were pooled (after counting) to obtain sufficient cells for flow cytometry. Directly conjugated antibodies used in this study were obtained from PharMingen, Inc. (San Diego, CA) unless otherwise indicated. Anti–mouse reagents: FITC-conjugated CD8 (clone 53-6.7) and CD44 (clone MJ-64, grown and conjugated by Butcher lab staff), APC-conjugated CD4 (clone GK1.5, a generous gift of S. Michie, Stanford University, Stanford, CA), biotinylated CD3ε (clone 145-2C11), and TCRαβ (clone H57-597). Anti–human reagents: FITC-conjugated CD3 (clone UCHT1) and CD45RA (clone HI100); PE-conjugated CD19 (clone B43), CD45RO (clone UCHL1), and CD56 (clone B159); biotinylated CD4 (clone RPA-T4), CD8 (clone RPA-T8), and CD16 (clone 3G8). The second stage reagent used for all biotinylated antibodies was streptavidin-conjugated PerCP (Becton Dickinson, San Jose, CA). Flow cytometry analysis was performed on a FACSCalibur® driven by Cell Quest software (Becton Dickinson).
CCR7 Transfectants
L1/2 cells were stably transfected with epitope-tagged receptors as described (10, 11, 16). The FLAGged muCCR7 cDNA was obtained from BALB/c lymph node RNA using PCR primers based on GenBank accession number L31580 and inserted into a cytomegalovirus-driven vector (pMRB-101). Human CCR7 (data available from GenBank/EMBL/DDBJ under accession number L31581) cDNA was tagged at the NH2 terminus with a hemagglutin in epitope (YPYDVPDYA) and subcloned into pCDNA3. For transient transfections, L1/2 cells were cotransfected by electroporation with these same constructs and the green fluorescent protein (GFP) expression vector pGreen Lantern (GIBCO BRL, Gaithersburg, MD).
Receptor Binding Studies
Interaction of 6CK/SLC with a common lymphocyte receptor was assessed by direct competition for radioactive MIP-3β binding. Mouse lymph node lymphocytes were incubated with 125I-MIP-3β (obtained from New England Nuclear [Boston, MA] and Amersham Corp. [Arlington Heights, IL]; 0.2 nM) in the presence or absence of 200 nM cold MIP-3β, 6CK/SLC, or MCP-1 (R&D Systems). Chemokine binding to mouse lymphocytes was carried out using a modification of a method described previously (38). Briefly, mouse lymphocyte cells were prepared from lymph nodes, washed, and resuspended in binding buffer (50 mM Hepes, pH 7.5, 1 mM CaCl2, 5 mM MgCl2, and 0.5% BSA) at 2 × 107 cells/ml. For each binding reaction (in a final volume of 100 ml), 25 μl of cell suspension (5 × 105 cells) was mixed with 0.2 nM labeled chemokine, in the presence or absence of 200 nM unlabeled chemokines. The reactions were incubated at room temperature for 60 min and stopped by transferring the mixture to GFB filter plates, which were then washed three times with binding buffer containing 0.5 M NaCl. The plates were dried, and MicroScint scintillation fluid was added before counting. The bound radioactivity was determined and used to calculate the percent inhibition by each of the competitors. Two independent experiments were performed showing inhibition by cold MIP-3β and 6CK/SLC, each with duplicate or triplicate determinations. MCP-1 inhibition was assessed in one experiment in triplicate.
Results and Discussion
6CK/SLC Mediates Chemotaxis of CCR7 Transfectants
In a method described previously (10, 11, 16), L1/2 cells (an Abelson-transformed murine pre-B cell line) were stably transfected with an epitope-tagged (FLAGged) version of mouse CCR7 (Fig. 1). As expected, FLAGged CCR7 transfectants, but not CCR1 transfectants, were attracted efficiently to the known CCR7 ligand MIP-3β. As shown in Fig. 1 D, mouse 6CK/SLC also attracted the CCR7 transfectants, but not the CCR1 transfectants.
When working with stably transfected cell lines, it is always formally possible that observed changes in cell behavior are brought about by selection pressures unrelated (or only indirectly related) to the transfected gene. To confirm that the 6CK/SLC-induced migration was mediated by the transfected CCR7, transient transfections were performed. Transient transfection of L1/2 cells creates a heterogeneous population of receptor-expressing cells successfully taking up and expressing transfected plasmid (Fig. 2 A): In this system, cotransfected plasmids are coordinately expressed (Bowman, E.P., unpublished data). Cells transiently expressing transfected human or mouse CCR7 sequences (GFP[+]) migrated efficiently towards MIP-3β and 6CK/SLC (Fig. 2 C), while those cells that did not take up the electroporated DNA (GFP[−]) remained unresponsive to these chemokines. We conclude that CCR7 is a receptor for 6CK/SLC as well as for MIP-3β. Importantly, CCR7, also known as EBI-1 or BLR-2 (3, 6), is constitutively expressed at the Northern level in human PBMC (3) and is highly represented in Southern blots of complex cDNA libraries from L-selectin+ naive T cells and Ig+ B cells from mouse spleen (Zlotnik, A., unpublished data). CCR7 is thus likely to be a prominent receptor for 6CK/SLC on circulating lymphocytes.
MIP-3β, as well as 6CK/SLC, Is a Potent Chemoattractant for Resting Lymphocytes
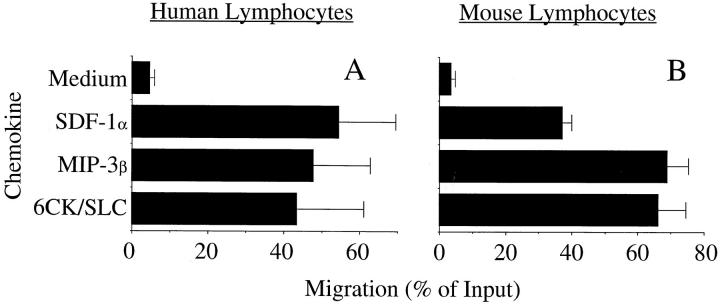
We reasoned that if 6CK/SLC and MIP-3β share CCR7 as a prominent receptor on lymphocytes, then MIP-3β should attract the major populations of circulating peripheral blood lymphocytes and mouse lymph node (LN) lymphocytes that respond to 6CK/SLC, including naive lymphocytes. Indeed, we found that the majority of both human and mouse lymphocytes responding efficiently in Transwell assays to MIP-3β, with chemotaxis comparable to that of 6CK/SLC (Fig. 3). Checkerboard analyses confirmed that the responses to MIP-3β and 6CK/SLC were chemotactic (not shown). Thus, both 6CK/SLC and MIP-3β are not only potent adhesion-triggering chemokines for most resting lymphocytes (12), but they are also potent circulating lymphocyte chemoattractants.
Figure 3.
MIP-3β, like 6CK/SLC, is a potent attractant for resting human and mouse lymphocytes. (A) Migration of freshly isolated human circulatory lymphocytes through 5-μm pores for 90 min. The optimal chemotactic dose was used for each chemokine (see Materials and Methods). Medium, the number of cells migrating into the bottom well in the absence of chemokine. Mean of two experiments (eight wells each) with individual healthy donors is shown; error bars indicate range. (B) Migration of freshly isolated mouse lymph node lymphocytes for 60 min through 5-μm pores. The optimal chemotactic dose was used. Mean of two experiments with different mouse strains (BALB/c and C57BL/6) is shown; error bars indicate range.
MIP-3β and 6CK/SLC Attract Similar Major Populations of Resting Human and Mouse Lymphocytes
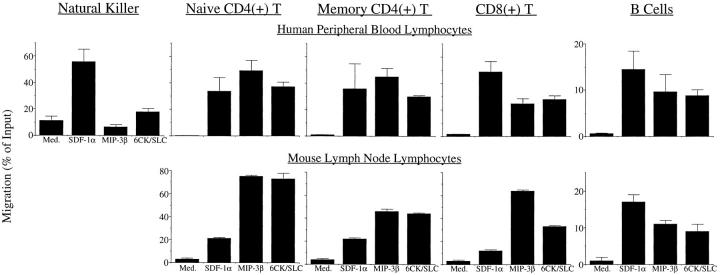
The relative migration efficiency of various lymphocyte types was determined by noting the number of each subtype in the starting population (as determined by flow cytometry) and observing what percentage of this specific population migrated to the bottom well in response to optimal chemotactic concentrations of MIP-3β, 6CK/SLC, or, for comparison, SDF-1α. CD4(+) helper T cells form the largest subpopulation of circulating human lymphocytes and can be divided into naive and memory phenotypes based on their expression of CD45RA vs. CD45RO (29). CD4(+) T cells are also a major subpopulation in mouse lymph node, and naive vs. memory subpopulations can be distinguished by relative CD44 levels in C57BL/6 mice (21). Both naive and memory helper T cells (Fig. 4) were attracted efficiently to all three chemokines, as were CD8(+) cytotoxic T and B cells. The NK population (defined as CD3[−], CD56[+], and CD16[+]), however, showed strong differences in its ability to respond to these chemokines. This lymphocyte subset was only studied in the human system, as it is not a major population in healthy mouse lymph nodes. It is interesting to note that the background migration of NK cells was higher than that of any other population studied in this report (up to 20% in some donors) and that NK cells comprised ∼80% of all human lymphocytes that migrated in the absence of chemokine in our system (not shown). Although the background migration was high, a gradient of SDF-1α more than tripled the number of migrating NK cells, whereas MIP-3β and 6CK/SLC had no effect.
Figure 4.
MIP-3β and 6CK/SLC attract similar lymphocyte subpopulations. (Top row) Percent migration of resting human peripheral blood lymphocytes. Subsets defined by multiparameter FACS® analysis as follows: natural killer = CD3(−), CD56(+), CD16(+); naive CD4(+) = CD4(+), CD45RA(+), CD45RO(−); memory CD4(+) = CD4(+), CD45RA(−), CD45RO(+); CD8(+) T cells = CD3(+), CD8(+); B cells = CD3(−), CD19(+). Migration through 5-μm pores for 90 min. Mean of two experiments (eight wells each) with individual healthy donors is shown; error bars indicate range. (Bottom row) Percent migration of resting mouse lymphocytes defined as follows: naive CD4(+) = CD3(+), CD4(+), CD44(lo); memory CD4(+) = CD3(+), CD4(+), CD44(hi); CD8(+) T cells = TCRαβ(+), CD8(+); B cells = TCRαβ(−), B220(+). Migration through 5-μm pores for 60 min. Mean of two individual experiments (six wells each) with 20-wk C57BL/6 is shown; error bars indicate range.
Thus, with the exception of NK cells (a population that does not normally traffic through secondary lymphoid tissue), MIP-3β and 6CK/SLC were potent attractors of each of the major human and mouse lymphocyte subpopulations. Different cell types were attracted with varying efficiency. However, in our opinion, the quantitative differences in B vs. T cell chemotaxis to these chemokines in vitro must be interpreted with caution since B cells also migrate less efficiently in response to SDF-1α. In the absence of a “control” B cell attractant that recruits the majority of B cells across Transwell membranes, one cannot exclude that mechanical limitations (e.g., deformability) or other cell properties limit in vitro B cell responses. On the other hand, additional chemokines may support B cell recruitment, and it is also worth pointing out that B cell recirculation is in general slower than that of T cells in vivo, as well.
It is interesting to note that the optimal chemotactic doses of MIP-3β and 6CK/SLC differed by 10-fold between human circulating lymphocytes and mouse lymph node lymphocytes in our system. However, when human or mouse CCR7 were expressed in L1/2 cells, the optimal chemotactic doses were indistinguishable (100 nM, similar to those for mouse lymph node cells). This suggests that the cellular environment in which the receptor is expressed may influence its interaction with ligand and/or its signaling.
MIP-3β Inhibits Lymphocyte Chemotaxis to 6CK/SLC but Not to SDF-1
Cross-desensitization assays are an established tool for assessing common receptor usage by chemokines: Binding of agonist to chemokine receptors induces rapid homologous desensitization, inhibiting responses to other agonists acting through the same receptor, but not to those interacting with independent receptors (for review see reference 1). Therefore, we postulated that if 6CK/SLC acts primarily through the MIP-3β receptor on lymphocytes, then MIP-3β preincubation should inhibit its activity. The mouse system was used for these studies because much lower concentrations of chemokines were needed for optimal chemotaxis to 6CK/SLC and MIP-3β than were required for the human system (see Materials and Methods).
Mouse lymphocytes were treated for 30 min with MIP-3β (at 5× the optimal chemotactic dose) or control medium, washed quickly by centrifugation through a layer of serum, and then immediately placed in a chemotaxis assay. Ability to migrate towards an optimal concentration of 6CK/ SLC was then tested. The desensitization process did not appreciably increase background migration (not shown). MIP-3β quite effectively desensitized migration to 6CK/ SLC, whereas the response to SDF-1α (which acts through an independent receptor CXCR4; references 4, 26) was not inhibited (Fig. 5). Similarly, 6CK/SLC preincubation abrogated lymphocyte chemotaxis to MIP-3β. In contrast, preincubation with SDF-1α inhibited its own responses, with only minor effects on migration to the CCR7 agonists.
Figure 5.

MIP-3β desensitizes lymphocyte chemotaxis to 6CK/ SLC. Mouse lymph node cells were pretreated with 5× the optimal chemotactic dose of each chemokine (indicated under each column) and then washed and allowed to migrate towards the same or another chemokine (at the optimal chemotactic dose) through 5-μm pores for 15 min. This short chemotactic period was used because resensitization of pretreated cells was quite rapid: ∼50% of homologous desensitization was lost if chemotaxis was extended to 30 min. Less migration was achieved at 15 min than when the experiments were allowed to go to completion, but the number of migrated cells was sufficiently high for reliable reproducibility: mean migration of control pretreated cells was ∼25% of input for MIP-3β, ∼15% for 6CK/SLC, and ∼10% for SDF-1α. Mean of two experiments, each with duplicate wells is presented; error bars indicate range.
6CK/SLC Inhibits MIP-3β Binding to Lymphocytes
We reasoned that if CCR7 is a prominent receptor for 6CK/SLC and MIP-3β on lymphocytes, then 6CK/SLC should inhibit MIP-3β binding to the lymphocyte surface. Mouse lymph node cells bound 125I–MIP-3β, and this binding was significantly inhibited by excess cold MIP-3β (62 and 78% in two separate experiments) and to a similar extent by 6CK/SLC (69 and 73%). In contrast, cold MCP-1 had no significant effect. Thus, the predominant MIP-3β receptor on resting lymphocytes is shared by 6CK/SLC.
Our results demonstrate that two major lymphocyte chemoattractants of lymphoid tissues—6CK/SLC, expressed by HEV(15), and MIP-3β, also prominently expressed in LN, Peyer's patch (PP), and spleen (27, 39)— share a common receptor and attract the major circulating lymphocyte subsets that traffic through secondary lymphoid organs. Both chemokines are potent attractants for naive T cells, but also recruit memory CD4(+) T cells and CD8(+) T cells and B cells, all populations that enter lymphoid tissues via HEV with varying efficiency (8, 9, 22, 33, 37). Although murine MIP-3β protein was not available for these experiments, a mouse sequence with 74% amino acid identity to human MIP-3β is found in the EST database (available from GenBank/EMBL/DDBJ under accession number AA444730), suggesting that a mouse version of MIP-3β protein does indeed exist in vivo. In light of the functional similarities between 6CK/SLC and MIP-3β, including preferential, high levels of mRNA expression in secondary lymphoid organs as well as shared receptor usage and chemoattractant activity for lymphocytes as shown here, it is of interest that these two CC chemokines share a common chromosomal location (24). Thus, even though they display only 33% amino acid identity in the chemokine domain, they may well have arisen from a relatively recent common lymphocyte chemoattractant precursor. The evolutionary advantage of having two related agonists for CCR7 is unclear at present, but such promiscuity is not uncommon among the chemokines and their receptors (1, 29). It will be of interest to determine the extent of specialization between 6CK/SLC and MIP-3β in controlling lymphocyte migration into and within lymphoid organs, and to ask how such specialization relates to the clear structural differences between them, especially the 38–amino acid COOH-terminal extension of 6CK/ SLC. Since 6CK/SLC is abundantly expressed by HEV by in situ analyses, one can postulate that its COOH-terminal extension may have a role in stabilizing or facilitating its presentation at the vascular lumen, where it may help trigger β2 integrin–dependent lymphocyte arrest.
Recent studies show that 6CK/SLC can also signal through CXCR3, a receptor that also supports chemotaxis to the α chemokines MIG and IP-10 (Zlotnik, A., unpublished data). Importantly, responses to MIG and IP-10 are reported to be predominantly limited to in vitro–activated T cells, with little effect on the bulk of freshly isolated PBL (20, 23). Moreover, MIP-3β does not signal though CXCR3. Thus, CXCR3 may contribute to the migration of activated T cells in vivo, but is unlikely to play a significant role in the major, especially naive, B and T cell responses to 6CK/SLC and MIP-3β discussed here.
It is important to consider the potential contribution of chemokines to the specificity of lymphocyte trafficking in the context of the specialized selectin, integrin, and other adhesion/homing receptors involved. As mentioned in the introduction, although most circulating white blood cells express L-selectin and β2 integrins required for rolling and arrest in lymph node HEV (36), and eosinophils and subsets of NK cells, at least, also display the α4β7 integrin intestinal homing receptor involved in homing to Peyer's patches, these cells do not normally enter LN or PP efficiently from the blood in the absence of significant inflammation. Such observations, in fact, led to the initial hypothesis of a generalized multistep model of leukocyte– endothelial recognition, in which an essential, rapid activation event required for integrin-mediated adhesion was postulated to control specificity of lymphocyte (vs. monocyte and granulocyte) arrest in HEV (7). In this context, the apparent selectivity of 6CK/SLC and MIP-3β for recirculating lymphocyte populations, and their lack of activity on monocytes, neutrophils, and NK cells, would allow them to make a critical contribution to the selectivity of lymphocyte–HEV recognition. On the other hand, each of these chemokines appears to be expressed in both intestinal (PP, appendix) and peripheral (LN, spleen) lymphoid organs (15, 17, 24, 27, 39). Thus, while they may permit lymphocyte but not other leukocyte arrest in these sites, differential adhesion/homing receptor requirements (e.g., α4β7-MAdCAM-1 in PPs) may remain the primary determinants for the targeted homing and segregation of intestinal vs. peripheral memory lymphocytes to PPs vs. peripheral lymphoid tissues (8).
Finally, the finding that CCR7 is a shared receptor for the HEV-expressed chemokine 6CK/SLC, as well as for MIP-3β, suggests that it may itself be an important lymphocyte “homing receptor” for secondary lymphoid organs. In vivo studies will be required, however, to explore further the involvement of CCR7 in physiological lymphocyte trafficking, to elucidate the relative contributions of 6CK/SLC and MIP-3β, and to define their roles in physiological lymphocyte recirculation and in lymphocyte homing during pathological inflammation.
Acknowledgments
We thank S. Michie for APC-conjugated GK1.5, J. Twelves for FITC-conjugated MJ-64, S. Grossman for administrative assistance, and L. Rott for expert flow cytometry support.
This work was supported by the National Institutes of Health (NIH) grant GM37743 (E.C. Butcher), Public Health Service grant 5T32CA09151 (E.P. Bowman), the NIH Cancer Etiology, Prevention, Detection, and Diagnosis Training Grant 5T32CA090302 (J.J. Campbell and K.R. Youngman), NIH Individual National Research Service Award 1F32AI08930 (J.J. Campbell), and by the FACS® Core facility of the Stanford Digestive Diseases Center under DK38707. E.P. Bowman and J.J. Campbell are recipients of Arthritis Foundation Postdoctoral Fellowships.
Abbreviations used in this paper
- 6CK
6-C-kine
- GFP
green fluorescent protein
- HEV
high endothelial venules
- LN
lymph node(s)
- NK
natural killer
- PP
Peyer's patch(es)
- SDF
stromal cell–derived factor
- SLC
secondary lymphoid-tissue chemokine
Note Added in Proof
Since submission of this manuscript, Kim et al. (J. Immunol. 1998. 160:2418) have reported that MIP-3β (CKβ-11) induced chemotaxis of blood B and T cells, and Yoshida et al. (J. Biol. Chem. 1998. 273:7118) have confirmed that CCR7 is a receptor of 6CK/SLC.
Footnotes
Address all correspondence to James J. Campbell, Ph.D., Department of Pathology, Stanford University School of Medicine, Stanford, CA 94305. Tel.: (650) 493-5000 (x6-3133). Fax: (650) 858-3986. E-mail: jcampbel@cmgm.stanford.edu
References
- 1.Baggiolini M, Dewald B, Moser B. Human chemokines: an update. Annu Rev Immunol. 1997;15:675–705. doi: 10.1146/annurev.immunol.15.1.675. [DOI] [PubMed] [Google Scholar]
- 2.Bargatze RF, Jutila MA, Butcher EC. Distinct roles of L-selectin and integrins α4β7 and LFA-1 in lymphocyte homing to Peyer's patch-HEV in situ: the multistep model confirmed and refined. Immunity. 1995;3:99–108. doi: 10.1016/1074-7613(95)90162-0. [DOI] [PubMed] [Google Scholar]
- 3.Birkenbach M, Josefsen K, Yalamanchill R, Lenoir G, Kieff E. Epstein-Barr virus-induced genes: first lymphocyte-specific G protein-coupled receptors. J Virol. 1993;67:2209–2220. doi: 10.1128/jvi.67.4.2209-2220.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Bleul CC, Farzan M, Choe H, Parolin C, Clark-Lewis I, Sodrosky J, Springer TA. The lymphocyte chemoattractant SDF-1 is a ligand for LESTR/FUSIN and blocks HIV entry. Nature. 1996;392:829–833. doi: 10.1038/382829a0. [DOI] [PubMed] [Google Scholar]
- 5.Bleul CC, Fuhlbrigge RC, Casanovas JM, Aiuti A, Springer TA. A highly efficacious lymphocyte chemoattractant, stromal cell– derived factor 1 (SDF-1) J Exp Med. 1996;184:1101–1109. doi: 10.1084/jem.184.3.1101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Burgstahler R, Kempkes B, Steube K, Lipp M. Expression of the chemokine receptor BLR2/EBI1 is specifically transactivated by Epstein-Barr virus nuclear antigen 2. Biochem Biophys Res Commun. 1995;215:738–743. doi: 10.1006/bbrc.1995.2525. [DOI] [PubMed] [Google Scholar]
- 7.Butcher EC. Leukocyte-endothelial cell recognition: three (or more) steps to specificity and diversity. Cell. 1991;67:1033–1037. doi: 10.1016/0092-8674(91)90279-8. [DOI] [PubMed] [Google Scholar]
- 8.Butcher EC, Picker LJ. Lymphocyte homing and homeostasis. Science. 1996;272:60–66. doi: 10.1126/science.272.5258.60. [DOI] [PubMed] [Google Scholar]
- 9.Butcher EC, Scollay RG, Weissman IL. Organ specificity of lymphocyte migration: mediation by highly selective lymphocyte interaction with organ-specific determinants on high endothelial venules. Eur J Immunol. 1980;10:556–561. doi: 10.1002/eji.1830100713. [DOI] [PubMed] [Google Scholar]
- 10.Campbell JJ, Qin S, Bacon KB, Mackay CR, Butcher EC. The biology of chemokine and classical chemoattractant receptors: differential requirements for adhesion-triggering versus chemotactic responses in lymphoid cells. J Cell Biol. 1996;134:255–266. doi: 10.1083/jcb.134.1.255. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Campbell JJ, Foxman EF, Butcher EC. Chemoattractant cross talk as a regulatory mechanism in leukocyte adhesion and migration. Eur J Immunol. 1997;27:2571–2578. doi: 10.1002/eji.1830271016. [DOI] [PubMed] [Google Scholar]
- 12.Campbell JJ, Hedrick J, Zlotnik A, Siani MA, Thompson DA, Butcher EC. Chemokines and the arrest of lymphocytes rolling under flow conditions. Science. 1998;279:391–394. doi: 10.1126/science.279.5349.381. [DOI] [PubMed] [Google Scholar]
- 13.Dawson PE, Muir TW, Clark-Lewis I, Kent SHB. Synthesis of proteins by native chemical ligation. Science. 1994;266:776–779. doi: 10.1126/science.7973629. [DOI] [PubMed] [Google Scholar]
- 14.Gunn MD, Tangemann K, Tam C, Cyster JG, Rosen SD, Williams LT. A chemokine expressed in lymphoid high endothelial venules promotes the adhesion and chemotaxis of naive lymphocytes. Proc Natl Acad Sci USA. 1998;95:258–263. doi: 10.1073/pnas.95.1.258. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Hedrick JA, Zlotnik A. Identification and characterization of a novel B chemokine containing 6 conserved cysteines. J Immunol. 1997;159:1589–1593. [PubMed] [Google Scholar]
- 16.Honda S, Campbell JJ, Andrew DP, Engelhardt B, Butcher B, Warnock RA, Ye R, Butcher EC. Ligand-induced adhesion to activated endothelium and to vascular cell adhesion molecule-1 in lymphocytes transfected with the N-formyl peptide receptor. J Immunol. 1994;152:4226–4235. [PubMed] [Google Scholar]
- 17.Hromas R, Kim CH, Klemsz M, Krathwohl M, Fife K, Cooper S, Schnizlein-Bick C, Broxmeyer HE. Isolation and characterization of exodus-2, a novel C-C chemokine with a unique 38-amino acid carboxyl-terminal extension. J Immunol. 1997;159:2554–2558. [PubMed] [Google Scholar]
- 18.Jutila, M.A., D.M. Lewinsohn, E. Lakey-Berg, L.J. Picker, and E.C. Butcher. 1988. Homing receptors in lymphocyte, neutrophil, and monocyte interactions with endothelial cells. In Leukocyte Adhesion Molecules: Structure, Function, and Regulation. T.A. Springer, editor. Springer-Verlag, New York. 227–235.
- 19.Lewinsohn D, Bargatze R, Butcher EC. Evidence of a common molecular mechanism shared by neutrophils, lymphocytes, and other leukocytes. J Immunol. 1987;139:4313–4321. [PubMed] [Google Scholar]
- 20.Loetscher M, Gerber B, Loetscher P, Jones SA, Piali L, Clark-Lewis I, Baggiolini M, Moser B. Chemokine receptor specific for IP10 and Mig: structure, function, and expression in activated T-lymphocytes. J Exp Med. 1996;184:963–969. doi: 10.1084/jem.184.3.963. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.MacDonald HR, Budd RC, Cerottini J C. Pgp-1 (Ly 24) as a marker of murine memory T lymphocytes. Curr Top Microbiol Immunol. 1990;159:97–112. doi: 10.1007/978-3-642-75244-5_6. [DOI] [PubMed] [Google Scholar]
- 22.Mackay CR. Immunological memory. Adv Immunol. 1993;53:217–265. doi: 10.1016/s0065-2776(08)60501-5. [DOI] [PubMed] [Google Scholar]
- 23.Mackay CR. Chemokine receptors and T cell chemotaxis. J Exp Med. 1996;184:799–802. doi: 10.1084/jem.184.3.799. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Nagira M, Imai T, Heishima K, Kusuda J, Ridanpaa M, Nishimura M, Kakizaki M, Nomiyama H, Yoshie O. Molecular cloning of a novel human CC chemokine secondary lymphoid-tissue chemokine that is a potent chemoattractant for lymphocytes and mapped to chromosome 9p13. J Biol Chem. 1997;272:19518–19524. doi: 10.1074/jbc.272.31.19518. [DOI] [PubMed] [Google Scholar]
- 25.Nielson CP, Vestal RE, Sturm RJ, Haeslip R. Effects of selective phosphodiesterase inhibitors on the polymorphonuclear leukocyte respiratory burst. J Allergy Clin Immunol. 1990;88:801–805. doi: 10.1016/s0091-6749(05)80186-1. [DOI] [PubMed] [Google Scholar]
- 26.Oberlin E, Amara A, Bachelerie F, Bessia C, Virelizier J-L, Arenzana-Seisdedos F, Schwartz O, Heard J-M, Clark-Lewis I, Legler DF, et al. The CXC chemokine SDF-1 is the ligand for LESTR/FUSIN and prevents infection by T-cell-line-adapted HIV-1. Nature. 1996;392:833–835. doi: 10.1038/382833a0. [DOI] [PubMed] [Google Scholar]
- 27.Rossi DL, Vicari AP, Franz-Bacon K, McClanahan T, Zlotnik A. Identification through bioinformatics of two new macrophage proinflammatory human chemokines. J Immunol. 1997;158:1033–1037. [PubMed] [Google Scholar]
- 28.Roth SJ, Carr MW, Springer TA. C-C chemokines, but not the C-x-C chemokines interleukin-8 and interferon-γ inducible protein-10, stimulate transendothelial chemotaxis of T lymphocytes. Eur J Immunol. 1995;25:3482–3488. doi: 10.1002/eji.1830251241. [DOI] [PubMed] [Google Scholar]
- 29.Sanders ME, Makgoba MW, Sharrow SO, Stephany D, Springer TA, Young HA, Shaw S. Human memory T lymphocytes express increased levels of three cell adhesion molecules, (LFA-3, CD2, and LFA-1) and three other molecules (UCHL1, CDw29, and PGP-1) and have enhanced IFN-γ production. J Immunol. 1988;140:1401–1407. [PubMed] [Google Scholar]
- 30.Schall TJ, Bacon KB. Chemokines, leukocyte trafficking, and inflammation. Curr Opin Immunol. 1994;6:865–873. doi: 10.1016/0952-7915(94)90006-x. [DOI] [PubMed] [Google Scholar]
- 31.Shimuzu Y, Newman W, Tanaka Y, Shaw S. Lymphocyte interactions with endothelial cells. Immunol Today. 1992;13:106–108. doi: 10.1016/0167-5699(92)90151-V. [DOI] [PubMed] [Google Scholar]
- 32.Springer TA. Traffic signals on endothelium for lymphocyte recirculation and leukocyte emigration. Annu Rev Physiol. 1995;57:827–872. doi: 10.1146/annurev.ph.57.030195.004143. [DOI] [PubMed] [Google Scholar]
- 33.Stevens SK, Weissman IL, Butcher EC. Differences in the migration of B and T lymphocytes, organ-selective localization in vivoand the role of lymphocyte-endothelial cell recognition. J Immunol. 1982;128:844–850. [PubMed] [Google Scholar]
- 34.Tanabe S, Lu Z, Luo Y, Quackenbush EJ, Berman MA, Collins-Racie LA, Mi S, Reilly C, Lo D, Jacobs KA, Dorf ME. Identification of a new mouse β-chemokine, thymus-derived chemotactic agent 4, with activity on T lymphocytes and mesangial cells. J Immunol. 1997;159:5671–5679. [PubMed] [Google Scholar]
- 35.Tashiro K, Tada H, Heilker R, Shirozu M, Nakano T, Honjo T. Signal sequence trap: a cloning strategy for secreted proteins and type I membrane proteins. Science. 1993;261:600–603. doi: 10.1126/science.8342023. [DOI] [PubMed] [Google Scholar]
- 36.Warnock RA, Askari S, Butcher EC, von Andrian UH. Molecular mechanisms of lymphocyte homing to peripheral lymph nodes. J Exp Med. 1998;187:205–216. doi: 10.1084/jem.187.2.205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Williams MB, Butcher EC. Homing of naive and memory T lymphocyte subsets to Peyer's patches, lymph nodes and spleen. J Immunol. 1997;159:1746–1752. [PubMed] [Google Scholar]
- 38.Wu L, LaRosa G, Kassam N, Gordon CL, Heath H, Ruffing N, Chen H, Humblias J, Samson M, Parmentier M, et al. Interaction of chemokine receptor CCR5 with its ligands: multiple domains for HIV-1 gp120 binding and a single domain for chemokine binding. J Exp Med. 1997;186:1373–1381. doi: 10.1084/jem.186.8.1373. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Yoshida R, Imai T, Hieshima K, Kusuda J, Baba M, Kitauri M, Nishimura M, Kakizaki M, Nomiyama H, Yoshie O. Molecular cloning of a novel human CC chemokine EBI-1-ligand chemokine that is a specific functional ligand for EBI-1, CCR7. J Biol Chem. 1997;272:13903–13909. doi: 10.1074/jbc.272.21.13803. [DOI] [PubMed] [Google Scholar]
- 40.Yoshie O, Imai T, Nomiyama H. Novel lymphocyte-specific CC chemokines and their receptors. J Leukocyte Biol. 1997;62:634–644. doi: 10.1002/jlb.62.5.634. [DOI] [PubMed] [Google Scholar]