Abstract
Pep 5 and nisin are cationic peptide antibiotics which in addition to their membrane-disruptive action induce autolysis in staphylococci. To investigate the mechanism of lysis induction, the influence of the peptides on the activity of the N-acetylmuramoyl-L-alanine amidase of Staphylococcus simulans 22 was studied. In experiments with isolated cell walls at low ionic strength, the amidase activity was stimulated by the addition of Pep 5 and nisin, as well as by polylysine, streptomycin, and mono- and divalent cations. The concentrations necessary for activation depended on the nature of the cation and ranged from 5 microM for poly-L-lysine (n = 17) to 150 mM for Na+ at a cell wall concentration of 100 micrograms of cell walls per ml. No effect was observed if the cell walls were devoid of polyanionic constituents. Kinetic data suggested that the amidase bound to the teichoic and teichuronic acids of the cell wall and was thereby inhibited. Cationic molecules reversed this inhibition, most likely by displacing the enzyme from the polyanions. If the concentrations of the larger peptides were high in relation to cell wall concentration, the activation turned into inhibition, presumably by interfering with the access of the enzyme to its substrate. These experiments demonstrate that the activity of the amidase is modulated by basic peptides in vitro and help to explain how Pep 5 and nisin may cause lysis of treated cells.
Full text
PDF

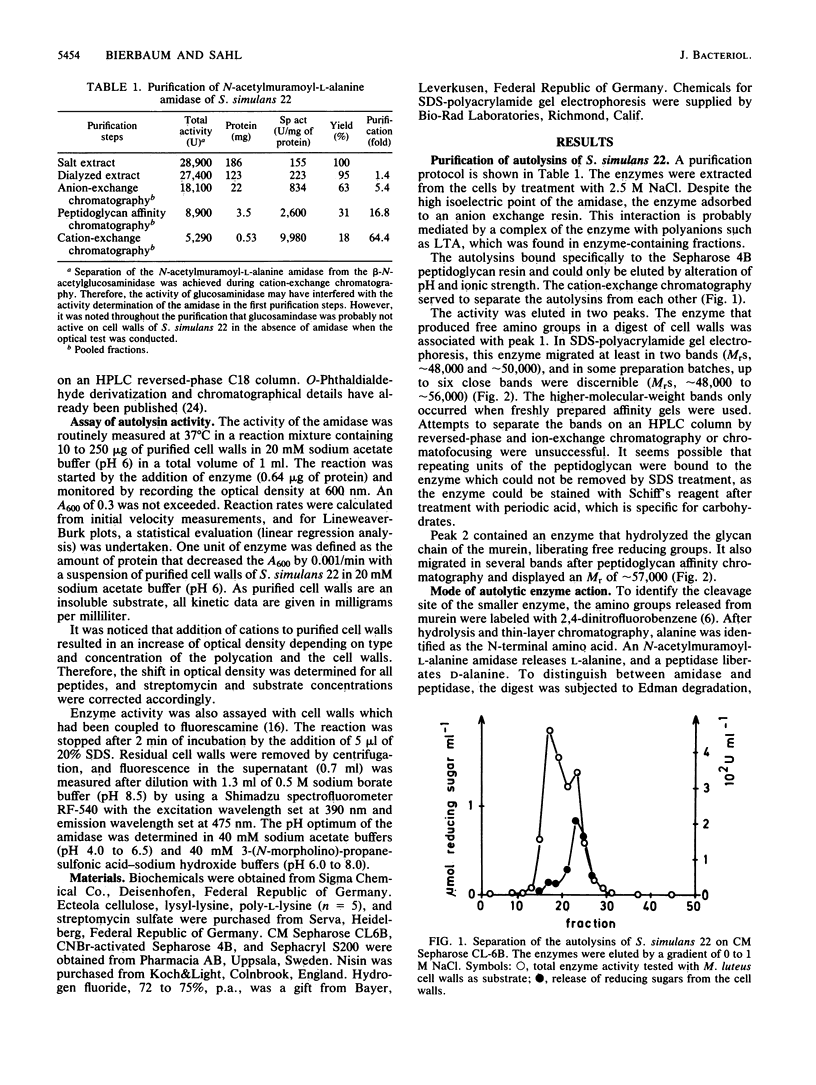
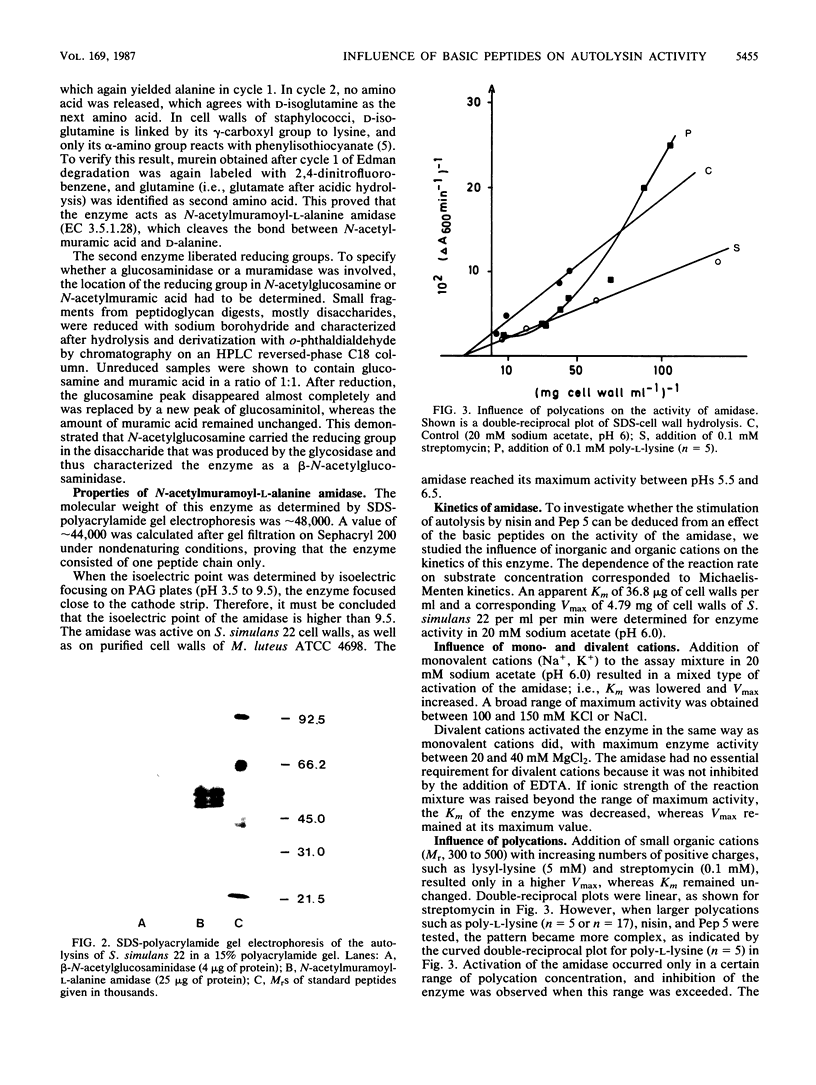
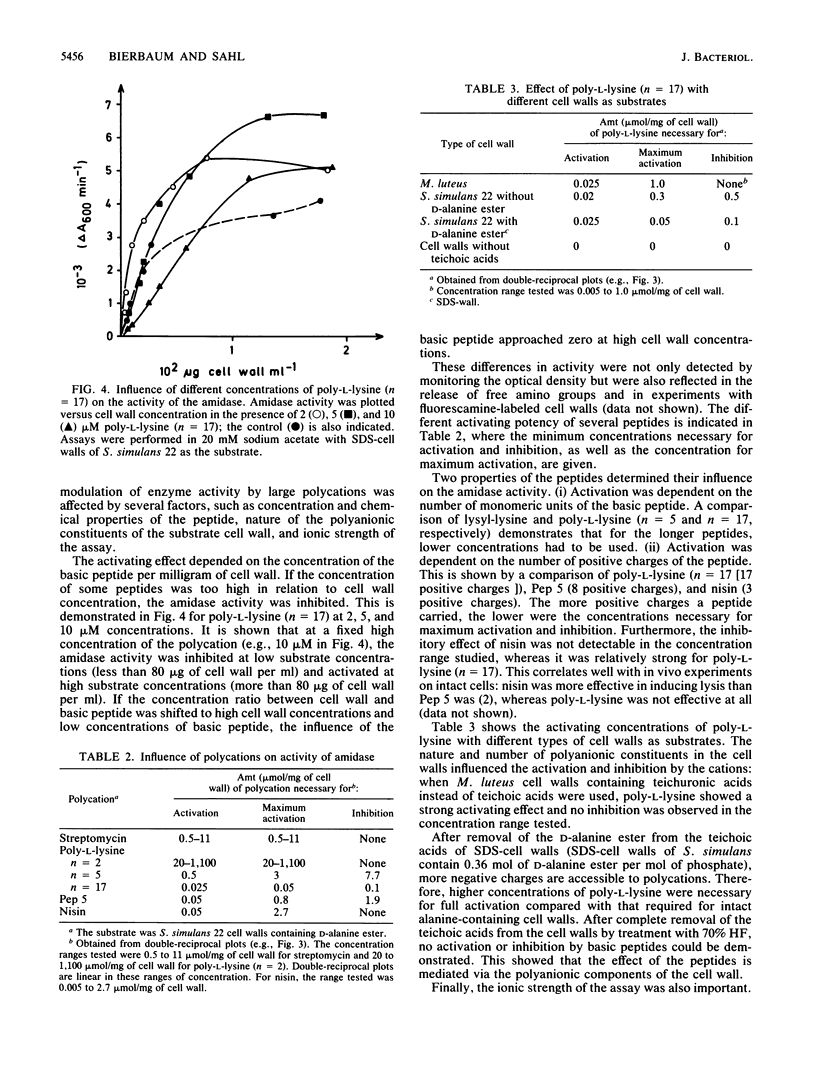


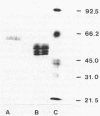
Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bierbaum G., Sahl H. G. Induction of autolysis of staphylococci by the basic peptide antibiotics Pep 5 and nisin and their influence on the activity of autolytic enzymes. Arch Microbiol. 1985 Apr;141(3):249–254. doi: 10.1007/BF00408067. [DOI] [PubMed] [Google Scholar]
- Ghuysen J. M. Use of bacteriolytic enzymes in determination of wall structure and their role in cell metabolism. Bacteriol Rev. 1968 Dec;32(4 Pt 2):425–464. [PMC free article] [PubMed] [Google Scholar]
- Gross E., Morell J. L. The structure of nisin. J Am Chem Soc. 1971 Sep 8;93(18):4634–4635. doi: 10.1021/ja00747a073. [DOI] [PubMed] [Google Scholar]
- Jolliffe L. K., Doyle R. J., Streips U. N. The energized membrane and cellular autolysis in Bacillus subtilis. Cell. 1981 Sep;25(3):753–763. doi: 10.1016/0092-8674(81)90183-5. [DOI] [PubMed] [Google Scholar]
- Kusser W., Zimmer K., Fiedler F. Characteristics of the binding of aminoglycoside antibiotics to teichoic acids. A potential model system for interaction of aminoglycosides with polyanions. Eur J Biochem. 1985 Sep 16;151(3):601–605. doi: 10.1111/j.1432-1033.1985.tb09146.x. [DOI] [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- Mintz G., Herbold D. R., Glaser L. A fluorescent assay for bacterial cell wall lytic enzymes. Anal Biochem. 1975 May 26;66(1):272–278. doi: 10.1016/0003-2697(75)90746-0. [DOI] [PubMed] [Google Scholar]
- Navaratnam N., Virk S. S., Ward S., Kuhn N. J. Cationic activation of galactosyltransferase from rat mammary Golgi membranes by polyamines and by basic peptides and proteins. Biochem J. 1986 Oct 15;239(2):423–433. doi: 10.1042/bj2390423. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rogers H. J., Taylor C., Rayter S., Ward J. B. Purification and properties of autolytic endo-beta-N-acetylglucosaminidase and the N-acetylmuramyl-L-alanine amidase from Bacillus subtilis strain 168. J Gen Microbiol. 1984 Sep;130(9):2395–2402. doi: 10.1099/00221287-130-9-2395. [DOI] [PubMed] [Google Scholar]
- Ruhr E., Sahl H. G. Mode of action of the peptide antibiotic nisin and influence on the membrane potential of whole cells and on cytoplasmic and artificial membrane vesicles. Antimicrob Agents Chemother. 1985 May;27(5):841–845. doi: 10.1128/aac.27.5.841. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sahl H. G. Bactericidal cationic peptides involved in bacterial antagonism and host defence. Microbiol Sci. 1985 Jul;2(7):212–217. [PubMed] [Google Scholar]
- Sahl H. G., Brandis H. Production, purification and chemical properties of an antistaphylococcal agent produced by Staphylococcus epidermidis. J Gen Microbiol. 1981 Dec;127(2):377–384. doi: 10.1099/00221287-127-2-377. [DOI] [PubMed] [Google Scholar]
- Sahl H. G., Grossgarten M., Widger W. R., Cramer W. A., Brandis H. Structural similarities of the staphylococcin-like peptide Pep-5 to the peptide antibiotic nisin. Antimicrob Agents Chemother. 1985 May;27(5):836–840. doi: 10.1128/aac.27.5.836. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sahl H. G., Hahn C., Brandis H. Interaction of the staphylococcin-like peptide Pep 5 with cell walls and isolated cell wall components of Gram-positive bacteria. Zentralbl Bakteriol Mikrobiol Hyg A. 1985 Oct;260(2):197–205. doi: 10.1016/s0176-6724(85)80115-2. [DOI] [PubMed] [Google Scholar]
- Sahl H. G. Influence of the staphylococcinlike peptide Pep 5 on membrane potential of bacterial cells and cytoplasmic membrane vesicles. J Bacteriol. 1985 May;162(2):833–836. doi: 10.1128/jb.162.2.833-836.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Valisena S., Varaldo P. E., Satta G. Purification and characterization of three separate bacteriolytic enzymes excreted by Staphylococcus aureus, Staphylococcus simulans, and Staphylococcus saprophyticus. J Bacteriol. 1982 Aug;151(2):636–647. doi: 10.1128/jb.151.2.636-647.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weber K., Pringle J. R., Osborn M. Measurement of molecular weights by electrophoresis on SDS-acrylamide gel. Methods Enzymol. 1972;26:3–27. doi: 10.1016/s0076-6879(72)26003-7. [DOI] [PubMed] [Google Scholar]
- Wecke J., Lahav M., Ginsburg I., Giesbrecht P. Cell wall degradation of Staphylococcus aureus by lysozyme. Arch Microbiol. 1982 Mar;131(2):116–123. doi: 10.1007/BF01053992. [DOI] [PubMed] [Google Scholar]
- Wecke J., Lahav M., Ginsburg I., Kwa E., Giesbrecht P. Inhibition of wall autolysis of staphylococci by sodium polyanethole sulfonate "liquoid". Arch Microbiol. 1986 Mar;144(2):110–115. doi: 10.1007/BF00414719. [DOI] [PubMed] [Google Scholar]