Abstract
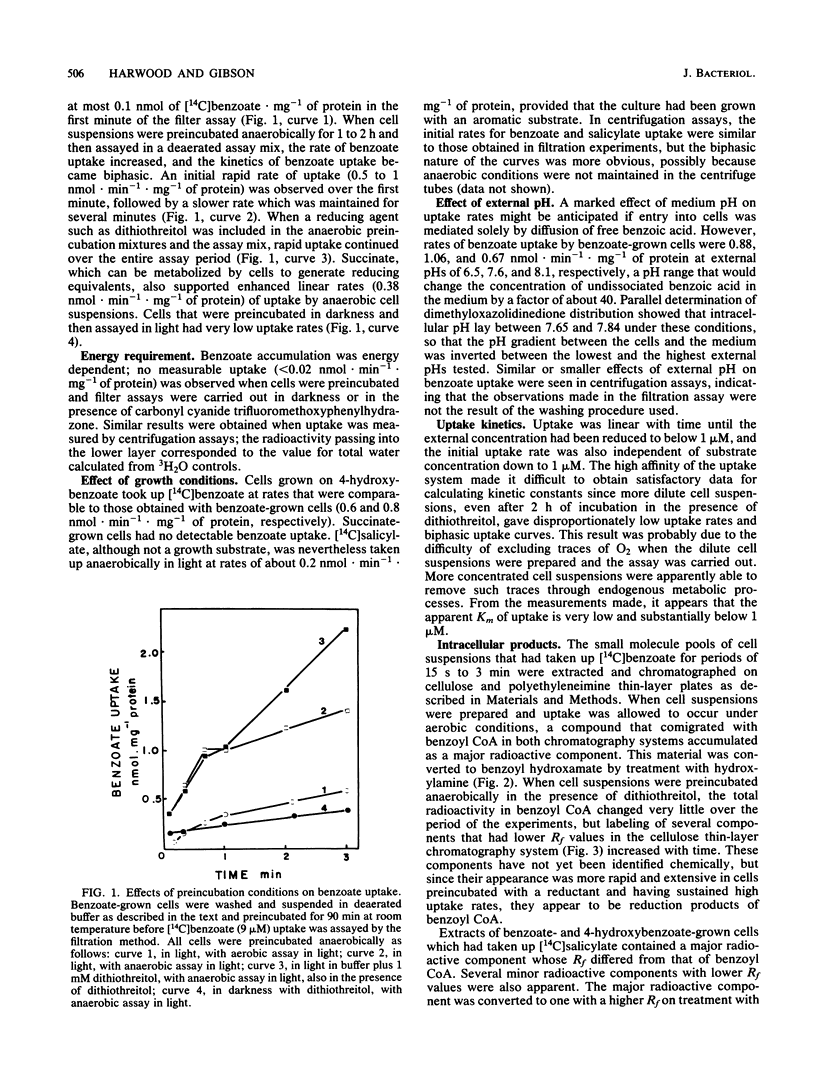
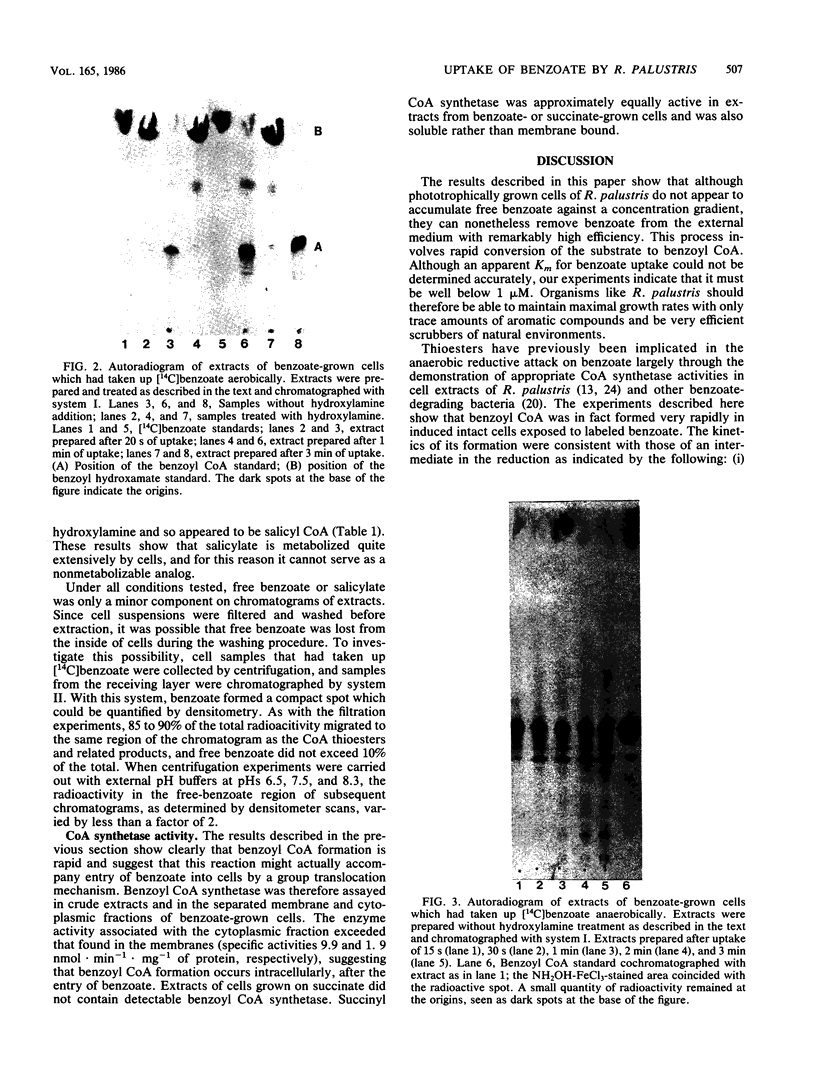
The uptake and anaerobic metabolism of benzoate were studied in short-term experiments with phototrophic cells of Rhodopseudomonas palustris. Cells that were preincubated and assayed anaerobically in the presence of 1 mM dithiothreitol accumulated [7-14C]benzoate at a rate of at least 0.5 nmol . min-1 . mg-1 of protein. Cells that were preincubated aerobically, or anaerobically in the absence of a reducing agent or an electron donor such as succinate, took up benzoate at reduced rates. Benzoate was removed from the external medium with remarkably high efficiency; initial uptake rates were independent of substrate concentration, and uptake remained linear down to concentrations of less than 1 microM. Uptake rates were not sensitive to external pH in the range of 6.5 to 8.1, and very little free benzoate was found associated with the cells. By contrast, benzoyl coenzyme A (CoA) was formed rapidly in cells exposed to labeled benzoate. Its appearance in such cells, together with the more gradual accumulation of other compounds tentatively identified as reduction products, is consistent with the identification of benzoyl CoA as an intermediate in the anaerobic reductive metabolism of benzoate. The very effective uptake of external benzoate can be explained by its conversion to benzoyl CoA immediately after its passage across the cell membrane by simple or facilitated diffusion. Such a chemical conversion would serve to maintain a downhill concentration gradient between the cell cytoplasm and the cell surroundings, even at very low external benzoate concentrations.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Blakley E. R. The microbial degradation of cyclohexanecarboxylic acid by a beta-oxidation pathway with simultaneous induction to the utilization of benzoate. Can J Microbiol. 1978 Jul;24(7):847–855. doi: 10.1139/m78-141. [DOI] [PubMed] [Google Scholar]
- Bradford M. M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem. 1976 May 7;72:248–254. doi: 10.1016/0003-2697(76)90527-3. [DOI] [PubMed] [Google Scholar]
- Dagley S. A biochemical approach to some problems of environmental pollution. Essays Biochem. 1975;11:81–138. [PubMed] [Google Scholar]
- Dutton P. L., Evans W. C. The metabolism of aromatic compounds by Rhodopseudomonas palustris. A new, reductive, method of aromatic ring metabolism. Biochem J. 1969 Jul;113(3):525–536. doi: 10.1042/bj1130525. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Evans W. C. Biochemistry of the bacterial catabolism of aromatic compounds in anaerobic environments. Nature. 1977 Nov 3;270(5632):17–22. doi: 10.1038/270017a0. [DOI] [PubMed] [Google Scholar]
- Frerman F. E. The role of acetyl coenzyme A: butyrate coenzyme A in the transferase uptake of butyrate by isolated membrane vesicles of Escherichia coli. Arch Biochem Biophys. 1973 Nov;159(1):444–452. doi: 10.1016/0003-9861(73)90472-4. [DOI] [PubMed] [Google Scholar]
- Gaensslen R. E., McCarty R. E. Amine uptake in chloroplasts. Arch Biochem Biophys. 1971 Nov;147(1):55–65. doi: 10.1016/0003-9861(71)90309-2. [DOI] [PubMed] [Google Scholar]
- Guyer M., Hegeman G. Evidence for a reductive pathway for the anaerobic metabolism of benzoate. J Bacteriol. 1969 Sep;99(3):906–907. doi: 10.1128/jb.99.3.906-907.1969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harwood C. S., Canale-Parola E. Adenosine 5'-triphosphate- yielding pathways of branched-chain amino acid fermentation by a marine spirochete. J Bacteriol. 1981 Oct;148(1):117–123. doi: 10.1128/jb.148.1.117-123.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kashket E. R. Stoichiometry of the H+-ATPase of growing and resting, aerobic Escherichia coli. Biochemistry. 1982 Oct 26;21(22):5534–5538. doi: 10.1021/bi00265a024. [DOI] [PubMed] [Google Scholar]
- Keith C. L., Bridges R. L., Fina L. R., Iverson K. L., Cloran J. A. The anaerobic decomposition of benzoic acid during methane fermentation. IV. Dearomatization of the ring and volatile fatty acids formed on ring rupture. Arch Microbiol. 1978 Aug 1;118(2):173–176. doi: 10.1007/BF00415726. [DOI] [PubMed] [Google Scholar]
- Kihara M., Macnab R. M. Cytoplasmic pH mediates pH taxis and weak-acid repellent taxis of bacteria. J Bacteriol. 1981 Mar;145(3):1209–1221. doi: 10.1128/jb.145.3.1209-1221.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Repaske D. R., Adler J. Change in intracellular pH of Escherichia coli mediates the chemotactic response to certain attractants and repellents. J Bacteriol. 1981 Mar;145(3):1196–1208. doi: 10.1128/jb.145.3.1196-1208.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schennen U., Braun K., Knackmuss H. J. Anaerobic degradation of 2-fluorobenzoate by benzoate-degrading, denitrifying bacteria. J Bacteriol. 1985 Jan;161(1):321–325. doi: 10.1128/jb.161.1.321-325.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thayer J. R., Wheelis M. L. Active transport of benzoate in Pseudomonas putida. J Gen Microbiol. 1982 Aug;128(8):1749–1753. doi: 10.1099/00221287-128-8-1749. [DOI] [PubMed] [Google Scholar]
- Thayer J. R., Wheelis M. L. Characterization of a benzoate permease mutant of Pseudomonas putida. Arch Microbiol. 1976 Oct 11;110(1):37–42. doi: 10.1007/BF00416966. [DOI] [PubMed] [Google Scholar]
- Whittle P. J., Lunt D. O., Evans W. C. Anaerobic photometabolism of aromatic compounds by Rhodopseudomonas sp. Biochem Soc Trans. 1976;4(3):490–491. doi: 10.1042/bst0040490. [DOI] [PubMed] [Google Scholar]
- Williams R. J., Evans W. C. The metabolism of benzoate by Moraxella species through anaerobic nitrate respiration. Evidence for a reductive pathway. Biochem J. 1975 Apr;148(1):1–10. doi: 10.1042/bj1480001a. [DOI] [PMC free article] [PubMed] [Google Scholar]