Abstract
Several eubacteria including Esherichia coli use an alternative nonmevalonate pathway for the biosynthesis of isopentenyl diphosphate instead of the ubiquitous mevalonate pathway. In the alternative pathway, 2-C-methyl-d-erythritol or its 4-phosphate, which is proposed to be formed from 1-deoxy-d-xylulose 5-phosphate via intramolecular rearrangement followed by reduction process, is one of the biosynthetic precursors of isopentenyl diphosphate. To clone the gene(s) responsible for synthesis of 2-C-methyl-d-erythritol 4-phosphate, we prepared and selected E. coli mutants with an obligatory requirement for 2-C-methylerythritol for growth and survival. All the DNA fragments that complemented the defect in synthesizing 2-C-methyl-d-erythritol 4-phosphate of these mutants contained the yaeM gene, which is located at 4.2 min on the chromosomal map of E. coli. The gene product showed significant homologies to hypothetical proteins with unknown functions present in Haemophilus influenzae, Synechocystis sp. PCC6803, Mycobacterium tuberculosis, Helicobacter pyroli, and Bacillus subtilis. The purified recombinant yaeM gene product was overexpressed in E. coli and found to catalyze the formation of 2-C-methyl-d-erythritol 4-phosphate from 1-deoxy-d-xylulose 5-phosphate in the presence of NADPH. Replacement of NADPH with NADH decreased the reaction rate to about 1% of the original rate. The enzyme required Mn2+, Co2+, or Mg2+ as well. These data clearly show that the yaeM gene encodes an enzyme, designated 1-deoxy-d-xylulose 5-phosphate reductoisomerase, that synthesizes 2-C-methyl-d-erythritol 4-phosphate from 1-deoxy-d-xylulose 5-phosphate, in a single step by intramolecular rearrangement and reduction and that this gene is responsible for terpenoid biosynthesis in E. coli.
Isoprenoids play important roles in all living organisms such as steroid hormones in mammals, carotenoids in plants, and ubiquinone or menaquinone in bacteria (1). Since the initial discovery of the mevalonate pathway in the 1950s, it was widely accepted that isopentenyl diphosphate (IPP), the fundamental unit in terpenoid biosynthesis, was only formed by condensation of acetyl CoA through the ubiquitous mevalonate pathway. However, it has been disclosed recently that many organisms including several bacteria, green algae, and chloroplasts of higher plants use an alternative mevalonate-independent pathway (nonmevalonate pathway) for the formation of IPP (2–9). Several recent experimental findings lend additional supports to the operation of this pathway in terpenoid biosynthesis (10–13).
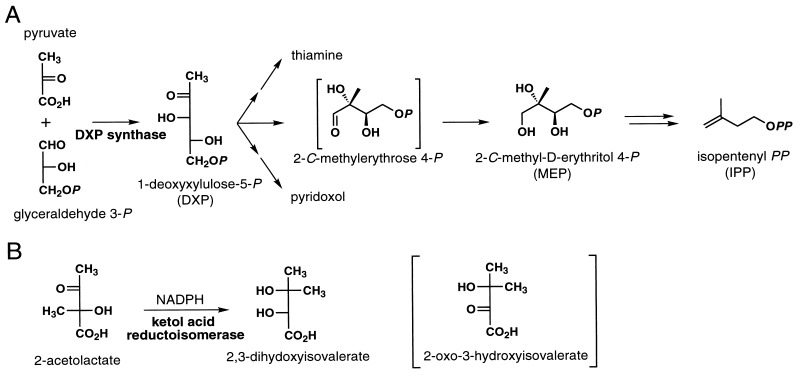
According to Rohmer and coworkers (4, 9), the initial step of this pathway is the formation of 1-deoxy-d-xylulose 4-phosphate (DXP) by condensation of pyruvate and glyceraldehyde 3-phosphate (Fig. 1A). In the second step the intramolecular rearrangement of DXP is assumed to give a hypothetical rearrangement intermediate 2-C-methylerythrose 4-phosphate, which is then converted to 2-C-methyl-d-erythritol 4-phosphate (MEP) by an unspecified reduction process (7). Thus, precise details about the reductive mechanism for the formation of MEP remained to be unclear.
Figure 1.
Hypothetical alternative nonmevalonate pathway for IPP biosynthesis (A) and ketol acid reductoisomerase reaction (B).
A precedent for this biosynthetic rearrangement reaction is found in the biosynthesis of valine, isoleucine, and leucine. The enzyme involved in this reaction ketol acid reductoisomerase (EC1.1.1.86) catalyzes rearrangement of 2-acetolactate to yield 2,3-dihydroxyisovalerate with simultaneous reduction in the presence of NADPH (Fig. 1B) (14–17). It should be noted that a hypothetical compound with a keto function corresponding to 2-C-methylerythrose 4-phosphate had been excluded as a possible reaction product intermediate because of its inhibitory action on this reaction (15). By analogy with the reaction by ketol acid reductoisomerase, MEP could be synthesized by rearrangement and reduction of DXP in a single step. Therefore, we initially considered the possibility that MEP might be synthesized from DXP by the same reaction mechanism used by ketol acid reductoisomerase. If the assumption were correct, an enzyme for synthesizing MEP from DXP might have sequence similarity to ketol acid reductoisomerase. However, we were unable to find other proteins homologous to ketol acid reductoisomerase in the total genome sequence of Escherichia coli (18). This result suggested that MEP may be synthesized in a different reaction mechanism from that of ketol acid reductoisomerase. Therefore, no information about the enzyme(s) for MEP formation has been obtained from our knowledge on ketol acid reductoisomerase.
DXP is a biosynthetic intermediate not only of IPP but also of thiamine and pyridoxol in E. coli (19–21). Therefore, the synthesis of MEP from DXP seems to be the committed step in IPP formation (Fig. 1A). This speculation has led us to the idea that auxotrophic mutants requiring MEP for growth and survival should be promising hosts for the cloning of the gene responsible for synthesis of MEP, because such mutants would possess a metabolic block(s) between DXP and MEP. To obtain such mutants, we used 2-C-methylerythritol (ME), the free alcohol of MEP, in place of MEP to overcome the problem associated with uptake of phosphorylated compounds into cells. Fortunately, ME is used by E. coli in the place of MEP (7). As an additional advantage, this strategy enabled us to avoid somewhat tedious procedures for preparation of the phosphate ester MEP. With chemically synthesized ME (7) in hand, we tried to isolate the mutants that could grow on a minimal medium containing ME but not in the absence of ME. Such mutants should not be able to synthesize MEP and, hence, should be unable to grow on the minimal medium that lacks ME.
We show in this study that the yaeM gene (GenBank accession no. AE000126.) at 4.2 min on the E. coli chromosome complemented the IPP biosynthesis coding region of all the isolated mutants. We also demonstrate that the yaeM gene product, designated DXP reductoisomerase, catalyzes intramolecular rearrangement and reduction of DXP to form MEP in a single step. Preliminary results of this work have been reported (22).
MATERIALS AND METHODS
Bacterial Strains.
The E. coli K-12 strains W3110 (wild-type) and JM109 (recA1, endA1, gyrA96, thi, hsdR17, supE44, relA1, Δ(lac-proAB)/F′[traD36, proAB+, LacIq, lacZΔM15]) (23) were used in this study. W3110 was provided by Hajime Tokuda (Institute of Molecular and Cellular Biosciences, University of Tokyo). JM109 was purchased from Takara Shuzo (Kyoto).
Mutagenesis of E. coli.
W3110 by N-Methyl-N′-nitro-N-nitrosoguanidine. N-Methyl-N′-nitro-N-nitrosoguanidine (Nacalai Tesque, Kyoto) was used as a mutagen to obtain mutants defective in biosynthesis of MEP. N-Methyl-N′-nitro-N-nitrosoguanidine-treated W3110 cells were grown on M9 plates (24) containing 0.1% ME, which was chemically synthesized as reported (7). Agarose (ultra PURE, GIBCO/BRL) was used for preparation of M9 plates. Well-separated colonies on the M9 plates were in duplicate inoculated on M9 plates with and without 0.1% ME. Mutants growing only on the M9 containing ME were selected. Three mutants thus isolated indeed required ME for growth and survival. These mutants were used as the hosts for the cloning a gene(s) responsible for synthesizing MEP.
Cloning of a Gene(s) that Complements the IPP Biosynthesis Coding Region of the Isolated Mutants.
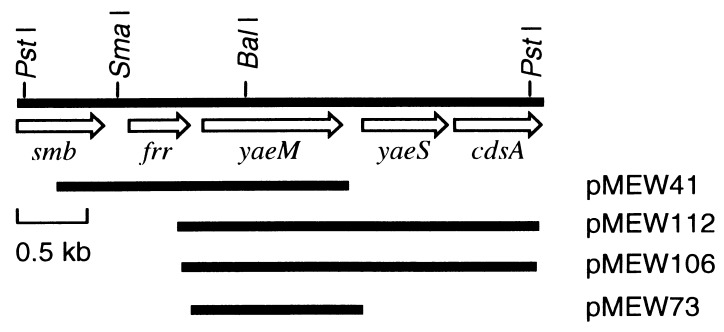
DNA manipulations and transformation in E. coli were done as described by Maniatis et al. (24). Chromosomal DNA extracted from E. coli W3110 was partially digested with Sau3AI and size-fractionated (2–4 kb) by agarose gel electrophoresis. The 2- to 4-kb DNA fragments were ligated to BamHI- and phosphatase-treated pMW118 (refs. 25 and 26; Nippon Gene, Toyama, Japan). The resulting plasmids were used as a genomic library of E. coli W3110 for cloning a gene(s) responsible for synthesizing MEP. The mutants described above were transformed with the genomic library, and then ampicillin-resistant transformants were selected on Luria–Bertani (LB) (24) plates without ME. Plasmids, designated pMEW41, 73, 106, and 112 (see Fig. 3), were extracted from these transformants and sequenced as described bellow.
Figure 3.
Restriction map around the yaeM gene and DNA fragments that complemented IPP biosynthesis coding region of the isolated mutants. The arrows indicate the extents and directions of the ORFs found in the database of E. coli genome sequence. smb, frr, and cdsA have been identified as MukB suppressor protein, ribosome recycling factor, and phosphatidate cytidylyltransferase, respectively. Both yaeM and yaeS have been reported as hypothetical proteins. pMEW73 contains only the yaeM gene in its inserted DNA. The plasmid overcame requirement of ME for growth of all the mutants.
DNA Sequence Analysis.
DNA sequence was determined by the dideoxynucleotide chain-termination method (27) with an automated sequencer (Li-cor, model 4000L) and the protocol of the supplier. A homology search with protein databases was performed by the fasta program (28, 29).
Construction of the Plasmid for Overexpression of the yaeM Gene.
On the basis of the entire nucleotide sequence of the yaeM gene from E. coli W3110, two oligonucleotide primers, 5′-GGGGGATCCAAGCAACTCACCATTCTGGGC-3′ (5′ of the yaeM gene) and 5′-GGGGGATCCGCTTGCGAGACGCATCACCTC-3′ (3′ of the yaeM gene), including BamHI restriction sites (italic) were synthesized (Amersham Pharmacia). These primers were then used with total DNA from W3110 to amplify the yaeM gene. By using Taq DNA polymerase (Boehringer) and the protocol of the supplier, a 1,209-bp fragment for the yaeM gene was amplified. The PCR fragment was cleaved with BamHI and cloned into pUC118 (Takara). Strain JM109 was used as a recipient in this transformation. Clones were analyzed for the correct insert by DNA sequencing as described above. The correct fragment was cloned into the multicloning site (BamHI) of the expression vector pQE30 (Qiagen, Chatsworth, CA) to give pQEDXR for the yaeM gene. Expression and purification of the recombinant yaeM gene product were done as described (22).
Electrophoresis.
Proteins were separated by SDS-PAGE on 8–25% gels or native PAGE on 8–25% gels with PhastSystem (Pharmacia). Molecular weight electrophoresis calibration kits (Pharmacia) were used as standards.
Determination of Molecular Mass.
The molecular mass of the native DXP reductoisomerase was estimated by gel filtration on a Toyopearl HW65 column (2 × 90 cm) equilibrated in 100 mM Tris⋅HCl (pH 7.5). The column was eluted at a flow rate of 0.3 ml/min and fractions of 2 ml were collected. The molecular mass was estimated by comparing the elution of DXP reductoisomerase with that of the standard proteins: ferritin (440 kDa), catalase (232 kDa), aldolase (158 kDa), and BSA (66 kDa).
Assay of DXP Reductoisomerase.
The assay system contained 100 mM Tris⋅HCl (pH 7.5), 1 mM MnCl2, 0.3 mM NADPH, and 0.3 mM enzymatically synthesized DXP (22) in a final volume of 1 ml. The reaction was initiated by adding DXP to the complete assay mixture. The oxidation of NADPH was monitored in a Shimadzu UV-160 spectrophotometer equipped with a cell holder adjusted at 37°C. One unit of DXP reductoisomerase activity is defined as the amount of enzyme that caused the oxidation of 1 μmol of NADPH per min.
RESULTS
Isolation of Mutants that Require ME for Growth.
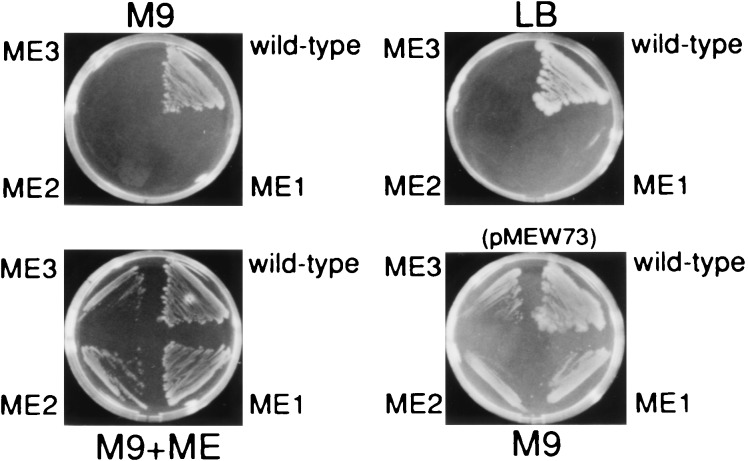
We initiated isolation of mutants that required ME for growth and survival. Some of these mutants would possess a metabolic block(s) between DXP and MEP. E. coli K-12 wild-type strain W3110 was treated with N-methyl-N′-nitro-N-nitrosoguanidine and then spread on the M9 plates containing chemically synthesized ME. A total of 2 × 104 well-separated colonies were transferred to the M9 plates with and without ME. We found three colonies that were able to grow on the M9 plates only in the presence of ME. As shown in Fig. 2, these mutants showed the expected properties; the addition of the free alcohol of MEP, but not of 1-deoxyxylulose (DX; data not shown), to the minimal medium M9 facilitated the growth of these mutants. In addition, these mutants did not grow on the LB plates or on the M9 plates, showing that the components in LB plates do not facilitate the growth of these mutants. These data proved that these three mutants were defective in synthesizing MEP.
Figure 2.
Phenotypes of the isolated mutants and the parent strain W3110. The three mutants designated ME1, ME2, and ME3 cannot grow on M9 and LB plates but do grow on M9 plates supplemented 0.1% ME (M9+ME). In addition, these mutants transformed with pMEW73 (Fig. 3) become able to grow on M9 without ME (M9) (Lower Right).
Cloning of a Gene that Complements the IPP Biosynthesis Coding Region of the Mutants.
With these mutants in hand, we next tried to clone a gene(s) that complements the IPP biosynthesis coding region of the isolated mutants. Each of these three mutants was transformed with a genomic library of W3110, and ampicillin-resistant transformants were at first selected on the LB plates containing ampicillin (50 μg/ml). The six selected transformants were then transferred to the M9 plates containing ampicillin (50 μg/ml). Fig. 3 shows a map of four plasmids, pMEW41, 73, 106, and 112, isolated from the six transformants. Sequence analysis of these plasmids revealed that the yaeM gene, which is located at 4.2 min on the chromosomal map of E. coli, was inserted in common into the vector plasmid. Therefore, all the mutants could be yaeM-deficient mutants. In fact, pMEW73 containing only the yaeM gene as its inserted DNA was able to overcome requirement of ME for growth of all the mutants (Fig. 2). In addition, the yaeM disruptants generated by homologous recombination were also auxotrophic for ME as were the mutants induced by N-methyl-N′-nitro-N-nitrosoguanidine treatment (data not shown).
yaeM Homologs Are Widespread in Eubacteria.
The yaeM gene product has 398 amino acid residues with a molecular mass of 43 kDa. The amino acid sequence was compared with other known amino acid sequences in the databases (Swiss-Prot and the Protein Identification Resource) of DNA Data Bank of Japan (DDBJ) by using the fasta program. Fig. 4 represents a multiple alignment of the amino acid sequences of the E. coli yaeM gene and other yaeM gene homologs. The amino acid sequence of the E. coli yaeM gene product shows 33.9% to 62.4% identities to other hypothetical proteins with unknown functions encoded in the genomes of Haemophilus influenzae, Synechocystis sp. (PCC6803), Mycobacterium tuberculosis, Helicobacter pyroli, and Bacillus subtilis.
Figure 4.
Multiple alignment of the amino acid sequences of E. coli yaeM gene and other yaeM gene homologs. Identical amino acids among six proteins are marked by asterisks. The putative NADPH binding motif is marked by #. Dashes indicate gaps introduced for the optimization of the alignment. Ec, E. coli (accession no. AB013300); Bs, Bacillus subtilis (accession no. B69881); Hi, Haemophilus influenzae (accession no. P44055); Hp, Helicobacter pyroli, (accession no. P56139); Mycobacterium tuberculosis (accession no. Q10798), Sy, Synechocystis sp. PCC6803 (accession no. Q55663).
Biochemical Properties of the yaeM Gene Product.
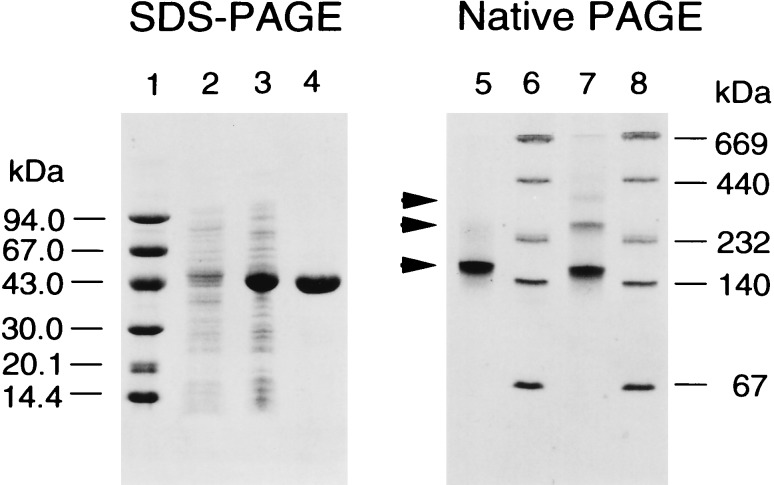
To demonstrate that the yaeM gene was actually involved in IPP biosynthesis, we constructed a plasmid for the overexpression of this gene product and producing a one-step purification of the recombinant enzyme by Ni-nitrilotriacetic acid resin. The purified recombinant enzyme gave a homogeneous protein band on SDS/PAGE with a subunit size of 42 kDa (Fig. 5). The relative molecular mass of the enzyme under native condition was determined to be 150 kDa by Toyopearl HW65 gel filtration. Thus the enzyme appears to form a homotetramer. On the other hand, native polyacrylamide gradient gel electrophoresis led to the appearance of three bands with estimated molecular mass of 355, 260, and 165 (predominant form) kDa (Fig. 5). We assume that these bands reflect octameric, hexameric, and tetrameric forms of the enzyme, respectively. When an enzyme solution including 10 mM DTT was applied to native PAGE, only the tetrameric form was observed (Fig. 5).
Figure 5.
Expression and purification of the recombinant DXP reductoisomerase. SDS/PAGE of the enzyme. Lanes: 1, low molecular mass marker; 2, the extract of uninduced E. coli M15 (pREP4, pQEDXR) culture; 3, the same extract after induction with isopropyl β-d-thiogalactoside; 4, purified enzyme after Ni-nitrilotriacetic acid agarose chromatography. Native PAGE of the purified enzyme. Lanes: 5, purified enzyme with 10 mM DTT; 6 and 8, high molecular mass markers; 7, purified enzyme without DTT. Positions of DXP reductoisomerase are indicated by arrows.
Incubation of this enzyme with enzymatically synthesized DXP in the presence of NADPH resulted in the oxidation of NADPH (22). Replacement of NADPH with NADH decreased the reaction rate to about 1% of the original rate. Thus the enzyme uses only NADPH as a coenzyme for this reaction. The enzyme required Mn2+, Co2+, or Mg2+, and the optimum concentrations of the divalent cations were 1 mM. The activity was maximal with Mn2+, whereas Co2+ or Mg2+ was less effective than Mn2+. When the concentrations of these divalent cations were increased to 5 mM, the activity was suppressed by 40–60%. No effects of other divalent cations such as Ca2+, Ni2+, or Zn2+ at 1 mM were detected (Table 1). Addition of EDTA, Cu2+, or Fe2+ resulted in the complete loss of the enzyme activity. The activity was optimal between pH 7.5 and 8.0 in Tris⋅HCl buffer.
Table 1.
Effects of divalent cations on DXP reductoisomerase activity
| Addition | Specific activity, units/mg protein |
|---|---|
| No | 0.3 |
| EDTA | ND |
| MnC12 | 11.8 |
| CoCl2 | 6.0 |
| MgCl2 | 4.0 |
| CaCl2 | 0.2 |
| NiSO4 | 0.2 |
| ZnSO4 | 0.3 |
| CuSO4 | ND |
| FeSO4 | ND |
Enzyme activity was assayed in the standard mixture described under Materials and Methods with 1 mM metal chlorides, metal sulfates, or EDTA. ND, not detected.
We have previously demonstrated that the yaeM gene product synthesized MEP in the presence of NADPH by rearrangement and reduction of DXP in a single step (22). In consideration of this result, we propose that the yaeM gene product is designated 1-deoxy-d-xylulose 5-phosphate reductoisomerase and that the yaeM gene is renamed dxr for 1-deoxy-d-xylulose 5-phosphate reductoisomerase.
DISCUSSION
DXP and MEP were reported to be metabolic intermediates of the alternative nonmevalonate pathway for terpenoid biosynthesis (Fig. 1A). MEP could be synthesized by rearrangement and reduction of DXP. Details about the reaction mechanism of the MEP formation from DXP, however, were not disclosed nor made available from earlier studies. To further elucidate the details of this interesting mechanism of the MEP formation, we initiated studies on the cloning of the gene responsible for synthesis of MEP. To this end, we used the useful strategy of preparing and selecting E. coli mutants auxotrophic for ME. This free alcohol of MEP has been proved to be incorporated into ubiquinone and menaquinone of E. coli in place of MEP. We succeeded in obtaining three mutants with the expected phenotypes (Fig. 2) from about 20,000 colonies screened and in cloning a gene that complemented the IPP biosynthesis coding region of these mutants. The gene yaeM (now renamed dxr) had been identified as a gene encoding a protein with unknown function. Homology searches using the fasta program in amino acid sequence databases (Swiss-Prot and the Protein Identification Resource), however, detected homologs of the dxr gene in other eubacteria such as B. subtilis, H. influenzae, H. pyroli, M. tuberculosis, and Synechocystis sp. PCC6803, whose complete genome sequences have been available. All these eubacteria were also reported to possess dxs homologs encoding DXP synthase, which catalyzes the formation of DXP from pyruvate and glyceraldehyde 3-phosphate (30, 31), showing that these organisms use the nonmevalonate pathway for terpenoid biosynthesis. We emphasize that no homolog of the dxr gene has been found in animals and higher plants. However, because chloroplasts of higher plants use the nonmevalonate pathway (8, 32), dxr gene homologs will be found in higher plants when these complete genome sequences are available.
To characterize the properties of the dxr gene product overexpressed in E. coli, we purified the recombinant enzyme to homogeneity. On native PAGE, the enzyme showed octameric, hexameric, and tetrameric forms. When an enzyme solution containing 10 mM DTT was incubated for 10 min at 4°C, only the tetrameric form was detected on native PAGE. This indicates the formation of disulfide bonds for the octameric and hexameric forms of the enzyme. We speculate that the formation of disulfide bonds is not significant for the expression of the enzyme activity, because the addition of 0.1–10 mM DTT to the enzyme had no effect on the enzyme activity.
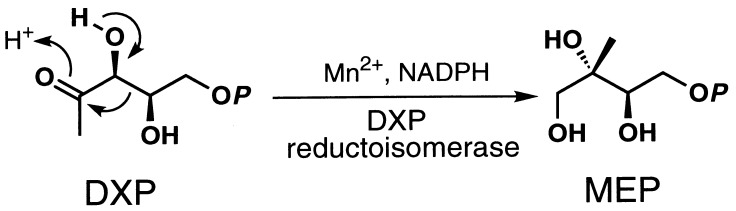
We have previously demonstrated the dxr gene product catalyzes rearrangement and reduction of DXP to yield MEP in the presence of NADPH in a single step (Fig. 6) (22). As described in the Introduction, ketol acid reductoisomerase catalyzes the rearrangement of 2-acetolactate to yield 2,3-dihydroxyisovalerate with simultaneous reduction in the presence of NADPH. Both enzymes have an optimum pH around 7.5 and used NADPH as a reducing regent. DXP reductoisomerase requires a divalent cation such as Mn2+, Co2+, or Mg2+ with Mn2+ being the most effective metal ion for its activity. On the other hand, ketol acid reductoisomerase absolutely requires Mg2+. In addition, Mn2+ did behave as a competitive inhibitor of ketol acid reductoisomerase with respect to Mg2+ (33). This difference between these two reductoisomerases may reflect the fact that a putative Mg2+ binding site (34, 35) for ketol acid reductoisomerase was not conserved in the DXP reductoisomerase sequence.
Figure 6.
DXP reductoisomerase reaction.
It is interesting to note that the recombinant DXP synthase synthesized DX from pyruvate and dl-glyceraldehyde (30, 31). In addition, DX was reported to be used by E. coli in place of DXP. Then we investigated whether DXP reductoisomerase was able to convert DX to ME. At concentrations up to 2 mM DX, no change in the A340 was detected when the incubation included Mn2+ and NADPH in our standard buffer (data not shown). This result indicates that DXP is an actual precursor for the IPP biosynthesis and that DX incorporated into E. coli cells needs to be converted to DXP by a certain kinase(s) to be used as the precursor.
The deduced amino acid sequence of the dxr gene of E. coli shows 33.9% to 62.4% identities to other dxr homologs. The amino acid sequence alignment shown in Fig. 4 reveals that the conserved amino acid residues are found in the total sequence. These conserved residues may function as important catalytic domains of the enzyme. In particular, a conserved motif (LGXTGSIG) in their N-terminal regions is deduced to be an NADPH-binding site. The motif is also found in the N-terminal region of ketol acid reductoisomerases isolated from Arabidopsis thaliana (35), Spinacia oleracea (36), Saccharomyces cerevisiae (37) and Neurospora crassa (34). Because the dxr gene product (DXP reductoisomerase) uses NADPH as a cofactor as well, the motif found in this enzyme could be an NADPH binding site. It is reasonably assumed that DXP reductoisomerase uses NADPH for reduction of 2-C-methylerythrose 4-phosphate, a hypothetical reaction intermediate in the formation of MEP proposed by Rohmer et al. (7). This process may be carried out at the enzyme bound stage, because the hypothetical intermediate could not be detected as a reaction product. In the ketol acid reductoisomerase reaction, it is inferred that 2-oxo-3-hydroxyisovalerate, a hypothetical compound corresponding to 2-C-methylerythrose 4-phosphate, could remain tightly bound to the enzyme during 2,3-dihydroxyisovalerate formation (38).
The dxr and the dxs genes locate at 4.2 min and 9.0 min on the chromosomal map of E. coli, respectively (18). This suggests that other unknown genes involved in the nonmevalonate pathway may be scattered on the genome of E. coli. However, the dxr gene and the immediately downstream gene yaeS (Swiss-Prot accession no. Q47675) could constitute an operon. In addition, yaeS homologs are found in H. influenzae, B. subtilis, Synechocystis sp. (PCC6803), H. pyroli, and M. leprae. Therefore, although the yaeS gene product has been identified as a hypothetical protein with an unknown function, its gene product could constitute the nonmevalonate pathway. Further investigation on this gene is now under way.
DXP is a biosynthetic intermediate not only for IPP but also for thiamine and pyridoxol in E. coli (19–21), and MEP is a precursor characteristic of the IPP biosynthesis (7). Therefore, our result on the in vitro formation of MEP from DXP provides evidence that the reaction catalyzed by DXP reductoisomerase is the committed step in the IPP formation (22).
Acknowledgments
Mass spectra were collected by Dr. N. Morisaki of Institute of Molecular and Cellular Biosciences, University of Tokyo. This work was supported in part by a Grant-in-Aid for Encouragement of Young Scientists (09760114 to T.K.) and by a Grant-in-Aid for Scientific Research (B) to H.S. from The Ministry of Education, Science, Sports and Culture, Japan.
ABBREVIATIONS
- IPP
isopentenyl diphosphate
- DX
1-deoxyxylulose
- DXP
1-deoxy-d-xylulose 5-phosphate
- ME
2-C-methylerythritol
- MEP
2-C-methyl-d-erythritol 4-phosphate
Footnotes
Data deposition: The sequence reported in this paper has been deposited in the GenBank database (accession no. AB013300).
References
- 1.Sacchettini J C, Poulter C D. Science. 1997;277:1788–1789. doi: 10.1126/science.277.5333.1788. [DOI] [PubMed] [Google Scholar]
- 2.Rohmer M, Knani M, Simonin P, Sutter B, Sahm H. Biochem J. 1993;295:517–524. doi: 10.1042/bj2950517. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Lichtenthaler H K, Schwender J, Disch A, Rohmer M. Biochem J. 1996;316:73–80. doi: 10.1042/bj3160073. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Rohmer M, Seeman M, Horbach S, Bringer-Meyer S, Sahm H. J Am Chem Soc. 1996;118:2564–2566. [Google Scholar]
- 5.Seto H, Watanabe H, Furihata K. Tetrahedron Lett. 1996;37:7979–7982. [Google Scholar]
- 6.Duvold T, Bravo J M, Pale-Grosdemange C, Rohmer M. Tetrahedron Lett. 1997;38:4769–4772. [Google Scholar]
- 7.Duvold T, Cali P, Bravo J M, Rohmer M. Tetrahedron Lett. 1997;38:6181–6184. [Google Scholar]
- 8.Lichtenthaler H K, Schwender J, Disch A, Rohmer M. FEBS Lett. 1997;400:271–274. doi: 10.1016/s0014-5793(96)01404-4. [DOI] [PubMed] [Google Scholar]
- 9.Arigoni D, Sagner S, Latzel C, Eisenreich W, Bacher A, Zenk M H. Proc Natl Acad Sci USA. 1997;94:10600–10605. doi: 10.1073/pnas.94.20.10600. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Putra S R, Lois L M, Campos N, Boronat A, Rohmer M. Tetrahedron Lett. 1998;39:23–26. [Google Scholar]
- 11.Sagner S, Eisenreich W, Fellermeier M, Latzel C, Bacher A, Zenk M H. Tetrahedron Lett. 1998;39:2091–2094. [Google Scholar]
- 12.Li S-M, Hennig S, Heide L. Tetrahedron Lett. 1998;39:2717–2720. [Google Scholar]
- 13.Orihara N, Kuzuyama T, Takahashi S, Furihata K, Seto H. J Antibiot. 1998;51:676–678. doi: 10.7164/antibiotics.51.676. [DOI] [PubMed] [Google Scholar]
- 14.Umbarger H E, Brown B, Erying E. J Biol Chem. 1960;235:1425–1432. [PubMed] [Google Scholar]
- 15.Satyanarayana T, Radhakrishnan A N. Biochim Biophys Acta. 1965;110:380–388. doi: 10.1016/s0926-6593(65)80045-5. [DOI] [PubMed] [Google Scholar]
- 16.Kiritani K, Narise S. J Biol Chem. 1966;241:2047–2051. [PubMed] [Google Scholar]
- 17.Arfin S M, Umbarger H E. J Biol Chem. 1969;244:1118–1127. [PubMed] [Google Scholar]
- 18.Blattner F R, Plunkett G, III, Bloch C A, Perna N T, Burland V, Riley M, Collado-Vides J, Glasner J D, Rode C K, Mayhew G F, et al. Science. 1997;277:1453–1474. doi: 10.1126/science.277.5331.1453. [DOI] [PubMed] [Google Scholar]
- 19.Julliard J H, Douce R. Proc Natl Acad Sci USA. 1991;88:2042–2045. doi: 10.1073/pnas.88.6.2042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Himmeldirk, K., Kennedy, I. A., Hill, R. E., Sayer, B. G. & Spencer, I. D. (1996) Chem. Commun. 1187–1188. [DOI] [PubMed]
- 21.Hill R E, Sayer B G, Spencer I D. J Am Chem Soc. 1989;111:1916–1917. [Google Scholar]
- 22.Kuzuyama T, Takahashi S, Watanabe H, Seto H. Tetrahedron Lett. 1998;39:4509–4512. [Google Scholar]
- 23.Yanisch-Perron C, Vieira J, Messing J. Gene. 1985;33:103–119. doi: 10.1016/0378-1119(85)90120-9. [DOI] [PubMed] [Google Scholar]
- 24.Sambrook J, Fritsch E F, Maniatis T. Molecular Clonning: A Laboratory Mannual. 2nd Ed. Plainview, NY: Cold Spring Harbor Lab. Press; 1989. [Google Scholar]
- 25.Bernardi A, Bernardi F. Nucleic Acids Res. 1984;12:9415–9426. doi: 10.1093/nar/12.24.9415. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Yamaguchi K, Masamune Y. Mol Gen Genet. 1985;200:362–367. doi: 10.1007/BF00425718. [DOI] [PubMed] [Google Scholar]
- 27.Sanger F, Nicklen S, Coulson A R. Proc Natl Acad Sci USA. 1977;74:5463–5467. doi: 10.1073/pnas.74.12.5463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Lipman D J, Person W R. Science. 1985;227:1435–1441. doi: 10.1126/science.2983426. [DOI] [PubMed] [Google Scholar]
- 29.Pearson W R, Lipman D J. Proc Natl Acad Sci USA. 1988;85:2444–2448. doi: 10.1073/pnas.85.8.2444. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Sprenger G A, Schorken U, Wiegert T, Grolle S, Graaf A A, Taylar S V, Begley T P, Bringer-Meyer S, Sahm H. Proc Natl Acad Sci USA. 1997;94:12957–12862. doi: 10.1073/pnas.94.24.12857. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Lois L M, Campos N, Putra S R, Danielsen K, Rohmer M, Boronat A. Proc Natl Acad Sci USA. 1998;95:2105–2110. doi: 10.1073/pnas.95.5.2105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Lange B M, Wildung M R, McCaskill D, Croteau R. Proc Natl Acad Sci USA. 1998;95:2100–2104. doi: 10.1073/pnas.95.5.2100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Chunduru S K, Mrachko G T, Calvo K C. Biochemistry. 1989;28:486–493. doi: 10.1021/bi00428a012. [DOI] [PubMed] [Google Scholar]
- 34.Sista H, Bowman B. Gene. 1992;120:115–118. doi: 10.1016/0378-1119(92)90018-k. [DOI] [PubMed] [Google Scholar]
- 35.Dumas R D, Curien G, Derose R T, Douce R. Biochem J. 1993;294:821–828. doi: 10.1042/bj2940821. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Dumas R, Lebrun M, Douce R. Biochem J. 1991;277:469–475. doi: 10.1042/bj2770469. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Petersen J G L, Holmberg S. Nucleic Acids Res. 1986;14:9631–9651. doi: 10.1093/nar/14.24.9631. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Walsh C. Enzyme Reaction Mechanisms. New York: Freeman; 1979. pp. 623–625. [Google Scholar]