Abstract
Identification of the processing/activation of multiple interleukin-1β converting enzyme (ICE)–like proteases and their target substrates in the intact cell is critical to our understanding of the apoptotic process. In this study we demonstrate processing/activation of at least four ICE-like proteases during the execution phase of apoptosis in human monocytic tumor THP.1 cells. Apoptosis was accompanied by processing of Ich-1, CPP32, and Mch3α to their catalytically active subunits, and lysates from these cells displayed a proteolytic activity with kinetics, characteristic of CPP32/Mch3α but not of ICE. Fluorescence-activated cell sorting was used to obtain pure populations of normal and apoptotic cells. In apoptotic cells, extensive cleavage of Ich-1, CPP32, and Mch3α was observed together with proteolysis of the ICE-like protease substrates, poly (ADP-ribose) polymerase (PARP), the 70-kD protein component of U1 small nuclear ribonucleoprotein (U170K), and lamins A/B. In contrast, no cleavage of CPP32, Mch3α or the substrates was observed in normal cells. In cells exposed to an apoptotic stimulus, some processing of Ich-1 was detected in morphologically normal cells, suggesting that cleavage of Ich-1 may occur early in the apoptotic process. The ICE-like protease inhibitor, benzyloxycarbonyl-Val-Ala-Asp (OMe) fluoromethyl ketone (Z-VAD.FMK), inhibited apoptosis and cleavage of Ich-1, CPP32, Mch3α, Mch2α, PARP, U1-70K, and lamins. These results suggest that Z-VAD.FMK inhibits apoptosis by inhibiting a key effector protease upstream of Ich-1, CPP32, Mch3α, and Mch2α. Together these observations demonstrate that processing/activation of Ich-1, CPP32, Mch3α, and Mch2α accompanies the execution phase of apoptosis in THP.1 cells. This is the first demonstration of the activation of at least four ICE-like proteases in apoptotic cells, providing further evidence for a requirement for the activation of multiple ICE-like proteases during apoptosis.
Apoptosis is a form of cell death that is essential for the control of cell populations during normal development and in many diseases (Arends and Wyllie, 1991; Raff, 1992). It is recognized by distinct morphological changes, including cell shrinkage, nuclear condensation, and fragmentation. Apoptotic cell death has been divided into two distinct phases: an initial condemned phase where cells receive a signal that results in commitment to cell death without any morphological changes, followed by an execution phase when all of the characteristic morphological and biochemical features of apoptosis occur (Earnshaw, 1995).
In the nematode, Caenorhabditis elegans genetically determined cell death has an essential requirement for the gene ced-3 (Ellis et al., 1991), which encodes a protein with sequence homology to the mammalian cysteine protease, interleukin-1β converting enzyme (ICE)1 (Yuan et al., 1993). ICE is required for the proteolytic processing of pro–interleukin-1β to the active cytokine (Thornberry et al., 1992). Overexpresssion of ICE-like proteases induces cell death by apoptosis (for review see Kumar, 1995), suggesting that ICE, or a related protease, is an essential component of the mammalian cell death pathway. However, such studies have to be interpreted with some caution as overexpression of a particular protease may result in the cleavage of low affinity protein substrates leading to cytotoxicity. More direct evidence for the involvement of ICE or ICE-like proteases in apoptosis has come from inhibitor studies with CrmA, a viral serpin inhibitor of ICE (Ray et al., 1992), and the baculovirus antiapoptotic protein p35 (Bump et al., 1995).
While initial studies highlighted a major role for ICE in apoptosis, recent investigations demonstrated that mice deficient in ICE fail to exhibit a prominent cell death– defective phenotype, except in Fas-induced apoptosis of thymocytes (Li et al., 1995; Kuida et al., 1995). This suggested that other ICE homologues may be required for apoptosis, and recent work has identified a family of such proteases including CPP32/apopain/Yama (Fernandes-Alnemri et al., 1994; Nicholson et al., 1995; Tewari et al., 1995), Ich-1 (Wang et al., 1994) and its mouse homologue Nedd-2 (Kumar et al., 1994), Mch2 (Fernandes-Alnemri et al., 1995a ), Mch3/ ICE-LAP3 (Duan et al., 1996; Fernandes-Alnemri et al., 1995b ), Mch4 (Fernandes-Alnemri et al., 1996), MACH/FLICE/ Mch5 (Boldin et al., 1996; FernandesAlnemri et al., 1996; Muzio et al., 1996), Mch6 (Srinivasula et al., 1996), Tx/Ich-2/ICErel-II (Faucheu et al., 1995; Kamens et al., 1995; Munday et al., 1995), and ICErel-III/ Ty (Munday et al., 1995; Faucheu et al., 1996).
ICE-like proteases are synthesized as inactive precursors requiring cleavage at specific Asp residues to yield two subunits of molecular mass ∼20 and 10 kD, which together form the active enzyme (Thornberry et al., 1992; Thornberry and Molineaux, 1995). Several studies have suggested that ICE-like proteases may autoprocess or, alternatively, some ICE-like proteases may activate other family members (for review see Takahashi and Earnshaw, 1996). During apoptosis, ICE-like proteases cleave several intracellular proteins including poly (ADP-ribose) polymerase (PARP), lamins, the 70-kD protein component of U1 small nuclear ribonucleoprotein (U1-70K), DNA-dependent protein kinase, and sterol regulatory element binding proteins (Casciola-Rosen et al., 1994, 1996; Kaufmann et al., 1993; Song et al., 1996; Wang et al., 1995). Although the precise substrate specificity of different ICE homologues is not known, CPP32, Mch2α, and Mch3α cleave PARP (Nicholson et al., 1995; Fernandes-Alnemri et al., 1995a ,b; Lazebnik et al., 1995), while Mch2α but not CPP32 can cleave lamins (Orth et al., 1996; Takahashi et al., 1996), and CPP32 can cleave U1-70K (Casciola-Rosen et al., 1996).
It remains to be determined whether activation of more than one ICE-like protease is required for the execution of apoptosis (Takahashi and Earnshaw, 1996). In the present study we have used peptide inhibitors to distinguish the potential roles of different ICE-like proteases in apoptosis. Acetyl-Tyr-Val-Ala-Asp-aldehyde (Ac-YVAD.CHO) and acetyl-Asp-Glu-Val-Asp-aldehyde (Ac-DEVD.CHO) are specific inhibitors of ICE (Thornberry et al., 1992) and CPP32/Mch3α, respectively (Fernandes-Alnemri et al., 1995b ; Nicholson et al., 1995). Although acetyl-Tyr-ValAla-Asp chloromethyl ketone (YVAD.CMK) was originally described as a specific inhibitor of ICE, it also inhibits other ICE-like proteases, including the proteolytic activity described as prICE (Lazebnik et al., 1994). Benzyloxycarbonyl-Val-Ala-Asp (OMe) fluoromethyl ketone (Z-VAD.FMK), a cell membrane–permeable inhibitor of ICE-like proteases, inhibits apoptosis in diverse systems including human monocytic tumor THP.1 cells (Chow et al., 1995; Fearnhead et al., 1995; Zhu et al., 1995). Z-VAD. FMK inhibits apoptosis at an early stage, as evidenced by inhibition of DNA fragmention, PARP proteolysis (Zhu et al., 1995), and processing of CPP32 (Jacobson et al., 1996; Slee et al., 1996). Using these inhibitors, we now demonstrate the cleavage/activation of at least four ICE-like proteases, Ich-1, CPP32, Mch3α, and Mch2α, in THP.1 cells during the execution phase of apoptosis.
Materials and Methods
Materials
Media and serum were purchased from Gibco (Paisley, UK). Pronase and N-α-p-tosyl-l-lysine chloromethyl ketone (TLCK) were from BoehringerMannheim UK (Lewes, UK). Z-VAD.FMK and benzyloxycarbonylAsp-Glu-Val-Asp-7-amino-4-trifluoromethyl coumarin (Z-DEVD.AFC) were from Enzyme Systems, Inc. (Dublin, CA). Ac-YVAD.CHO, Ac-DEVD.CHO, and YVAD.CMK were from Bachem (Bubendorf, Switzerland). Acetyl-Tyr-Val-Ala-Asp-7-amino-4-methyl coumarin (AcYVAD.AMC) and all other chemicals were from Sigma Chemical Co. (Poole, UK).
Cell Culture
The human monocytic tumor cell line, THP.1, was obtained from ECACC (Wiltshire, UK) and maintained in RPMI 1640 supplemented with 10% heat-inactivated FCS, 100 U/ml penicillin, and 100 μg/ml streptomycin in an atmosphere of 5% CO2 in air at 37°C. The cells were maintained in logarithmic growth phase by routine passage every 2–3 d. To induce apoptosis, 1 × 10 6 cells per ml were incubated either alone or in the presence of cycloheximide (25 μM) and TLCK (100 μM) or etoposide (25 μM) as previously described (Zhu et al., 1995). Both these stimuli induce extensive apoptosis in THP.1 cells. To assess the effect of the ICE-like protease inhibitors, THP.1 cells were pretreated for 1 h with Z-VAD.FMK (50 μM) or YVAD.CMK (5–25 μM) before exposure to the apoptotic stimulus. The peptide inhibitors were prepared as stock solutions in DMSO, and aliquots were added to the control cultures.
Preparation of Cell and Bacterial Lysates
For preparation of lysates, THP.1 cells were incubated as required and then placed on ice, washed twice with ice-cold PBS, and resuspended in Pipes buffer (50 mM Pipes/KOH (pH 6.5), 2 mM EDTA, 0.1% (wt/vol) CHAPS, 5 mM DTT, 20 μg/ml leupeptin, 10 μg/ml pepstatin A, 10 μg/ml aprotinin, and 2 mM PMSF) at a concentration of 6 × 10 6 cells per 10 μl. The cells were frozen and thawed three times in liquid nitrogen, and then centrifuged at 20,000 g for 30 min at 4°C. The supernatant fraction was then centrifuged for an additional 45 min at 100,000 g. The protein concentration in the supernatant fractions (the lysate) was determined by the Bradford assay (Bio Rad Laboratories, Hercules, CA). Recombinant CPP32 and Mch3α were expressed in bacteria and bacterial extracts prepared as previously described (Fernandes-Alnemri et al., 1995a ,b).
Separation of Normal and Apoptotic Cells by Flow Cytometry
Apoptosis was assessed by flow cytometry using Hoechst 33342 as previously described (Sun et al., 1992; Zhu et al., 1995). The basis of this method is the increased permeability to Hoechst 33342 of the apoptotic cells compared with normal cells (Ormerod et al., 1993). We have previously shown that the high blue fluorescent cells (indicative of increased Hoechst 33342 staining) are apoptotic based on a number of criteria including ultrastructure and analysis of DNA by conventional agarose gel electrophoresis (Sun et al., 1992; Zhu et al., 1995). In the present study we used fluorescence-activated cell sorting to separate pure populations of normal and apoptotic cells. When examined by fluorescence microscopy, cells with low blue fluorescence exhibited normal morphology, and those with high blue fluorescence exhibited distinctive apoptotic morphology.
Western Blot Analysis
Samples of 0.5 × 106 cells were prepared as described (Harlow and Lane, 1988). Proteins were resolved on 13% (CPP32, Mch3α, Mch2α, and Ich-1) or 7% (PARP, U1-70K, and lamins A/B) SDS polyacrylamide gels and blotted onto nitrocellulose (Hybond-C extra; Amersham, Little Chalfont, UK). Membranes were blocked before detection with rabbit polyclonal antibodies directed to the p17 subunit of CPP32 (Nicholson et al., 1995), or to the p12 subunit of Mch2α (both kindly provided by D. Nicholson, Merck Frosst, Quebec, Canada); a rabbit polyclonal antibody to the carboxy terminus of Ich-1L (Santa Cruz Biotechnology, Santa Cruz, CA); or a rabbit polyclonal antibody directed to the p19 subunit of Mch3α (see below). The CPP32 antibody detects both pro-CPP32 and the p17 subunit; the antibody to Ich-1 detects both pro–Ich-1 and the p12 subunit; and the antibody to Mch3α detects both pro-Mch3α and the p19 subunit, while the antibody to Mch2α detects only pro-Mch2α. The rabbit polyclonal antibodies to PARP (318) and U1-70K were kind gifts from Dr. G. Poirier (Laval University, Quebec, Canada) and Dr. K. Luhrmann (University of Marburg, Germany), respectively. The PARP antibody recognizes both intact PARP (116 kD) and a cleavage product of 85 kD, whereas the U170K antibody detects only intact U1-70K, which, in THP.1 cells, was calculated to have a molecular mass of ∼64 kD. A mouse mAb to lamins A/B, which detects both intact (66 kD) and cleaved (46 kD) lamins, was kindly provided by Dr. E. Nigg (University of Geneva, Switzerland). Detection was achieved using the appropriate secondary antibody (goat anti–rabbit IgG or goat anti–mouse IgG) conjugated to HRP and by enhanced chemiluminescence (Amersham).
Fluorimetric Measurement of Proteolytic Cleavage
The proteolytic cleavage activities of THP.1 lysates and recombinant CPP32 and Mch3α were measured using a continuous fluorimetric assay modified from the method of Thornberry (1994). Liberation of 7-amino-4trifluoromethylcoumarin from Z-DEVD.AFC as a measure of CPP32like activity was assayed at excitation and emission wavelengths of 400 and 505 nm, respectively. ICE activity was measured by the liberation of 7-amino-4-methylcoumarin from Ac-YVAD.AMC at excitation and emission wave lengths of 380 and 460 nm, respectively. Lysates were assayed at 37°C in a modified thermostatted cuvette holder in 1.25 ml of 100 mM Hepes, 10% sucrose, 0.1% CHAPS, 10 mM DTT, pH 7.5, in an LS50B luminescence fluorimeter (Perkin-Elmer Corp., Norwalk, CT). Routinely, assays contained 100 μg of lysate protein and 20 μM substrate. Calibration was carried out with standard solutions of 7-amino-4-trifluoromethylcoumarin and 7-amino-4-methylcoumarin. Lysates, from THP.1 cells exposed for 1 h to an apoptotic stimulus, were preincubated for 15 min with various concentrations of inhibitors to ensure equilibrium, and the reaction, initiated by addition of Z-DEVD.AFC, was followed for 5 min. Substrates and inhibitors were added in DMSO, whose final concentration never exceeded 1% and had no effect on enzymic activity.
Generation of a Polyclonal Antibody to Mch3α
A polyclonal antibody to Mch3α was raised against the p17 subunit of recombinant Mch3α, obtained using a glutathione-S-transferase–Mch3α cDNA (Fernandes-Alnemri et al., 1995). Briefly, the p17 fragment of Mch3α (amino acids 54–198) was subcloned in-frame into the BamH1 site of the bacterial expression vector pGEX2T (Pharmacia Biotech, Uppsala, Sweden). Bacterial extracts were prepared, and glutathione-S-transferase–p17 Mch3α protein was purified by glutathione–agarose beads (Sigma Chemical Co.) before immunization. The rabbit polyclonal antibody obtained was characterized by ELISA and Western blot analysis, which verified that the antibody obtained recognized both intact Mch3α (∼36 kD) and the p19 subunit but did not recognize CPP32 (data not shown).
Results
Processing of CPP32 and Ich-1 Accompanies the Induction of Apoptosis in Intact THP.1 Cells
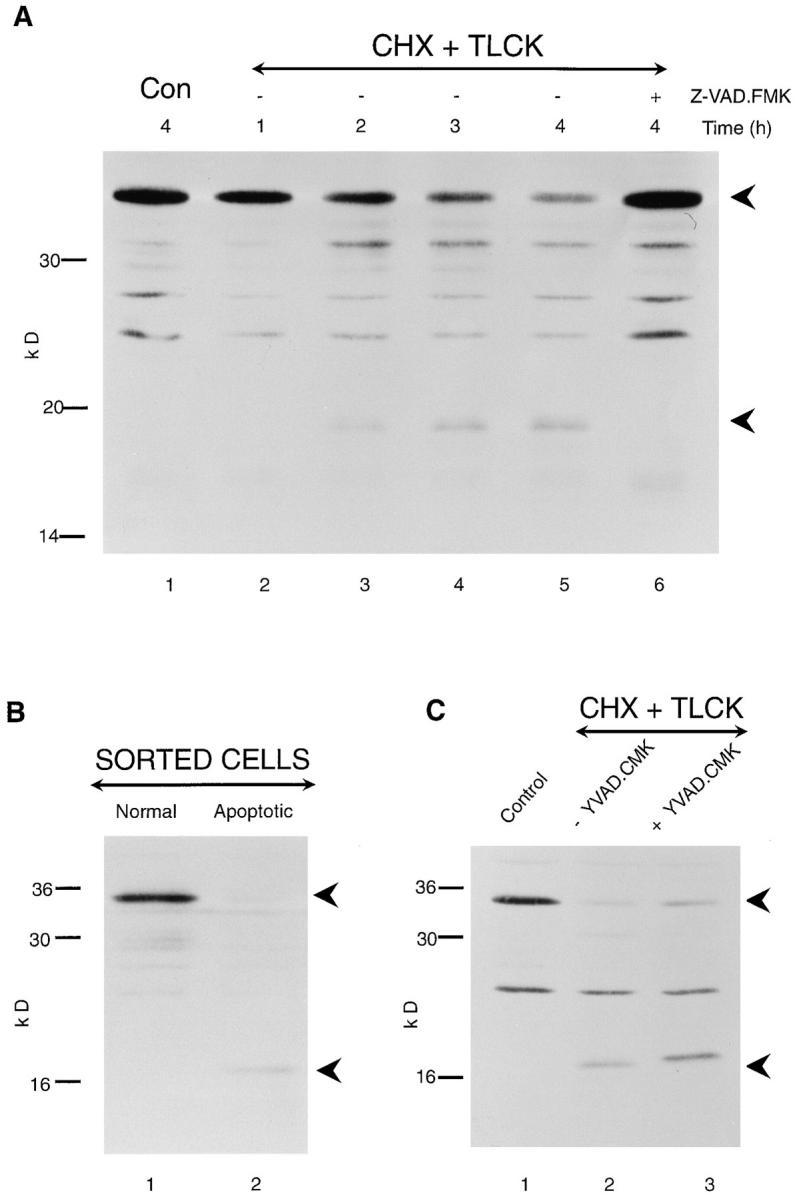
Exposure of THP.1 cells to a number of stimuli, including etoposide, a DNA topoisomerase II inhibitor, or cotreatment with the protein synthesis inhibitor cycloheximide and TLCK, an inhibitor of trypsin-like proteases, induces apoptosis (Zhu et al., 1995). THP.1 cells were coincubated with cycloheximide (25 μM) and TLCK (100 μM) for up to 4 h, and the amount of apoptosis was quantified by flow cytometry (Fig. 1 A). A time-dependent increase in apoptosis was observed, which was first detected at 2 h (Fig. 1 A). To determine whether a time-dependent processing of ICE-like proteases occurred, Western blot analysis was performed using antibodies to the p17 fragment of CPP32 and the p12 fragment of Ich-1. In untreated cells, immunoblots showed the presence of the 32-kD precursor of CPP32 and the 48-kD precursor of Ich-1 (Fig. 1 B, lane 1). After induction of apoptosis, the p17 subunit of the mature CPP32 enzyme was first detected by 2 h and remained elevated until 4 h (Fig. 1 B, lanes 3–5). In parallel, a timedependent decrease in the level of the 32-kD precursor of CPP32 was observed. A time-dependent decrease in the 48-kD precursor protein of Ich-1 accompanied by the formation of its p12 subunit was also observed (Fig. 1 B, lanes 2–5). A small amount of the p12 fragment of Ich-1 was already evident at 1 h (Fig. 1 B, lane 2). Similar results were obtained when apoptosis was induced by etoposide (data not shown).
Figure 1.

More than one ICE homologue is cleaved in cells exposed to an apoptotic stimulus. THP.1 cells were incubated for up to 4 h, either alone (Con) or in the presence of cycloheximide (CHX) (25 μM) and TLCK (100 μM). (A) The time course of induction of apoptosis was determined by flow cytometry as described in Materials and Methods. (B) The time course of cleavage of the proforms of CPP32 (upper panel) and Ich-1 (lower panel) was determined by Western blot analysis as described in Materials and Methods. The 32-kD pro-CPP32 is indicated by the upper arrowhead, and the 17-kD cleavage product is indicated by the lower arrowhead in the upper panel. The 48-kD pro–Ich-1 is indicated by the upper arrowhead, and the 12-kD cleavage product is indicated by the lower arrowhead in the lower panel.
Extensive Processing of CPP32 and Ich-1 in Apoptotic THP.1 Cells But Not in Cells That Exhibit Normal Morphology
As apoptosis is a stochastic process, cells at all stages of the apoptotic process, including normal, condemned, and apoptotic, are present after exposure to an apoptotic stimulus (Earnshaw, 1995). Thus, the data describing the status of the ICE-like proteases in Fig. 1 and in all similar studies in the literature refer to results obtained with a mixed population of cells. To overcome this problem, we have used a flow cytometric method to sort pure populations of normal and apoptotic cells for further analysis (Zhu et al., 1995). THP.1 cells were exposed for 4 h to etoposide (25 μM), and cells displaying either normal or apoptotic morphology were separated by fluorescence-activated cell sorting. Dramatic differences were observed in the level of the proforms of both CPP32 and Ich-1 between these normal and apoptotic cells. Cells displaying normal morphology contained only the proform of CPP32 and predominantly the proform of Ich-1, whereas apoptotic cells contained almost no pro-CPP32 or pro–Ich-1 (Fig. 2 A, compare lanes 1 and 2). In the case of CPP32, the catalytically active subunit p17 was detected in apoptotic cells but not in normal cells (Fig. 2 A, compare lanes 1 and 2). For Ich-1, significant levels of the p12 subunit were detected in apoptotic cells with only very low levels detected in normal cells (Fig. 2 A). As low levels of the p12 subunit were observed in morphologically normal cells obtained after exposure to an apoptotic stimulus, we examined similar morphologically normal cells, which had not been exposed to an apoptotic stimulus. No p12 subunit of Ich-1 was seen in these sorted cells (data not shown). No cleavage of CPP32 was observed until cells displaying normal morphology (low Hoechst fluorescence) acquired apoptotic morphology (high blue fluorescence). In contrast, a very small amount of cleavage of Ich-1 was evident in morphologically normal cells exposed to an apoptotic stimulus, with extensive cleavage only observed when these cells acquired apoptotic morphology. Thus, extensive cleavage/activation of at least two ICE homologues occurred during the execution phase of apoptosis.
Figure 2.

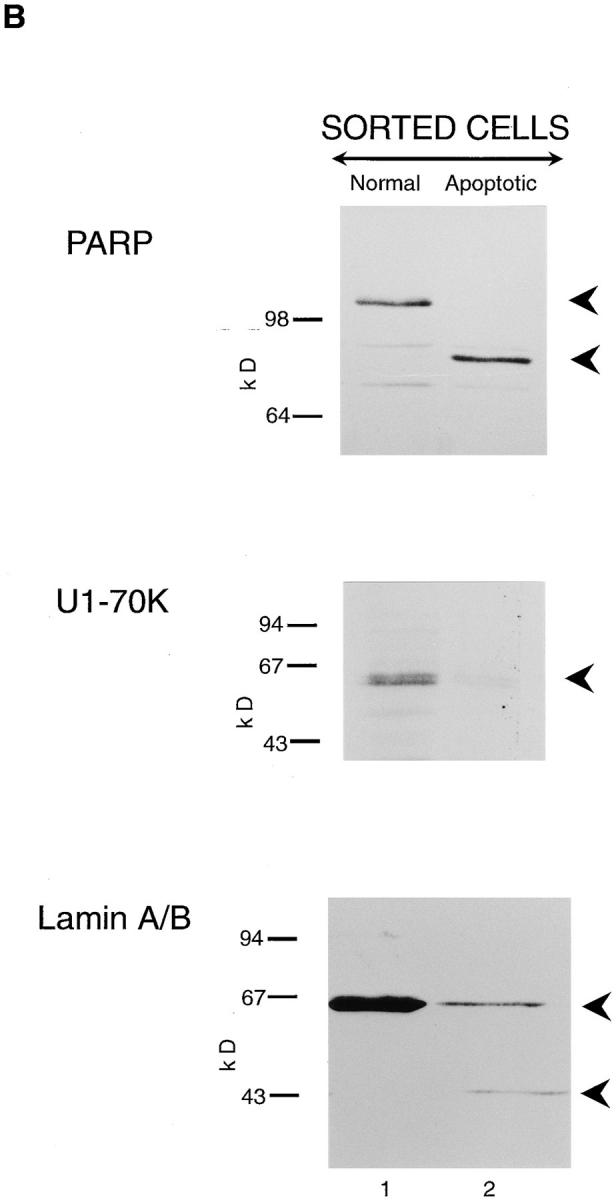
CPP32 and Ich-1 are extensively cleaved in apoptotic but not morphologically normal THP.1 cells, and their processing is concomitant with the cleavage of PARP, U1-70K, and lamins A/ B. THP.1 cells were incubated for 4 h in the presence of etoposide (25 μM), stained with Hoechst 33342 and propidium iodide, and then sorted by flow cytometry as described previously (Zhu et al., 1995). Cells with low blue fluorescence were morphologically normal and those with high blue fluorescence exhibited distinctive apoptotic morphology when examined by fluorescence microscopy. Cells with either normal (lane 1) or apoptotic (lane 2) morphology were analyzed by Western blot analysis as described in Materials and Methods. (A) Cells were analyzed using antibodies to CPP32 (upper panel) and Ich-1 (lower panel). (B) Cells were analyzed using antibodies to PARP, U1-70K, and lamins A/B (upper, middle and lower panels, respectively). The proforms of CPP32 and Ich-1 are indicated by the upper arrowheads (A), and intact PARP, U170K, and lamins A/B are indicated by the upper arrowheads (B). The lower arrowheads represent either processed enzymes or cleaved proteolytic fragments. Cells displaying normal morphology contain primarily the intact forms of all the proteins analyzed (lane 1), whereas, in apoptotic cells, processing of more than one ICE-like protease is detected and associated with cleavage of PARP, U1-70K, and lamins A/B.
Cleavage of PARP, U1-70K, and Lamins A/B Accompanied the Processing of CPP32 and Ich-1 in Apoptotic Cells
To obtain further evidence for the activation of more than one ICE-like protease during apoptosis, we used antibodies to PARP, U1-70K, and lamins A/B to determine which putative ICE-like protease substrates were cleaved in apoptotic cells. Pure populations of normal and apoptotic cells were sorted by flow cytometry as described in the previous section. In cells displaying normal morphology, only the intact form of all three proteins was detected, namely 116-kD PARP protein, 64-kD U1-70K protein, and 66-kD lamin A/B protein (Fig. 2 B, lane 1). In marked contrast, in a pure population of apoptotic cells, all three proteins were processed, resulting in their disappearance together with the appearance of, in the case of PARP and lamin A/B, protein fragments of 85 and 46 kD, respectively (Fig. 2 B, lane 2). Therefore, several target proteins, which previously have been reported to be substrates for ICE-like proteases, are cleaved in the execution phase of apoptosis. Cleavage of PARP and U1-70K suggested activation of CPP32 and/or Mch3α (Casciola-Rosen et al., 1996; Fernandes-Alnemri et al., 1995b ; Nicholson et al., 1995). However, as Mch2α is the only ICE-like protease known to cleave lamins (Orth et al., 1996; Takahashi et al., 1996), the cleavage of lamins A/B in apoptotic cells supports the activation of Mch2α in THP.1 cell apoptosis.
Z-VAD.FMK Inhibits Both Apoptosis and the Processing of CPP32 and Ich-1
Incubation of THP.1 cells for 4 h with etoposide (25 μM) resulted in the induction of apoptosis (33.5 ± 2.5%, mean ± SEM, n = 3). Etoposide also induced the processing of the proforms of both CPP32 and Ich-1. This accompanied the appearance of the catalytically active p17 subunit of CPP32 and the p12 subunit of Ich-1, both of which were apparent when compared with control cells (Fig. 3, compare lanes 1 and 2). Pretreatment for 1 h with Z-VAD.FMK (50 μM) inhibited not only apoptosis (1.7 ± 0.5%) but also the loss of both pro–Ich-1 and pro-CPP32 and the formation of the p17 subunit of CPP32 and the p12 subunit of Ich-1 (Fig. 3, lanes 2 and 3). Pretreatment of THP.1 cells for 1 h with Z-VAD.FMK also completely prevented etoposide-induced cleavage of PARP, U1-70K, and lamins A/B (data not shown). These results demonstrated that the proforms of both CPP32 and Ich-1 were processed in THP.1 cells undergoing apoptosis to yield their catalytically active subunits, and that inhibition of their processing by Z-VAD.FMK corresponded with an inhibition of apoptosis.
Figure 3.
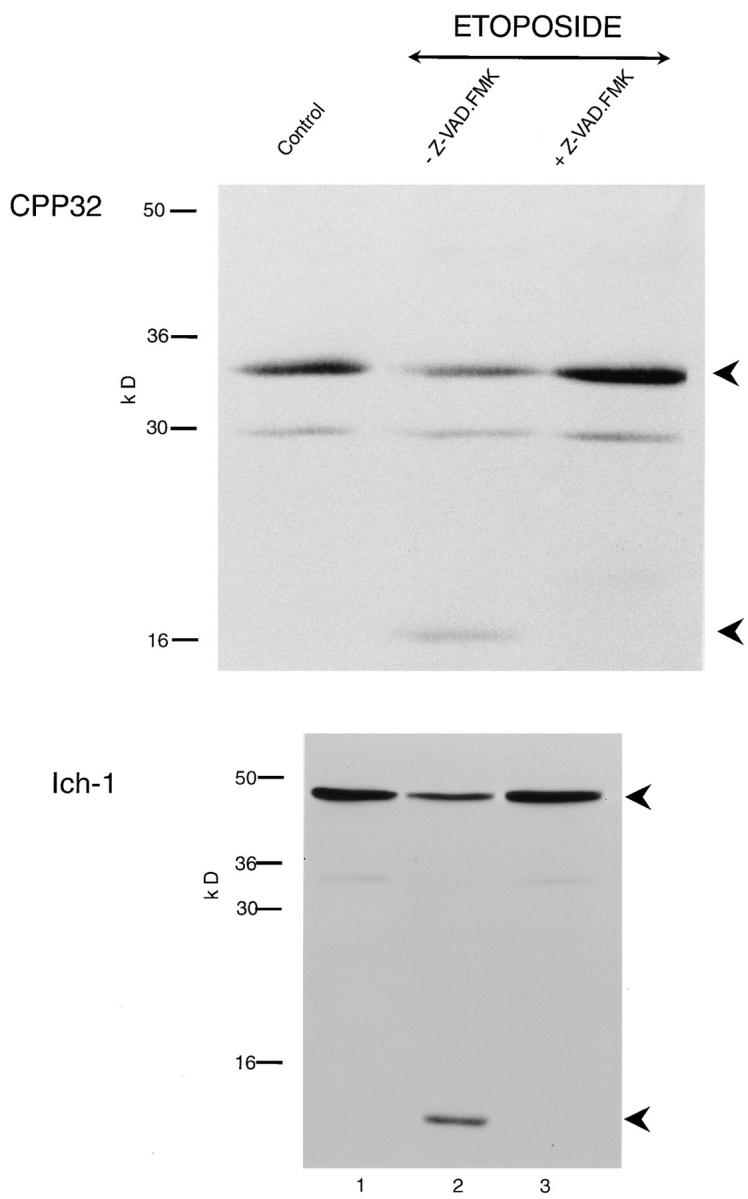
Z-VAD.FMK inhibits the cleavage of CPP32 and Ich-1. THP.1 cells were incubated for 4 h, either alone (lane 1) or with etoposide (25 μM) in the presence (lane 3) or absence (lane 2) of Z-VAD.FMK (50 μM) as described in Materials and Methods. CPP32 and Ich-1 were detected by Western blot analysis. (Upper panel) pro-CPP32 is indicated by the upper arrowhead, and the 17-kD cleavage product is indicated by the lower arrowhead; (lower panel) 48-kD pro–Ich-1 is indicated by the upper arrowhead, and the 12-kD cleavage product is indicated by the lower arrowhead. On induction of apoptosis, intact CPP32 (upper panel) and Ich-1 (lower panel) in control cells (lane 1) were cleaved (lane 2), and these were inhibited by Z-VAD.FMK (lane 3).
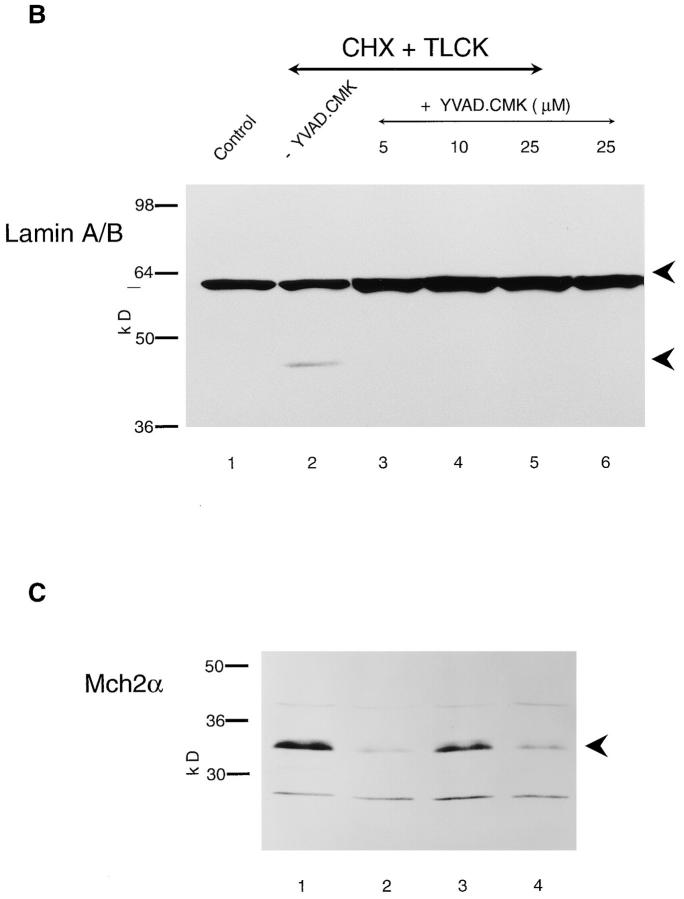
YVAD.CMK Inhibits Lamin Cleavage But Not Apoptosis
THP.1 cells were preincubated for 1 h either alone or with YVAD.CMK (5–25 μM), and then further incubated for 4 h with cycloheximide (25 μM) and TLCK (100 μM). Apoptosis was assessed by flow cytometry. Cycloheximide in the presence of TLCK induced ∼48% apoptosis that was not affected by pretreatment with YVAD.CMK (∼46%). Thus, YVAD.CMK did not inhibit apoptosis in THP.1 cells, assessed either by flow cytometry or by internucleosomal cleavage of DNA (Zhu et al., 1995). Furthermore, no significant inhibition of the processing of pro-CPP32 or pro– Ich-1 was observed in cells pretreated with YVAD.CMK (25 μM) (Fig. 4 A). The processed fragment of CPP32 detected in the presence of YVAD.CMK (Fig. 4 A, lane 3) was slightly larger and did not comigrate with the fragments obtained after treatment with the apoptotic stimulus alone (Fig. 4 A, lane 2). This suggested that, while YVAD.CMK did not significantly inhibit the loss of proCPP32, it did inhibit further degradation of the initial cleaved product. In contrast to its inability to inhibit processing of CPP32 and Ich-1, YVAD.CMK was a very good inhibitor of the cleavage of lamins A/B (Fig. 4 B). Control cells contained only intact 66-kD lamin A/B (Fig. 4 B, lane 1), which was cleaved to a 46-kD fragment in the presence of cycloheximide and TLCK (Fig. 4 B, lane 2). This cleavage was almost totally inhibited by YVAD.CMK (5–25 μM) (Fig. 4 B, lanes 3–5). Essentially similar results were obtained with etoposide (data not shown). In contrast with the observed inhibition of lamin cleavage, these concentrations of YVAD.CMK did not inhibit PARP cleavage (Browne, S., M. MacFarlane, G.M. Cohen, and C. Paraskeva, manuscript submitted for publication).
Figure 4.

YVAD.CMK inhibits cleavage of lamins A/B but not of CPP32 and Ich-1. THP.1 cells were incubated for 4 h, either alone (lane 1) or with cycloheximide (CHX) (25 μM) and TLCK (100 μM) (lane 2) in the presence of YVAD.CMK (25 μM) (lane 3). (A) Cell samples were analyzed by Western blot analysis using antibodies to CPP32 and Ich-1 as described in Materials and Methods. (B) Cells were incubated for 4 h, either alone (lane 1) or with cycloheximide (CHX) (25 μM) and TLCK (100 μM) (lane 2) in the presence of the indicated concentrations of YVAD.CMK (5–25 μM) (lanes 3–5). The cleavage of intact lamin A/B (66 kD) to a fragment of 46 kD was detected by Western blot analysis using a lamin A/B antibody. (C) THP.1 cells were incubated for 4 h, either alone (lane 1) or with cycloheximide (CHX) (25 μM) and TLCK (100 μM) (lane 2) in the presence of Z-VAD.FMK (50 μM) (lane 3) or YVAD.CMK (25 μM) (lane 4). Cell samples were analyzed by Western blot analysis using an antibody to Mch2α.
Mch2α Is Cleaved during the Execution Phase of Apoptosis and This Cleavage Is Inhibited by Z-VAD.FMK
The ability of YVAD.CMK to inhibit lamin cleavage, but not other features of apoptosis, provided indirect evidence for activation of the lamin protease, Mch2α (Orth et al., 1996; Takahashi et al., 1996). To obtain direct evidence for the involvement of Mch2α in THP.1 cell apoptosis, we used an antibody that recognizes pro-Mch2α. Immunoblots showed the presence of the 34-kD pro-Mch2α in control cells incubated for 4 h (Fig. 4 C, lane 1). Induction of apoptosis by cycloheximide in the presence of TLCK resulted in extensive loss of pro-Mch2α (Fig. 4 C, lane 2), which was totally prevented by the ICE-like protease inhibitor, Z-VAD.FMK (Fig. 4 C, lane 3). These data demonstrate the activation of Mch2α during the execution phase of apoptosis in THP.1 cells. Concentrations of YVAD.CMK (25 μM), which did not inhibit apoptosis, did not significantly inhibit cleavage of pro-Mch2α (Fig. 4 C, lane 4). As this concentration of YVAD.CMK inhibited lamin cleavage (Fig. 4 B), our data suggested that YVAD.CMK preferentially inhibited Mch2α activity rather than the processing of Mch2α.
Induction of Apoptosis Activates Z-DEVD.AFC Cleavage in Cell Lysates
The processing/cleavage of CPP32, Ich-1, Mch2α, PARP, U1-70K, and lamins A/B strongly suggested that activation of an ICE-like proteolytic activity was an important effector of the cell death program. This was further investigated using lysates from unsorted control and apoptotic cells to assay for proteolytic activity with Z-DEVD.AFC. This model substrate was chosen because the DEVD tetrapeptide sequence is identical to the cleavage site of PARP, i.e., DEVD216-G217. In addition, CPP32 (Nicholson et al., 1995), Mch2α, and Mch3α (Fernandes-Alnemri et al., 1995a ,b) cleave the similar substrate analogue, Ac-DEVD.AMC. The Z-DEVD.AFC cleaving activity of lysates obtained from control cells was very low and did not increase after incubation of cells for 4 h (Fig. 5 A). In contrast, increased proteolytic activity was observed in lysates prepared from cells exposed to an apoptotic stimulus (Fig. 5 A). Within 1 h of exposure to the apoptotic stimulus, a small increase in Z-DEVD.AFC cleavage activity was detected in lysates (Fig. 5 A), which preceded the onset of apoptosis in intact cells, as measured by flow cytometry (Figs. 1 A and 5 A). The maximal increase in proteolytic activity occurred at 2 h and was ∼15-fold greater than the activity in control lysates (Fig. 5 A).
Figure 5.

Z.DEVD-AFC cleavage activity in lysates derived from THP.1 cells committed to apoptosis precedes the morphological characteristics of cell death. THP.1 cells were incubated for up to 4 h, either alone (Con) (○–○) or in the presence of cycloheximide (CHX) and TLCK (•–•; ▪–▪), and lysates were prepared at the indicated time points as described in Materials and Methods. Z-DEVD.AFC (○–○; •–•) and Ac-YVAD.AMC (▪–▪) hydrolysis activities were determined as described in Materials and Methods and are shown in A. The extent of apoptosis at each time point, as assessed by flow cytometry, is indicated in brackets. Lysates from both untreated (Con) and THP.1 cells exposed to an apoptotic stimulus (CHX + TLCK) were analyzed by Western blot analysis using antibodies to CPP32 as described in Materials and Methods. (B) pro-CPP32 is indicated by the upper arrowhead, and the 17-kD cleavage product is indicated by the lower arrowhead. The time course of cleavage of CPP32 (B) paralleled the hydrolysis activity of the lysates towards Z-DEVD.AFC.
We wished to investigate whether these changes in proteolytic activity correlated with cleavage of CPP32. Lysates obtained from control cells incubated for 4 h contained only the proform of CPP32 (Fig. 5 B, lane 1). In contrast, lysates from cells exposed to an apoptotic stimulus displayed a time-dependent loss of the proform of CPP32 (Fig. 5 B, lanes 2–4). This was accompanied by the appearance of the p17 subunit of CPP32, which was first observed at 1 h, was maximal at 2 h, but decreased at 4 h (Fig. 5 B). In the same lysates, cleavage or loss of the proform of Ich-1 was almost maximal at 1 h, the first time point examined (data not shown).
In the present study lysates obtained from THP.1 cells that had been pretreated with Z-VAD.FMK before induction of apoptosis exhibited neither Z-DEVD.AFC proteolytic activity nor cleavage of CPP32 or Ich-1 (data not shown). Thus, Z-VAD.FMK inhibits the generation of a CPP32-like protease activity in addition to blocking the cleavage of both Ich-1 and CPP32.
Induction of Apoptosis in THP.1 Cells Results in Cleavage of Z-DEVD.AFC But Not of Ac-YVAD.AMC
To further characterize the proteolytic activity in THP.1 cells lysates, we investigated its susceptibility to specific inhibitors. Ac-YVAD.CHO is a specific inhibitor of ICE (Thornberry et al., 1992), and Ac-DEVD.CHO is a relatively specific inhibitor of CPP32 (Nicholson et al., 1995) although it also inhibits Mch3α (Fernandes-Alnemri et al., 1995b ) and Mch4 (Fernandes-Alnemri et al., 1996). Hydrolysis of Z-DEVD.AFC was inhibited in a time-dependent manner by Ac-DEVD.CHO, Z-VAD.FMK, and AcYVAD.CHO (data not shown). When IC50 values were determined from the inhibition curves (Table I), AcDEVD.CHO was the most potent inhibitor with an IC50 of 3 nM. Z-VAD.FMK was a more potent inhibitor than Ac-YVAD.CHO, but it was not as effective as AcDEVD.CHO (Table I). Ac-YVAD.CHO was 10,000-fold less potent than Ac-DEVD.CHO. These differing potencies suggested that the ICE-like protease activity in the lysate was due to a CPP32-like activity but not to ICE. Further evidence was provided by our finding that these lysates did not exhibit an increased ability to hydrolyze Ac-YVAD.AMC, a model substrate for ICE activity (Fig. 5 A). These data were consistent with the Z-DEVD.AFC hydrolysis activity of lysates, from cells exposed to an apoptotic stimulus, being due to a CPP32-like activity but not to ICE.
Table I.
Kinetic Properties of Lysates Prepared from THP.1 Cells Exposed to an Apoptotic Stimulus
| Kinetic parameter | THP.1 lysate | |
|---|---|---|
| K m (μM) | 55.4 ± 7.1 | |
| Vmax (pmol/mg/min) | 411 ± 19.7 | |
| I50 Ac-YVAD.CHO (μM) | 38 | |
| I50 Z-VAD.FMK (μM) | 1.2 | |
| I50 Ac-DEVD.CHO (μM) | 0.003 |
Lysates were prepared from THP.1 cells exposed for 1 h to cycloheximide (25 μM) and TLCK (100 μM), and the kinetic properties were investigated using a continuous fluorimetric assay with Z-DEVD.AFC as substrate. Enzyme assays were carried out as described in Materials and Methods. K m determinations were analyzed by a nonlinear curve fitting program. I50 (defined as the concentration producing 50% inhibition) values were determined in experiments where the lysates were preincubated with the inhibitor for 15 min at 37°C before adding the substrate (20 μM).
Kinetic Characterization of Z-DEVD.AFC Cleavage
To further characterize this CPP32-like proteolytic activity, we investigated its kinetic characteristics. Lysates, from cells exposed for 1 h to cycloheximide and TLCK, were incubated with Z-DEVD.AFC (10–200 μM) and proteolysis was assessed. The lysates exhibited Michaelis-Menten kinetics with a K m of 55.4 ± 7.1 μM (mean ± SEM) and Vmax = 411 ± 19.7 pmol/mg/min (mean ± SEM). This K m is higher than that for pure or recombinant CPP32 (FernandesAlnemri et al., 1995b; Nicholson et al., 1995) and is very similar to that reported for recombinant Mch3α (FernandesAlnemri et al., 1995b). To determine whether lysates, obtained from cells exposed to an apoptotic stimulus, contained a proteolytic activity characteristic of both CPP32 and Mch3α, the kinetic characteristics of these lysates were compared with those of recombinant CPP32 and Mch3α. Bacterial extracts of CPP32 and Mch3α were incubated with Z-DEVD.AFC (10–200 μM) and proteolysis was assessed. Both CPP32 and Mch3α exhibited MichaelisMenten kinetics with a K m of 21.9 ± 2.0 μM and 61.5 ± 8.8 μM, respectively. Thus, lysates, obtained from THP.1 cells exposed to an apoptotic stimulus, exhibited kinetic characteristics suggestive of the activation of both CPP32 and Mch3α in the execution phase of apoptosis in THP.1 cells.
Processing of Mch3α during the Execution Phase of Apoptosis Is Inhibited by ZVAD.FMK But Not by YVAD.CMK
Characterization of the proteolytic activity present in lysates suggested the activation of another CPP32-like protease, in addition to CPP32 itself, in the execution phase of apoptosis in THP.1 cells. Kinetic data suggested that this protease was Mch3α, and to verify we raised an antibody to the p17 subunit of Mch3α. THP.1 cells were coincubated with cycloheximide (25 μM) and TLCK (100 μM) for up to 4 h, and the cleavage of Mch3α was assessed (Fig. 6 A). In untreated cells, immunoblots showed the presence of the 34-kD precursor form of Mch3α (Fig. 6 A, lane 1). After induction of apoptosis, the p19 subunit of Mch3α was first detected by 2 h and remained evident until 4 h (Fig. 6 A, lanes 2–5). In parallel, a time-dependent decrease in the 34-kD proform of Mch3α was observed, coincident with the induction of apoptosis as previously assessed by flow cytometry (Fig. 1 A). Similar results were obtained when apoptosis was induced by etoposide (data not shown). To determine whether processing of Mch3α correlated with apoptotic execution, pure populations of normal and apoptotic cells were obtained and analyzed. THP.1 cells were exposed for 4 h to etoposide (25 μM), and cells displaying either normal or apoptotic morphology were separated by fluorescence-activated cell sorting. Cells displaying normal morphology contained only the proform of Mch3α (Fig. 6 B, lane 1), whereas apoptotic cells contained the catalytically active subunit p19 with almost no intact Mch3α (Fig. 6 B, lane 2). Thus, no cleavage of Mch3α was observed until cells displaying normal morphology acquired apoptotic morphology, suggesting that activation of Mch3α had occurred during the execution phase of apoptosis.
Figure 6.
Processing of Mch3α in apoptotic THP.1 cells: inhibition by ZVAD.FMK but not by YVAD.CMK. (A) THP.1 cells were incubated for up to 4 h, either alone (Con) or in the presence of cycloheximide (CHX) (25 μM) and TLCK (100 μM). Where indicated, cells were pretreated for 1 h with Z-VAD.FMK (50 μM), and then incubated for 4 h in the presence of the apoptotic stimulus. The time course of processing of the proform of Mch3α (lanes 1–5) and its inhibition by Z-VAD.FMK (lane 6) was determined by Western blot analysis as described in Materials and Methods. (B) THP.1 cells were incubated for 4 h in the presence of etoposide (25 μM), stained with Hoechst 33342 and propidium iodide, and then sorted by flow cytometry as previously described (see Materials and Methods and Fig. 2). Cells with either normal (lane 1) or apoptotic (lane 2) morphology were analyzed by Western blot analysis. (C) THP.1 cells were incubated for 4 h, either alone (lane 1) or with cycloheximide (25 μM) and TLCK (100 μM) (lane 2) in the presence of YVAD.CMK (25 μM) (lane 3), and samples were analyzed by Western blot analysis. The proforms of Mch3α and its cleavage product are indicated by the upper and lower arrowheads, respectively (A–C).
To investigate whether the ability of Z-VAD.FMK to inhibit apoptosis (see above) corresponded with an inhibition of the processing of Mch3α, THP.1 cells were pretreated for 1 h with ZVAD.FMK (50 μM), and then coincubated with cycloheximide (25 μM) and TLCK (100 μM) for 4 h. Pretreatment with Z-VAD.FMK inhibited both the processing of proMch3α and the formation of the catalytically active p19 subunit (Fig. 6 A, lane 6). Pretreatment of THP.1 cells for 1 h with YVAD.CMK (25 μM) before treatment with etoposide (25 μM) did not inhibit apoptosis (see above) or the processing of the proform of Mch3α (Fig. 6 C, compare lanes 2 and 3). These results demonstrated that processing of the proform of Mch3α to yield the catalytically active subunit p19 occurred in THP.1 cells undergoing apoptosis, and that inhibition of this processing was concomitant with an inhibition of apoptosis.
Discussion
Processing/Activation of At Least Four ICE-like Proteases Accompanies the Execution of Apoptosis
Apoptosis in THP.1 cells was accompanied by processing of the proforms of three ICE homologues, Ich-1, CPP32, and Mch3α, together with the appearance of their catalytically active p12, p17, and p19 subunits, respectively (Figs. 1 and 6). While we have demonstrated that the loss of CPP32 and Mch3α was due to processing to an active protease (Fig. 5), we cannot state unequivocally that Ich-1 was similarly activated, as an intracellular substrate for Ich-1 has yet to be identified. However, cleavage of Ich-1 to yield its p12 subunit and the ability of Z-VAD.FMK to prevent the loss of Ich-1 (Fig. 3) suggested that Ich-1 was also cleaved by an ICE-like protease to an active enzyme. This is the first demonstration of the processing of Ich-1 in apoptotic cells; thus the identification/development of either natural or synthetic substrates is required to further elucidate the role(s) of Ich-1 in apoptosis. In this regard, it is significant that Z-DEVD.AFC is a very poor substrate for recombinant Ich-1/Nedd2 (Kumar, S., personal communication).
The data derived from pure populations of morphologically normal and apoptotic cells, obtained by fluorescenceactivated cell sorting (Figs. 2 and 6), provided further evidence for the involvement of at least four ICE-like proteases in apoptosis in THP.1 cells. Normal cells contained only the proforms of CPP32 and Mch3α and predominantly pro-Ich-1 together with intact PARP, U1-70K, and lamins A/B. In marked contrast, almost all of the Ich-1, CPP32, and Mch3α was processed in apoptotic cells and the ICE-like protease substrates were extensively cleaved (Figs. 2 and 6). Although it is probable that different ICElike proteases may have overlapping substrate specificities, recent studies with cloned recombinant ICE-like proteases have shown that certain ICE-like proteases cleave specific substrates. Of relevance to the present work, two studies have shown that proteolysis of lamins is mediated by Mch2α but not by CPP32 or Mch3α (Orth et al., 1996; Takahashi et al., 1996). Cleavage of PARP is mediated primarily by CPP32/Mch3α with a possible contribution from Mch2α (Nicholson et al., 1995; Tewari et al., 1995; Fernandes-Alnemri et al., 1995a ,b), while cleavage of U170K is mediated by CPP32 (Casciola-Rosen et al., 1996). Thus, the cleavage in the apoptotic cells of PARP and U170K and in particular lamins A/B (Fig. 2) suggested the activation of Mch2α in addition to CPP32/Mch3α. Further indirect evidence for the activation of Mch2α was provided by the sensitivity of the lamin protease to YVAD. CMK (Fig. 4) (Lazebnik et al., 1995). Direct evidence for the activation of Mch2α during the execution phase of apoptosis was provided by the loss of pro-Mch2α and its prevention by Z-VAD.FMK (Fig. 4 C). These results provide strong evidence for the activation of Ich-1, CPP32, Mch3α, and Mch2α in apoptosis in intact THP.1 cells. This study demonstrates for the first time the activation of at least four ICE-like proteases during the execution phase of apoptosis.
Z-VAD.FMK Inhibits Apoptosis and the Processing of Multiple ICE-like Proteases
The ability of Z-VAD.FMK to inhibit apoptosis in different cell systems (Chow et al., 1995; Fearnhead et al., 1995; Zhu et al., 1995; Cain et al., 1996; Jacobson et al., 1996; Pronk et al., 1996; Slee et al., 1996) has shown that this compound is a valuable tool in the study of apoptosis. Its precise site(s) of action is still unknown, but the present study shows that both apoptosis and the processing of CPP32, Ich-1, Mch3α, and Mch2α were blocked by Z-VAD.FMK (Figs. 3, 4, and 6). As the most likely target for Z-VAD.FMK is an ICE-like protease, it is probable that this unidentified protease is responsible for activation of Ich-1, CPP32, Mch3α, and/or Mch2α. It is notable that YVAD.CMK, designed as a specific inhibitor of ICE, did not significantly inhibit the processing of Ich-1, CPP32, Mch3α, or Mch2α (Figs. 4, A and C, and 6 C), suggesting that the protease upstream of Ich-1, CPP32, Mch3α, and/or Mch2α is not ICE. It appears that Mch4 and/or Mch5/MACH/FLICE might be the most upstream proteases responsible for the activation of multiple CED-3/ICE homologues (Boldin et al., 1996; Fernandes-Alnemri et al., 1996; Muzio et al., 1996). For example, recombinant Mch4 and Mch5 can process both pro-CPP32 and pro-Mch3 (Fernandes-Alnemri et al., 1996; unpublished observations), but whether this occurs in intact cells undergoing apoptosis is not known. Activation of MACH/FLICE occurs as a consequence of tumor necrosis factor– and Fas receptor–mediated cell death (Boldin et al., 1996; Muzio et al., 1996). It remains to be determined whether other forms of apoptosis, such as drug-induced apoptosis, require activation of MACH/ FLICE/Mch5 or other cellular homologues such as Mch4.
Lysates from Apoptotic Cells Contain a CPP32/ Mch3α-like Protease Activity
Lysates, from THP.1 cells exposed to an apoptotic stimulus, exhibited Z-DEVD.AFC cleavage activity, which was potently inhibited by Ac-DEVD.CHO, an inhibitor of CPP32 and Mch3α (Nicholson et al., 1995; FernandesAlnemri et al., 1995b) (Table I). Z-DEVD.AFC hydrolysis activity was due, in part, to cleavage of CPP32, as shown by the appearance of its catalytically active subunit p17 (Fig. 5). Using Z-DEVD.AFC as a substrate, the lysates had a K m of 55 μM, which is intermediate between that for recombinant CPP32 (21.9 μM) and Mch3 (61.5 μM). The K m values found in this study for recombinant CPP32 and Mch3 using Z-DEVD.AFC as a substrate were similar to those reported using Ac-DEVD.AMC (Nicholson et al., 1995; Fernandes-Alnemri et al., 1995b ). Thus, our data suggested that, in addition to CPP32, activation of at least one other CPP32-like protease, most likely Mch-3α, accompanied the execution of apoptosis in THP.1 cells and was responsible for the Z-DEVD.AFC cleavage activity found in the lysates. Activation of Mch3α in THP.1 cell lysates was demonstrated by Western blot analysis (data not shown). Thus, our data demonstrating activation of both CPP32 and Mch3α in THP.1 cell apoptosis is in agreement with a recent study showing processing of these homologues in apoptosis in Jurkat T cells (Chinnaiyan et al., 1996).
ICE Is Not Activated in the Execution Phase of Apoptosis in THP.1 Cells
Our data from both cell lysates and intact cells did not support the involvement of ICE per se in the execution phase of apoptosis in THP.1 cells. Lysates, from THP.1 cells exposed to an apoptotic stimulus, did not exhibit any significant increase in YVAD.AMC cleaving activity (Fig. 5 A). Two additional pieces of evidence suggested that, under our experimental conditions, ICE itself was not activated in THP.1 cells. First, no cleavage of ICE into its catalytically active p20/p10 subunits was observed with an antibody specific to the p10 subunit of ICE (data not shown). Second, YVAD.CMK, designed as a specific inhibitor of ICE, did not inhibit apoptosis in THP.1 cells. Our results, which demonstrate that the execution of apoptosis resulted in a CPP32/Mch3α-like protease activity but not ICE per se, are in agreement with others (Nicholson et al., 1995; Jacobson et al., 1996; Schlegel et al., 1996; Slee et al., 1996; Nett-Fiordalisi et al., 1996). Our observations do not exclude a role for ICE in certain forms of apoptosis (Li et al., 1995; Yuan et al., 1993) such as Fas-mediated apoptosis when a transient increase in ICE activity preceded the appearance of a CPP32/Mch3α-like activity (Enari et al., 1996).
We have demonstrated activation of four ICE-like proteases during the execution phase of apoptosis in THP.1 cells. The inhibition of lamin cleavage, but not apoptosis, by YVAD.CMK (Fig. 4 B) suggested that Mch2α-induced lamin cleavage occurred late in the apoptotic process, in agreement with other studies (Lazebnik et al., 1995; Takahashi et al., 1996). This is supported by the observation that recombinant CPP32 activates pro-Mch2α (Srinivasula et al.,1996). Two pieces of data suggested that Ich-1 is processed early in the apoptotic process: the p12 subunit was observed after 1 h exposure to an apoptotic stimulus (Fig. 1 B), and a small amount of the p12 subunit was also detected in the morphologically normal cells obtained by fluorescence-activated cell sorting (Fig. 2 A). As we cannot exclude the possibility that the Ich-1 antibody may be more sensitive than the other antibodies, we cannot say whether Ich-1 is activated before CPP32 and/or Mch3α. It remains to be determined whether activation of Ich-1 may lead either directly or indirectly to activation of these other homologues.
In summary, we demonstrate that processing/activation of Ich-1, CPP32, Mch3α, and Mch2α accompanies the generation of the apoptotic phenotype in THP.1 cells. Furthermore, we show that Z-VAD.FMK blocks apoptosis, most likely through inhibition of an unidentified key effector ICE-like protease, possibly MACH/FLICE/Mch5 or Mch4, thereby preventing cleavage/processing of Ich-1, CPP32, Mch3α, and Mch2α. This is the first demonstration of the activation of at least four ICE-like proteases during the execution phase of apoptosis in intact cells.
Acknowledgments
We thank R. Snowden for help with the flow cytometry.
This work was supported in part by research grant AG 13847 from the National Institutes of Health (to E.S. Alnemri).
Abbreviations used in this paper
- ICE
interleukin-1β converting enzyme
- PARP
poly(ADP-ribose) polymerase
- TLCK
N-α-p-tosyl-l-lysine chloromethyl ketone
- U1-70K
70-kD protein component of U1 small nuclear ribonucleoprotein
Footnotes
Please address all correspondence to Gerald M. Cohen, MRC Toxicology Unit, Centre for Mechanisms of Human Toxicity, Hodgkin Building, University of Leicester, PO Box 138, Lancaster Road, Leicester LE1 9HN, United Kingdom. Tel.: (44) 116-252-5589/5600. Fax: (44) 116-252-5616. e-mail: gmc2@leicester.ac.uk
References
- Arends MJ, Wyllie AH. Apoptosis: mechanisms and roles in pathology. Int Rev Exp Pathol. 1991;32:223–254. doi: 10.1016/b978-0-12-364932-4.50010-1. [DOI] [PubMed] [Google Scholar]
- Boldin MP, Goncharov TM, Goltsev YV, Wallach D. Involvement of MACH, a novel MORT1/FADD-interacting protease, in Fas/APO1- and TNF receptor-induced cell death. Cell. 1996;85:803–815. doi: 10.1016/s0092-8674(00)81265-9. [DOI] [PubMed] [Google Scholar]
- Bump NJ, Hackett M, Hugunin M, Seshagiri S, Brady K, Chen P, Ferenz C, Franklin S, Ghayur T, Li P, et al. Inhibition of ICE family proteases by baculovirus antiapoptotic protein p35. Science (Wash DC) 1995;269:1885–1888. doi: 10.1126/science.7569933. [DOI] [PubMed] [Google Scholar]
- Cain K, Inayat-Hussain SH, Couet C, Cohen GM. A cleavage site directed inhibitor of interleukin-1β-converting enzyme-like proteases inhibits apoptosis in primary cultures of rat hepatocytes. Biochem J. 1996;314:27–32. doi: 10.1042/bj3140027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Casciola-Rosen LA, Miller DK, Anhalt GJ, Rosen A. Specific cleavage of the 70-kDa protein component of the U1 small nuclear ribonucleoprotein is a characteristic biochemical feature of apoptotic cell death. J Biol Chem. 1994;269:30757–30760. [PubMed] [Google Scholar]
- Casciola-Rosen L, Nicholson DW, Chong KR, Rowan KR, Thornberry NA, Miller DK, Rosen A. Apopain/CPP32 cleaves proteins that are essential for cellular repair: a fundemental principle of apoptotic death. J Exp Med. 1996;183:1957–1964. doi: 10.1084/jem.183.5.1957. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chinnaiyan AM, Orth K, O'Rourke K, Duan H, Poirier GG, Dixit VM. Molecular ordering of the cell death pathway. Bcl-2 and Bcl-xLfunction upstream of the CED-3-like apoptotic protease. J Biol Chem. 1996;271:4573–4576. doi: 10.1074/jbc.271.9.4573. [DOI] [PubMed] [Google Scholar]
- Chow SC, Weis M, Kass GEN, Holmström TH, Eriksson JE, Orrenius S. Involvement of multiple proteases during Fas-mediated apoptosis in T lymphocytes. FEBS Lett. 1995;364:134–138. doi: 10.1016/0014-5793(95)00370-o. [DOI] [PubMed] [Google Scholar]
- Duan H, Chinnaiyan AM, Hudson PL, Wing JP, He W-W, Dixit VM. ICE-LAP3, a novel mammaliam homologue of the Caenorhabditis eleganscell death protein CED-3 is activated during Fas- and tumour necrosis factor-induced apoptosis. J Biol Chem. 1996;271:1621–1625. doi: 10.1074/jbc.271.3.1621. [DOI] [PubMed] [Google Scholar]
- Earnshaw WC. Nuclear changes in apoptosis. Curr Opin Cell Biol. 1995;7:337–343. doi: 10.1016/0955-0674(95)80088-3. [DOI] [PubMed] [Google Scholar]
- Ellis RE, Yuan J, Horvitz HR. Mechanisms and functions of cell death. Annu Rev Cell Biol. 1991;7:663–698. doi: 10.1146/annurev.cb.07.110191.003311. [DOI] [PubMed] [Google Scholar]
- Enari M, Talanian RV, Wong WW, Nagata S. Sequential activation of ICE-like and CPP32-like proteases during Fas-mediated apoptosis. Nature (Lond) 1996;380:723–726. doi: 10.1038/380723a0. [DOI] [PubMed] [Google Scholar]
- Faucheu C, Diu A, Chan AW, Blanchet AM, Miossec C, Herve F, Collard V, Dutilleul, Gu Y, Aldape RA, Lippke JA, et al. A novel human protease similar to the interleukin-1β converting enzyme induces apoptosis in transfected cells. EMBO (Eur Mol Biol Organ) J. 1995;14:1914–1922. doi: 10.1002/j.1460-2075.1995.tb07183.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Faucheau C, Blanchet AM, Collard-Dutilleul V, Lalanne JL, DiuHercend A. Identification of a cysteine protease closely related to interleukin-1β-converting enzyme. Eur J Biochem. 1996;236:207–213. doi: 10.1111/j.1432-1033.1996.t01-1-00207.x. [DOI] [PubMed] [Google Scholar]
- Fearnhead HO, Dinsdale D, Cohen GM. An interleukin-1β-converting enzyme-like protease is a common mediator of apoptosis in thymocytes. FEBS Lett. 1995;375:283–288. doi: 10.1016/0014-5793(95)01228-7. [DOI] [PubMed] [Google Scholar]
- Fernandes-Alnemri T, Litwack G, Alnemri ES. CPP32, a novel human apoptotic protein with homology to Caenorhabditis eleganscell death protein Ced-3 and mammalian interleukin-1β-converting enzyme. J Biol Chem. 1994;269:30761–30764. [PubMed] [Google Scholar]
- Fernandes-Alnemri T, Litwack G, Alnemri ES. Mch2, a new member of the apoptotic Ced-3/ICE cysteine protease gene family. Cancer Res. 1995a;55:2737–2742. [PubMed] [Google Scholar]
- Fernandes-Alnemri T, Takahashi A, Armstrong R, Krebs J, Fritz L, Tomaselli K, Wang L, Yu Z, Croce C, Salveson G, et al. Mch3, a novel human apoptotic cysteine protease highly related to CPP32. Cancer Res. 1995b;55:6045–6052. [PubMed] [Google Scholar]
- Fernandes-Alnemri T, Armstrong RC, Krebs J, Srinivasula SM, Wang L, Bullrich F, Fritz LC, Trapani JA, Tomaselli KJ, Litwack G, et al. In vitroactivation of CPP32 and Mch3 by Mch4, a novel human apoptotic cysteine protease containing two FADD-like domains. Proc Natl Acad Sci USA. 1996;93:7464–7469. doi: 10.1073/pnas.93.15.7464. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harlow, E., and D. Lane. 1988. Antibodies: A Laboratory Manual. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, NY. 726 pp.
- Jacobson MD, Weil M, Raff MC. Role of CED-3/ICE-family proteases in staurosporine-induced programmed cell death. J Cell Biol. 1996;133:1041–1051. doi: 10.1083/jcb.133.5.1041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kamens J, Paskind M, Hugunin M, Talanian RV, Allen H, Banach D, Bump N, Hackett M, Johnston CG, Li P, et al. Identification and characterization of ICH-2, a novel member of the interleukin-1β-converting enzyme family of cysteine proteases. J Biol Chem. 1995;270:15250–15256. doi: 10.1074/jbc.270.25.15250. [DOI] [PubMed] [Google Scholar]
- Kaufmann SH, Desnoyers S, Ottaviano Y, Davidson NE, Poirier GG. Specific proteolytic cleavage of poly (ADP-ribose) polymerase: an early marker of chemotherapy-induced apoptosis. Cancer Res. 1993;53:3976–3985. [PubMed] [Google Scholar]
- Kuida K, Lippke JA, Ku G, Harding MW, Livingston DJ, Su MS, Flavell RA. Altered cytokine export and apoptosis in mice deficient in interleukin-1β converting enzyme. Science (Wash DC) 1995;267:2000–2003. doi: 10.1126/science.7535475. [DOI] [PubMed] [Google Scholar]
- Kumar S. ICE-like proteases in apoptosis. Trends Biochem Sci. 1995;20:198–202. doi: 10.1016/s0968-0004(00)89007-6. [DOI] [PubMed] [Google Scholar]
- Kumar S, Kinoshita M, Noda M, Copeland NG, Jenkins NA. Induction of apoptosis by the mouse Nedd2 gene, which encodes a protein similar to the product of the Caenorhabditis elegans cell death gene ced-3and the mammalian IL-1β-converting enzyme. Genes & Dev. 1994;8:1613–1626. doi: 10.1101/gad.8.14.1613. [DOI] [PubMed] [Google Scholar]
- Lazebnik YA, Kaufmann SH, Desnoyers S, Poirer GG, Earnshaw WC. Cleavage of poly (ADP-ribose) polymerase by a proteinase with properties like ICE. Nature (Lond) 1994;371:346–347. doi: 10.1038/371346a0. [DOI] [PubMed] [Google Scholar]
- Lazebnik YA, Takahashi A, Goldman RD, Poirier GG, Kaufmann SH, Earnshaw WC. Studies of the lamin proteinase reveal multiple parallel biochemical pathways during apoptotic execution. Proc Natl Acad Sci USA. 1995;92:9042–9046. doi: 10.1073/pnas.92.20.9042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li P, Allen H, Banerjee S, Franklin S, Herzog L, Johnston C, McDowell J, Paskind M, Rodman L, Salfeld J, et al. Mice deficient in 1L-1β-converting enzyme are defective in production of mature 1L-1β and resistant to endotoxic shock. Cell. 1995;80:401–411. doi: 10.1016/0092-8674(95)90490-5. [DOI] [PubMed] [Google Scholar]
- Munday NA, Vaillancourt JP, Ali A, Casano FJ, Miller DK, Molineaux SM, Yamin TT, Yu VL, Nicholson DW. Molecular cloning and pro-apoptotic activity of ICEre1II and ICEre1III, members of the ICE/ CED-3 family of cysteine proteases. J Biol Chem. 1995;270:15870–15876. doi: 10.1074/jbc.270.26.15870. [DOI] [PubMed] [Google Scholar]
- Muzio M, Chinnaiyan AM, Kischkel FC, O'Rourke K, Shevchenko A, Ni J, Scaffidi C, Bretz JD, Zhang M, Gentz R, et al. FLICE, a novel FADD-homologous ICE CED-3-like protease, is recruited to the CD95 (FAS/APO-1) death-inducing signaling complex. Cell. 1996;85:817–827. doi: 10.1016/s0092-8674(00)81266-0. [DOI] [PubMed] [Google Scholar]
- Nett-Fiordalisi M, Tomaselli K, Russell JH, Chaplin DD. Macrophage apoptosis in the absence of active interleukin-1β-converting enzyme. J Leukocyte Biol. 1995;58:717–724. doi: 10.1002/jlb.58.6.717. [DOI] [PubMed] [Google Scholar]
- Nicholson DW, Ali A, Thornberry NA, Vaillancourt JP, Ding CK, Callant M, Gareau Y, Griffin PR, Labelle M, Lazebnik YA, et al. Identification and inhibition of the ICE/CED-3 protease necessary for mammalian apoptosis. Nature (Lond) 1995;376:37–43. doi: 10.1038/376037a0. [DOI] [PubMed] [Google Scholar]
- Ormerod M, Sun X-M, Snowden RT, Davies R, Fearnhead H, Cohen GM. Increased membrane permeability of apoptotic thymocytes: a flow cytometric study. Cytometry. 1993;14:595–602. doi: 10.1002/cyto.990140603. [DOI] [PubMed] [Google Scholar]
- Orth K, Chinnaiyan AM, Garg M, Froelich CJ, Dixit VM. The CED-3/ICE-like protease Mch2 is activated during apoptosis and cleaves the death substrate lamin A. J Biol Chem. 1996;271:16443–16446. [PubMed] [Google Scholar]
- Pronk GJ, Ramer K, Amiri P, Williams LT. Requirement of an ICE-like protease for induction of apoptosis and ceramide generation by REAPER. Science (Wash DC) 1996;271:808–810. doi: 10.1126/science.271.5250.808. [DOI] [PubMed] [Google Scholar]
- Raff MC. Social controls on cell survival and cell death. Nature (Lond) 1992;356:397–400. doi: 10.1038/356397a0. [DOI] [PubMed] [Google Scholar]
- Ray CA, Black RA, Kronheim SR, Greenstreet TA, Sleath PR, Salvesen GS, Pickup DJ. Viral inhibition of inflammation: cowpox virus encodes an inhibitor of the interleukin-1β converting enzyme. Cell. 1992;69:597–604. doi: 10.1016/0092-8674(92)90223-y. [DOI] [PubMed] [Google Scholar]
- Schlegel J, Peters I, Orrenius S, Miller DK, Thornberry NA, Yamin T-T, Nicholson DW. CPP32/Apopain is a key interleukin 1β converting enzyme-like protease involved in Fas-mediated apoptosis. J Biol Chem. 1996;271:1841–1844. doi: 10.1074/jbc.271.4.1841. [DOI] [PubMed] [Google Scholar]
- Slee EA, Zhu H, Chow SC, MacFarlane M, Nicholson DW, Cohen GM. Benzyloxycarbonyl-val-ala-asp (OMe) fluoromethylketone (ZVAD.FMK) inhibits apoptosis by blocking the processing of CPP32. Biochem J. 1996;315:21–24. doi: 10.1042/bj3150021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Song Q, Lees-Miller SP, Kumar S, Zhang N, Chan DW, Smith GCM, Jackson SP, Alnemri ES, Litwack G, Khanna KK, Lavin MF. DNA-dependent protein kinase catalytic subunit: a target for an ICE-like protease in apoptosis. EMBO (Eur Mol Biol Organ) J. 1996;15:3238–3246. [PMC free article] [PubMed] [Google Scholar]
- Srinivasula SM, Fernandes-Alnemri T, Zangrill J, Robertson N, Armstrong RC, Wang L, Trapani JA, Tomaselli KJ, Litwack G, Alnemri ES. The Ced-3/interleukin 1β converting enzyme-like homolog Mch6 and the lamin-cleaving enzyme Mch2α are substrates for the apoptotic mediator CPP32. J Biol Chem. 1996;271:27099–27106. doi: 10.1074/jbc.271.43.27099. [DOI] [PubMed] [Google Scholar]
- Sun X-M, Snowden RT, Skilleter DN, Dinsdale D, Ormerod MG, Cohen GM. A flow cytometric method for the separation and quantitation of normal and apoptotic thymocytes. Anal Biochem. 1992;204:351–356. doi: 10.1016/0003-2697(92)90251-2. [DOI] [PubMed] [Google Scholar]
- Takahashi A, Earnshaw WC. ICE-related proteases in apoptosis. Curr Opin Gen Dev. 1996;6:50–55. doi: 10.1016/s0959-437x(96)90010-6. [DOI] [PubMed] [Google Scholar]
- Takahashi A, Alnemri ES, Lazebnik YA, Fernandes-Alnemri T, Litwack G, Moir RD, Goldman RD, Poirier GG, Kaufmann SH, Earnshaw WC. Cleavage of lamin A by Mch2α but not CPP32: multiple interleukin 1β-converting enzyme-related proteases with distinct substrate recognition properties are active in apoptosis. Proc Natl Acad Sci USA. 1996;93:1220–1225. doi: 10.1073/pnas.93.16.8395. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tewari M, Quan LT, O'Rourke K, Desnoyers S, Zeng Z, Beidler DR, Poirier GG, Salvesen GS, Dixit VM. Yama/CPP32 β, a mammalian homolog of CED-3, is a CrmA-inhibitable protease that cleaves the death substrate poly(ADP-ribose) polymerase. Cell. 1995;81:801–809. doi: 10.1016/0092-8674(95)90541-3. [DOI] [PubMed] [Google Scholar]
- Thornberry NA. Interleukin-1β converting enzyme converting enzyme. Methods Enzymol. 1994;244:615–631. doi: 10.1016/0076-6879(94)44045-x. [DOI] [PubMed] [Google Scholar]
- Thornberry NA, Molineaux SM. Interleukin 1β converting enzyme. A novel cysteine protease required for IL-1β production and implicated in programmed cell death. Protein Sci. 1995;4:3–12. doi: 10.1002/pro.5560040102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thornberry NA, Bull HG, Calaycay JR, Chapman KT, Howard AD, Kostura MJ, Miller DK, Molineaux SM, Weidner JR, Aunins J, et al. A novel heterodimeric cysteine protease is required for interleukin-1β processing in monocytes. Nature (Lond) 1992;356:768–774. doi: 10.1038/356768a0. [DOI] [PubMed] [Google Scholar]
- Wang L, Miura M, Bergeron L, Zhu H, Yuan J. Ich-1, an Ice/ced3-related gene, encodes both positive and negative regulators of programmed cell death. Cell. 1994;78:739–750. doi: 10.1016/s0092-8674(94)90422-7. [DOI] [PubMed] [Google Scholar]
- Wang X, Pai J-T, Wiedenfeld EA, Medina JC, Slaughter CA, Goldstein JL, Brown MS. Purification of an interleukin-1β converting enzyme-related cysteine protease that cleaves sterol regulatory element-binding proteins between the leucine zipper and transmembrane domains. J Biol Chem. 1995;270:18044–18050. doi: 10.1074/jbc.270.30.18044. [DOI] [PubMed] [Google Scholar]
- Yuan J, Shaham S, Ledoux S, Ellis HM, Horvitz HR. The C. elegans cell death gene ced-3encodes a protein similar to mammalian interleukin-1β-converting enzyme. Cell. 1993;75:641–652. doi: 10.1016/0092-8674(93)90485-9. [DOI] [PubMed] [Google Scholar]
- Zhu H, Fearnhead HO, Cohen GM. An ICE-like protease is a common mediator of apoptosis induced by diverse stimuli in human monocytic THP.1 cells. FEBS Lett. 1995;374:303–308. doi: 10.1016/0014-5793(95)01116-v. [DOI] [PubMed] [Google Scholar]