Abstract
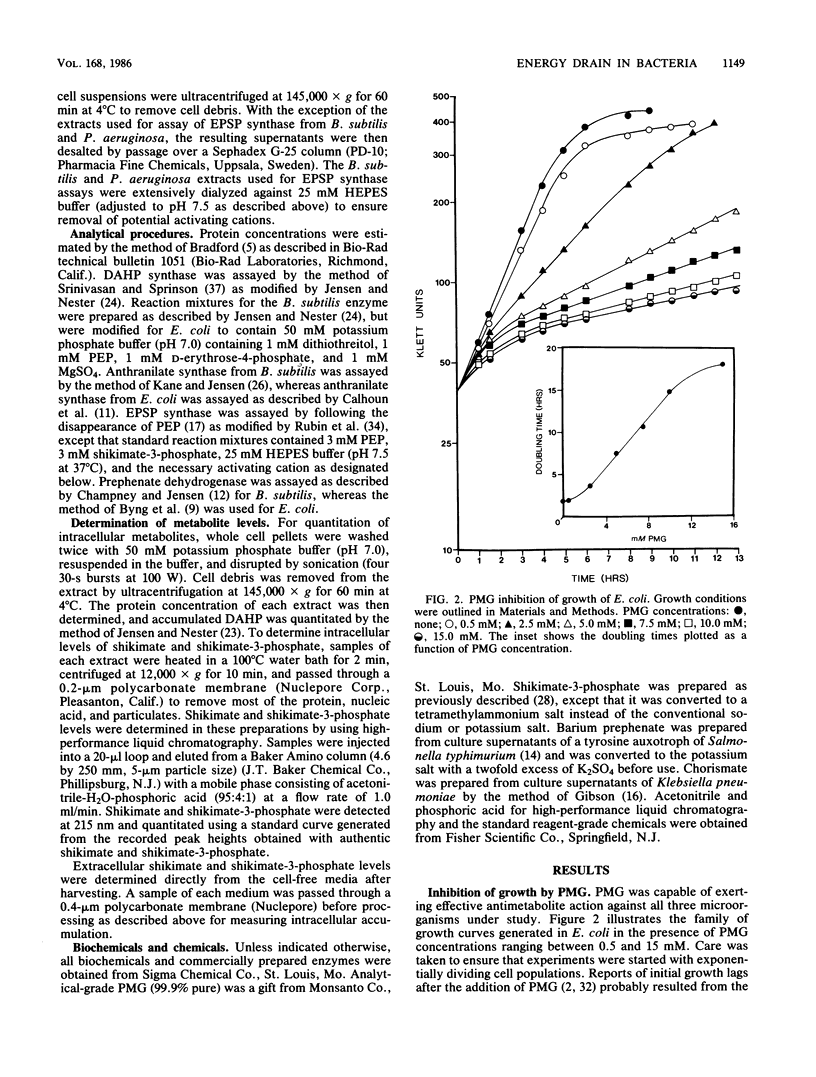
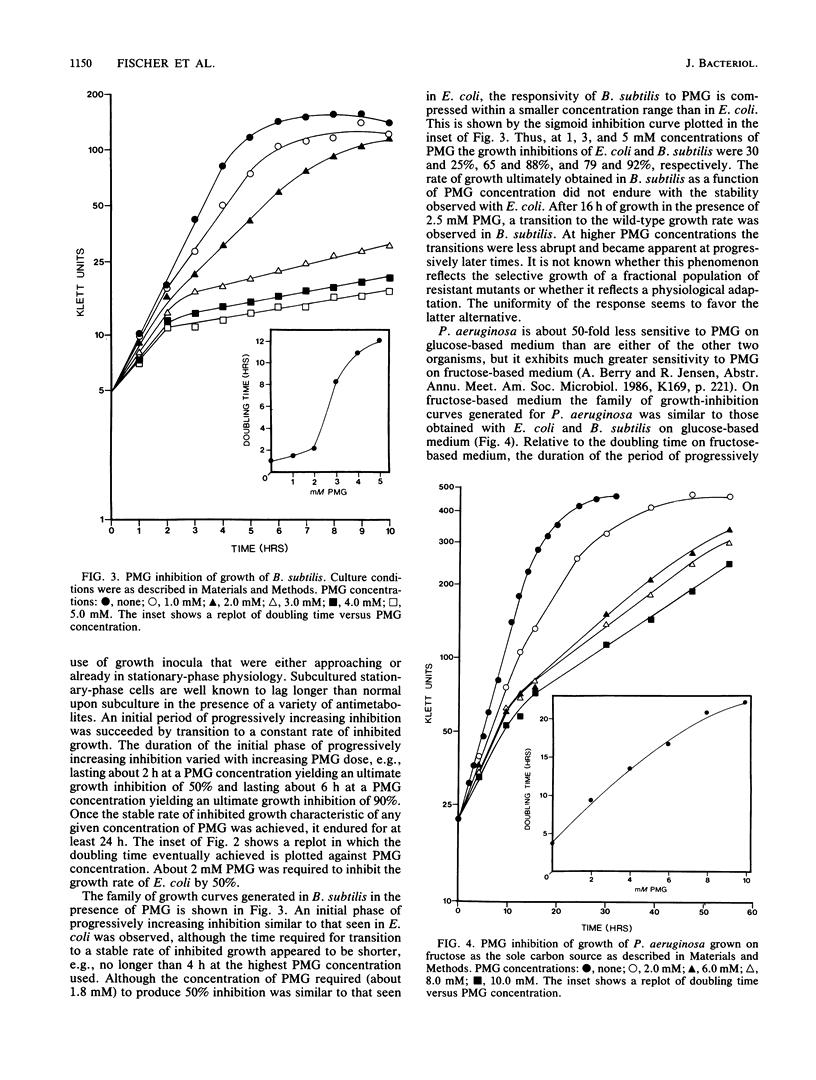
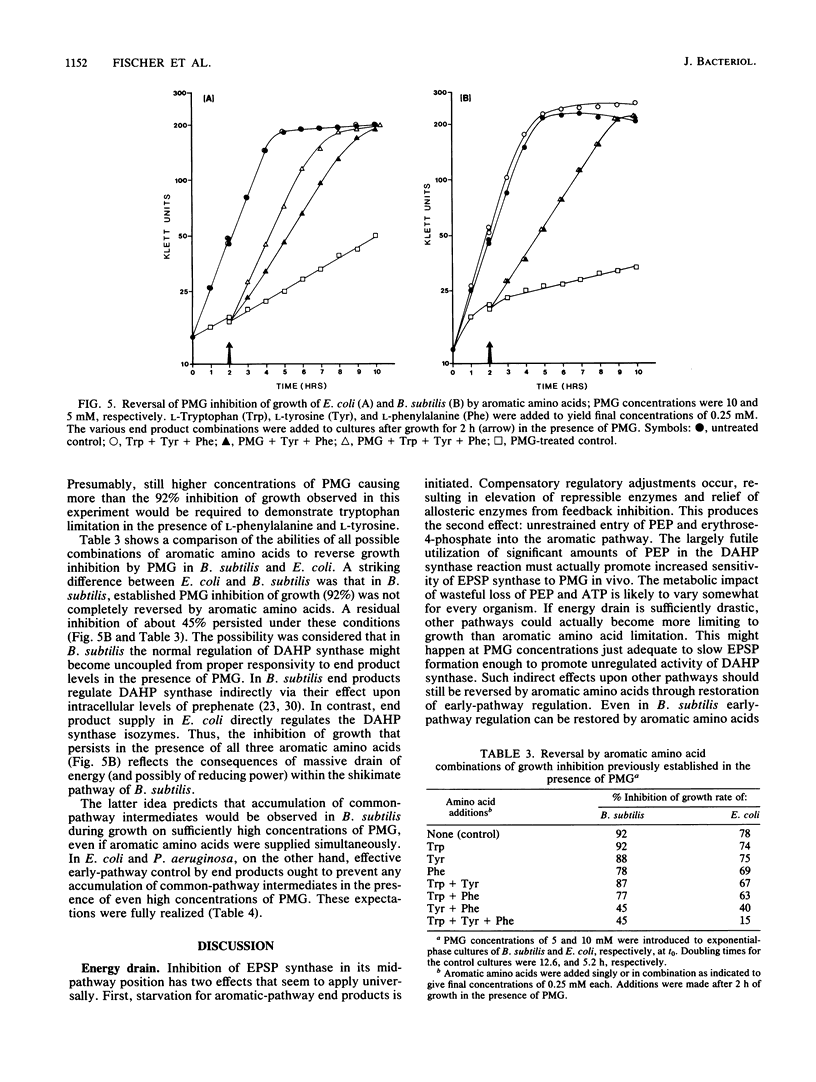
Escherichia coli, Bacillus subtilis, and Pseudomonas aeruginosa, each possessing a 5-enolpyruvylshikimate 3-phosphate synthase that is sensitive to inhibition by glyphosate [N-(phosphonomethyl)glycine], provide a good cross-section of organisms exemplifying the biochemical diversity of the aromatic pathway targeted by this potent antimicrobial compound. The pattern of growth inhibition, the alteration in levels of aromatic-pathway enzymes, and the accumulation of early-pathway metabolites after the addition of glyphosate were distinctive for each organism. Substantial intracellular shikimate-3-phosphate accumulated in response to glyphosate treatment in all three organisms. Both E. coli and P. aeruginosa, but not B. subtilis, accumulated near-millimolar levels of shikimate-3-phosphate in the culture medium. Intracellular backup of common-pathway precursors of shikimate-3-phosphate was substantial in B. subtilis, moderate in P. aeruginosa, and not detectable in E. coli. The full complement of aromatic amino acids prevented growth inhibition and metabolite accumulation in E. coli and P. aeruginosa where amino acid end products directly control early-pathway enzyme activity. In contrast, the initial prevention of growth inhibition in the presence of aromatic amino acids in B. subtilis was succeeded by progressively greater growth inhibition that correlated with rapid metabolite accumulation. In B. subtilis glyphosate can decrease prephenate concentrations sufficiently to uncouple the sequentially acting loops of feedback inhibition that ordinarily link end product excess to feedback inhibition of 3-deoxy-D-arabino-heptulosonate 7-phosphate synthase by prephenate. The consequential unrestrained entry is an energy-rich substrates into the aromatic pathway, even in the presence of aromatic amino acid end products, is an energy drain that potentially accounts for the inability of end products to fully reverse glyphosate inhibition in B. subtilis. Even in E. coli after glyphosate inhibition and metabolite accumulation were allowed to become fully established, a transient period where end products were capable of only partial reversal of growth inhibition occurred. The distinctive metabolism produced by dissimilation of different carbon sources also profound effects upon glyphosate sensitivity.
Full text
PDF




Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Ahmad S., Rightmire B., Jensen R. A. Evolution of the regulatory isozymes of 3-deoxy-D-arabino-heptulosonate 7-phosphate synthase present in the Escherichia coli genealogy. J Bacteriol. 1986 Jan;165(1):146–154. doi: 10.1128/jb.165.1.146-154.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- BROWN K. D., DOY C. H. END-PRODUCT REGULATION OF THE GENERAL AROMATIC-PATHWAY IN ESCHERICHIA COLI W. Biochim Biophys Acta. 1963 Sep 3;77:170–172. doi: 10.1016/0006-3002(63)90489-x. [DOI] [PubMed] [Google Scholar]
- Bachmann B. J. Pedigrees of some mutant strains of Escherichia coli K-12. Bacteriol Rev. 1972 Dec;36(4):525–557. doi: 10.1128/br.36.4.525-557.1972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bode R., Melo C., Birnbaum D. Mode of action of glyphosate in Candida maltosa. Arch Microbiol. 1984 Nov;140(1):83–85. doi: 10.1007/BF00409776. [DOI] [PubMed] [Google Scholar]
- Bradford M. M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem. 1976 May 7;72:248–254. doi: 10.1016/0003-2697(76)90527-3. [DOI] [PubMed] [Google Scholar]
- Byng G. S., Jensen R. A. Impact of isozymes upon partitioning of carbon flow and regulation of aromatic biosynthesis in prokaryotes. Isozymes Curr Top Biol Med Res. 1983;8:115–140. [PubMed] [Google Scholar]
- Byng G. S., Kane J. F., Jensen R. A. Diversity in the routing and regulation of complex biochemical pathways as indicators of microbial relatedness. Crit Rev Microbiol. 1982 May;9(4):227–252. doi: 10.3109/10408418209104491. [DOI] [PubMed] [Google Scholar]
- Byng G. S., Whitaker R. J., Gherna R. L., Jensen R. A. Variable enzymological patterning in tyrosine biosynthesis as a means of determining natural relatedness among the Pseudomonadaceae. J Bacteriol. 1980 Oct;144(1):247–257. doi: 10.1128/jb.144.1.247-257.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Calhoun D. H., Pierson D. L., Jensen R. A. The regulation of tryptophan biosynthesis in Pseudomonas aeruginosa. Mol Gen Genet. 1973 Mar 1;121(2):117–132. doi: 10.1007/BF00277526. [DOI] [PubMed] [Google Scholar]
- Champney W. S., Jensen R. A. The enzymology of prephenate dehydrogenase in Bacillus subtilis. J Biol Chem. 1970 Aug 10;245(15):3763–3770. [PubMed] [Google Scholar]
- Duncan K., Lewendon A., Coggins J. R. The purification of 5-enolpyruvylshikimate 3-phosphate synthase from an overproducing strain of Escherichia coli. FEBS Lett. 1984 Jan 2;165(1):121–127. doi: 10.1016/0014-5793(84)80027-7. [DOI] [PubMed] [Google Scholar]
- Gibson F. Chorismic acid: purification and some chemical and physical studies. Biochem J. 1964 Feb;90(2):256–261. doi: 10.1042/bj0900256. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gollub E., Zalkin H., Sprinson D. B. Correlation of genes and enzymes, and studies on regulation of the aromatic pathway in Salmonella. J Biol Chem. 1967 Nov 25;242(22):5323–5328. [PubMed] [Google Scholar]
- HOLLOWAY B. W. Genetic recombination in Pseudomonas aeruginosa. J Gen Microbiol. 1955 Dec;13(3):572–581. doi: 10.1099/00221287-13-3-572. [DOI] [PubMed] [Google Scholar]
- JENSEN R. A., NESTER E. W. THE REGULATORY SIGNIFICANCE OF INTERMEDIARY METABOLITES: CONTROL OF AROMATIC ACID BIOSYNTHESIS BY FEEDBACK INHIBITION IN BACILLUS SUBTILIS. J Mol Biol. 1965 Jun;12:468–481. doi: 10.1016/s0022-2836(65)80270-4. [DOI] [PubMed] [Google Scholar]
- Jensen R. A. Biochemical pathways in prokaryotes can be traced backward through evolutionary time. Mol Biol Evol. 1985 Mar;2(2):92–108. doi: 10.1093/oxfordjournals.molbev.a040338. [DOI] [PubMed] [Google Scholar]
- Jensen R. A., Nasser D. S. Comparative regulation of isoenzymic 3-deoxy-D-arabino-heptulosonate 7-phosphate synthetases in microorganisms. J Bacteriol. 1968 Jan;95(1):188–196. doi: 10.1128/jb.95.1.188-196.1968. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jensen R. A., Nester E. W. Regulatory enzymes of aromatic amino acid biosynthesis in Bacillus subtilis. I. Purification and properties of 3-deoxy-D-arabino-heptulosonate 7-phosphate synthetase. J Biol Chem. 1966 Jul 25;241(14):3365–3372. [PubMed] [Google Scholar]
- Kane J. F., Holmes W. M., Jensen R. A. Metabolic interlock. The dual function of a folate pathway gene as an extra-operonic gene of tryptophan biosynthesis. J Biol Chem. 1972 Mar 10;247(5):1587–1596. [PubMed] [Google Scholar]
- Kane J. F., Jensen R. A. Metabolic interlock. The influence of histidine on tryptophan biosynthesis in Bacillus subtilis. J Biol Chem. 1970 May 10;245(9):2384–2390. [PubMed] [Google Scholar]
- Kane J. F., Stenmark S. L., Calhoun D. H., Jensen R. A. Metabolic interlock. The role of the subordinate type of enzyme in the regulation of a complex pathway. J Biol Chem. 1971 Jul 10;246(13):4308–4316. [PubMed] [Google Scholar]
- Nester E. W., Jensen R. A. Control of aromatic acid biosynthesis in Bacillus subtilis: sequenial feedback inhibition. J Bacteriol. 1966 Apr;91(4):1594–1598. doi: 10.1128/jb.91.4.1594-1598.1966. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nester E. W., Jensen R. A., Nasser D. S. Regulation of enzyme synthesis in the aromatic amino acid pathway of Bacillus subtilus. J Bacteriol. 1969 Jan;97(1):83–90. doi: 10.1128/jb.97.1.83-90.1969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roisch U., Lingens F. Zum Wirkungsmechanismus des Herbizids N-(Phosphonomethyl)glycin. Einfluss von N-(Phosphonomethyl)glycin auf das Wachstum und auf die Enzyme der Aromatenbiosynthese von Escherichia coli. Hoppe Seylers Z Physiol Chem. 1980 Jul;361(7):1049–1058. [PubMed] [Google Scholar]
- Rubin J. L., Gaines C. G., Jensen R. A. Enzymological basis for herbicidal action of glyphosate. Plant Physiol. 1982 Sep;70(3):833–839. doi: 10.1104/pp.70.3.833. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rubin J. L., Gaines C. G., Jensen R. A. Glyphosate Inhibition of 5-Enolpyruvylshikimate 3-Phosphate Synthase from Suspension-Cultured Cells of Nicotiana silvestris. Plant Physiol. 1984 Jul;75(3):839–845. doi: 10.1104/pp.75.3.839. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rubin J. L., Jensen R. A. Differentially Regulated Isozymes of 3-Deoxy-d-arabino-Heptulosonate-7-Phosphate Synthase from Seedlings of Vigna radiata [L.] Wilczek. Plant Physiol. 1985 Nov;79(3):711–718. doi: 10.1104/pp.79.3.711. [DOI] [PMC free article] [PubMed] [Google Scholar]
- SRINIVASAN P. R., SPRINSON D. B. 2-Keto-3-deoxy-D-arabo-heptonic acid 7-phosphate synthetase. J Biol Chem. 1959 Apr;234(4):716–722. [PubMed] [Google Scholar]
- Spizizen J. TRANSFORMATION OF BIOCHEMICALLY DEFICIENT STRAINS OF BACILLUS SUBTILIS BY DEOXYRIBONUCLEATE. Proc Natl Acad Sci U S A. 1958 Oct 15;44(10):1072–1078. doi: 10.1073/pnas.44.10.1072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tribe D. E., Camakaris H., Pittard J. Constitutive and repressivle enzymes of the common pathway of aromatic biosynthesis in Escherichia coli K-12: regulation of enzyme synthesis at different growth rates. J Bacteriol. 1976 Sep;127(3):1085–1097. doi: 10.1128/jb.127.3.1085-1097.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Whitaker R. J., Fiske M. J., Jensen R. A. Pseudomonas aeruginosa possesses two novel regulatory isozymes of 3-deoxy-D-arabino-heptulosonate 7-phosphate synthase. J Biol Chem. 1982 Nov 10;257(21):12789–12794. [PubMed] [Google Scholar]



