Abstract
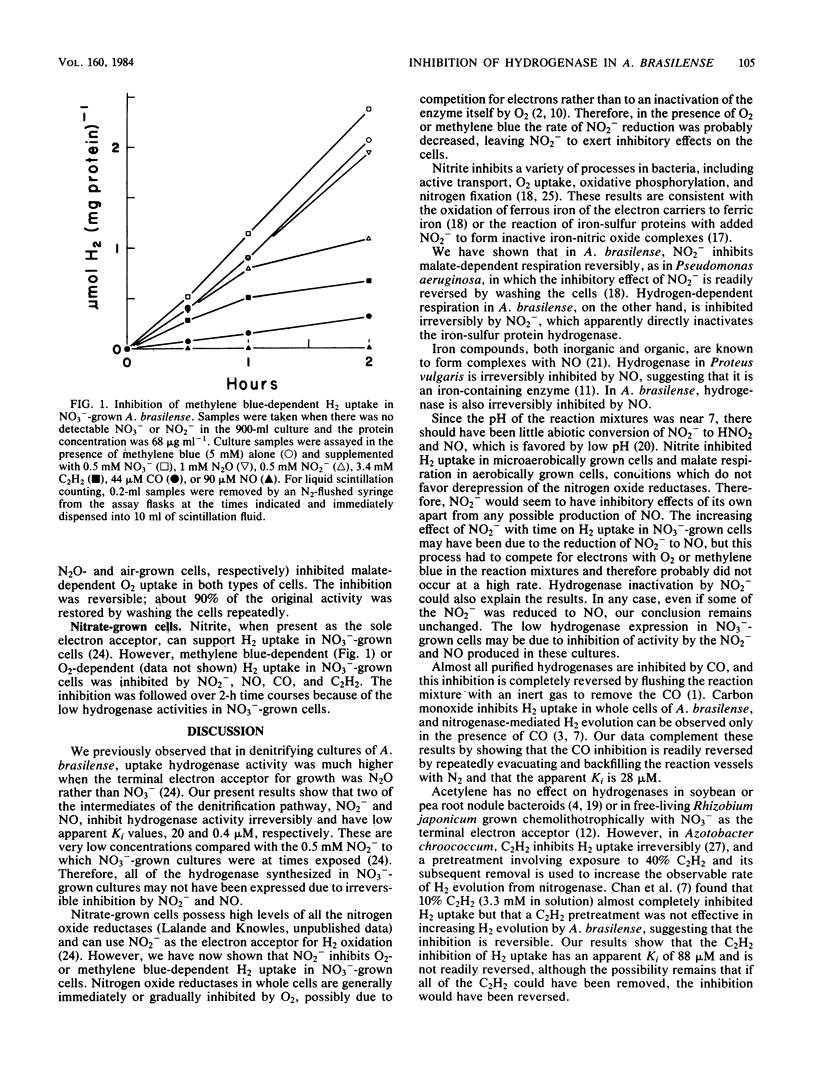
Nitrite, NO, CO, and C2H2 inhibited O2-dependent H2 uptake (H3H oxidation) in denitrifying Azospirillum brasilense Sp7 grown anaerobically on N2O or NO3-. The apparent Ki values for inhibition of O2-dependent H2 uptake were 20 microM for NO2-, 0.4 microM for NO, 28 microM for CO, and 88 microM for C2H2. These inhibitors also affected methylene blue-dependent H2 uptake, presumably by acting directly on the hydrogenase. Nitrite and NO inhibited H2 uptake irreversibly, whereas inhibition due to CO was easily reversed by repeatedly evacuating and backfilling with N2. The C2H2 inhibition was not readily reversed, partly due to difficulty in removing the last traces of this gas from solution. The NO2- inhibition of malate-dependent respiration was readily reversed by repeatedly washing the cells, in contrast to the effect of NO2- on H2-dependent respiration. These results suggest that the low hydrogenase activities observed in NO3(-)-grown cultures of A. brasilense may be due to the irreversible inhibition of hydrogenase by NO2- and NO produced by NO3- reduction.
Full text
PDF


Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Adams M. W., Mortenson L. E., Chen J. S. Hydrogenase. Biochim Biophys Acta. 1980 Dec;594(2-3):105–176. doi: 10.1016/0304-4173(80)90007-5. [DOI] [PubMed] [Google Scholar]
- Bethlenfalvay G. J., Phillips D. A. Variation in nitrogenase and hydrogenase activity of alaska pea root nodules. Plant Physiol. 1979 May;63(5):816–820. doi: 10.1104/pp.63.5.816. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bothe H., Tennigkeit J., Eisbrenner G. The utilization of molecular hydrogen by the blue-green alga Anabaena cylindrica. Arch Microbiol. 1977 Jul 26;114(1):43–49. doi: 10.1007/BF00429628. [DOI] [PubMed] [Google Scholar]
- Chan Y. K., Nelson L. M., Knowles R. Hydrogen metabolism of Azospirillum brasilense in nitrogen-free medium. Can J Microbiol. 1980 Sep;26(9):1126–1131. doi: 10.1139/m80-186. [DOI] [PubMed] [Google Scholar]
- Eskew D. L., Focht D. D., Ting I. P. Nitrogen fixation, denitrification, and pleomorphic growth in a highly pigmented Spirillum lipoferum. Appl Environ Microbiol. 1977 Nov;34(5):582–585. doi: 10.1128/aem.34.5.582-585.1977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Knowles R. Denitrification. Microbiol Rev. 1982 Mar;46(1):43–70. doi: 10.1128/mr.46.1.43-70.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Krasna A. I., Rittenberg D. THE INHIBITION OF HYDROGENASE BY NITRIC OXIDE. Proc Natl Acad Sci U S A. 1954 Apr;40(4):225–227. doi: 10.1073/pnas.40.4.225. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nelson L. M., Knowles R. Effect of oxygen and nitrate on nitrogen fixation and denitrification by Azospirillum brasilense grown in continuous culture. Can J Microbiol. 1978 Nov;24(11):1395–1403. doi: 10.1139/m78-223. [DOI] [PubMed] [Google Scholar]
- Neyra C. A., Döbereiner J. Denitrification by N2-fixing Sprillum lipoferum. Can J Microbiol. 1977 Mar;23(3):300–305. doi: 10.1139/m77-044. [DOI] [PubMed] [Google Scholar]
- Okon Y., Houchins J. P., Albrecht S. L., Burris R. H. Growth of Spirillum lipoferum at constant partial pressures of oxygen, and the properties of its nitrogenase in cell-free extracts. J Gen Microbiol. 1977 Jan;98(1):87–93. doi: 10.1099/00221287-98-1-87. [DOI] [PubMed] [Google Scholar]
- Reddy D., Lancaster J. R., Jr, Cornforth D. P. Nitrite inhibition of Clostridium botulinum: electron spin resonance detection of iron-nitric oxide complexes. Science. 1983 Aug 19;221(4612):769–770. doi: 10.1126/science.6308761. [DOI] [PubMed] [Google Scholar]
- SHANK J. L., SILLIKER J. H., HARPER R. H. The effect of nitric oxide on bacteria. Appl Microbiol. 1962 May;10:185–189. doi: 10.1128/am.10.3.185-189.1962. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smith L. A., Hill S., Yates M. G. Inhibition by acetylene of conventional hydrogenase in nitrogen-fixing bacteria. Nature. 1976 Jul 15;262(5565):209–210. doi: 10.1038/262209a0. [DOI] [PubMed] [Google Scholar]
- Tibelius K. H., Knowles R. Uptake hydrogenase activity in denitrifying Azospirillum brasilense grown anaerobically with nitrous oxide or nitrate. J Bacteriol. 1984 Jan;157(1):84–88. doi: 10.1128/jb.157.1.84-88.1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Trinchant J. C., Rigaud J. Nitrite and nitric oxide as inhibitors of nitrogenase from soybean bacteroids. Appl Environ Microbiol. 1982 Dec;44(6):1385–1388. doi: 10.1128/aem.44.6.1385-1388.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Walker C. C., Yates M. G. The hydrogen cycle in nitrogen-fixing Azotobacter chroococcum. Biochimie. 1978;60(3):225–231. doi: 10.1016/s0300-9084(78)80818-9. [DOI] [PubMed] [Google Scholar]
- van Berkum P., Bohlool B. B. Evaluation of nitrogen fixation by bacteria in association with roots of tropical grasses. Microbiol Rev. 1980 Sep;44(3):491–517. doi: 10.1128/mr.44.3.491-517.1980. [DOI] [PMC free article] [PubMed] [Google Scholar]


